Abstract
Purpose
Infection following knee replacement is an important cause of failure despite rigorous prophylaxis antibiotic protocols. The two-stage reimplantation procedure is considered the gold standard for treatment of subacute and chronic deep periprosthetic infections. The purpose of this study was to determine whether or not a preformed articulated spacer would allow comparable eradication of infection equal to rates reported in published studies and to see whether there is a resulting improvement in postoperative function with an acceptable quality of life, reducing postoperative pain and limiting surgical complications, thus simplifying the second stage of the procedure.
Methods
We retrospectively reviewed 50 patients with infected TKA who underwent a two-stage exchange arthroplasty using an articulating preformed spacer. The device, designed like an ultracongruent condylar knee prosthesis, is composed of acrylic cement impregnated with antibiotic, with tested and standardised mechanical properties and antibiotic content and release mechanism.
Results
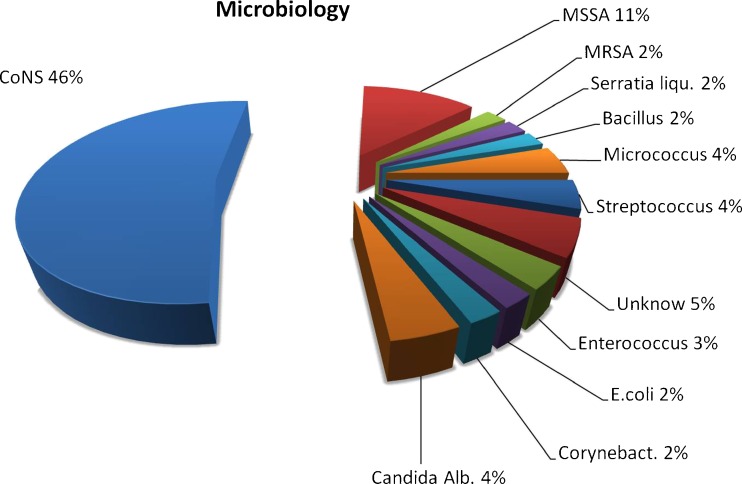
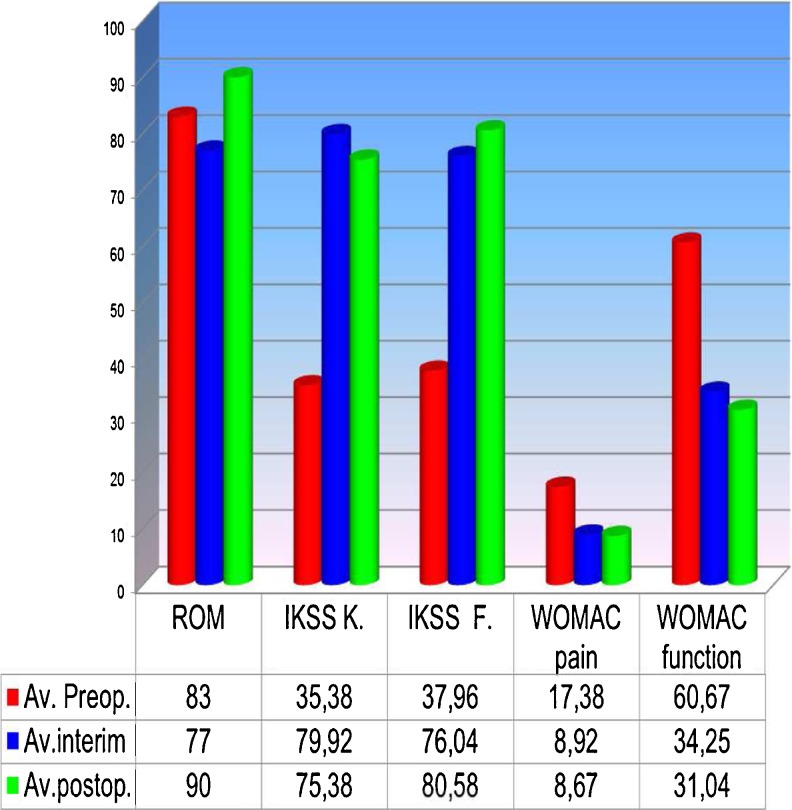
The median follow-up period was seven (two to 13) years. Two-stage exchange arthroplasty was successful in controlling the infection in 92 % of patients; 64% of patients where women, and median patient age was 68 (54–80) years. Median implantation time of the preformed spacer was 16 (four to 60) weeks; 4 % of infections were delayed, and 96 % were late. Forty-six percent were caused by coagulase-negative Staphylococcus (CoNS). Mean Knee Society Score (KSS) was 35.38 (clinical) and 37.96 (function) on presentation; it improved to a mean of 72.92 (clinical) and 76.04 (function) after the first stage and to a mean of 75.38 (clinical) and 80.58 (function) at the final review. Bone loss was unchanged between stages, and range of motion remained unchanged or improved after definitive reimplantation.
Conclusion
The use of preformed articulated knee spacer during a two stage technique for infected TKA improves patient QOL between stages and increases patient compliance and cooperation, reducing social costs.
Introduction
Total knee replacement (TKR) is one of the most common and successful procedures in orthopaedic surgery. Excellent long-term results are reported both in young adult and elderly patients [1–3]. However, infection remains an important cause of failure, notwithstanding rigorous antibiotic prophylactic protocols. The success of our prostheses depends upon the winner of “the race for the surface”: This elegant pathogenetic metaphor for the situation occurring shortly after the insertion of implants was proposed by Gristina et al. [4]. The race is influenced by prosthetic surface characteristics, host factors, and number of bacteria and their generic characteristics. Orthopaedic biomaterials are foreign bodies, and their surfaces can provide a point of attachment for bacteria colonies, which use slime and biofilm as a means of protection and permits their survival. Biofilm inhibits immune activity by multiple mechanisms: it decreases or inhibits polymorphonuclear chemotaxis, complement activation, opsonisation, phagocytosis and antibody susceptibility. All these mechanisms facilitate infection chronicity [5]. The incidence of prosthetic joint infection is grossly underestimated by current literature [59]. The risk increases in revision surgery or in patients with certain risk factors, such as patients with rheumatoid arthritis under cortisone treatment, diabetes mellitus or compromised immune status [6]. Data regarding these complications differ depending whether they are collected by a selected series, large series or open registry and range from 1.5 % to 2.5 % in primary TKA up to 6 % in revision of selected series [36, 56]. In large series of early revisions, infection is a cause of failure in 15 % –40 % of cases [7, 8, 40]. Treating this devastating complication is controversial, and many therapeutic options are described in the literature—simple parenteral antibiotic therapy, repeated joint irrigation, surgical toilette for saving the prosthetic implant, resection arthroplasty, one- or two-stage reimplantation (early or late) and arthrodesis or amputation [9–11]. The choice of the treatment depends upon many variables, such as infection type, responsible organism and patient general condition and life expectancy. Prosthetic infections are classified using chronological criteria [6]:
Early: onset within 12 weeks from surgery
Delayed: onset within two years from surgery
Late: onset after a longer period from surgery and caused by hematogenous spread from a distant infection site
Acute infection with early onset can be treated with extensive and accurate surgical toilette together with substitution of the polyethylene component. This type of treatment is insufficient in delayed or late infections involving the bone–implant interface [12]. In such cases, the best results, both for disease eradication and appropriate functional recovery, have been obtained with either one- or two-stage reimplantation. One-stage surgery has been successful in small series and has the advantage of less surgical risk and discomfort, ability to maintain joint function and lower hospital costs [13]. Generally, however, the effectiveness doubtful when considering reports of larger series with longer follow-up times [14–16]. The best results in terms of disease eradication and functional recovery have been obtained with two-stage reimplantation. [17–23, 25, 26, 47, 50]. The first stage of the procedure involves removing the original prosthesis and inserting an antibiotic cement spacer. Different types of spacers have been advocated for treatment of chronically infected knee arthroplasties: block spacers, articulated spacers such as Prostalac®, prosthesis re-sterilised (Hoffmann technique) and the antibiotic-loaded acrylic cement (ALAC) spacer (handmade with or without using a mould; preformed). We present additional follow-up of a technique described earlier [23, 25, 34, 57] using in the first stage a preformed, articulating spacer that allows delivery of high local concentrations of antibiotics while giving patients a functional joint before a second-stage reimplantation. The purpose of the study was to determine whether or not a preformed articulated spacer would allow comparable eradication of infection equal to rates reported in published studies and to see whether there is a resulting improvement in postoperative function with an acceptable quality of life (QOL), reducing postoperative pain and limiting surgical complications, thus simplifying the second stage of the procedure.
Materials and methods
From March 2000, we routinely used the preformed, articulated spacer in a two-stage procedure. The device is an industrially preformed polymethylmethacrylate (PMMA) spacer available in two versions: impregnated with gentamicin (Spacer K®) or with gentamicin plus vancomycin (Vancogenx-Space Knee®) Tecres, Italy. The single-antibiotic version contains 2.8 % w/w of active gentamicin (equivalent to 1.1 g/40 g PMMA); the double antibiotic spacer contains 2.8 % w/w of active vancomycin. The total amount of antibiotic is in relation to the device size. The spacer is composed of two independent elements ready to use: the first is a flat base (tibial component) upon which the second (femoral component) articulates. The two components, designed as an ultracongruent condylar knee prosthesis, must be fixed to the bone with bone cement (Fig. 1). The components are manufactured in different sizes, with tested and standardised mechanical properties and antibiotic content, and a release mechanism that functions during the period following implantation [23–25, 34, 57]. The device was mechanically tested following the International Standards Organization (ISO) and ASTM standards used for definitive prostheses (500,000 load cycles, corresponding to 6 months of walking) without failure during the test. [31] The in vitro release was tested according to the European Pharmacopoeia (agar-well diffusion test). [23, 24] With approval from the ethical committee board, 50 consecutive patients with mainly type A or B [26] affected by delayed and late infection were enrolled in our prospective study, which had no control group. All patients with infected TKA requiring two-stage exchange underwent preoperative aspiration [culture and white blood cell (WBC) count] and specimen histology (bone and synovial fluid) from any area with suspected infection. No patient received pre-operative or intra-operative antibiotics (at least three weeks of antibiotic washout) [26, 27]. Local anaesthetics were not used because of their bacteriostatic properties. After periprosthetic infection was diagnosed, patients underwent a two-stage procedure. During the first stage of treatment, the knee was approached through a midline incision incorporating the prior incision, and a standard medial parapatellar arthrotomy was performed. All components were removed, taking care to ensure minimal bone loss but with aggressive debridement of infected and devitalised tissues [60].
Fig. 1.
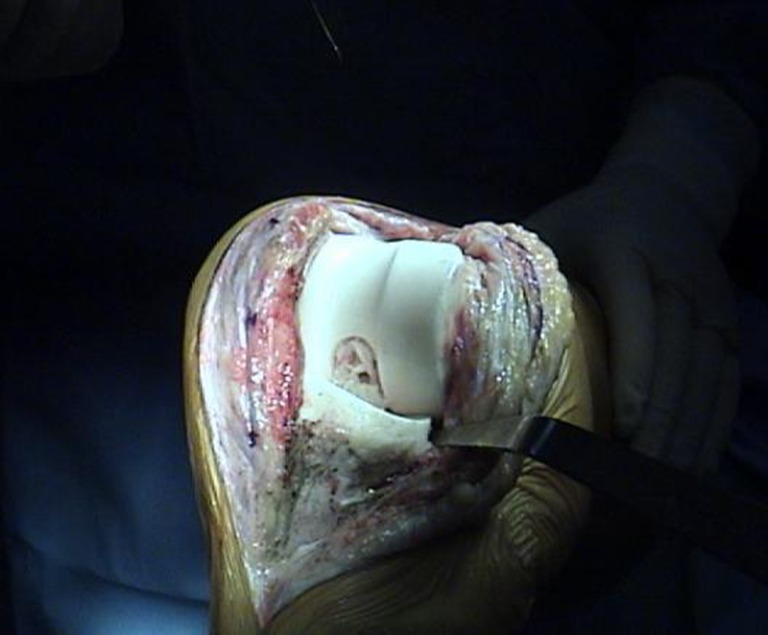
Industrially preformed device with standardised mechanical and pharmacological performance in situ
All PMMA material was removed with hand and power tools, including high-speed burrs. The wound was irrigated with extensive pulsatile lavage using saline solution. The moulded dowels were attached if necessary with extra cement to the prepared femoral and tibial spacer components. The preformed articulating spacer was implanted securely with appropriate antibiotic-loaded cement based upon microbiological cultures. A working extensor mechanism and ligamentous apparatus, with bone loss type I or II [29], was mandatory. During the procedure, synovial fluid samples were taken for microbiological analysis WBC count, and periprosthetic tissue samples were used for microbiological examination. When it was not possible to identify the infecting germ preoperatively, the hardware removed was subjected to scraping with a double-scalpel blade [one blade was sent to microbiology and and one for scanning electron microscopy (SEM) analysis] [28]. Individual prosthetic components were sent to microbiology for sonication in most cases but not in all. All patients received six weeks of tailored antibiotic therapy IV (minimum two types of antibiotic) suggested by the infectious disease consultant. Postoperative standard rehabilitation was the same as for the primary TKA: patients were encouraged to actively mobilise the knee immediately after surgery, with partial weight bearing permitted. The spacer was left in place until clinical healing of soft tissue and normal laboratory parameters. The second stage of the procedure was undertaken after a mean of 16 (median average four to 60) weeks. Range of motion (ROM) was recorded before arthroplasty removal, while the device was working, and at the last follow-up of the final implant. Western Ontario and McMaster Osteoarthritis Index (WOMAC) and Knee Society Scoere (KSS) outcomes were assessed on the basis of the patient response. We decided to introduce an original clinical evaluation between stages to help determine the potential advantages of our method to determine QOL: satisfaction score was evaluated using subjective patient evaluation regarding daily activities (scored on four different levels: excellent, good, fair, poor), postoperative pain, use of crutches ( two, one, none) and ability to walk (unlimited: up to 1 km; good: up to 800 m less than 1 km; fair: <400 m; poor: only at home). Bone loss was assessed radiographically and defined intra-operatively according to the Anderson Orthopaedic Research Institute (AORI) classification [29] at both the first and second stage of the revision. We also considered surgical approach for the second stage, type of revision implant [60], infection recurrence and complications.
Results
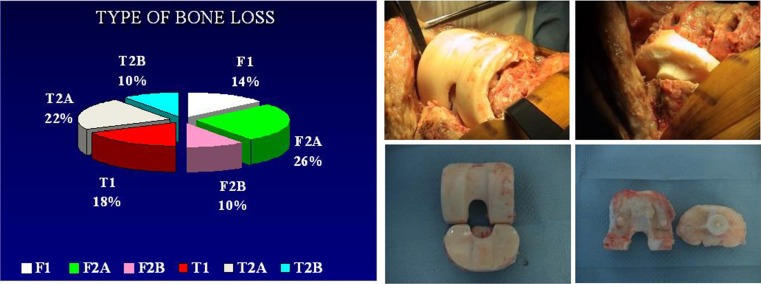
Diagnosis of infection was confirmed on the basis of positive cultures of pre-operative aspirates, intra-operative tissue specimens and increased C-reactive protein levels. [27, 28] In 5 % of patients, it was not possible to identify the pathogen. Sixty-four percent of patients were women, and mean patient age was 68 (54–80) years. Using chronological criteria, infections were delayed in 4 % and late in 6 %. The most common infecting organism was coagulase-negative Staphylococcus (CoNS), in 46 % of cases (Fig. 2). Comorbidities based upon McPherson’s classification[26] indicated that 35 % of patients were type B and 8 % were type C. Body mass index (BMI) was <30 >25 in 48 % and <40 >30 in 7 % of patients. All patients were followed up for a minimum of two and a maximum of 13 (mean seven) years. Four patients died within an average follow-up of 39 months (car accident), 42 months (gastric cancer) and 77 and 90 months (age related); these patients where clinically free of infection. Mean spacer implantation time was 16 (four to 60) weeks. Two-stage exchange arthroplasty was successful in controlling the infection in 92 % of patients. Persistent original infection (two patients) or recurrent infection with a new organism (two patients) was diagnosed after 17, 15, five and 14 months following previous reimplantation. These patients were not healthy enough to undergo a second reimplantation procedure and were managed with chronic antibiotic suppression and irrigation (three patients) and débridement with substitution of the polyethylene component and retention of prosthesis (one patient). (Table 1) Mean preoperative ROM was 59° (range 5°–90°), was almost unchanged during the interstage period, with the articulating spacer being 77° (range 10°–100°); ROM improved after insertion of the final prosthesis to 94° (0°–120°). Mean KSS was 35.38 (clinical) and 37.62 (function) on presentation, and improved to a mean of 72.92 (clinical) and 76.04 (function) after the first stage and a mean of 75.38 (clinical) and 80.58 (function) at final review. Mean WOMAC (function and pain scores) were 17.38 and 60.67 on presentation, respectively, improving to a mean of 8.92 and 34.25, respectively, after the first stage and 8.67 and 31.04, respectively, at final review (Fig. 3). Patients judged the result as excellent or good in 76 % of cases, fair in 16 % and poor in 8 %; 71 % reported no pain, and 29 % reported mild discomfort. During the interim period, the use of crutches is mandatory, and the patients began walking two days postsurgery. Two weeks later, 77 % of patients used only one crutch, but one third of this percentage stated they were capable of walking without crutches. A brace was used for the first postoperative week four patients only. The ability to walk was unlimited in 24 % of patients, good in 52 %, fair in 16 % and poor in 8 %. All knees had anatomical and functional integrity of the extensor apparatus and adequate mediolateral stability except one. Eleven knees were rated type I, 35 type II and four type III for bone loss according to AORI criteria [29] (Fig. 4). Histology indicated periprosthetic tissues in the first stage showed a slight inflammatory reaction at the ultrastructural level, with giant plurinuclear cells and histiocyte elements (Fig. 5). No spacer wear or breakage was reported. Spacer removal was easy, with no change in previous AORI classification[29] or in the shape of the bone itself. At the time of revision TKA in 20 % of knees, flexion was <50° and required a quadriceps snip or V-Y turndowns [30]. Tibial tubercle osteotomy was performed for exposure. During the first stage, the following prosthesis types were removed: 62 % posterior-stabilized (PS), 33 % cruciate-retaining (CR), 5 % hinge; for the second stage, the following types were required: PS in 30 %, varus/valgus constrained (VVC) in 55 % and hinge in 15 %. No patient had evidence of renal toxicity. No above-knee amputation was necessary in this series. Without apparent correlation to the device, we found one asymptomatic heterotopic ossification of the quadriceps, one temporary skin-healing problem in a patient with numerous previous surgical scars (this complication required us to maintain the K spacer in place for 24 weeks; no mechanical problems arose) and one spacer dislocation due to extensor apparatus instability (incorrect pre-operative indication).
Fig. 2.
The infecting organism determined by culture result
Table 1.
Failed infection control
| Gender | Patient type* | First infection | Second infection | Follow-up (months) | Treatment |
|---|---|---|---|---|---|
| Relapse | |||||
| Male | B | CoNS | Doubtful (1 sample over 8 positive for CoNS) | 17 | AB suppression |
| Female | C | Polymicrobial | Polymicrobial | 15 | Arthroscopic lavage/AB suppression |
| Reinfection | |||||
| Male | C | Streptococcus | CoNS | 5 | Arthroscopic lavage/AB suppression |
| Female | B | CoNS | MSSA (sepsis) | 14 | Debridement with retention of prosthesis, arthroscopic lavage/AB suppression |
CoNS coagulase-negative Staphylococcus, MSSA methicillin-sensitive Staphylococcus aureus, AB antibiotic
* = Mc. Phearson cl. [26]
Fig. 3.
Range of motion (ROM) and functional outcome
Fig. 4.
Type of bone loss (unchanged during the procedure). Nomacroscopic signs of wear or breakage of the device
Fig. 5.
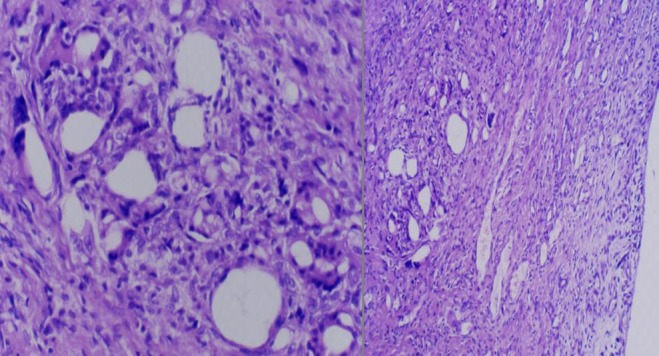
Histology shows giant plurinuclear cells and histiocytet elements
Discussion
Two-stage reimplantation has proven to be the most successful method of treating chronically infected TKAs. Different types of spacers have been advocated to treat such infections. Borden and Gearen[37] first used antibiotic-impregnated cement beads or block spacers for two-stage delayed reimplantation. Despite encouraging results in infection eradication, the block spacer presents several disadvantages: the patient is not allowed to move the knee, cast immobilisation is required between stages and the spacer can dislodge and cause bone erosion [57, 60]. Moreover, second-stage surgery is difficult due to a scar formation, tissue adherence and quadriceps shortening. To overcame the disadvantages of block spacers and facilitate reimplantation surgery, several authors independently introduced articulated spacers. A custom-made articulated spacer of metal and plastic coated with antibiotic-impregnated cement was proposed by Duncan et al. [19, 39] (Prostalac®). McPherson et al. [26] proposed the use of a cement spacer moulded intra-operatively. Hofmann et al. [20, 43] described the use of an articulated spacer created from the removed and re-sterilised femoral component in association with a new polyethylene component fixed with antibiotic-impregnated cement. Castelli et al. [23–25, 34] introduced a industrially preformed articulating spacer. MacAvoy et al. [44] used a relatively constrained ball-and-socket articulating antibiotic-impregnated cement spacer. Cerulli et al. [58]. proposed the use of an articulated antibiotic-loaded cement spacer assembled with two unicompartmental implants.
The purpose of the study reported here was to determine whether a preformed articulated spacer would allow comparable eradication of infection equal to rates reported in published studies, improved postoperative function and acceptable QOL while reducing postoperative pain and limiting surgical complications. To date our reinfection rate is 8 %, similar to that achieved by other surgeons with a follow-up comparable with ours. Duncan and colleagues [19, 39], based on 45 patients with knee infection, found that recurrence of infection occurred in 9 % at mean follow-up of 48 months. Hofmann et al. [20, 43] reported on 50 patients with a 12 % recurrence rate of infection at a mean follow-up of 73 months. Emerson [38], only assessing patients with the static block spacer, report a reinfection rate of 30 % in a 12-year follow-up. We agree that the ability to bend the knee between stages improves patient satisfaction without compromising infection eradication, but the definition of complete healing of the infectious process would, however, require a longer period of observation. Also, it is remarkable that reinfection rate depends more on time elapsed rather than on surgical technique [41]. PMMA cement is the most useful, suitable and studied local antibiotic delivery system [17, 33, 35]; however, data from the literature are difficult to compare because of the difference in elution methods, experimental conditions, cements and in vitro models used. An important aspect that must be considered is the compatibility between antibiotic type and dose and PMMA. Not all antibiotics work in the same way, and there is a strict relationship between mechanical performance, antibiotic load and release. High-dose spacers and antibiotic combinations are often advocated [32] (Table 2); however, some authors [46] do not support this opinion. Release is affected by antibiotic dose only in the very initial phase [33]. Therefore, it seems more appropriate to replace the concept of antibiotic dose with the concept of antibiotic release: antibiotic dose in a spacer is not relevant; more relevant is the possibility of a high and prolonged release of antibiotic from the spacer. QOL using a preformed spacer between stages is quite satisfactory, and the ability to bend the knee between stages improves patient satisfaction without compromising infection eradication. With the exception of our first three cases in which a brace was used for the first 7 days postoperatively, we never immobilised the knee. Knee motion was pain free in most cases, with an ROM that remained unchanged between the first and second stages or improved after definitive reimplantation in the stiff knee during the first stage. Of the 77% of patients who used only one crutch, at least one third admitted that they could walk without crutches. Few authors report details of findings regarding ROM and QOL between first- and second-stage surgery. Duncan et al. [19, 39] reported that the average Hospital for Special Surgery (HSS) score during the interim period was 55.9, with a mean flexion of 76.1°. However, cement spacers moulded in the operating theatre do not have reproducible mechanical characteristics, and there is a potential risk of component fracture [48]. We believe that the preformed spacer works in a similar fashion to the intermediate prosthesis, but the major drawback of the intermediate prosthesis is the presence of hardware that could theoretically favour glycocalyx formation and bacterial adhesion [52, 53]. Moreover, with the preformed spacer, it is not necessary to wait until the hardware is sterilised.
Table 2.
Antibiotic polymethylmethacrylate (PMMA) spacer doses reported in the literature
| Author | Year | AB/40 U PMMA | Dose (g) |
|---|---|---|---|
| Hofmann et al. [20] | 1995 | Tobramycin | 4.8 |
| Haddad et al. [22] | 2000 | Tobramycin | 1.2–3.6 |
| Vancomycin | 0.5–1.5 | ||
| Emerson et al. [38] | 2002 | Tobramycin | 3.6 |
| Vancomycin | 2.0 | ||
| Durbhakula et al. [42] | 2004 | Tobramycin | 2.4 |
| Vancomycin | 1 | ||
| Springer et al. [61] | 2004 | Gentamicin | 3.6–9.2 |
| Vancomycin | 3–16 | ||
| Meek et al. [62] | 2004 | Tobramycin | 3.6 |
| Vancomycin | 1.5 | ||
| Castelli and Ferrari [64] | 2006 | Gentamicin | 1 + (0.8–2.7)a |
| Vancomycin | 2 + (0.9–2.7)b | ||
| Ghanem et al. [63] | 2007 | Tobramycin | 3.6 |
| Vancomycin | 4 |
AB antibiotic
aIndustrially preformed spacer Genta
bIndustrially preformed spacer Genta/Vanco
Removal of the preformed spacer proved easy, and in no case was there progression in osseous defect on either the femoral or tibial side. Other authors [22, 38] did not specifically evaluate bone loss; however, Fehring et al. [54, 55] performed a comparison of static spacer blocks with an articulating spacer and reported that 60 % of patients with static spacers had either tibial or femoral bone loss. At the same time, the authors found no bone loss between stages in the 30 patients who underwent reconstruction with an articulated spacer. Regarding surgical technique in our study, all knees were surgically exposed at the second stage through the previous incision using a medial parapatellar arthrotomy. The quadriceps snip was always used at the second stage by other surgeons [30, 49]. We used it at the first stage in knees with flexion <50°. A tibial tubercle osteotomy at the second stage was never necessary. For the revision implant, we used a PS type in 30 % of patients; in 55 %, we used VVC and in 15 % a hinge implant was required. These results demonstrate that good ligament balance is possible at the second stage. The hinge implant was used in knees that were stiff from the first stage.
One limitation of our study is the absence of a control group managed with either a static spacer or an intra-operatively moulded spacer. Such a study would require a consistent sample size, which is a difficult task with this kind of pathology.
Two-stage revision is considered the gold standard in treating TKA infections. Following an accurate and thorough surgical debridement, along with the selection of the correct systemic antibiotic therapy, the high and prolonged antibiotic release guaranteed by the ALAC spacer is key [17–23, 25, 26]. Articulating spacers offer a better functional outcome compared with static spacers [45, 51] while showing similar infection eradication rate. Infection recurrence depends upon microbiology results, patient classification type and follow-up length. We chose an industrially preformed knee spacer in order to obtain a high, prolonged, standardised antibiotic release [35] while allowing the excellent mechanical performance [31] guaranteed by the industrial manufacturing process.
Acknowledgement
We thank the entire team in this study, especially C.Farina, A.Raglio and A. Grigis, Department of Microbiology; S. Buoro, Department of Clinical Biochemistry laboratory, G. Virotta, Department of Nuclear Medicine, G. Gregis, Departement of Infectious Diseases; Ospedale Papa Giovanni XXII Bergamo, Italy; A. Remuzzi, Engineering, Mario Negri Institute, Italy; G. Calonego, Tecres, Italy
Conflict of interest
No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article.
References
- 1.Ranawat CS, Flynn WF, Saddler S, et al. Long-term results of the total condylar knee arthroplasty. Clin. Orthop. 1993;286(94):1993. [PubMed] [Google Scholar]
- 2.Diduch DR, Insall JN, Scott WN, et al. Total knee replacement in young, active patients. Long term of follow-up and functional outcome. J.B.J.S. Am. 1997;79:575. doi: 10.2106/00004623-199704000-00015. [DOI] [PubMed] [Google Scholar]
- 3.Robertsson O, Knutson K, Lewold S, et al. Knee arthroplasty in reumatoid arthritis. A report from the Swedish Knee Arthroplasty Register on 4381 primary operations 1985–1995. Acta Orthop. Scand. 1997;68(6):545. doi: 10.3109/17453679708999024. [DOI] [PubMed] [Google Scholar]
- 4.Gristina AG, Naylor P, Myrvik Q. Infections from biomaterials and implants: a race for the surface. Med Prog Technol. 1988;14(3–4):205–224. [PubMed] [Google Scholar]
- 5.Costerton JW, Stewart PS, Greenberg EP. Bacterial biofilms: a common cause of persistent infections. Science. 1999;284(5418):1318–1322. doi: 10.1126/science.284.5418.1318. [DOI] [PubMed] [Google Scholar]
- 6.Segawa H, Tsukayama DT, Kyle RF, Becker DA, Gustilo RB. Infection after total knee arthroplasty. A retrospective study of the treatment of eighty-one infections. J Bone Joint Surg Am. 1999;81:1434–1445. doi: 10.2106/00004623-199910000-00008. [DOI] [PubMed] [Google Scholar]
- 7.Sharkey PF, Hozack WJ, Rothman RH, Shastri S, Jacoby SM Insall Award paper Why are total knee arthroplasties failing today? Clin Orthop Relat Res. 2002;404:7–13. doi: 10.1097/00003086-200211000-00003. [DOI] [PubMed] [Google Scholar]
- 8.Fehring TK, Odum S, Griffin WL, Mason JB, Nadaud M. Early failures in total knee arthroplasty. Clin Orthop Relat Res. 2001;392:315–318. doi: 10.1097/00003086-200111000-00041. [DOI] [PubMed] [Google Scholar]
- 9.Cuckler JM, Star AM, Alavi A, Noto RB. Diagnosis and management of the infected joint arthroplasty. Clin Orthop North Am. 1991;22:523. [PubMed] [Google Scholar]
- 10.Rand JA. Alternatives to reimplantation for salvage of the total knee arthroplasty complicated by infection. J Bone Joint Surg. 1993;75:282. doi: 10.2106/00004623-199302000-00018. [DOI] [PubMed] [Google Scholar]
- 11.Insall JN (1986) Infection of total knee arthroplasty. AAOS Instr Course Lect, St. Louis, CV Mosby, p 319 [PubMed]
- 12.Borden LS, Gearen PF. Infected total knee arthroplasty. J Arthroplasty. 1987;2:27–36. doi: 10.1016/S0883-5403(87)80028-1. [DOI] [PubMed] [Google Scholar]
- 13.Goksan SB, Freeman MAR. One stage reimplantation for infected total knee arthroplasty. J Bone Joint Surg Br. 1992;74:78. doi: 10.1302/0301-620X.74B1.1732271. [DOI] [PubMed] [Google Scholar]
- 14.Johnson DP, Bannister GC. The outcome of infected arthroplasty of the knee. J.B.J.S. 1986;68B:289–291. doi: 10.1302/0301-620X.68B2.3958017. [DOI] [PubMed] [Google Scholar]
- 15.Romanò CL, Gala L, Logoluso N. Two-stage revision of septic knee prosthesis with articulating knee spacers yields better infection eradication rate than one-stage or two-stage revision with static spacers. KSSTA. 2012;2012:2445–2453. doi: 10.1007/s00167-012-1885-x. [DOI] [PubMed] [Google Scholar]
- 16.Masters JPM, Smith NA, Foguet P, Reed M, Parsons H, Sprowson PA. A systematic review of the evidence for single stage and two stage revision of infected knee replacement. BMC Musculoskelet Disord. 2013;2013(14):222. doi: 10.1186/1471-2474-14-222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Buchholz HW, Elson RA, Heinert K. Antibiotic-loaded acrylic cement: Current concepts. Clin. Orthop. 1984;190:96. [PubMed] [Google Scholar]
- 18.Wilde AH, Ruth JT. Two-stage reimplantation in infected total knee arthroplasty. Clin Orthop. 1988;236:23–35. [PubMed] [Google Scholar]
- 19.Duncan CP, Beauchamp CP, Masri B, et al. The antibiotic loaded joint replacement system: a novel approach to the management of the infected knee replacement. J.B.J.S. Br. 1992;74(suppl. III):296. [Google Scholar]
- 20.Hofmann AA, Kane KR, Tkach TK, Plaster RL, Camargo MR. Treatment of infected total knee arthroplasty using an articulating spacer. Clin. Orthop. 1995;321(45–54):1995. [PubMed] [Google Scholar]
- 21.Calton TF, Fehring TK, Griffin WL. Bone loss associated with the use of spacer blocks in infected total knee arthroplasty. Clin Orthop Relat Res. 1997;345:148–154. doi: 10.1097/00003086-199712000-00020. [DOI] [PubMed] [Google Scholar]
- 22.Haddad FA, Masri BA, Campbell D, McGraw RW, Beauchamp CP Duncan CP. The Prostalac functional spacer in two-stage revision for infected knee replacement. J.B.J.S. 2000;82-B:807––812. doi: 10.1302/0301-620X.82B6.10486. [DOI] [PubMed] [Google Scholar]
- 23.Castelli C, Robotti PF, Calonego G. (2002) Preformed all-cement knee spacer in two-stage revision for infected knee replacement: mechanical and pharmacological behaviour. Proceedings of the SICOT XXII World Congress, Abstract P1415
- 24.Bertazzoni Minelli E, Benini A, Magnan B, Bartolozzi P. Release of gentamicin and vancomycin from temporary human hip spacers in two-stage revision of infected arthroplasty. J Antimicrob Chemother. 2003;53(2):329–334. doi: 10.1093/jac/dkh032. [DOI] [PubMed] [Google Scholar]
- 25.Pitto RP, Castelli CC, Ferrari R, Munro J. Pre-formed articulating knee spacer in two-stage revision for the infected total knee arthroplasty. Int Orthop. 2005;29(5):305–308. doi: 10.1007/s00264-005-0670-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.McPherson EJ, Lewonoswski K, Don LD. Use of an articulated PMMA spacer in the infected total knee arthroplasty. Clin. Arthroplasty. 1995;10(1):87–89. doi: 10.1016/S0883-5403(05)80105-6. [DOI] [PubMed] [Google Scholar]
- 27.Trampuz A, Hanssen AD, Osmon DR, Mandrekar J, Steckelberg JM, Patel R. Sinovial fluid leukocyte count and differential for the diagnosis of prosthetic knee infection. Am J Med. 2004;117:556–62. doi: 10.1016/j.amjmed.2004.06.022. [DOI] [PubMed] [Google Scholar]
- 28.Ferrari R, Goglio A, Grigis G, Raglio A, Buoro SG, Virotta A, Remuzzi Castelli C. From swab to SEM: Our protocol on tka infection suspicion. Eur Cells and Mater. 2011;21(2):32. [Google Scholar]
- 29.Engh GA, Parks NL. The management of bone defects in revision total knee arthroplasty. Instr Course Lect. 1997;46:227–236. [PubMed] [Google Scholar]
- 30.Scott RD, Siliski JM. The use of a modified V-Y quadricepsplasty during total knee replacement to gain exposure and improve flexion in the ankylosed knee. Orthopedics. 1985;8(45–48):1985. doi: 10.3928/0147-7447-19850101-07. [DOI] [PubMed] [Google Scholar]
- 31.Villa T, Carnelli D. Experimental evaluation of the biomechanical performances of a PMMA-based knee spacer. Knee. 2007;14(2):145–53. doi: 10.1016/j.knee.2006.11.010. [DOI] [PubMed] [Google Scholar]
- 32.Leone JM, Hanssen AD. Management of Infection at the Site of a Total KneeArthroplasty. J Bone Joint Surg Am. 2005;87(10):2335–2348. doi: 10.2106/00004623-200510000-00026. [DOI] [PubMed] [Google Scholar]
- 33.Dunne N, Hill J, McAfee P, Todd K, Kirkpatrick R, Tunney M, Patrick S. In vitro study of the efficacy of acrylic bone cementloaded with supplementary amounts of gentamicinEffect on mechanical properties, antibiotic release, and biofilm formation. Acta Orthopaedica. 2007;78(6):774–785. doi: 10.1080/17453670710014545. [DOI] [PubMed] [Google Scholar]
- 34.Castelli C, Martinelli R, Ferrari R. The articulated knee spacer. In: Pipino F, editor. Bone cement and cemented fixation of implants. Italy: Finto Di Stampare Nel Mese; 2001. pp. 181–191. [Google Scholar]
- 35.Mutimer J, Gillespie G, Lovering AM, Porteous AJ. Measurements of in vivo intra-articular gentamicin levels from antibiotic loaded articulating spacers in revision total knee replacement. Knee. 2009;16(2009):39–41. doi: 10.1016/j.knee.2008.07.009. [DOI] [PubMed] [Google Scholar]
- 36.Bozic KJ, Kurtz SM, Lau E, Ong K, Chiu V, Vail TP, Rubash HE, Berry DJ. The epidemiology of revision total knee arthroplastyIn the united states. Clin Orthop Relat Res. 2010;468:45–51. doi: 10.1007/s11999-009-0945-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Borden LS, Gearen PF, Infected total knee arthroplasty A protocol for management. J Arthroplasty. 1987;1987(2):27–36. doi: 10.1016/S0883-5403(87)80028-1. [DOI] [PubMed] [Google Scholar]
- 38.Emerson RH, Jr, Muncie M, Tarbox TR, Higgins LL. Comparison of a static with a mobile spacer in total knee infection. Clin Orthop Relat Res. 2002;404:132–138. doi: 10.1097/00003086-200211000-00023. [DOI] [PubMed] [Google Scholar]
- 39.Duncan CP, Beauchamp CP. A temporary antibiotic-loaded joint replacement system for management of complex infections involving the hip. Orthop. Clin. North. Am. 1993;24(751–9):1993. [PubMed] [Google Scholar]
- 40.Mortazavi SM, Molligan J, Austin MS, Purtill JJ, Hozack WJ, Parvizi J. Failure following revision total knee arthroplasty: infection is the major cause. Int Orthop. 2010;35(8):1157–1164. doi: 10.1007/s00264-010-1134-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kubista B, Hartzler RU, Wood CM, Osmon DR, Hanssen AD, Lewallen DG. Reinfection after two-stage revision for periprosthetic infection of total knee arthroplasty. Int Orthop. 2012;36(1):65–71. doi: 10.1007/s00264-011-1267-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Durbhakula SM, Czajka J, Fuchs MD, Uhl RL (2004) Antibiotic-loaded articulating cement spacer in the 2-stage exchange of infected total knee arthroplasty. J Arthroplasty 19:768–774 [DOI] [PubMed]
- 43.Hofmann AA, Goldberg T, Tanner AM, Kurtin SM. Treatment of infected total knee arthroplasty using an articulating spacer: 2- to 12-year experience. Clin Orthop Relat Res. 2005;430:125–1314. doi: 10.1097/01.blo.0000149241.77924.01. [DOI] [PubMed] [Google Scholar]
- 44.MacAvoy MC, Ries MD. The ball and socket articulating spacer for infected total knee arthroplasty. J Arthroplasty. 2005;2005(20):757–762. doi: 10.1016/j.arth.2004.11.014. [DOI] [PubMed] [Google Scholar]
- 45.Masters JPM, Smith NA, Foguet P, Reed M, Parsons H, Sprowson PA. A systematic review of the evidence for single stage and two stage revision of infected knee replacement. BMC Musculoskeletal Disorders. 2013;14:222. doi: 10.1186/1471-2474-14-222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Larikov D, Demian H, Rubin D, Alexander J, Nambiar S (2012) The choice and doses of antibacterial agents for cement spacers in treatment of prosthetic joint infections: review of published studies Clinical Infectious Diseases Advance Access published August 23, 2012 [DOI] [PubMed]
- 47.Munro JT, Garbuz BDS, Masri A, Duncan CP. Articulating antibiotic impregnated spacers in two-stage revision of infected total knee arthroplasty. J Bone Joint Surg Br. 2012;94-B(Supple A):123–125. doi: 10.1302/0301-620X.94B11.30747. [DOI] [PubMed] [Google Scholar]
- 48.Struelens B, Claes S, Bellemans J. Spacer-related problems in two-stage revision knee arthroplasty Acta Orthop. Belg. 2013;79:422–426. [PubMed] [Google Scholar]
- 49.Garvin KL, Scuderi G, Insall JN. Evolution of quadriceps snip. Clin. Orthop. 1995;321:131–137. [PubMed] [Google Scholar]
- 50.Voleti PB, Baldwin KD, Lee GC. Use of static or articulating spacers for infection following total knee arthroplasty: a systematic literature review. J Bone Joint Surg Am. 2013;95(17):1594–1599. doi: 10.2106/JBJS.L.01461. [DOI] [PubMed] [Google Scholar]
- 51.Goldman RT, Scuderi GR, Insall JN. (1996) 2-stage reimplantation for infected total knee replacement. Clin Orthop. 1996;331:118–124. doi: 10.1097/00003086-199610000-00016. [DOI] [PubMed] [Google Scholar]
- 52.Pitto RP, Spika I. A Antibiotic-loaded bone cement spacers in twostage management of infected total knee arthroplasty. Int Orthop (SICOT) 2004;28:129–133. doi: 10.1007/s00264-004-0545-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Fehring TK, Odum S, Calton TF, Mason JB. Articulating versus static spacers in revision total knee arthroplasty for sepsis. The Ranawat Award. Clin Orthop Relat Res. 2000;380:9–16. doi: 10.1097/00003086-200011000-00003. [DOI] [PubMed] [Google Scholar]
- 54.Calton T, Fehring TK, Griffin WL. Bone Loss Associated With the Useof Spacer Blocks in Infected Total Knee Arthroplasty. Clin Orthop Relat Res. 1994;345:148–154. [PubMed] [Google Scholar]
- 55.Jafari SM, Casper DS, Camilo Restrepo BS, Zmistowski B, Parvizi J, Sharkey PF. Periprosthetic Joint Infection: Are Patients With Multiple Prosthetic Joints at Risk? J Arthroplasty. 2012;27(6):877–880. doi: 10.1016/j.arth.2012.01.002. [DOI] [PubMed] [Google Scholar]
- 56.Castelli C, Ferrari R, Martinelli R. (2002) Preformed all-cement knee spacer in two-stage revision for infected knee replacement: early clinical results. Proceedings of the SICOT XXII World Congress, Abstract 327c
- 57.Carulli C, Villano M, Civinini R, Matassi F, Nistri L, Innocenti M. A novel technique to preserve range of motion in two-stage revision of infected total knee arthroplasty. Int Orthop. 2013;37(6):1069–1074. doi: 10.1007/s00264-013-1880-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Meijer MF, Reininga IH, Boerboom AL, Stevens M, Bulstra SK. Poorer survival after a primary implant during revision total knee arthroplasty. Int Orthop. 2013;37(3):415–419. doi: 10.1007/s00264-012-1739-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Sternheim A, Lochab J, Drexler M, Kuzyk P, Safir O, Gross A, Backstein D. The benefit of revision knee arthroplasty for component malrotation after primary total knee replacement. Int Orthop. 2012;36(12):2473–2478. doi: 10.1007/s00264-012-1675-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Jensen CL, Petersen MM, Schrøder HM, Lund B. Bone mineral density changes of the proximal tibia after revision total knee arthroplasty.A randomised study with the use of porous tantalum metaphyseal cones. Int Orthop. 2012;36(9):1857–1863. doi: 10.1007/s00264-012-1601-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Springer BD, Lee GC, Osmon D, Haidukewych GJ, Hanssen AD, Jacofsky DJ (2004) Systemic safety of high-dose antibiotic-loaded cement spacers after resection of an infected total knee arthroplasty. Clin Orthop Relat Res 427:47–51 [DOI] [PubMed]
- 62.Meek RM, Dunlop D, Garbuz DS, McGraw R, Greidanus NV, Masri BA (2004) Patient satisfaction and functional status after aseptic versus septic revision total knee arthroplasty using the PROSTALAC articulating spacer. J Arthroplasty 19(7):874–879 [DOI] [PubMed]
- 63.Ghanem E, Kurd M, Pulido L, Sharkey PF, Hozack WJ, Parvizi J (2007) Surgical Treatment of Periprosthetic Infection. A Declining Success. European Bone and Joint Infection Society EBJIS. Corfu, Greece, September 20–22
- 64.Castelli C., Ferrari R. (2006) Preformed articulated all-cement spacer in two stage revision for infected knee replacement. Rationale and clinical result. J Bone Joint Surg Br vol. 88-B no. SUPP I 84