Abstract
Response selection dysfunction contributes to processing speed impairment in schizophrenia. However, it is unclear if response selection impairment transcends sensory and motor modalities or is modality specific. To address this question, healthy subjects and individuals with schizophrenia completed reaction time (RT) experiments with different combinations of sensory cues (i.e. visual, auditory) and motor response (i.e. manual, vocal). We found that response selection impairment in schizophrenia was present regardless of the sensory and motor modality of the tasks and correlated with performance on neuropsychological tests of processing speed. These results implicate dysfunction of amodal response selection brain regions in schizophrenia. Interventions that reduce the length of response selection stage processing may improve processing speed in schizophrenia.
1. Introduction
Processing speed is impaired in schizophrenia and an important cognitive predictor of functional outcome (Dickinson et al., 2007;Sanchez et al., 2009). Standardized neuropsychological tests of processing speed, such as the digit-symbol coding subtest from the Wechsler Adult Intelligence Scales (WAIS), require the integration of a number of cognitive operations, such as perception, sustained attention, decision making, and graphomotor abilities (Wechsler, 1997a). Consequently, it is unclear if processing speed impairment in schizophrenia results from dysfunction of a specific cognitive operation or a combination of abilities. This poses a significant challenge to developing effective treatments and behavioral interventions for improving processing speed in schizophrenia.
Investigators have attempted several different approaches to overcome these barriers. For example, by manipulating demands placed on visual scanning and memory, Bachman and colleagues found that reduced performance on a computerized version of the digit-symbol coding test in schizophrenia was due, in part, to impaired relational memory (Bachman et al., 2010). Alternatively, applying factor analysis to a battery of neuropsychological tests of processing speed, Knowles et al. (2012) found that processing speed could be broken down into three factors, psychomotor speed, verbal fluency, and sequencing and shifting, and that schizophrenia patients relied more on verbal fluency to perform the digit symbol coding task compared to healthy subjects who relied on sequencing and shifting.
Here, rather than manipulating neuropsychological tests or factor analyzing a battery of neuropsychological measures, which are themselves complex, we use cognitive neuroscience-based models of processing speed to investigate processing speed impairment in schizophrenia. One prominent cognitive model, derived largely from experiments using single and dual-task reaction time (RT) paradigms, posits that information processing speed is comprised of 3 stages: perceptual analysis, response selection, and response production (Pashler, 1994). The available evidence, while modest, suggests that slowed processed speed in schizophrenia results from dysfunction at the response selection stage, where stimulus-response translation and decision-making occurs (Woodward et al., 2013; Krieger et al., 2001b; Krieger et al., 2001a; Pellizzer and Stephane, 2007). Consistent with this, electrophysiological investigations have found that the lateralized readiness potential (LRP), an indicator of stimulus-response translation, is abnormal in schizophrenia (Kappenman et al., 2012; Luck et al., 2009). However, it is not known if response selection impairment reflects dysfunction of a unitary process that cuts across sensory and motor modalities or is modality specific. Resolving this issue is crucial to understanding the neural basis and treatment of processing speed impairment. Indeed, the contribution of perceptual disturbances to deficits in “higher order” cognitive abilities is an area of active debate in schizophrenia (Javitt, 2009).
In an effort to address this question, we conducted a study in which subjects completed 4 single-task RT experiments, each having a different combination of sensory cues (visual, auditory) and motor responses (manual, vocal). This allowed us to determine if slowed overall processing speed and response selection impairment, based on simple RT (SRT) and choice RT (CRT) conditions respectively, is modality specific or amodal in schizophrenia. Generalized response selection impairment and RT slowing would imply dysfunction of a central amodal information processing network and suggest that training programs shown to improve neural processing speed in amodal processing brain regions should be attempted in schizophrenia (i.e. Dux et al., 2009). Conversely, modality-specific impairment would implicate dysfunction of modality-specific neural machinery and suggest that interventions targeting specific sensory and/or motor processes might be more effective at improving processing speed in schizophrenia, as has been shown for other cognitive abilities such as verbal memory (Fisher et al., 2009).
2. Method
2.1. Study Participants
25 healthy subjects and 25 patients with schizophrenia meeting DSM-IV criteria according to chart review and the Structured Clinical Interview for DSM Disorders (First et al., 1996) were recruited into this study. This study was approved by the Vanderbilt Institutional Review Board (IRB) and all participants provided written informed consent. Individuals with schizophrenia were recruited through the Vanderbilt Psychotic Disorders Program. Healthy subjects were recruited from Nashville and surrounding area via internet and print advertising, mass emailing to Vanderbilt University employees, and word-of-mouth. Exclusion criteria included the following: estimated pre-morbid IQ less than 80 based on the Wechsler Test of Adult Reading (Wechsler, 2001), age less than 18 or greater than 55, presence of a systemic medical illness or central nervous system disorder that would affect study participation, history of significant head trauma, and active substance abuse disorder within the last three months.
2.2. Study Procedures
Each subject completed four single-task RT experiments, each of which included an SRT condition, and 2 and 4- CRT conditions. The modality of the cues (visual or auditory) and responses (manual, vocal) was crossed to create visual-manual (i.e. visual cues-manual responses), auditory-manual, visual-vocal, and auditory-vocal RT experiments. Sensory stimuli consisted of sounds and visual cues. Auditory stimuli, described previously (Dux et al., 2006), consisted of complex tones and man-made natural sounds edited by adding noise and/or reversing the waveform. Visual cues included colored circles (red, green, blue, yellow) and black geometric shapes (circle, diamond, square, triangle) presented centrally and measuring 6 cm in diameter/width. Motor responses consisted of manual button presses executed with the index and middle fingers of the left and right hands, and vocal responses (i.e. pseudo-syllables: “bah,” “koe,” “tay,” “dee,”). Subjects were trained to 90% accuracy on the stimulus-response mappings during 4 training blocks at the beginning of each experiment. Only subjects meeting this threshold went on to complete the rest of the experiment. Following training, each RT experiment included 168 trials comprised of 56 trials of each condition, SRT, 2-CRT, and 4-CRT. Trials were divided into blocks that began with a cue indicating which type of trials would appear during the upcoming block; SRT, 2-CRT, or 4-CRT. Stimulus-response pairings were counterbalanced across subjects, as was the order of RT conditions within experiment and order in which RT experiments were completed. Participants were also administered the WAIS-III Processing Speed Index (PSI) and Wechsler Memory Scale- 3rd Edition (WMS-III: Wechsler, 1997b) Working Memory Index (WMI).
2.3. Statistical Analyses
Median RT for the SRT and CRT conditions (correct trials only) for each experiment served as the dependent variables. As not all subjects reached the 90% accuracy criterion during training for every experiment, the RT data were analyzed using linear mixed models which provide a more powerful approach than traditional repeated measured analysis in situations where some subjects have incomplete data (Hoffman and Rovine, 2007). SRT data were analyzed first with experiment entered as repeated measure and group a between subjects variable. CRT data were analyzed in a similar manner with response uncertainty (i.e. 2-CRT, 4-CRT) entered as an additional repeated measure and mean SRT included as a covariate to control for overall processing speed.
3. Results
25 patients and 25 healthy controls met the 90% accuracy criterion for at least one of the four RT experiments during training and were included in the analyses reported below. 15 patients and 22 healthy subjects completed all four RT experiments. Demographics are presented in Table 1. Of the 25 patients, 4 were unmedicated and antipsychotic medication dosage could not be determined for 2 individuals. Average antipsychotic dose (in chlorpromazine equivalents) for the remaining 19 patients was 492.4 ± 289.3 mg/day.
Table 1. Demographic Characteristics and Cognitive Tests Scores.
| Healthy Subjects | Schizophrenia | Statistics | ||||
|---|---|---|---|---|---|---|
| Variable | x2 | p | ||||
| N | 25 | 25 | ||||
| Sex (male:female) | 12:13 | 12:13 | -- | -- | ||
| Ethnicity (White:AA) | 13:12 | 14:11 | 0.81 | .777 | ||
|
| ||||||
| Mean | SD | Mean | SD | t | p | |
|
| ||||||
| Age | 41.5 | 9.6 | 42.0 | 11.3 | 0.15 | .883 |
| Education | 15.9 | 2.0 | 13.5 | 2.0 | 4.30 | .001 |
| Mother Education | 13.2 | 2.4 | 13.5 | 3.1 | 0.40 | .688 |
| Father Education | 13.1 | 3.4 | 13.3 | 4.3 | 0.18 | .860 |
| Estimated Premorbid IQ | 104.9 | 13.2 | 99.2 | 16.0 | 1.37 | .178 |
| WAIS Processing Speed Index | 105.3 | 17.1 | 85.2 | 11.8 | 4.82 | .001 |
| WAIS Working Memory Index | 102.9 | 17.3 | 91.0 | 15.4 | 2.55 | .014 |
|
| ||||||
| Clinical Symptoms | ||||||
| PANSS Positive | -- | -- | 12.6 | 6.8 | -- | -- |
| PANSS Negative | -- | -- | 14.8 | 6.5 | -- | -- |
| PANSS General | -- | -- | 25.0 | 6.8 | -- | -- |
3.1. Simple and Choice RT
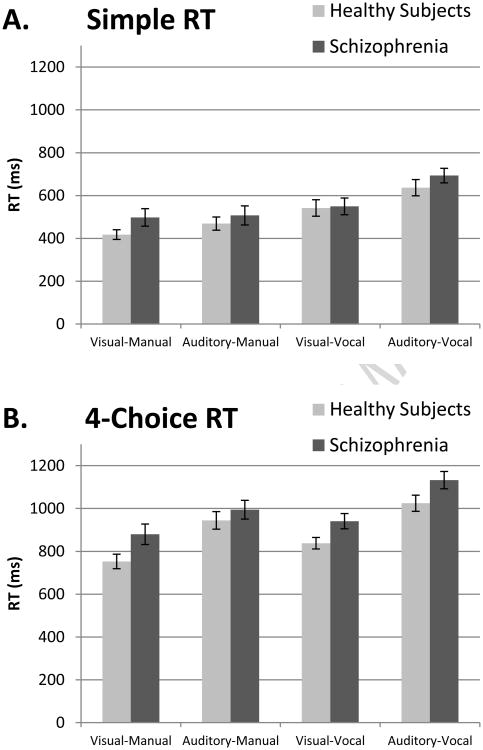
For SRT, there was a robust main effect of experiment (F(3,37.3)=34.6, p≪.001), but no effect of group (F(1,46.7)=0.693, p=.409) and no group by experiment interaction (F(3,37.3)=0.96, p=.421). Similar results were obtained when the analyses was restricted to the sub-set of individuals that completed all four experiments. Specifically, repeated measures ANOVA revealed a main effect of experiment (F(3,34)=40.59, p≪.001), but no effect of group (F(1,36)=2.64, p=.113) and no experiment × group interaction (F(3,34)=0.15, p=.931). As shown in Figure 1A, SRT was fastest for the visual-manual experiment and slowest for the auditory-vocal experiment.
Figure 1.
Simple and 4-choice reaction time (RT) in healthy subjects and individuals with schizophrenia across sensory and motor modalities. Panel A: Simple RT was fastest for the experiment with visual cues and manual button press responses (i.e. visual-manual) and slowest for the experiment with auditory cues and vocal responses (i.e. auditory-vocal). Simple RT did not differ between groups. Panel B: 4-choice RT was significantly longer for all experiments in schizophrenia compared to healthy subjects after adjusting for simple RT.
With respect to CRT performance, linear mixed models analysis revealed a main effect of CRT condition (F(1,33.3)=227.48, p<.001), reflecting the fact that 4-CRT was longer than 2-CRT, and, consistent with SRT, a main effect of experiment (F(3,40.2)=31.81, p<.001) with CRTs being shortest for the visual-manual experiment and longest for the auditory-vocal experiment. Importantly, the group by CRT condition interaction was significant (F(1,33.3)=4.34, p=.045) indicating that the increase in RT from 2-CRT to 4-CRT differed between groups after correcting for overall processing speed (i.e. mean SRT). Follow-up analysis revealed that schizophrenia patients had longer RTs that healthy subjects at the 4-choice condition (987 ms vs. 890 ms; F(1,46.3)=4.98, p=.031), but not 2-choice condition (770 ms vs. 719 ms; F(1,47.1)=1.38, p=.246).
In addition, an experiment by CRT condition interaction was also observed (F(3,36.8)=4.11, p=.013) due to the fact that the increase in RT between 2 and 4 CRT conditions was greater for the auditory-manual and auditory-vocal tasks compared to visual-manual and visual-vocal experiments (all p-values<.054). Similar results were obtained when the analyses was restricted to the sub-set of individuals that completed all four experiments. Specifically, repeated measures ANOVA revealed a main effect of CRT condition (F(1,34)=68.93, p≪.001), reflecting the fact that 4-CRT was longer than 2-CRT, and a trend towards a group by CRT condition interaction (F(1,34)=3.94, p=.055). Follow-up univariate analysis revealed that the interaction was due to the fact that 4-CRT was longer in schizophrenia patients (F(1,34)=4.76, p=.036), whereas 2-CRT did not differ between groups (F(1,34)=0.39, p=.536).
Accuracy was very high and highly skewed in both groups across all RT conditions and experiments (i.e. 83-99%). As such, these data were not analyzed.
3.2. Correlations between RT, Neuropsychological Test Scores, and Clinical Variables
Controlling for the effect of group, WAIS PSI scores inversely correlated with 2-CRT (r=-34, p=.019) and 4-CRT (r=-.30, p=.037), but not SRT (r=-.23, p=.116). The correlation between CRT and WAIS PSI was most pronounced for the visual-manual task (2-CRT: r=-.43, p=.009; 4-CRT=-.37, p=.026). However, auditory-vocal CRT, which does not overlap at all with WAIS PSI in terms of sensory and motor modality, also correlated with WAIS PSI scores at the trend significance level (2-CRT: r=-.32, p=.055; 4-CRT: r=-.30, p=.073). WMS WMI scores did not correlate with SRT and CRT. In patients, positive, negative, and general symptoms from the PANSS were unrelated to SRT and CRT (all Pearson's r's<|.17|, p>.445). Similarly, antipsychotic medication dosage did not correlate with SRT and CRT (all Pearson's r's<|.25|, p>.293).
4. Discussion
Using single-task RT experiments with different combinations of sensory stimuli and motor responses, we sought to determine if response selection impairment in schizophrenia transcends sensory and motor modalities or is modality specific. Answering this question will help to determine if amodal or modality specific brain networks are affected in schizophrenia and what types of interventions might be useful for ameliorating processing speed impairment. We found that response selection impairment in schizophrenia cuts across sensory and motor modalities. As expected, the impairment was most pronounced at the 4-CRT condition which placed the most demands on response selection, a finding consistent with prior investigations (Woodward et al., 2013; Pellizzer and Stephane, 2007). Moreover, although not statistically significant, SRT slowing was similar regardless of the sensory and motor modalities of the experiment providing further evidence that slowed processing speed in schizophrenia results from dysfunction of an amodal information processing system.
Prior work in healthy subjects has shown that training can shorten the duration of response selection stage processing and increase the speed of neural responses in amodal information processing brain regions, such as the prefrontal cortex (Dux et al., 2009). Our results strongly suggest that training programs such as this might be useful for improving response selection in schizophrenia. Indeed, prior work by our group has shown abnormal activity in prefrontal cortex during response selection in schizophrenia (Woodward et al., 2013). Moreover, the findings from our current study showing that CRT correlates with performance on standardized tests of processing speed, even for RT tasks with different sensory and motor modalities than typical neuropsychological tests, suggests that response selection training may generalize to neuropsychological tests and perhaps other cognitive domains relying on response selection, such as working memory encoding (Tombu et al., 2011).
One limitation of the study is the relatively high number of schizophrenia patients that failed to meet the 90% accuracy criterion during training (10/25 patients); although this limitation was mitigated to some extent by using multi-level statistical methods that allow subjects with incomplete data to be included in the analysis. We note that this was a particularly challenging study to implement as each experiment required subjects to learn 4 sets of novel stimulus-response pairings. It is perhaps not surprising that patients had trouble learning the stimulus-response pairings given evidence that schizophrenia is associated with deficits in paired learning (Armstrong et al., 2012). Moreover, the fact that all experiments had to be completed within a single session limited the length of training and likely contributed to problem.
Acknowledgments
This research was supported by NIMH 5R21MH096177 (awarded to NDW) and the Jack Martin, M.D., Research Professorship (awarded to NDW). The authors would like to thank Kristan Armstrong for her assistance with recruiting subjects to participate in this study. The authors are indebted to the individuals who participated in the study.
Role of Funding Source: This research was supported by NIMH 5R21MH096177 (awarded to NDW) and the Jack Martin, M.D., Research Professorship (awarded to NDW). Neither funding source had any further role in designing the study; collection, analysis, or interpretation of the data; writing the manuscript; or in the decision to submit the manuscript for publication.
Footnotes
Contributors: Author NDW conceived and designed the research reported on, secured funding for the project, analyzed the data, and wrote the final draft of the manuscript. Author BD recruited subjects, assisted with running the experiments and collecting data, assisted with analyzing the data, and contributed to an initial draft of the manuscript. Author HK assisted with analyzing the data.
Conflict of Interest: No commercial support was received for this manuscript and the authors have no conflicts of interest to report.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Armstrong K, Kose S, Williams L, Woolard A, Heckers S. Impaired associative inference in patients with schizophrenia. Schizophr Bull. 2012;38:622–629. doi: 10.1093/schbul/sbq145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bachman P, Reichenberg A, Rice P, Woolsey M, Chaves O, Martinez D, Maples N, Velligan DI, Glahn DC. Deconstructing processing speed deficits in schizophrenia: application of a parametric digit symbol coding test. Schizophr Res. 2010;118:6–11. doi: 10.1016/j.schres.2010.02.1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dickinson D, Ramsey ME, Gold JM. Overlooking the obvious: a meta-analytic comparison of digit symbol coding tasks and other cognitive measures in schizophrenia. Arch Gen Psychiatry. 2007;64:532–542. doi: 10.1001/archpsyc.64.5.532. [DOI] [PubMed] [Google Scholar]
- Dux PE, Ivanoff J, Asplund CL, Marois R. Isolation of a central bottleneck of information processing with time-resolved FMRI. Neuron. 2006;52:1109–1120. doi: 10.1016/j.neuron.2006.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dux PE, Tombu MN, Harrison S, Rogers BP, Tong F, Marois R. Training improves multitasking performance by increasing the speed of information processing in human prefrontal cortex. Neuron. 2009;63:127–138. doi: 10.1016/j.neuron.2009.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- First MB, Spitzer RL, Gibbon M, Williams J. Structured Clinical Interview for DSM-IV Axis I Disorders, Clinical Version (SCID-CV) American Psychiatric Press Inc.; Washington, D.C.: 1996. [Google Scholar]
- Fisher M, Holland C, Merzenich MM, Vinogradov S. Using neuroplasticity-based auditory training to improve verbal memory in schizophrenia. Am J Psychiatry. 2009;166:805–811. doi: 10.1176/appi.ajp.2009.08050757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffman L, Rovine MJ. Multilevel models for the experimental psychologist: foundations and illustrative examples. Behav Res Methods. 2007;39:101–117. doi: 10.3758/bf03192848. [DOI] [PubMed] [Google Scholar]
- Javitt DC. When doors of perception close: bottom-up models of disrupted cognition in schizophrenia. Annu Rev Clin Psychol. 2009;5:249–275. doi: 10.1146/annurev.clinpsy.032408.153502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kappenman ES, Kaiser ST, Robinson BM, Morris SE, Hahn B, Beck VM, Leonard CJ, Gold JM, Luck SJ. Response activation impairments in schizophrenia: evidence from the lateralized readiness potential. Psychophysiology. 2012;49:73–84. doi: 10.1111/j.1469-8986.2011.01288.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knowles EE, Weiser M, David AS, Dickinson D, Glahn D, Gold J, Davidson M, Reichenberg A. Dedifferentiation and substitute strategy: deconstructing the processing-speed impairment in schizophrenia. Schizophr Res. 2012;142:129–136. doi: 10.1016/j.schres.2012.08.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krieger S, Lis S, Gallhofer B. Cognitive subprocesses and schizophrenia. A. Reaction-time decomposition. Acta Psychiatr Scand Suppl. 2001a:18–27. doi: 10.1034/j.1600-0447.2001.104s408018.x. [DOI] [PubMed] [Google Scholar]
- Krieger S, Lis S, Gallhofer B. Reaction-times and bioelectrical brain signals of drug-naive schizophrenic first-onset patients in identification and classification tasks. Acta Psychiatr Scand Suppl. 2001b:42–59. doi: 10.1034/j.1600-0447.2001.104s408042.x. [DOI] [PubMed] [Google Scholar]
- Luck SJ, Kappenman ES, Fuller RL, Robinson B, Summerfelt A, Gold JM. Impaired response selection in schizophrenia: evidence from the P3 wave and the lateralized readiness potential. Psychophysiology. 2009;46:776–786. doi: 10.1111/j.1469-8986.2009.00817.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pashler H. Dual-task interference in simple tasks: data and theory. Psychol Bull. 1994;116:220–244. doi: 10.1037/0033-2909.116.2.220. [DOI] [PubMed] [Google Scholar]
- Pellizzer G, Stephane M. Response selection in schizophrenia. Exp Brain Res. 2007;180:705–714. doi: 10.1007/s00221-007-0892-5. [DOI] [PubMed] [Google Scholar]
- Sanchez P, Ojeda N, Pena J, Elizagarate E, Yoller AB, Gutierrez M, Ezcurra J. Predictors of longitudinal changes in schizophrenia: the role of processing speed. J Clin Psychiatry. 2009;70:888–896. doi: 10.4088/JCP.08m04294. [DOI] [PubMed] [Google Scholar]
- Tombu MN, Asplund CL, Dux PE, Godwin D, Martin JW, Marois R. A Unified attentional bottleneck in the human brain. Proc Natl Acad Sci U S A. 2011;108:13426–13431. doi: 10.1073/pnas.1103583108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wechsler D. Wechsler Adult Intelligence Scale-3rd Edition. Third. The Psychological Corporation; 1997a. [Google Scholar]
- Wechsler D. Wechsler Memory Scale: 3rd Edition. The Psychological Corporation; 1997b. [Google Scholar]
- Wechsler D. Wechsler Test of Adult Reading. Pearson Education; 2001. [Google Scholar]
- Woodward ND, Duffy B, Karbasforoushan H. Prefrontal Cortex Activity during Response Selection Predicts Processing Speed Impairment in Schizophrenia. J Int Neuropsychol Soc. 2013;19:782–791. doi: 10.1017/S1355617713000532. [DOI] [PMC free article] [PubMed] [Google Scholar]