Abstract
This issue marks the 50th Anniversary of the release of the U.S. Surgeon General’s Report on Smoking and Health. Perhaps no other singular event has done more to highlight the effects of smoking on the development of cancer. Tobacco exposure is the leading cause of cancers involving the oral cavity, conductive airways and the lung. Owing to the many carcinogens in tobacco smoke, smoking-related malignancies have a high genome-wide burden of mutations, including in the gene encoding for p53. The p53 protein is the most frequently mutated tumor suppressor in cancer, responsible for a range of critical cellular functions that are compromised by the presence of a mutation. Herein we review the epidemiologic connection between tobacco exposure and cancer, the molecular basis of p53 mutation in lung cancer, and the normal molecular and cellular roles of p53 that are abrogated during lung tumor development and progression as defined by in vitro and in vivo studies. We also consider the therapeutic potential of targeting mutant p53 in a clinical setting based upon the cellular role of mutant p53 and data from genetic murine models.
Keywords: Tobacco, Smoking, Surgeon General, Lung Cancer, p53, Metastasis, Animal models
Introduction
Smoking and Lung Cancer
Although smoking confers increased risk of multiple tumor types, perhaps no malignancy is more closely linked with smoking than lung cancer. Prior to the commercial introduction of cigarettes, the incidence of lung cancer was extremely low, but small epidemiologic studies and clinical observations as early as the 1930s suggested a potential causative relationship between tobacco exposure and a rise in lung cancer cases (reviewed in [1, 2]). These observations spurred large, definitive studies, along with the development of the appropriate methodology required for epidemiologic studies of chronic diseases, which by the 1950s provided strong epidemiologic evidence for the relationship between the amount of tobacco exposure and risk of cancer, the beneficial effects of cessation, and an association with tumor histology [3, 4].
However, these studies provided epidemiologic association without causation. Since no clear evidence for specific carcinogens in tobacco smoke had been found, and since experimental data recapitulating tobacco as a cancerous agent or providing a mechanistic biochemical explanation was lacking, the effect of smoking on lung cancer was not broadly endorsed and instead engendered significant debate in the medical community. This attitude began to change in the medical community on both sides of the Atlantic with statements by the British Medical Research Council in 1957 that endorsed tobacco as a direct cause of cancer and by the Surgeon General of the United States, Dr. Leroy E. Burney, in 1959, which defined smoking as the “principal etiologic factor in the increased incidence of lung cancer [5, 6].” The weight of the epidemiologic evidence was sufficient enough that the release of the first report of the Surgeon General’s Advisory Committee on Smoking and Health on January 11, 1964, by Dr. Luther L. Terry set into motion significant legislation by the U.S. Congress, and great public debate over how best to address smoking cessation and the health issues caused by smoking [7]. Towards this end, the U.S. Congress adopted the Federal Cigarette Labeling and Advertising Act of 1965 and the Public Health Cigarette Smoking Act of 1969, which required health warnings on cigarette packaging, banned advertising in the media and called for the issuance of an annual report on the health consequences of smoking. In September 1965, the Public Health Service established a unit called the National Clearinghouse for Smoking and Health, which was succeeded by the Centers for Disease Control and Prevention’s Office on Smoking and Health. Since 1965 these two agencies have been responsible for 29 reports on the health consequences of smoking and have provided on-going support for tobacco cessation and prevention efforts on the part of all levels of government and local communities. These efforts have produced substantial reductions in the number of current smokers in the United States, and recent estimates are that roughly 800,000 deaths from lung cancer were prevented due to these efforts [8].
Smoking and Genetic Mutations
It was not until the late 1980s that the data was presented demonstrating that lung tumors have high rates of mutation in known oncogenes like KRAS and in emerging tumor suppressors such as TP53. It was reported from analyses of both primary tumors and cell lines of many tumor types (including non-small cell and small cell lung cancer) that regions of the short arm of chromosome 17 (containing TP53) are frequently deleted, often along with point mutations in the remaining TP53 allele [9–13]. At about the same time it was identified that patients with the Li-Fraumeni syndrome, who are susceptible to multiple tumor types at an early age, including lung carcinoma in the absence of tobacco exposure, have germline TP53 mutations [14]. A database was established in the early 1990’s of the documented TP53 mutations from all tumor types and cell lines [15], which continues to be maintained and updated by the International Agency for Research on Cancer for use by the scientific community (http://www.iarc.fr/p53/Index.html).
Tobacco smoke contains thousands of vapor phase and particulate phase compounds, at least 60 of which have been classified as carcinogens (Hoffman 2001), including PAH (polycyclic aromatic hydrocarbons) such as BaP (benzo[a]pyrene), dibenz[a,h]anthracene, 5-methylchrysene and dibenzo[a,i]pyrene, and the N-nitrosamines [such as NNK (nicotine-derived nitrosamine ketone) and NNN (N′-nitrosonornicotine)]. Chronic exposure of the lung epithelium to this mixture of compounds confers increased cancer susceptibility due to the formation of DNA adducts that produce oncogenic mutations (reviewed in [16]). The heightened mutation rate is observed in smoking-related cancers versus other tumor types, in lung cancers from smokers versus non-smokers, and an increase in mutations is even found in the non-cancerous lung tissues of smokers [17]. Additionally, the mutations observed in relation to tobacco exposure are commonly due to the presence of G to T transversions. This mutational pattern is consistent with the mechanism of DNA adduct formation from carcinogens in tobacco smoke, most prominently PAH and NNK [18]. Of the multitude of carcinogens found in tobacco smoke, these are the best documented to produce lung tumors in experimental conditions and to produce DNA adducts at the same DNA sites found in patient tumors. These findings provide a mechanistic link between the epidemiologic association of smoking with lung cancer and the observed mutation profiles in lung tumors.
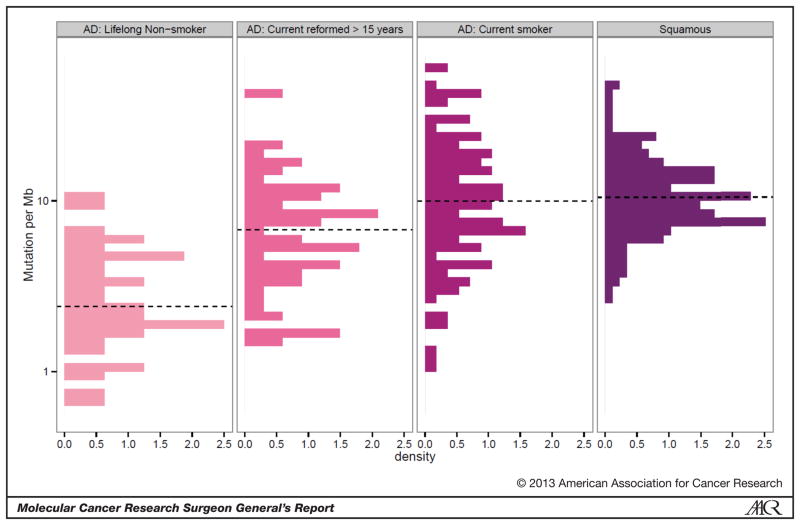
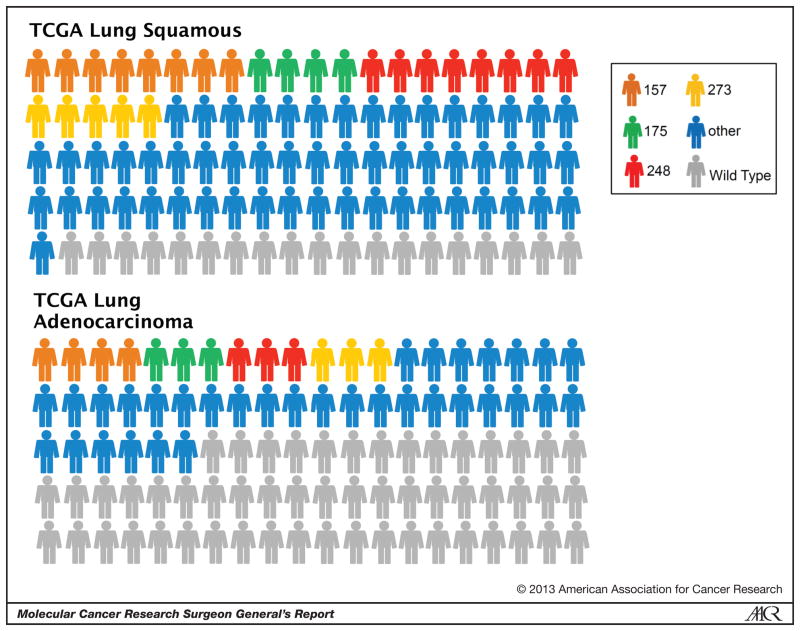
More recently, whole-genome, exome and RNA sequencing of patient tumors from individual institutions and The Cancer Genome Atlas (TCGA) projects on lung adenocarcinoma, lung squamous cell carcinoma and squamous cell carcinomas of the head and neck have confirmed several of these points in regard to the effect of tobacco exposure [19–21]. First, the genome-wide mutational burden is significantly higher in lung adenocarcinoma from patients who are current or former smokers, similar to that found in patients with squamous cell carcinoma, a group where the percentage of smokers is much higher (Figure 1). Second, lung cancers have a high p53-specific mutation rate (46% in lung adenocarcinoma and 81% in squamous cell) and display a high percentage of mutations in hotspots regions (Figure 2) [19].
Figure 1.
Genome-wide mutation density from the TCGA datasets for lung adenocarcinoma and squamous cell carcinoma. Plot of mutations/megabase of the genome, with the X-axis representing the probability density. Current smokers were grouped with those reformed for less than 15 years, while those reformed greater than 15 years and lifelong non-smokers are presented separately. Since 96% of the squamous cell lung cancer group was identified as a smoker, this group was not further segregated.
Figure 2.
p53 mutation spectrum from the TCGA lung squamous and lung adenocarcinoma datasets. A graphical representation is shown of the percent with p53 mutations (colored) versus wild-type p53 (grey). The mutations within a 5 residue stretch are grouped and colored to represent one of four hotspot regions, around amino acid 157 (orange), 175 (green), 248 (red) and 273 (yellow). Mutations outside of these four hotspots are grouped and colored blue.
Normal cellular functions of p53 and potential roles in cancer
The early data on p53 arose from studies of tumorigenic DNA viruses such as simian virus 40 (SV40) and human papilloma virus (HPV), which revealed that p53 is often the target of viral oncoproteins [22]. The p53 protein was originally discovered and the gene subsequently cloned by investigation of proteins interacting with the SV40 large T antigen that account for the transforming capability of the virus [23, 24]. The unique nature of p53 function generated initial confusion about whether it is an oncogene or a tumor suppressor [22]. Analysis of tumor tissues and tumor cell lines revealed that the half-life of p53 and the amounts of protein found were greater than in non-transformed cells. Along with the initial in vitro data that demonstrated transforming capability for p53, this evidence suggested p53 as a new oncoprotein similar to the previously discovered Ras and Myc. But it was subsequently found that the DNA clones used in these early experiments contained mutant TP53 sequences, and that wild-type TP53 not only lacked transforming capability, but was able to suppress Ras-induced transformation in vitro in complementation assays and the formation of tumors in animals [25, 26]. Conversely, mutant TP53 was able to transform rodent and human cell lines or primary cultured cells, and both null and point mutant alleles were able to cooperate with Ras to transform cultured cells, although the point mutation produced a stronger effect [27]. The conclusion from these results was that p53 normally serves as a tumor suppressor, the function of which is lost upon allelic loss or mutation.
The TP53 gene encodes for a protein of 53 kDa, which is a sequence-specific transcription factor found at low levels under normal cellular conditions due to the regulatory action of the E3 ubiquitin ligase MDM2 [28, 29]. In response to many types of cellular stress, including DNA replication stress or damage [30], p53 is rapidly stabilized and the accumulated protein localizes to the nucleus. The outcome of p53 function is highly context-dependent, depending on its complex effects in activating transcriptional activity at some sites while repressing others, the balance of which may result in cell cycle arrest and repair of the damaged DNA, cellular apoptosis, senescence, metabolic changes or autophagy [31, 32]. Multiple factors affect this outcome, including the affinity of gene promoters for p53 binding, the ability of p53 to cooperatively bind to DNA, and the effects of co-factor binding at p53 promoter sites [33–35]. Recent genome-wide chromatin immunoprecipitation and sequencing (ChIP-seq) studies have demonstrated a core p53 default program that is significantly modified by the presence of co-factor binding at p53 sites to generate composite response elements and the affinity of p53 oligomers for binding to high- or low-affinity sites [34–36].
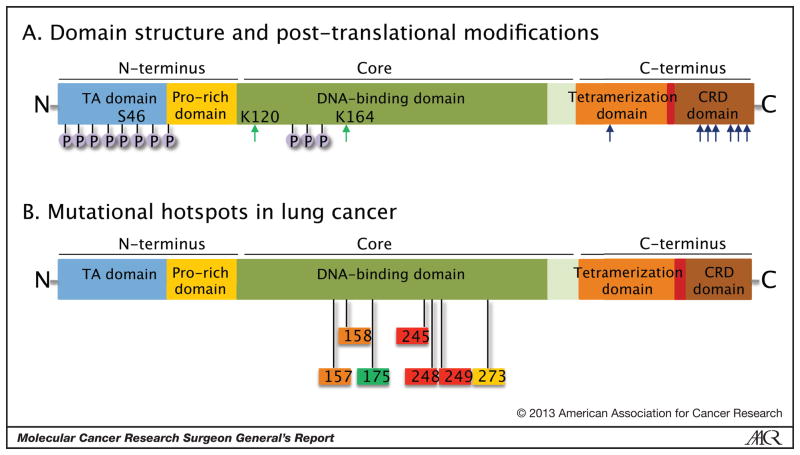
The domain structure of p53 reveals an N-terminal transactivation (TA) domain, a core DNA-binding domain, and a C-terminus containing both a tetramerization and a C-terminal regulatory domain (Figure 3A). Extensive post-translational modification occurs in each of the domains of p53, including serine/threonine phosphorylation (most prominently in the N-terminus), lysine acetylation, ubiquitination, neddylation, sumoylation and methylation. The schema depicted here is a simplified version and the reader is referred to several detailed reviews for a thorough treatment of this subject [31, 32, 36]. The post-translational modifications modulate p53 protein stability, cellular localization, its complement of interacting proteins and its subsequent target promoter selectivity. Acetylation or ubiquitination of the same amino acid residues has counteracting roles to respectively stabilize or destabilize the protein and thereby regulate activity. Although many of the post-translational modifications have pronounced effects when tested in vitro, testing in mice by knock-in approaches instead demonstrates that they fine-tune p53 function, likely due to functional redundancy of many of the sites. Of particular interest in this regard is a recent report comparing p53 null mice with the 3KR mutant, in which three acetylation cites of the DNA binding domain (K117, K161, K162; mouse protein numbering) were replaced with arginine, which demonstrated a lack of cell-cycle arrest, apoptosis or senescence in vivo during thymic lymphoma formation [37]. Nevertheless, the 3KR mutant p53 retained the ability to regulate energy metabolism (glucose uptake and glycolysis) and antioxidant function, and thereby suppress formation of thymic lymphomas. These findings challenge the prevailing models of p53 centered around cell cycle arrest and apoptosis/senescence, and highlight other cellular functions regulated by p53 that are critical to regulating in vivo tumorigenesis.
Figure 3.
Domain structure of the p53 protein. (A) The functional domains and some of the sites of post-translational modification are illustrated. The concentration of N-terminal Ser/Thr phosphorylation sites is schematically shown. The major sites of lysine acetylation are shown in green arrows at K120 and K164, while the cluster of 6 lysines in the TD and CRD that can be ubiquitinated or acetylated are schematically indicated with blue arrows. (B) The amino acid residues in the four mutational hotspot regions of the DNA binding domain are shown.
In addition to the direct effects of tobacco carcinogens on the cellular DNA of bronchial epithelial cells, tobacco smoke indirectly effects the overall tumor microenvironment by eliciting chronic inflammation in the lungs [38]. Even the inflammation from short-term experimental tobacco smoke exposure has been shown to promote lung cancer progression in both carcinogen- and genetic Kras-initiated models [39]. These findings are complemented by work from several groups demonstrating that Kras and p53 mutation drive NF-kappa B-dependent signaling in murine lung tumors [40, 41]. Within the setting of chronic inflammation, mutant p53 in tumor cells could potentially abrogate the normal p53-mediated check on cellular response to the inflammatory signals.
The DNA damage checkpoint serves as a potent activator of p53 function and allelic dilution of wild-type p53 by chromosomal deletion, along with point mutation in the remaining copy, is frequently observed in diverse human tumor types as a mechanism to bypass this critical checkpoint [30]. Although mutations have been found at almost all amino acid positions in the protein, several hotspot regions have been identified (V157, R158, R175, G245, R248, R249, R273), which highlight the critical function of the core DNA-binding domain (Figure 3B). Two classes of mutations emerge: structural mutants that affect the folding and stability of the protein, and mutations that critically affect direct amino acid-DNA contacts. Interestingly, besides classic tumor suppressor functions that are lost upon mutation of the protein, both classes of mutants have also been shown to produce gain-of-function phenotypes. Since p53 monomers oligomerize to form functional tetramers, and interact with many other proteins and sequence-specific promoter elements to produce target gene transcription, the presence of mutations can alter the protein-protein interactions that define its proper function [42]. The missense mutant p53 alleles demonstrate dominant-negative activity through their ability to form p53 tetramers or other protein-protein complexes, and the altered DNA binding of the mutant proteins can produce gain-of-function activity. Many of the effects of mutant p53 may be through its ability to oligomerize to form mixed tetramers with the p53 family members, p63 and p73 [43].
Early work with autopsy-derived explants of human bronchial epithelial tissues demonstrated the ability to study these tissues in culture over many weeks and derive primary normal human bronchial epithelial (NHBE) cells [44, 45]. Further work showed that these primary cells could be transformed with viral oncogenes from SV40, allowing extended study in culture to test for genetic changes that would reproduce the multistage bronchial carcinogenesis in vitro [46]. Work with the tissues and primary cells confirmed that components of cigarette smoke condensate (CSC), including BAP and PAH, bind to the cellular DNA and produce phenotypic changes [47–49]. Long-term exposure to NNK or CSC of the immortalized non-tumorigenic human bronchial epithelial cells grown in de-epithelialized rat tracheas in vivo produced neoplastic transformation to invasive adenocarcinomas [48]. More recently, Minna’s group produced human bronchial epithelial cell lines (HBEC) that are immortalized without viral oncogenes by overexpression of wild-type cyclin-dependent kinase 4 (CDK4), to prevent the p16INK4a-mediated growth arrest, and the catalytic subunit of telomerase (hTERT), to bypass telomere-dependent senescence [50]. The CDK4/hTERT-immortalized cells have an intact p53 checkpoint and display gene expression profiles that cluster with normal non-immortalized bronchial cells, distinct from both lung cancer cell lines and HBECs immortalized with the HPV-16 viral oncoproteins E6/7. In-line with the multistage carcinogenesis model, multiple additional oncogenic changes (mutant KRASV12 expression, loss of p53, c-MYC and/or serum-induced epithelial-mesenchymal transition) are required to confer a full malignant phenotype on these cells, including in vivo tumor growth [51]. Strikingly, a partial malignant phenotype was produced by moderate mutant KRASV12 expression, but full malignant transformation was dependent on high levels of KRASV12 expression, which required p53 loss to bypass oncogene-induced senescence [52].
Animal models of p53 loss and mutation
Although in vitro studies with cultured cell lines have revealed some of the functions of p53, description of the more nuanced in vivo biology relied upon the generation of animal models. Many labs have generated animal models to test the in vivo consequences of p53 loss or mutation. The first animal data was reported by Lavigueur and colleagues [53], in which cDNA fragments from Friend leukemia viruses were electroporated into embryos to obtain transgenic animals carrying mutated p53 fragments along with the two copies of wild-type p53. Multiple types of tumors developed in these transgenic animals, including lung, sarcoma and lymphoid malignancies.
The second generation of animal models that were created relied upon gene targeting to produce a null p53 allele, which could then be studied in heterozygous or homozygous conditions [54]. Surprisingly, these animals had normal development, but in the homozygous state were found to have a pronounced increase in the susceptibility for early tumor onset (~20 weeks) of many tumor types (~75% of animals), mostly sarcomas and T-cell lymphomas, although a few epithelial tumors were described. Further work with these and similar models revealed that the animals had enhanced progression of carcinogen-induced malignancies [55], increased susceptibility to radiation-induced sarcomas and lymphomas and accumulation of chromosomal abnormalities [56].
Despite the findings from these TP53 knockout models, most human tumors have a missense mutation in p53. To better understand the potential dominant negative and gain-of-function effects of mutant TP53, a transgenic animal model was generated carrying the TP53Ala135Val mutant as a single autosomal allele. To test the role of mutant p53 in different backgrounds, these animals were crossed with the wild-type or p53 null alleles [57]. In this study the presence of the mutant transgene both enhanced tumorigenesis and also shifted the tumor spectrum, with a strikingly high occurrence of lung adenocarcinomas (20%) in the heterozygous animals carrying the point mutant transgene (p53+/−;Tg), but not in the heterozygous animals without the transgene (p53+/−), nor in the p53 null animals. This report clearly demonstrated that mutant p53 can produce a striking gain-of-function phenotype.
Because human tumors frequently have multiple genes affected by mutation and because early in vitro models revealed synergy between p53 and other oncogenes, murine models were generated with a combination of the p53 null alleles and other activated oncogenes or tumor suppressors, e.g. Rb. The combination of Rb and TP53 knockout produced a faithful model of small cell lung cancer, consistent with the fact that ~90% of human SCLC have simultaneous loss of both genes [58–60]. In non-small cell lung cancer the two most frequently mutated genes are KRAS and TP53. The presence of an activating KrasG12D or KrasG12V allele in the lung epithelia of animals produces adenomatous hyperplasia, adenomas, and histologic progression to non-metastatic adenocarcinomas [61–63]. In contrast, combination of a mutant Kras allele with either TP53 loss or mutation (R172H, structural mutation; R270H, contact mutation) produces faster growing tumors with higher tumor grade, and greater histopathologic similarity to the human disease [64, 65]. Additionally, tumors in these animals display invasive and metastatic capability. The synergy between the Kras and TP53 mutations was demonstrated by the phenotypically more advanced lung tumors in the combination and the shift in the tumor spectrum from a preponderance of sarcomas and lymphomas in the animals with only p53 mutation to epithelial tumors, such as lung adenocarcinoma.
The gain-of-function phenotype observed with mutant p53 in cell and animal models may result from the fact that p63 and p73 preferentially bind to mutant rather than wild-type p53 [43, 66]. These results have been confirmed by several groups using the murine models of Li-Fraumeni containing a TP53R172H allele [67, 68]. Additionally, transfection of siRNA against p63 or p73 into the p53 null mouse embryo fibroblasts (MEFs) produced transformation potential similar to that of TP53R172H homozygous MEFs. Further evidence for this gain-of-function mechanism from missense mutant p53 has been presented from analysis of compound mutant mice containing p53 loss along with p63+/− or p73+/− [69, 70]. Similar to the effect of Kras and p53 combination, the p63/p53 or p73/p53 combinations exhibited a shift in tumor spectra, with a high rate of epithelial tumor types. Additionally, the p63+/−;p73+/− mice developed many of the same tumor types, suggesting a p53-independent tumor suppressive role for these family members.
Role of p53 on tumor progression and metastasis
Multiple alleles of p53 have been generated over the years to better study the many aspects of p53 function in vivo, including invasion and metastasis. Both the p53 null and the p53 mutant models (R172H and R270H) in combination with mutant Kras recapitulate the tumor progression and metastatic phenotype observed in lung cancer patients [64, 65]. The pattern of metastatic spread and compromised longevity of the animals is similar to that found in lung cancer patients and transcriptome expression profiling of these primary and metastatic tumors recapitulates features of the patient tumors [71, 72]. As such, these models have been useful to identify oncogenic dependencies in established tumors, such as NF-kappaB activation [40, 41], transcriptional or epigenetic drivers of histologic progression and metastasis, such as Nkx2-1 or microRNA changes [72, 73], and signaling pathway altered during tumor invasion, e.g. Notch signaling [74].
Work by our group and others has shown that there is a prominent role for microRNA reprogramming in these models that is necessary for tumor cell progression and metastasis. The epithelial-mesenchymal transition occurs in a subset of tumor cells in this model, producing invasion and metastasis [73, 74]. This phenotype is dependent upon the loss of expression of the miR-200 family members, the de-repression of the EMT-inducing transcription factors such as Zeb1, and the concomitant reprogramming of the protein expression in the cells [75]. Similar changes in the miR-200 family members were seen in human NSCLC cell lines and more recently in tumors from the TCGA dataset [73, 76]. The up-regulation of transcription factors such as Zeb1 also produces concomitant changes in the expression of other microRNAs such as miR-34a [77, 78]. These complementary microRNA changes are required for the full invasive and metastatic phenotype. Interestingly, work from the Belinsky lab has shown that treatment of HBECs with stressful, but not genotoxic, doses of tobacco carcinogens (methylnitrosourea & benzo(a)pyrene-diolepoxide) produces epigenetic repression of miR-200 family members by promoter hypermethylation [79]. The cells then display mesenchymal features, including increased anchorage-independent growth and invasion in 3D culture.
Clinical Implications of p53 Mutation
There is certainly a role in Li-Fraumeni kindreds for genetic testing of individuals, with regular medical check-ups and early screening for breast and colon cancer recommended for family members with a documented TP53 mutation. However, there is no role for TP53 mutation screening in the general population. In patients with a diagnosed lung cancer, molecular profiling of tumor tissue is frequently obtained, but the p53 status of the tumor is not currently used in therapeutic decision-making. Review of historical data is controversial about the predictive or prognostic role of p53 in lung cancer, but more recent data from a meta-analysis reported that TP53 mutations are a marker of poor prognosis [80], which has been confirmed by retrospective immunohistochemical (IHC) analysis of samples from two independent clinical trials (CALGB 9633 and JBR.10) where p53-positive staining by IHC conferred inferior survival, with a HR of 2.3 and 1.89, respectively, and was also predictive of benefit from chemotherapy [81, 82]. However, it is unclear if all p53 mutants confer the same clinical risk, whether the status of the second p53 allele is important for outcome and whether IHC is the best assay for assessing the p53 status of tumors. Our clinical thinking on this point will likely evolve as further analyses emerge from more complete datasets of sequenced patient tumors, such as found in The Cancer Genome Atlas and similar projects.
Multiple groups have generated murine models with conditional null or hypomorphic alleles of p53 to test the effect of re-activating wild-type p53 in established tumors. The results of these studies were surprising in several ways. First, the effect on tumor cells is dependent upon the tumor type, with lymphomas responding differently than sarcomas and carcinomas. The Evan lab used the Eμ-Myc model in combination with an allele encoding a p53-estrogen receptor fusion protein [83]. Upon activation of p53 by treatment with tamoxifen, induction of apoptosis occurred in the established lymphomas, with subsequent tumor regression. Using their p53LSL/LSL model, the Jacks lab showed that reactivation of wild-type p53 in lymphomas produced rapid induction of apoptosis and tumor regression, while sarcomas displayed delayed tumor regression due to reduced proliferation, cell cycle arrest and senescence [84]. The Lowe lab used a Tet-responsive promoter to drive shRNA-mediated p53 repression in a mutant Kras-driven model of liver carcinoma [85]. Upon even temporally limited re-expression of p53 with doxycycline treatment, senescence occurred in the liver carcinoma cells, followed by tumor cell clearance by the immune system. These studies also demonstrated that p53 reactivation had no apparent effects on the wild-type tissues in the animals, an important finding in thinking about the possible side-effects from therapeutic applications and in understanding the role of p53 activation in different tumor types. This point was further illustrated in reports from two different labs using the KrasG12D allele along with a re-activatable p53 [86, 87], which produces lung adenoma and adenocarcinoma. The response of tumor cells to wild-type p53 reactivation differed depending upon the stage of tumor progression and the degree of oncogenic signaling through the MAPK pathway, as measured by levels of p-ERK. The more advanced, later stage tumors, with elevated MAPK signaling provided a greater oncogenic activation, thereby enhancing p53 stability and producing a more pronounced effect of re-activation in this population of cells. These results highlight the heterogeneous response that is likely to be seen in clinical targeting of p53.
Since p53 is seldom null in human tumors, but frequently develops loss-of-heterozygosity at one allele and a missense mutation in the second allele, the presence of a mutant allele with gain-of-function activity becomes a potentially important therapeutic issue. To compare the effect of reactivating wild-type p53 in the setting of either a null second allele or missense mutation, the Lozano lab used the p53R172H allele, combined with a p53 wild-type allele containing a Lox-PGKneo-Lox cassette in intron 4, which makes it functionally hypomorphic [88]. Upon Cre recombinase removal of the neo cassette, wild-type p53 expression was restored to normal levels, while maintaining continued expression of the mutant p53. Reactivation of the wild-type on the background of a null second allele produced regression of established lymphomas and sarcomas, consistent with the findings from other groups. However, when the wild-type was restored in the presence of the R172H mutant, the tumors stopped growing, but did not undergo apoptosis, senescence, or tumor regression. They further demonstrated by chromatin immunoprecipitation that these results were a consequence of the mutant p53R172H protein binding the wild-type p53 and blunting its transcriptional activity of pro-apoptotic genes such as puma, but not its ability to bind the p21 promoter and block the cell cycle. Unfortunately in this pure model of p53 function the animals developed almost exclusively sarcomas and lymphomas, so it is unknown if the same results would be found in a combined model that develops lung cancer or another epithelial tumor type (e.g. the mutant Kras/p53 model).
Clinical practice with standard and combination therapies may also be influenced by the work emerging from animal models. A recently published co-clinical trial in lung cancer described the results of treating mouse models containing single or combined alleles (mutant Kras, mutant Kras+TP53 null, or mutant Kras+LKB1 null) with docetaxel alone or docetaxel combined with the MEK inhibitor selumetinib (AZD6244), a combination being explored in clinical trials [89]. This animal trial demonstrated that Kras mutant tumors respond better to treatment than tumors with concomitant mutations (either TP53 or LKB1 null), and that the Kras and Kras/p53 tumor types responded better to the combination than to docetaxel alone. The Kras/LKB1 tumors displayed primary resistance to the addition of selumetinib. These results demonstrate that the underlying survival pathways differ depending upon the combination of mutations found in the tumor. However, given the gain-of-function activity of mutant TP53 it is unclear how the results might have differed if the animal model contained mutant TP53 rather than the null allele combined with mutant Kras.
Early studies using adenovirus early region 1A (E1A) and Ras transformed TP53 null MEF cell lines and a syngeneic tumor model of fibrosarcoma demonstrated that p53 is required for tumor cell killing by gamma irradiation or several different chemotherapy agents [90, 91]. These findings supported the concept that therapy-induced apoptosis in vitro or in vivo requires active p53, the loss of which produces tumor resistance. However, a recent study addressed the more complex question of how mutant or null p53 status predicts for chemotherapy response in a spontaneous model of breast cancer [92]. In this report the MMTV-Wnt1 mice were crossed onto three different p53 backgrounds, wild-type, p53 null and the R172H mutant. These animals all formed breast carcinomas that were treated with doxorubicin. The animals with wild-type p53 had minimal tumor shrinkage, with rapid relapse. By contrast the animals with the R172H allele, and LOH of the second wild-type allele, had the best response to doxorubicin and longer time to relapse. At the cellular level this phenotype was due to cell cycle arrest/senescence in the tumors retaining wild-type p53 versus progression through the cell cycle in tumors with mutant p53, which resulted in aberrant mitoses and induction of apoptosis. These results may explain the paradoxical findings that mutant TP53 is a poor prognostic factor, but a good predictive factor for response to chemotherapy in some tumor types. They also highlight the importance of full TP53 genotyping if clinical decisions are to made on this information, as the LOH status of the tumors in this study was critical to their response to chemotherapy.
Targeted therapies for p53
Multiple strategies have been advocated to restore wild-type p53 function and target the p53 missense mutants frequently found in cancers. These include, viral vector delivery of wild-type p53, small molecules designed to alter the mutant protein conformation or targeted disruption of its interaction with other proteins. Due to the difficulties of appropriately delivering vector-based TP53, and the modest results in Phase I and II trials [93, 94], the efforts in the field have shifted. Nutlin (RG7112) was the first compound described in its class, capable of targeting the interaction of p53 and MDM2 [95]. By blocking the p53 binding cleft of MDM2, nutlins inhibit MDM2-mediated degradation of wild-type p53, increasing p53 transcriptional activity. However, because many tumors do not retain any wild-type p53, these agents have found limited potential clinical application.
Other agents, such as PRIMA-1 and RETRA, were discovered in screens specifically designed to find compounds able of suppressing tumor cell growth in a mutant p53-dependent manner. The compound 2,2-bis(hydroxymethyl)-1-azabicyclo[2,2,2]octan-3-one was identified more than 10 years ago and named PRIMA-1 (p53 re-activation and induction of massive apoptosis) [96]. The reported mechanism of action relies upon binding of the molecule to the core domain of mutant p53, stabilizing its conformation via alkylation of thiol groups, producing a p53 molecule with wild-type conformation that is capable of appropriately binding its transcriptional targets, leading to cell cycle arrest and apoptosis in tumor cells. It has been reported to have efficacy in cell culture and animal xenograft models of mutant p53 cancers. The PRIMA-1 structural analog, APR-246 (PRIMA-1MET), is a methylated derivative that is the first drug of this class to reach clinical testing. Recently a Phase 1 trial was completed and reported on the dosing and safety profile of APR-246 in 22 patients with hematologic malignancies or hormone refractory prostate cancer [97]. Given by intravenous infusion once per day for four consecutive days, it was well tolerated by patients, with predictable pharmacokinetic properties. Evidence from global expression profiling of circulating hematologic tumor cells from treated patients demonstrated cell cycle arrest, apoptosis, and up-regulation of p53 target genes, NOXA, PUMA, BAX. Whether all of the gene expression changes induced by APR-246 in patient tumors can be attributed to p53-specific mechanisms versus off-target effects is currently unclear.
RETRA (reactivation of transcriptional reporter activity) (2-(4,5-dihydro-1,3-thiazol-2-ylthio)-1-(3,4-dihyrdrophyenyl)ethanone hydrobromide) is reportedly the best in a small class of compounds with p53-mutant specific activity, that produces slower growth, reactivation of apoptosis and repression of tumor formation in a xenograft model. The reported mechanism of action is that treatment with RETRA releases p73 from its complex with mutant p53, producing reactivation of many downstream p73 transcriptional targets, such as CDKN1A and the effector caspases 3 and 7 [98].
Perspective and Conclusions
Tobacco exposure produces a heavy burden of genomic mutations in lung cancer, including mutation of the tumor suppressor TP53. Both loss of the wild-type p53 function and gain of mutant p53 function are important to the tumorigenic process. TP53 alterations are very frequent in squamous cell carcinoma, adenocarcinoma and small cell carcinoma of the lung. Both in vitro and in vivo models of lung cancer reveal the critical role of p53 alteration to malignant transformation, histologic progression, invasion and metastasis. Clinical treatment paradigms will need to evolve to incorporate the role of p53 loss and mutation, accounting for the differential effect on outcomes and our current inability to directly target p53 status in tumors. Pre-clinical models will remain an invaluable set of tools for elucidating a more complete understanding of p53 biology and the continued investigation of how to best target aberrant p53 function clinically.
Acknowledgments
We would like to thank Lixia Diao and Dr. Jing Wang for their assistance in analyzing the TCGA datasets for the information in Figures 1 and 2. This work was supported by the MD Anderson Cancer Center Physician-Scientist Program (D.L.G. and L.A.B.), NCI K08 CA151651 (D.L.G.), and the Elza and Ina A. Schackelford Endowed Professorship in Lung Cancer Research (J.M.K.). We would like to apologize to the authors whose work we have not been able to cite owing to space limitations.
References
- 1.White C. Research on smoking and lung cancer: a landmark in the history of chronic disease epidemiology. Yale J Biol Med. 1990;63(1):29–46. [PMC free article] [PubMed] [Google Scholar]
- 2.Ochsner A, DeBakey M. Primary Pulmonary Malignancy: Treatment by Total Pneumonectomy Analysis of 70 Collected Cases and Presentation of 7 Personal Cases. Surgery, Gynecology and Obstetrics. 1939;68:435–451. [PMC free article] [PubMed] [Google Scholar]
- 3.Doll R, Hill AB. Smoking and carcinoma of the lung; preliminary report. Br Med J. 1950;2(4682):739–48. doi: 10.1136/bmj.2.4682.739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wynder EL, Graham EA. Tobacco smoking as a possible etiologic factor in bronchiogenic carcinoma; a study of 684 proved cases. J Am Med Assoc. 1950;143(4):329–36. doi: 10.1001/jama.1950.02910390001001. [DOI] [PubMed] [Google Scholar]
- 5.Burney LE. Smoking and lung cancer: a statement of the Public Health Service. J Am Med Assoc. 1959;171:1829–37. doi: 10.1001/jama.1959.73010310005016. [DOI] [PubMed] [Google Scholar]
- 6.MEDICAL Research Council’s statement on tobacco smoking and cancer of the lung. Lancet. 1957;272(6983):1345–7. [PubMed] [Google Scholar]
- 7.United States Public Health Service: Smoking and Health. Report of the Advisory Committee to the Surgeon General of the Public Health Service. 1964;Chapter 5 PHS Publication No. 1103. [Google Scholar]
- 8.Moolgavkar SH, et al. Impact of reduced tobacco smoking on lung cancer mortality in the United States during 1975–2000. J Natl Cancer Inst. 2012;104(7):541–8. doi: 10.1093/jnci/djs136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Brauch H, et al. Molecular analysis of the short arm of chromosome 3 in small-cell and non-small-cell carcinoma of the lung. N Engl J Med. 1987;317(18):1109–13. doi: 10.1056/NEJM198710293171803. [DOI] [PubMed] [Google Scholar]
- 10.Chiba I, et al. Mutations in the p53 gene are frequent in primary, resected non-small cell lung cancer. Lung Cancer Study Group. Oncogene. 1990;5(10):1603–10. [PubMed] [Google Scholar]
- 11.Nigro JM, et al. Mutations in the p53 gene occur in diverse human tumour types. Nature. 1989;342(6250):705–8. doi: 10.1038/342705a0. [DOI] [PubMed] [Google Scholar]
- 12.Takahashi T, et al. p53: a frequent target for genetic abnormalities in lung cancer. Science. 1989;246(4929):491–4. doi: 10.1126/science.2554494. [DOI] [PubMed] [Google Scholar]
- 13.Iggo R, et al. Increased expression of mutant forms of p53 oncogene in primary lung cancer. Lancet. 1990;335(8691):675–9. doi: 10.1016/0140-6736(90)90801-b. [DOI] [PubMed] [Google Scholar]
- 14.Malkin D, et al. Germ line p53 mutations in a familial syndrome of breast cancer, sarcomas, and other neoplasms. Science. 1990;250(4985):1233–8. doi: 10.1126/science.1978757. [DOI] [PubMed] [Google Scholar]
- 15.Hollstein M, et al. Database of p53 gene somatic mutations in human tumors and cell lines. Nucleic Acids Res. 1994;22(17):3551–5. [PMC free article] [PubMed] [Google Scholar]
- 16.Pfeifer GP, et al. Tobacco smoke carcinogens, DNA damage and p53 mutations in smoking-associated cancers. Oncogene. 2002;21(48):7435–51. doi: 10.1038/sj.onc.1205803. [DOI] [PubMed] [Google Scholar]
- 17.Hussain SP, et al. Mutability of p53 hotspot codons to benzo(a)pyrene diol epoxide (BPDE) and the frequency of p53 mutations in nontumorous human lung. Cancer Res. 2001;61(17):6350–5. [PubMed] [Google Scholar]
- 18.Hecht SS. Tobacco smoke carcinogens and lung cancer. J Natl Cancer Inst. 1999;91(14):1194–210. doi: 10.1093/jnci/91.14.1194. [DOI] [PubMed] [Google Scholar]
- 19.Comprehensive genomic characterization of squamous cell lung cancers. Nature. 2012;489(7417):519–25. doi: 10.1038/nature11404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Govindan R, et al. Genomic landscape of non-small cell lung cancer in smokers and never-smokers. Cell. 2012;150(6):1121–34. doi: 10.1016/j.cell.2012.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lawrence MS, et al. Mutational heterogeneity in cancer and the search for new cancer-associated genes. Nature. 2013;499(7457):214–8. doi: 10.1038/nature12213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Levine AJ, Momand J, Finlay CA. The p53 tumour suppressor gene. Nature. 1991;351(6326):453–6. doi: 10.1038/351453a0. [DOI] [PubMed] [Google Scholar]
- 23.Linzer DI, Levine AJ. Characterization of a 54K dalton cellular SV40 tumor antigen present in SV40-transformed cells and uninfected embryonal carcinoma cells. Cell. 1979;17(1):43–52. doi: 10.1016/0092-8674(79)90293-9. [DOI] [PubMed] [Google Scholar]
- 24.Lane DP, Crawford LV. T antigen is bound to a host protein in SV40-transformed cells. Nature. 1979;278(5701):261–3. doi: 10.1038/278261a0. [DOI] [PubMed] [Google Scholar]
- 25.Chen PL, et al. Genetic mechanisms of tumor suppression by the human p53 gene. Science. 1990;250(4987):1576–80. doi: 10.1126/science.2274789. [DOI] [PubMed] [Google Scholar]
- 26.Finlay CA, Hinds PW, Levine AJ. The p53 proto-oncogene can act as a suppressor of transformation. Cell. 1989;57(7):1083–93. doi: 10.1016/0092-8674(89)90045-7. [DOI] [PubMed] [Google Scholar]
- 27.Eliyahu D, et al. Participation of p53 cellular tumour antigen in transformation of normal embryonic cells. Nature. 1984;312(5995):646–9. doi: 10.1038/312646a0. [DOI] [PubMed] [Google Scholar]
- 28.Haupt Y, et al. Mdm2 promotes the rapid degradation of p53. Nature. 1997;387(6630):296–9. doi: 10.1038/387296a0. [DOI] [PubMed] [Google Scholar]
- 29.Kubbutat MH, Jones SN, Vousden KH. Regulation of p53 stability by Mdm2. Nature. 1997;387(6630):299–303. doi: 10.1038/387299a0. [DOI] [PubMed] [Google Scholar]
- 30.Halazonetis TD, V, Gorgoulis G, Bartek J. An oncogene-induced DNA damage model for cancer development. Science. 2008;319(5868):1352–5. doi: 10.1126/science.1140735. [DOI] [PubMed] [Google Scholar]
- 31.Kruse JP, Gu W. Modes of p53 regulation. Cell. 2009;137(4):609–22. doi: 10.1016/j.cell.2009.04.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Dai C, Gu W. p53 post-translational modification: deregulated in tumorigenesis. Trends Mol Med. 2010;16(11):528–36. doi: 10.1016/j.molmed.2010.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Resnick-Silverman L, et al. Identification of a novel class of genomic DNA-binding sites suggests a mechanism for selectivity in target gene activation by the tumor suppressor protein p53. Genes Dev. 1998;12(14):2102–7. doi: 10.1101/gad.12.14.2102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nikulenkov F, et al. Insights into p53 transcriptional function via genome-wide chromatin occupancy and gene expression analysis. Cell Death Differ. 2012;19(12):1992–2002. doi: 10.1038/cdd.2012.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Schlereth K, et al. Characterization of the p53 cistrome--DNA binding cooperativity dissects p53’s tumor suppressor functions. PLoS Genet. 2013;9(8):e1003726. doi: 10.1371/journal.pgen.1003726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Carvajal LA, Manfredi JJ. Another fork in the road--life or death decisions by the tumour suppressor p53. EMBO Rep. 2013;14(5):414–21. doi: 10.1038/embor.2013.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li T, et al. Tumor suppression in the absence of p53-mediated cell-cycle arrest, apoptosis, and senescence. Cell. 2012;149(6):1269–83. doi: 10.1016/j.cell.2012.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Vlahos R, et al. Differential protease, innate immunity, and NF-kappaB induction profiles during lung inflammation induced by subchronic cigarette smoke exposure in mice. Am J Physiol Lung Cell Mol Physiol. 2006;290(5):L931–45. doi: 10.1152/ajplung.00201.2005. [DOI] [PubMed] [Google Scholar]
- 39.Takahashi H, et al. Tobacco smoke promotes lung tumorigenesis by triggering IKKbeta- and JNK1-dependent inflammation. Cancer Cell. 2010;17(1):89–97. doi: 10.1016/j.ccr.2009.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Meylan E, et al. Requirement for NF-kappaB signalling in a mouse model of lung adenocarcinoma. Nature. 2009;462(7269):104–7. doi: 10.1038/nature08462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Xue W, et al. Response and resistance to NF-kappaB inhibitors in mouse models of lung adenocarcinoma. Cancer Discov. 2011;1(3):236–47. doi: 10.1158/2159-8290.CD-11-0073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Muller PA, Vousden KH. p53 mutations in cancer. Nat Cell Biol. 2013;15(1):2–8. doi: 10.1038/ncb2641. [DOI] [PubMed] [Google Scholar]
- 43.Gaiddon C, et al. A subset of tumor-derived mutant forms of p53 down-regulate p63 and p73 through a direct interaction with the p53 core domain. Mol Cell Biol. 2001;21(5):1874–87. doi: 10.1128/MCB.21.5.1874-1887.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Barrett LA, et al. Long-term organ culture of human bronchial epithelium. Cancer Res. 1976;36(3):1003–10. [PubMed] [Google Scholar]
- 45.Lechner JF, et al. Clonal growth of epithelial cells from normal adult human bronchus. Cancer Res. 1981;41(6):2294–304. [PubMed] [Google Scholar]
- 46.Reddel RR, et al. Transformation of human bronchial epithelial cells by infection with SV40 or adenovirus-12 SV40 hybrid virus, or transfection via strontium phosphate coprecipitation with a plasmid containing SV40 early region genes. Cancer Res. 1988;48(7):1904–9. [PubMed] [Google Scholar]
- 47.Harris CC, et al. Interindividual variation in binding of benzo[a]pyrene to DNA in cultured human bronchi. Science. 1976;194(4269):1067–9. doi: 10.1126/science.982061. [DOI] [PubMed] [Google Scholar]
- 48.Klein-Szanto AJ, et al. A tobacco-specific N-nitrosamine or cigarette smoke condensate causes neoplastic transformation of xenotransplanted human bronchial epithelial cells. Proc Natl Acad Sci U S A. 1992;89(15):6693–7. doi: 10.1073/pnas.89.15.6693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Willey JC, et al. Biochemical and morphological effects of cigarette smoke condensate and its fractions on normal human bronchial epithelial cells in vitro. Cancer Res. 1987;47(8):2045–9. [PubMed] [Google Scholar]
- 50.Ramirez RD, et al. Immortalization of human bronchial epithelial cells in the absence of viral oncoproteins. Cancer Res. 2004;64(24):9027–34. doi: 10.1158/0008-5472.CAN-04-3703. [DOI] [PubMed] [Google Scholar]
- 51.Sato M, et al. Human lung epithelial cells progressed to malignancy through specific oncogenic manipulations. Mol Cancer Res. 2013;11(6):638–50. doi: 10.1158/1541-7786.MCR-12-0634-T. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sato M, et al. Multiple oncogenic changes (K-RAS(V12), p53 knockdown, mutant EGFRs, p16 bypass, telomerase) are not sufficient to confer a full malignant phenotype on human bronchial epithelial cells. Cancer Res. 2006;66(4):2116–28. doi: 10.1158/0008-5472.CAN-05-2521. [DOI] [PubMed] [Google Scholar]
- 53.Lavigueur A, et al. High incidence of lung, bone, and lymphoid tumors in transgenic mice overexpressing mutant alleles of the p53 oncogene. Mol Cell Biol. 1989;9(9):3982–91. doi: 10.1128/mcb.9.9.3982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Donehower LA, et al. Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature. 1992;356(6366):215–21. doi: 10.1038/356215a0. [DOI] [PubMed] [Google Scholar]
- 55.Kemp CJ, et al. Reduction of p53 gene dosage does not increase initiation or promotion but enhances malignant progression of chemically induced skin tumors. Cell. 1993;74(5):813–22. doi: 10.1016/0092-8674(93)90461-x. [DOI] [PubMed] [Google Scholar]
- 56.Lee JM, et al. Susceptibility to radiation-carcinogenesis and accumulation of chromosomal breakage in p53 deficient mice. Oncogene. 1994;9(12):3731–6. [PubMed] [Google Scholar]
- 57.Harvey M, et al. A mutant p53 transgene accelerates tumour development in heterozygous but not nullizygous p53-deficient mice. Nat Genet. 1995;9(3):305–11. doi: 10.1038/ng0395-305. [DOI] [PubMed] [Google Scholar]
- 58.Gouyer V, et al. Mechanism of retinoblastoma gene inactivation in the spectrum of neuroendocrine lung tumors. Am J Respir Cell Mol Biol. 1998;18(2):188–96. doi: 10.1165/ajrcmb.18.2.3008. [DOI] [PubMed] [Google Scholar]
- 59.Meuwissen R, et al. Induction of small cell lung cancer by somatic inactivation of both Trp53 and Rb1 in a conditional mouse model. Cancer Cell. 2003;4(3):181–9. doi: 10.1016/s1535-6108(03)00220-4. [DOI] [PubMed] [Google Scholar]
- 60.Williams BO, et al. Cooperative tumorigenic effects of germline mutations in Rb and p53. Nat Genet. 1994;7(4):480–4. doi: 10.1038/ng0894-480. [DOI] [PubMed] [Google Scholar]
- 61.Jackson EL, et al. Analysis of lung tumor initiation and progression using conditional expression of oncogenic K-ras. Genes Dev. 2001;15(24):3243–8. doi: 10.1101/gad.943001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Johnson L, et al. Somatic activation of the K-ras oncogene causes early onset lung cancer in mice. Nature. 2001;410(6832):1111–6. doi: 10.1038/35074129. [DOI] [PubMed] [Google Scholar]
- 63.Meuwissen R, et al. Mouse model for lung tumorigenesis through Cre/lox controlled sporadic activation of the K-Ras oncogene. Oncogene. 2001;20(45):6551–8. doi: 10.1038/sj.onc.1204837. [DOI] [PubMed] [Google Scholar]
- 64.Jackson EL, et al. The differential effects of mutant p53 alleles on advanced murine lung cancer. Cancer Res. 2005;65(22):10280–8. doi: 10.1158/0008-5472.CAN-05-2193. [DOI] [PubMed] [Google Scholar]
- 65.Zheng S, et al. A genetic mouse model for metastatic lung cancer with gender differences in survival. Oncogene. 2007;26(48):6896–904. doi: 10.1038/sj.onc.1210493. [DOI] [PubMed] [Google Scholar]
- 66.Di Como CJ, Gaiddon C, Prives C. p73 function is inhibited by tumor-derived p53 mutants in mammalian cells. Mol Cell Biol. 1999;19(2):1438–49. doi: 10.1128/mcb.19.2.1438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lang GA, et al. Gain of function of a p53 hot spot mutation in a mouse model of Li-Fraumeni syndrome. Cell. 2004;119(6):861–72. doi: 10.1016/j.cell.2004.11.006. [DOI] [PubMed] [Google Scholar]
- 68.Olive KP, et al. Mutant p53 gain of function in two mouse models of Li-Fraumeni syndrome. Cell. 2004;119(6):847–60. doi: 10.1016/j.cell.2004.11.004. [DOI] [PubMed] [Google Scholar]
- 69.Flores ER, et al. Tumor predisposition in mice mutant for p63 and p73: evidence for broader tumor suppressor functions for the p53 family. Cancer Cell. 2005;7(4):363–73. doi: 10.1016/j.ccr.2005.02.019. [DOI] [PubMed] [Google Scholar]
- 70.Flores ER, et al. p63 and p73 are required for p53-dependent apoptosis in response to DNA damage. Nature. 2002;416(6880):560–4. doi: 10.1038/416560a. [DOI] [PubMed] [Google Scholar]
- 71.Gibbons DL, et al. Expression signatures of metastatic capacity in a genetic mouse model of lung adenocarcinoma. PLoS One. 2009;4(4):e5401. doi: 10.1371/journal.pone.0005401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Winslow MM, et al. Suppression of lung adenocarcinoma progression by Nkx2-1. Nature. 2011;473(7345):101–4. doi: 10.1038/nature09881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Gibbons DL, et al. Contextual extracellular cues promote tumor cell EMT and metastasis by regulating miR-200 family expression. Genes Dev. 2009;23(18):2140–51. doi: 10.1101/gad.1820209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Yang Y, et al. The Notch ligand Jagged2 promotes lung adenocarcinoma metastasis through a miR-200-dependent pathway in mice. J Clin Invest. 2011;121(4):1373–85. doi: 10.1172/JCI42579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Schliekelman MJ, et al. Targets of the tumor suppressor miR-200 in regulation of the epithelial-mesenchymal transition in cancer. Cancer Res. 2011;71(24):7670–82. doi: 10.1158/0008-5472.CAN-11-0964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Pecot CV, et al. Tumour angiogenesis regulation by the miR-200 family. Nat Commun. 2013;4:2427. doi: 10.1038/ncomms3427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Ahn YH, et al. ZEB1 drives prometastatic actin cytoskeletal remodeling by downregulating miR-34a expression. J Clin Invest. 2012;122(9):3170–83. doi: 10.1172/JCI63608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Kasinski AL, Slack FJ. miRNA-34 prevents cancer initiation and progression in a therapeutically resistant K-ras and p53-induced mouse model of lung adenocarcinoma. Cancer Res. 2012;72(21):5576–87. doi: 10.1158/0008-5472.CAN-12-2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Tellez CS, et al. EMT and stem cell-like properties associated with miR-205 and miR-200 epigenetic silencing are early manifestations during carcinogen-induced transformation of human lung epithelial cells. Cancer Res. 2011;71(8):3087–97. doi: 10.1158/0008-5472.CAN-10-3035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Mitsudomi T, et al. Prognostic significance of p53 alterations in patients with non-small cell lung cancer: a meta-analysis. Clin Cancer Res. 2000;6(10):4055–63. [PubMed] [Google Scholar]
- 81.Graziano SL, et al. Prognostic significance of mucin and p53 expression in stage IB non-small cell lung cancer: a laboratory companion study to CALGB 9633. J Thorac Oncol. 2010;5(6):810–7. doi: 10.1097/jto.0b013e3181d89f95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Tsao MS, et al. Prognostic and predictive importance of p53 and RAS for adjuvant chemotherapy in non small-cell lung cancer. J Clin Oncol. 2007;25(33):5240–7. doi: 10.1200/JCO.2007.12.6953. [DOI] [PubMed] [Google Scholar]
- 83.Christophorou MA, et al. Temporal dissection of p53 function in vitro and in vivo. Nat Genet. 2005;37(7):718–26. doi: 10.1038/ng1572. [DOI] [PubMed] [Google Scholar]
- 84.Ventura A, et al. Restoration of p53 function leads to tumour regression in vivo. Nature. 2007;445(7128):661–5. doi: 10.1038/nature05541. [DOI] [PubMed] [Google Scholar]
- 85.Xue W, et al. Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature. 2007;445(7128):656–60. doi: 10.1038/nature05529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Feldser DM, et al. Stage-specific sensitivity to p53 restoration during lung cancer progression. Nature. 2010;468(7323):572–5. doi: 10.1038/nature09535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Junttila MR, et al. Selective activation of p53-mediated tumour suppression in high-grade tumours. Nature. 2010;468(7323):567–71. doi: 10.1038/nature09526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Wang Y, et al. Restoring expression of wild-type p53 suppresses tumor growth but does not cause tumor regression in mice with a p53 missense mutation. J Clin Invest. 2011;121(3):893–904. doi: 10.1172/JCI44504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Chen Z, et al. A murine lung cancer co-clinical trial identifies genetic modifiers of therapeutic response. Nature. 2012;483(7391):613–7. doi: 10.1038/nature10937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Lowe SW, et al. p53 status and the efficacy of cancer therapy in vivo. Science. 1994;266(5186):807–10. doi: 10.1126/science.7973635. [DOI] [PubMed] [Google Scholar]
- 91.Lowe SW, et al. p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell. 1993;74(6):957–67. doi: 10.1016/0092-8674(93)90719-7. [DOI] [PubMed] [Google Scholar]
- 92.Jackson JG, et al. p53-mediated senescence impairs the apoptotic response to chemotherapy and clinical outcome in breast cancer. Cancer Cell. 2012;21(6):793–806. doi: 10.1016/j.ccr.2012.04.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Roth JA, et al. Retrovirus-mediated wild-type p53 gene transfer to tumors of patients with lung cancer. Nat Med. 1996;2(9):985–91. doi: 10.1038/nm0996-985. [DOI] [PubMed] [Google Scholar]
- 94.Schuler M, et al. Adenovirus-mediated wild-type p53 gene transfer in patients receiving chemotherapy for advanced non-small-cell lung cancer: results of a multicenter phase II study. J Clin Oncol. 2001;19(6):1750–8. doi: 10.1200/JCO.2001.19.6.1750. [DOI] [PubMed] [Google Scholar]
- 95.Vassilev LT, et al. In vivo activation of the p53 pathway by small-molecule antagonists of MDM2. Science. 2004;303(5659):844–8. doi: 10.1126/science.1092472. [DOI] [PubMed] [Google Scholar]
- 96.Bykov VJ, et al. Restoration of the tumor suppressor function to mutant p53 by a low-molecular-weight compound. Nat Med. 2002;8(3):282–8. doi: 10.1038/nm0302-282. [DOI] [PubMed] [Google Scholar]
- 97.Lehmann S, et al. Targeting p53 in vivo: a first-in-human study with p53-targeting compound APR-246 in refractory hematologic malignancies and prostate cancer. J Clin Oncol. 2012;30(29):3633–9. doi: 10.1200/JCO.2011.40.7783. [DOI] [PubMed] [Google Scholar]
- 98.Kravchenko JE, et al. Small-molecule RETRA suppresses mutant p53-bearing cancer cells through a p73-dependent salvage pathway. Proc Natl Acad Sci U S A. 2008;105(17):6302–7. doi: 10.1073/pnas.0802091105. [DOI] [PMC free article] [PubMed] [Google Scholar]