Abstract
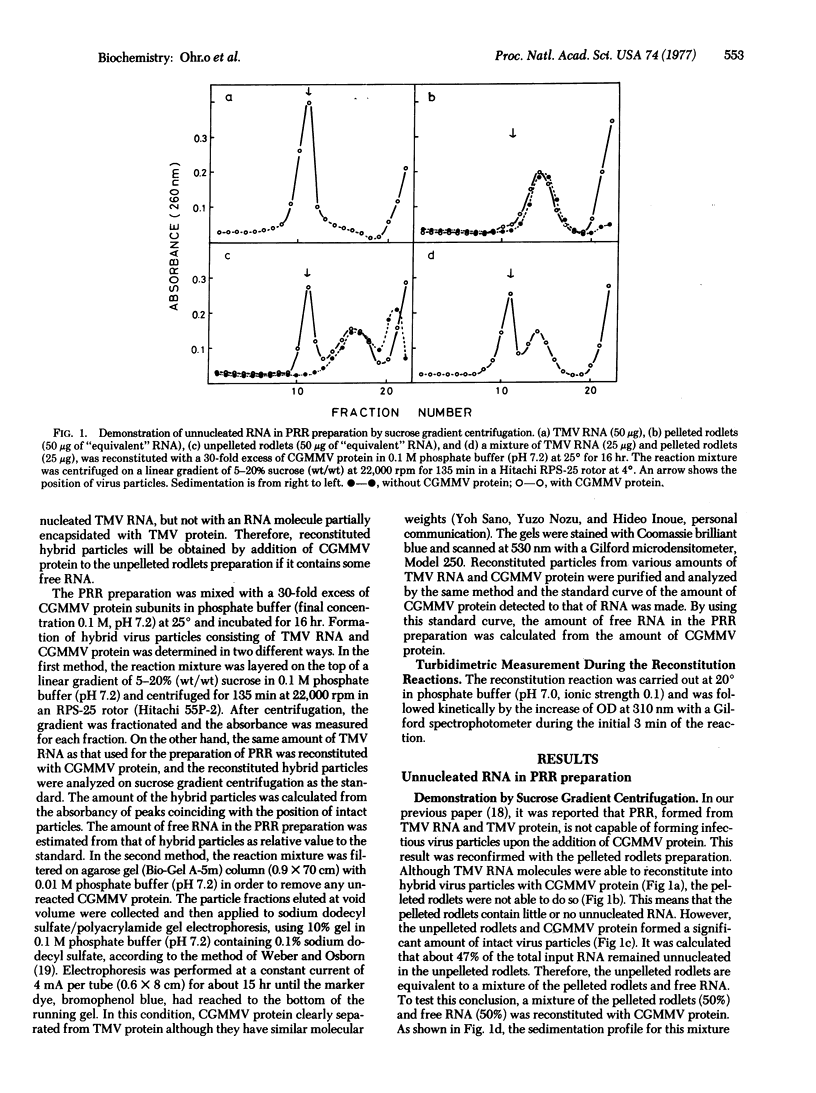
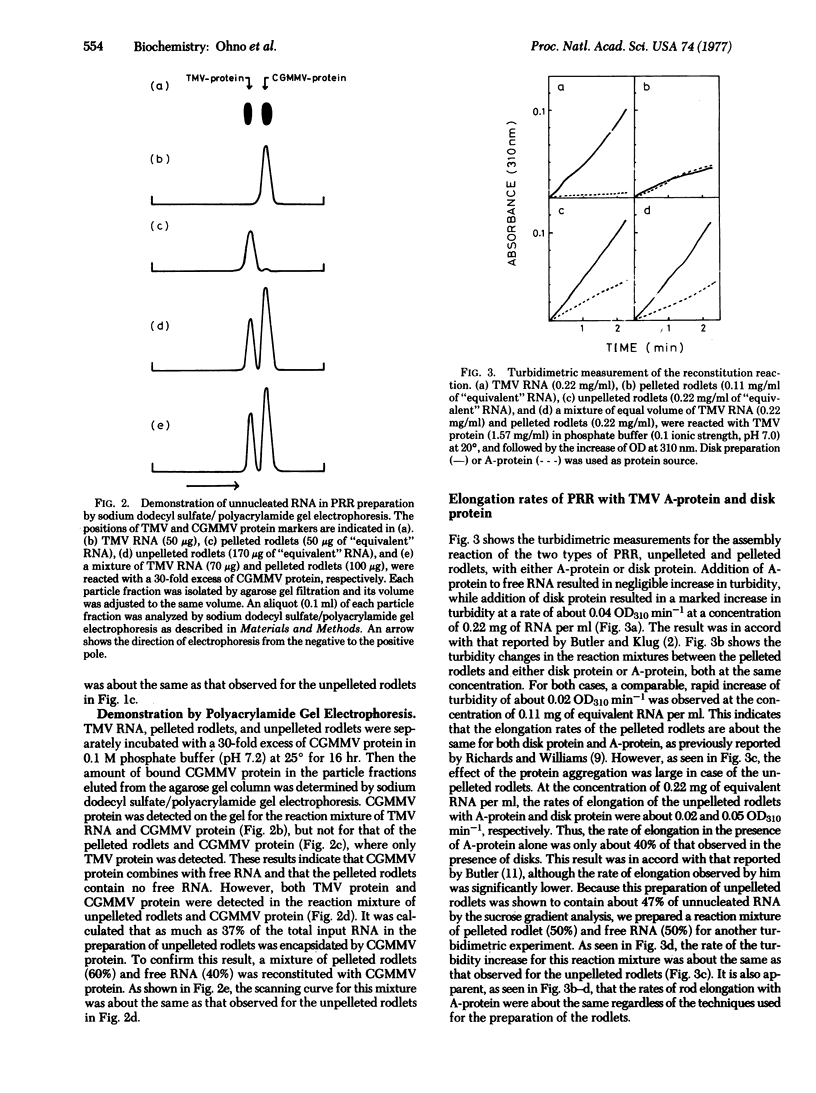
It was reported that the rate of elongation of partially reconstituted RNA, if prepared without centrifugation, showed dependency upon the state of protein aggregation like that found when free RNA was used as the nucleic acid source [Butler, P. J. G. (1974) J. Mol. Biol. 82, 333-341]. Butler concluded from this result that disks are the kinetically favored protein source for the elongation of the tobacco mosaic virus rod. We show that partially reconstituted RNA prepared without centrifugation always contains a significant amount of free RNA and conclude that the dependency of its elongation rate upon the state of protein aggregation is due to contamination of his partially reconstituted RNA preparation by free RNA. The elongation rate of partially reconstituted RNA, which had been pelleted and contained no free RNA, was found to be about the same regardless of the state of aggregation of the added protein.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Butler P. J., Klug A. Assembly of the particle of tobacco mosaic virus from RNA and disks of protein. Nat New Biol. 1971 Jan 13;229(2):47–50. doi: 10.1038/newbio229047a0. [DOI] [PubMed] [Google Scholar]
- Butler P. J. Structures and roles of the polymorphic forms of tobacco mosaic virus protein. 8. Elongation of nucleoprotein rods of the virus RNA and protein. J Mol Biol. 1974 Jan 25;82(3):333–341. doi: 10.1016/0022-2836(74)90594-4. [DOI] [PubMed] [Google Scholar]
- Durham A. C. Structures and roles of the polymorphic forms of tobacco mosaic virus protein. I. Sedimentation studies. J Mol Biol. 1972 Jun 20;67(2):289–305. doi: 10.1016/0022-2836(72)90242-2. [DOI] [PubMed] [Google Scholar]
- FRAENKEL-CONRAT H. Degradation of tobacco mosaic virus with acetic acid. Virology. 1957 Aug;4(1):1–4. doi: 10.1016/0042-6822(57)90038-7. [DOI] [PubMed] [Google Scholar]
- FRAENKEL-CONRAT H., SINGER B., TSUGITA A. Purification of viral RNA by means of bentonite. Virology. 1961 May;14:54–58. doi: 10.1016/0042-6822(61)90131-3. [DOI] [PubMed] [Google Scholar]
- Fraenkel-Conrat H., Williams R. C. RECONSTITUTION OF ACTIVE TOBACCO MOSAIC VIRUS FROM ITS INACTIVE PROTEIN AND NUCLEIC ACID COMPONENTS. Proc Natl Acad Sci U S A. 1955 Oct 15;41(10):690–698. doi: 10.1073/pnas.41.10.690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leberman R. The isolation of plant viruses by means of "simple" coacervates. Virology. 1966 Nov;30(3):341–347. doi: 10.1016/0042-6822(66)90112-7. [DOI] [PubMed] [Google Scholar]
- Nozu Y., Ono T., Okada Y. Amino acid sequences of some common Japanese strains of tobacco mosaic virus. J Biochem. 1970 Jul;68(1):39–52. [PubMed] [Google Scholar]
- Nozu Y., Tochihara H., Komuro Y., Okada Y. Chemical and immunological characterization of cucumber green mottle mosaic virus (watermelon strain) protein. Virology. 1971 Sep;45(3):577–585. doi: 10.1016/0042-6822(71)90173-5. [DOI] [PubMed] [Google Scholar]
- Okada Y., Oashi Y., Ono T., Nozu Y. Sequential reconstitution of tobacco mosaic virus. Virology. 1970 Sep;42(1):243–245. doi: 10.1016/0042-6822(70)90261-8. [DOI] [PubMed] [Google Scholar]
- Okada Y., Ohno T., Nonomura Y. Assembly of tobacco mosaic virus in vitro. Improved model for the elongation process by protein subunits. J Biochem. 1975 Jun;77(6):1157–1163. [PubMed] [Google Scholar]
- Ono T., Inoue H., Okada Y. Assembly of rod-shaped virus in vitro: reconstitution with cucumber green mottle mosaic virus protein and tobacco mosaic virus RNA. Proc Natl Acad Sci U S A. 1972 Dec;69(12):3680–3683. doi: 10.1073/pnas.69.12.3680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ono T., Okada Y., Nonomura Y., INOUE H. Assembly of a rod-shaped virus. Disk aggregate of cucumber green mottle mosaic virus protein and its function. J Biochem. 1975 Feb;77(2):313–319. doi: 10.1093/oxfordjournals.jbchem.a130728. [DOI] [PubMed] [Google Scholar]
- Ono T., Yamaura R., Kuriyama K., Inoue H., Okada Y. Structure of N-bromosuccinimide-modified tobacco mosaic virus protein and its function in the reconstitution process. Virology. 1972 Oct;50(1):76–83. doi: 10.1016/0042-6822(72)90347-9. [DOI] [PubMed] [Google Scholar]
- Richards K. E., Williams R. C. Assembly of tobacco mosaic virus in vitro: effect of state of polymerization of the protein component. Proc Natl Acad Sci U S A. 1972 May;69(5):1121–1124. doi: 10.1073/pnas.69.5.1121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richards K. E., Williams R. C. Assembly of tobacco mosaic virus rods in vitro. Elongation of partially assembled rods. Biochemistry. 1973 Nov 6;12(23):4574–4581. doi: 10.1021/bi00747a005. [DOI] [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]


