Abstract
Hypertensive pregnancy disorders complicate 6–8% of pregnancies and cause significant maternal and fetal morbidity and mortality. The goal of treatment is to prevent significant cerebrovascular and cardiovascular events in the mother, without compromising fetal well-being. Current guidelines differentiate between the treatment of women with acute hypertensive syndromes of pregnancy and women with preexisting chronic hypertension in pregnancy. This review will address the management of hypertension in pregnancy, review the various pharmacologic therapies, and discuss the future directions in this field.
Keywords: hypertension, anti-hypertensive agents, preeclampsia, pregnancy, gestational hypertension
Introduction
Hypertensive pregnancy disorders cover a spectrum of conditions, including preeclampsia/eclampsia, gestational hypertension, chronic hypertension, and preeclampsia superimposed on chronic hypertension (Table 1). According to the National High Blood Pressure Education Program (NHBPEP) Working Group Report on High Blood Pressure (BP) in Pregnancy, hypertension occurs in 6–8% of pregnancies in the United States. 1 Hypertensive pregnancy disorders represent the most significant complications of pregnancy and contribute significantly to maternal and perinatal morbidity and mortality. 2 Most of the current recommendations for the treatment of these disorders are based on expert opinion and observational studies, with a lack of evidence from randomized controlled trials. The overall strategy in the treatment of hypertension in pregnancy is to prevent maternal cerebrovascular and cardiac complications, while preserving the uteroplacental and fetal circulation and limiting medication toxicity to the fetus.
Table 1.
Hypertensive Pregnancy Disorder classification
| Preeclampsia/Eclampsia | Hypertension and proteinuria of ≥ 300 mg/24 hours after 20 weeks gestation. Eclampsia (the convulsive form of preeclampsia) affects 0.1% of all pregnancies. |
| Gestational hypertension | Hypertension occurring for the first time after 20 weeks of pregnancy in the absence of proteinuria. |
| Chronic hypertension | Blood pressure greater than or equal to 140/90 mm Hg prior to pregnancy, or before the 20th week of gestation. |
| Preeclampsia superimposed on chronic hypertension | Up to 30% of women with chronic hypertension develop preeclampsia, heralded by proteinuria, which occurs for the first time during the third trimester, and/or a sudden increase in previous level of proteinuria, sudden increase in blood pressure in a woman with previously controlled BP, thrombocytopenia (<100,000 cells/mm3), or increase in alanine aminotransferase or aspartate aminotransferase to abnormal levels |
Treatment strategies fall into two general categories – the management of acute hypertensive syndromes of pregnancy, such as preeclampsia/eclampsia, and the management of chronic hypertension. While the definitive treatment for acute hypertensive syndromes of pregnancy is delivery, expectant management with close observation may be appropriate in carefully selected patients, especially before 32 weeks gestation. Women with chronic hypertension should ideally be evaluated prior to pregnancy, with a focus on the presence of end-organ damage, evidence of secondary causes of hypertension (such as renal artery stenosis due to fibromuscular dysplasia, primary hyperaldosteronism and pheochromocytoma), medication adjustments, and counseling regarding the risks of preeclampsia and adverse fetal events.
Women with hypertensive pregnancy disorders should have a comprehensive plan of care, which includes prenatal counseling, frequent visits during pregnancy, timely delivery, appropriate intrapartum monitoring and care, and postpartum follow up. Care of these patients involves counseling at every step of the pregnancy to ensure that the woman is aware of the risks to her and her fetus such that she can make informed decisions.
Blood Pressure Measurement
Hypertension in pregnancy is defined as a systolic BP ≥ 140 mm Hg and a diastolic BP ≥ 90 mm Hg on two separate measurements at least 4–6 hours apart. However, the diagnosis of hypertension, in pregnancy or otherwise, requires first and foremost an accurate measurement of BP. Many automated BP cuffs have not been tested during pregnancy, and therefore obtaining a manual BP is the preferred technique. The 2000 NHBPEP Working Group Report on High BP in Pregnancy recommends that the Korotkoff phase V (disappearance) sound be used to determine the diastolic BP.1 In the outpatient setting, proper BP technique is essential and includes the subject being in a seated position, legs uncrossed, back supported, and no tobacco or caffeine for 30 minutes prior. In recumbent, hospitalized patients, the provider should measure the BP in the left lateral decubitus position to minimize the BP change caused by the compression of the inferior vena cava by the gravid uterus.
Blood pressure measurements should be interpreted in the context of the stage of pregnancy and the expected changes in blood pressure for each trimester. BP drops during the first and second trimesters, nadirs at around 20 weeks of gestation, and returns to preconception levels by the third trimester. Women who have not had regular medical care prior to pregnancy may be labeled as ‘gestational hypertension’ based on elevated BPs in the third trimester, when in reality, they were hypertensive prior to pregnancy, which was masked by the physiologic changes during mid-pregnancy. If a woman has gestational hypertension that does not resolve after delivery, she will subsequently be diagnosed as having chronic hypertension.
Ambulatory blood pressure monitoring (ABPM) and the hyperbaric index (HBI) have been suggested as alternative methods for diagnosing elevated blood pressure in pregnancy.3 The HBI is defined as the amount of BP excess during a given time period above a 90% tolerance limit, with units of mm Hg X hours. One promising study suggested that HBI calculated from a 48-hour ABPM performed in the first trimester had a 93% sensitivity and 100% specificity for predicting preeclampsia,4 although other researchers have not been able to replicate this high degree of accuracy and reliability. 5,6 There is currently no official role of ABPM in the diagnosis of hypertensive pregnancy disorders. 7 Home monitoring of blood pressure by automated cuffs in pregnancy has not been validated and some monitors have been shown to inaccurate in pregnancy and, therefore, in-office, manual BPs remain the gold-standard for the diagnosis and monitoring of hypertension in pregnancy. 8,9 This may involve frequent outpatient visits, especially in those with severe hypertension.
Blood Pressure Management in Pregnancy
Hypertension in Preeclamptic Patients
The NHBPEP Working Group Report on High BP in Pregnancy and the American College of Obstetrics and Gynecology (ACOG) guidelines recommend treatment in preeclampsia when the diastolic BP (DBP) is persistently above 105–110 mm Hg, 1 but there is no official recommendation regarding a systolic BP threshold for treatment. Most experts agree that pharmacologic therapy should be initiated when the BP approaches 150/100 mm Hg, 10 with the goal of preventing cerebral and cardiovascular events in the mother. If a woman has mild preeclampsia (DBP<100 mm Hg) with normal laboratory tests, other than low-level proteinuria, management as an outpatient can be appropriate, provided that there are frequent outpatient visits and that fetal nonstress testing (NST) is favorable. The frequency of formal ultrasound testing depends on the clinical condition and is at the discretion of an obstetrician. In the setting of severe preeclampsia that is being managed expectantly in the hospital, daily ultrasounds for fetal well-being may be indicated.
While treatment of hypertension may improve the risk profile of the mother, and therefore delay delivery, it does not cure preeclampsia, nor does it delay the progression to preeclampsia. 11 The diagnosis of severe preeclampsia includes greater than 1 of the following criteria – severe hypertension (defined as DBP>100 mm Hg), proteinuria > 5 g/24 hours or > 3+ on 2 random urine samples 4 hours apart, oliguria, cerebral or visual disturbances, pulmonary edema, epigastric or right upper quadrant pain, impaired liver function, thrombocytopenia or fetal growth restriction. The only definitive therapy for preeclampsia is delivery. When urgent control of BP is necessary, or when delivery is expected within the next 48 hours, intravenous agents, such as labetalol or hydralazine, are the drugs of choice. Oral agents may be considered if delivery is not imminent, and the choices of medications will be discussed further below.
Eclampsia may occur in the absence of gestational hypertension or preeclampsia in up to 20% of cases.12 Magnesium sulfate has been shown to decrease the risk of eclampsia and maternal death without evidence of significant harm to the mother or baby.13 Therefore, intravenous magnesium sulfate should be administered for seizure prophylaxis both during delivery and for 24 hours after delivery. The rate of continuous infusion, but not the loading dose, should be decreased for women with renal failure (as magnesium is renally excreted), and serum magnesium should be checked every 1–2 hours, as compared to women with normal renal function, for whom the level can be checked every 4–6 hours.
Timing of Delivery
The decision regarding the timing of delivery should be made after a careful assessment of the risks to the fetus and the mother. In appropriately selected patients, especially those before 32 weeks gestation, delivery can be postponed to allow for fetal maturation, particularly of the respiratory system. Women with mild preeclampsia, i. e., those with a diastolic BP < 100 mm Hg, without evidence of cerebral involvement, hemolysis with elevated liver enzymes and low platelets (HELLP) syndrome, or significant proteinuria (> 1 g/24 hours), may be candidates for this approach.
In women with severe preeclampsia before 34 weeks gestation, the timing of delivery is more complicated. In an interventional trial of 38 women with severe preeclampsia between 28–34 weeks gestation who were randomized to either aggressive therapy (betamethasone and delivery 48 hours later) or expectant management (betamethasone, and delivery only for prespecified indications, including low urine output, thrombocytopenia, abnormal liver function tests, imminent eclampsia, pulmonary edema or severe hypertension despite therapy), there was no difference in the maternal complications between the two groups. There was advanced gestational age at delivery (an addition of 7.1 vs. 1.3 days, p<0.05), with fewer neonatal complications in the expectant management group (33% vs. 75%, p<0.05).14. Another larger trial of 95 women with severe preeclampsia between 28–32 weeks gestation, randomized to aggressive versus expectant management, also showed advanced gestational age at delivery in the expectant group, with fewer visits to the neonatal intensive care unit and fewer episodes of respiratory distress in the infants.15 However, this study excluded women with underlying medical disease or obstetrical complications. A meta-analysis published in 2002 evaluating expectant versus interventional strategies in the management of women with early onset severe preeclampsia found insufficient evidence to recommend one approach over the other. 16 One approach that has been suggested by the Society for Maternal-Fetal Medicine is to admit women with early (< 34 weeks gestation), severe preeclampsia for observation and corticosteroid administration if delivery is not imminent, followed by daily laboratory tests, including liver and renal function tests, daily fetal assessments by ultrasound, and delivery at 34 weeks or earlier if any of the following develop – severe hypertension despite therapy, HELLP syndrome, pulmonary edema, eclampsia, severe renal dysfunction, disseminated intravascular coagulation, placental abruption, fetal growth restriction, oligohydramnios or abnormal fetal stress testing.17 In cases of severe hypertension (BP > 160/110 mm Hg), a trial of anti-hypertensive therapy may be undertaken, but if the BP is not reduced within 24–48 hours, delivery should be strongly considered. Given the complexity of such cases, including the risks to both mother and fetus, the decision must be made on an individual basis after thoughtful discussion with the mother. A nephrology consultation should be considered, especially in the management of severe preeclampsia, hypertension, and medication choices. In addition, a nephrologist can follow proteinuria levels and help risk-stratify patients into those who need closer follow-up for monitoring of renal disease in the post-partum setting.
If a woman is at greater than 34 weeks gestation and develops severe preeclampsia, delivery is still the treatment of choice, while expectant management may be reasonable in those with mild preeclampsia. At greater than 36–37 weeks gestation, induction of labor should pursued. Recent evidence for this approach comes from the Hypertension and Preeclampsia Intervention Trial at Term (HYPITAT) trial published in 2009 that compared the induction of labor versus expectant monitoring for gestational hypertension or mild preeclampsia after 36 weeks gestation. The HYPITAT trial was a multicenter randomized-controlled trial that included 756 women with a singleton pregnancy at 36–41 weeks, with mild gestational hypertension or mild preeclampsia. 18 The subjects were randomized to either expectant monitoring or induction of labor, with the primary outcomes being progression to severe disease, HELLP syndrome, eclampsia, pulmonary edema, placental abruption, postpartum hemorrhage, thromboembolic disease or death. There were no maternal or neonatal deaths and no cases of eclampsia or placental abruption in either group. The women randomized to the induction group had a relative risk reduction of 0.71 (95% CI 0.59–0.86) for the primary outcome, mainly due to a difference in the rate of progression to severe disease. The study was not powered to compare differences in primary outcomes between those with preeclampsia versus gestational hypertension. However, it does support the induction of labor in preeclamptic pregnancies beyond 36 weeks gestation.
Chronic Hypertension in Pregnancy
Data from the National Health and Nutrition Examination Survey (1999–2008), indicates that the prevalence of hypertension in women aged 20–44 years is 7.7%, and an estimated 4.9% of women use anti-hypertensive pharmacologic therapy, 19 with the two most common categories of medications being diuretics (47.9%) and angiotension-converting enzyme (ACE) inhibitors (44.0%). Prenatal counseling in women with chronic hypertension is an important component of their care. The goals of the visit should be to evaluate for end-organ damage, to adjust medications as necessary, to discuss appropriate lifestyle modifications, and to consider secondary causes of hypertension. Given the teratogenic potential of ACE inhibitors (see below), women should be counseled on the importance of birth control while on ACE inhibitors, and this class of medication should be changed prior to attempting to conceive.
There is continued controversy over whether women with mild-moderate chronic hypertension on anti-hypertensive therapy prior to pregnancy should stop these medications, be kept under close observation, and only reinstitute therapy for blood pressure elevations of 140–160/90–100 mm Hg, versus continuing their usual anti-hypertensive regimen.1 Currently, both approaches are used and there is insufficient evidence to recommend one approach over the other. Central to this controversy is the concern that anti-hypertensive therapy for mild-moderate hypertension may provide no maternal benefits, while, at the same time, harming the fetus (a) from the intrauterine exposure to anti-hypertensive medications with their possible adverse effects, and (b) as a result of unfavorable hemodynamics, whereby a decrease in the maternal blood pressure may impair uteroplacental perfusion, and, ultimately, fetal growth. The evidence for an association between fetoplacental growth and the use of oral anti-hypertensive medications is limited. In the absence of well-designed and adequately powered studies, a meta-analysis of 14 treatment trials is frequently cited. 20 This meta-analysis concluded that a 10 mm Hg decrease in mean arterial blood pressure was associated with a 145 gram decrease in neonatal birth weight. However, only 16% of the variation in birth weight was accounted for by the change in blood pressure, as several other variables may have contributed independently to the fetal growth impairment. These negative effects of anti-hypertensive therapy on birth weight have not been demonstrated consistently 21 and, in turn, the presence of hypertension itself, irrespective of blood pressure medication use, has been shown to result in lower birth weights. Those in favor of treatment support their view by citing evidence that treatment of chronic hypertension may prevent progression to severe hypertension and that chronic hypertension has been associated with increased feto-maternal risks, including perinatal mortality and placental abruption 22. In general, most experts agree with the use of anti-hypertensive therapy for a blood pressure ≥ 150/100 mm Hg. 10 Evaluation for end-organ damage is important in that it helps to guide therapy decisions. In the presence of renal disease, proteinuria, left ventricular hypertrophy, hypertensive retinopathy, microvascular disease, stroke and age greater than 40, therapy should be initiated for a diastolic BP ≥ 90 mm Hg. 1,23 It should be noted that therapeutic guidelines as to when to institute treatment and subsequent BP targets vary among different obstetrical and medical societies due to the lack of evidence supporting a single BP goal. 24,25
Secondary hypertension should be considered in women with difficult to control BP requiring three or more anti-hypertensive agents, including a diuretic, and/or indicative laboratory and clinical findings, such as hypokalemia or an abdominal bruit. A thorough history and physical exam should be performed by a nephrologist or hypertension specialist and should include a review of lifestyle factors, such as nicotine use, caffeine intake and recreational drug use, in particular cocaine and methamphetamines, which can mimic conditions such as pheochromocytoma. The primary differential for secondary causes of hypertension includes primary hyperaldosteronism, pheochromocytoma and renal artery stenosis (primarily due to fibromuscular dysplasia in this age group). Women with secondary hypertension are at a particularly high risk for adverse pregnancy outcomes. 26 Pheochromocytoma, in particular, is associated with significant maternal and fetal mortality, mainly due to hypertensive crisis in the mother and intrauterine growth restriction in the fetus.27 The diagnosis may be missed, mainly due to the fact that patients may present with atypical features, such as gestational diabetes and difficult to control hypertension (Figure 1), and may be inappropriately diagnosed as having preeclampsia. 28 Primary hyperaldosteronism is also difficult to diagnose during pregnancy, mainly due to the stimulation of the renin-angiotensin-aldosterone axis in normal pregnancy, resulting in false negatives when measuring the renin-aldosterone ratio. 29
Figure 1.
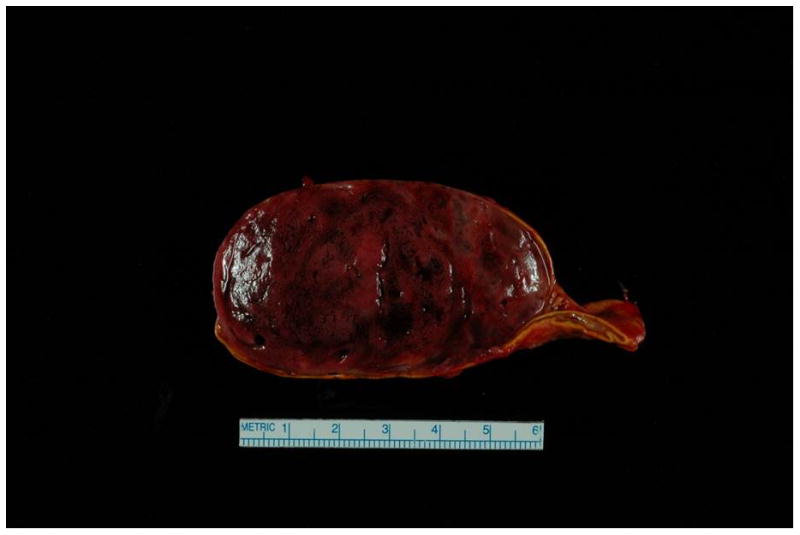
Surgical specimen of the adrenal pheochromocytoma.
Case presentation - Patient was a 31 year old P6G5 with a history of chronic hypertension requiring medications intermittently in the past, and a history of preeclampsia with her first pregnancy, with subsequent unremarkable pregnancies. She was admitted at 34 weeks due to systolic BP > 200 mm Hg, as well as 3+ protein on dipstick, elevated AST (57 U/L), serum creatinine of 1.2 mg/dL and uric acid of 10.1 mg/dL. The constellation of clinical findings and laboratory abnormalities was considered to be consistent with preeclampsia. She received IV magnesium sulfate and labetalol, and underwent C-section at 34 and 4/7 weeks, and gave birth to a healthy male infant. Systolic BPs remained in the 180s mm Hg post-delivery, and she was transferred to the ICU for hypertensive urgency. Work-up for secondary causes revealed a 3.2 × 6.2 × 5.3 cm heterogenous lesion in the right adrenal gland, and MRI was consistent with pheochromocytoma. The level of plasma free normetanephrine was 26.2 nmol/L (normal < 0.90 nmol/L), and the 24-hour urine showed significantly elevated levels of norepinephrine and normetanephrine. She received phenoxybenzamine, nicardipine and propranolol. She underwent a right total adrenalectomy (photograph of mass shown) and was able to be tapered off her BP medications.
Specific interventions to treat the underlying cause should be pursued, when possible, prior to pregnancy, whether it is surgery for pheochromocytoma or revascularization for renal artery stenosis, which may result in cure of the hypertension. In the case that the diagnosis is not made before pregnancy, there are no official guidelines as to the management of these conditions, and decisions must be made on a case-by-case basis. Renal artery revascularization has been successfully used during pregnancy, 26 and surgical resection of pheochromocytoma has been performed in all trimesters, after the initiation of 10–14 days of alpha blockage prior to surgery. 28
Lastly, women with chronic hypertension should be aware of the risks associated with pregnancy. In a study of 763 women with chronic hypertension enrolled in a multi-center trial of low-dose aspirin for the prevention of preeclampsia, 25% developed superimposed preeclampsia. 30 In addition, the women in this study who developed preeclampsia were at increased risk for placental abruption, versus those with chronic hypertension alone (3% vs. 1%, p=0.04). 30 Among women with severe or high risk hypertension (BP>160/110), placental abruption may affect as many as 5–10% of women.11 One study by Sibai and colleagues looked at the maternal and fetal outcomes of 44 women with severe chronic hypertension in the first trimester of pregnancy and found that while there were no maternal deaths, morbidity, including hospitalizations and reduction in renal function, was high. 11 Of the twenty women with deterioration in renal function during pregnancy, 19 had returned to their baseline renal function by their post-partum visit, although one patient continued to have significant renal impairment. Nearly one-half of these women developed superimposed preeclampsia, and the perinatal mortality was 25% (ten stillbirths and 1 neonatal death), predominantly in the group of women with preeclampsia, and all in infants less than 800 grams at delivery and less than 29 weeks of age. The risk of recurrence of preeclampsia in subsequent pregnancies after a pregnancy complicated by severe preeclampsia is 25%, but the relative risk is 2 times greater in those with a history of chronic hypertension. 31
Complications of hypertensive pregnancy
The most significant short term complications of hypertensive pregnancy in the mother are cerebrovascular complications, including cerebral hemorrhage and seizures, renal impairment and cardiovascular complications, such as pulmonary edema (Table 2). Women with chronic hypertension and evidence of end-organ damage prior to pregnancy are at increased risk of pulmonary edema, hypertensive encephalopathy, retinopathy, cerebral hemorrhage and acute renal failure. 1
Table 2.
Possible acute complications of hypertensive pregnancy by organ system
| Organ System | Clinical Signs and Symptoms |
|---|---|
| Cardiovascular | Hypertension Pulmonary Edema Reduced Cardiac Output |
| Renal | Reduced Glomerular Filtration Rate Proteinuria Cortical Necrosis |
| Hepatic (HELLP syndrome) | Elevated Liver Enzymes Hepatic Dysfunction Subcapsular Bleeding Hepatic Rupture |
| Central Nervous System | Cerebral hemorrhage Headache/blurred vision Scotoma Cortical Blindness Posterior Reversible Encephalopathy Syndrome (PRES) Seizure |
HELLP syndrome occurs in 10–20% of severe preeclamptic/eclamptic pregnancies.32,33 The syndrome was first described in 1982 by Weinstein and colleagues34 as being characterized by hemolysis, low platelets and elevated liver enzymes. It is associated with significant maternal morbidity and mortality. In a prospective study of 442 patients with HELLP syndrome, there were 5 maternal deaths, 3 attributed to diffuse hypoxic encephalopathy, and significant morbidity – including placental abruption (16%), acute renal failure (7.7%), pulmonary edema (6%) and subcapsular hematoma (0.9%). 32 Hypertension and proteinuria may not be present,32 and so providers must be alert to the deceptive signs and symptoms of HELLP syndrome – including midepigastric or right upper quadrant pain, nausea, vomiting and general malaise. Women with HELLP syndrome should typically be delivered as soon as possible due to the significant morbidity and mortality. They are not candidates for expectant management. 35,36 HELLP syndrome must be differentiated from hemolytic uremic syndrome (HUS) and thrombotic thrombocytopenia purpura (TTP), two forms of thrombotic microangiopathy that may present during pregnancy. 33 TTP and HUS, either typical or atypical, are not characterized by elevated liver enzymes and, in addition, TTP is associated with deficiency in the enzyme ADAMTS13, a metalloproteinase that degrades von Willebrand factor multimers.
Eclampsia is the convulsive form of preeclampsia and occurs in 0.5% of patients with mild preeclampsia, and in 2–3% of those with severe preeclampsia.37 The most feared complication and cause of maternal death in eclampsia is stroke. 38 In a population-based study in France in 1995, 31 cases of stroke in pregnancy were identified and eclampsia accounted for nearly half of both hemorrhagic and non-hemorrhagic strokes. 39 A more recent study by Martin and colleagues looked at 28 women who had suffered strokes in the setting of severe preeclampsia and eclampsia 40 and found that their systolic blood pressures were > 155 mm Hg immediately before their cerebrovascular accidents. Only 5 patients reached a diastolic blood pressure of 105 mm Hg, and therefore would not necessarily be candidates for treatment according to the current NHBPEP and ACOG guidelines. 1 In this study, maternal mortality was 53.6%, and only 3 of the 28 patients had no residual deficits post-stroke. The authors concluded that a paradigm shift is needed toward treatment of SBP of 155–160 mm Hg in severe preeclamptic and eclamptic patients.
Posterior reversible encephalopathy syndrome (PRES) was first described in the setting of eclampsia in 1996. 41 PRES is a syndrome characterized by neurologic signs and symptoms, such as headache, impaired consciousness, visual changes and seizures, in combination with neuroimaging findings of vasogenic edema in the posterior circulation. In a retrospective study of patients with eclampsia at Mayo Clinic, Rochester, 13 cases of eclampsia were identified, of which 7 had neuroimaging studies, and all of the women had characteristic findings of PRES syndrome. Importantly, pregnant patients developed PRES at lower peak systolic BPs (mean 173 mm Hg) than the non-pregnant PRES subjects. 42 The authors hypothesize that the reduction in BP in these women may delay progression of vasogenic to cytotoxic edema and cerebral infarction. The true incidence of PRES in eclampsia, or even in severe preeclampsia, has not been established, but the association is one that requires future study.
Long-term complications
It was long believed that if hypertension and proteinuria resolve after delivery, there would not be any long term risk of cardiovascular or renal complications in the mother. However, recent studies have shown that a history of hypertensive pregnancy is a risk factor for hypertension, 43,44 as well as cardiovascular disease, 44,45 with an approximate relative risk of 2, compared to those without hypertensive pregnancies. The risk of future kidney disease is not clear, although several studies have suggested an association. It has been shown that a history of preeclampsia is associated with a higher frequency of microalbuminuria up to 5 years after delivery. 46 A study using registry data from Norway found that preeclampsia occurring during a first pregnancy increased the risk of future end-stage renal disease (ESRD). 47 The study population consisted of women with a first singleton birth between 1967 and1991, and follow-up extended until December 2005. The absolute risk of ESRD after any preeclamptic pregnancy was low, 14.5/100,000 person-years, but the adjusted relative risk was 4.3 (95% CI 3.3–5.6), and in women with greater than 2 preeclamptic pregnancies, the adjusted relative risk increased to 10.9 (95% CI 5.0–23.8). It should be noted that as this was a registry study, and patients with renal disease prior to pregnancy were not able to be excluded, which would certainly impact the associated risk. As to the mechanisms that underlie this association, it is not clear whether it is the hypertensive pregnancy itself that leads to the increased risk of these complications, or whether there is underlying endothelial dysfunction in the mother that manifests differently at varying stages of life. Either way, women with a history of preeclampsia should be advised regarding life style modifications and should be monitored closely for traditional risk factors.
Treatment of hypertensive pregnancy disorders
Non-pharmacological therapeutic approach
Lifestyle interventions, such as weight loss and a reduction in salt intake, are of proven benefit in non-pregnant hypertensive patients. There is currently no evidence from prospective, randomized trials that instituting an exercise program during pregnancy is effective in preventing preeclampsia in at risk individuals,1,48, although some benefit has been seen in an animal model.49 Similarly, there is no current evidence that instituting a weight loss program in pregnancy can prevent preeclampsia,48 although obesity is a risk factor for gestational hypertension and preeclampsia.50 In 2009, the Institute of Medicine revised their guidelines for gestational weight gain and recommended that women who are overweight prior to pregnancy (body-mass index (BMI) 25–29.9) gain only 15–25 lbs during pregnancy, as opposed those with normal weight (BMI 18.5–24.9) prior to pregnancy, who should gain 25–35 lbs.51 Obese women with a BMI > 30 should gain only 11–20 lbs, according to the new recommendations.
As volume contraction is common in preeclampsia, salt restriction is not routinely recommended. However, bed rest is frequently advised, and has been shown to lower blood pressure, promote diuresis, and reduce premature labor 52,53.
Medications
Pharmacologic therapy during pregnancy may prevent progression to severe hypertension and maternal complications (such as heart failure and cerebrovascular events) while improving fetal maturity by permitting prolongation of pregnancy. During both induction and titration of anti-hypertensive medications, fetal well-being and safety should be closely monitored by several methods that are available in daily clinical practice. A discussion of the most common classes of anti-hypertensive therapy is presented below and summarized in Table 3.
Table 3.
Medication choices for pharmacologic treatment of hypertension in pregnancy
| Benefits | Risks | U.S. Federal Drug Administration Pregnancy Risk Category* | ||
|---|---|---|---|---|
| Central agents | ||||
| Preferred | Methyldopa | Proven safety and efficacy | Neuro-depressant side effects | B/C in injectable form |
| Alternative | Clonidine | Efficacy similar to methyldopa | Unproven safety | C |
| Beta blockers | ||||
| Preferred | Labetalol | Safety and efficacy similar to methyldopa. May be used for hypertensive urgency. | Fetal bradycardia, neonatal hypoglycemia, decreased uteroplacental flow | C |
| Contraindicated | Atenolol | None compared to Labetalol | Intra-uterine growth retardation | D |
| Calcium channel blockers | ||||
| Preferred | Nifedipine | Lowers BP without affecting umbilical artery flow | Fetal distress, profound hypotension with magnesium | C |
| Alternative | Verapamil | Similar efficacy to other oral agents | Untested safety profile, risk of interaction with magnesium | C |
| Direct vasodilators | ||||
| Preferred | Hydralazine | Most efficacious oral agent | Maternal neuropathy, drug-induced lupus, neonatal thrombocytopenia and lupus | C |
| Alternative | Nitroprusside | Effective in severe hypertension | Cyanide and thiocyanate toxicity | C |
| Diuretics | ||||
| Preferred | Thiazide | Useful in chronic hypertension, renal failure, congestive heart failure | Volume contraction, electrolyte abnormalities | B |
| Contraindicated | Spironolactone | None | Possible fetal anti-androgen effects | C |
| RAAS blockade | ||||
| Contraindicated | ACE inhibitors/ARBs | None | Associated with congenital heart and kidney defects | D |
| Contraindicated | Aliskerin | None | Oligohydramnios and other defects associated with RAAS blockade in the fetus | D |
A – Controlled human studies show no risk, B- No evidence of risk in studies, C- Risk cannot be ruled out, D- Positive evidence of risk
α-adrenergic agonists
One of the medications with the longest track record in pregnancy is methyldopa. A long-term follow-up study on children born to women treated with methyldopa during pregnancy could find no increased incidence of general health problems or cognitive problems. 54 This record of safety makes it the first line agent recommended by the NHBPEP working group. 1 Methyldopa acts centrally by decreasing sympathetic tone, and therefore can have many side effects, including sedation and impaired sleep patterns. One potential side effect is that it may cause mild elevations of liver enzymes, which can lead to diagnostic confusion with HELLP syndrome. Although it is relatively safe, methyldopa is not a potent BP lowering agent and side effects, which are commensurate with the dose, can limit its use. Methyldopa can be combined with other anti-hypertensives, such as a diuretic (discussed below), to achieve target blood pressure values.
Clonidine has a similar mode of action to methyldopa, but has a much stronger effect in lowering BP. Clonidine may impair fetal growth, especially if the mother has a reduction in heart rate after therapy is initiated. 21 It can cause significant rebound hypertension and does not have as strong a record of safety as methyldopa. It should be considered in cases of intolerance to methyldopa.
Beta-blockers
Beta-blockers are generally well-tolerated and safe in pregnancy. Labetalol is becoming one of the favored therapies for hypertension in pregnancy. It is a non-selective beta blocker that antagonizes both beta and alpha-1 receptors. Its side effects include fatigue, decreased exercise tolerance, as well as bronchospasm in individuals with reactive airway disease. Labetalol has been compared to methyldopa in prospective trials and neither medication was associated with adverse maternal or fetal outcomes. 25,55 It is available in both oral and intravenous forms, so it may be used for both outpatient and inpatient management.
Atenolol has been shown to have minimal effects on systolic BP in preeclamptic women, and it is also associated with intrauterine growth retardation.56 Given the availability of other more effective medications, including labetalol, atenolol should be avoided in pregnancy.
Calcium channel blockers
In a small trial of preeclamptic mothers who received nifedipine versus placebo, there were significant reductions in maternal BP, serum creatinine and urea values, and 24-hour urinary protein measurements, without a reduction in umbilical artery blood flow. 57 According to a prospective, multicenter cohort study of 78 women exposed to calcium channel blockers in the first trimester, mainly nifedipine and verapamil, there was no increase in major congenital malformations. There was an increase in preterm delivery in those who received calcium channel blockers versus controls, matched for age and smoking status (28% vs. 9%, p=0.003), although this was attributed to underlying maternal disease by stepwise regression.58 There is little data available on diltiazem, although it may be used as a rate control agent in pregnancy,59 and has been shown to lower BP and proteinuria in pregnant patients with underlying renal disease in a small study of 7 patients.60 Calcium channel blockers are also potent tocolytics and can affect the progression of labor. A major concern with calcium channel blockers in pregnancy is the concurrent use of magnesium sulfate for seizure prophylaxis, as co-administration of these agents has been reported to cause circulatory collapse and neuromuscular blockade.16 Despite these issues, calcium channel blockers, nifedipine in particular, as there is more data available for it, is an effective and safe alternative to methyldopa as a first-line agent for the treatment of hypertension in pregnancy.
Diuretics
As noted above, diuretics are the most commonly used medication among women of child-bearing age with chronic hypertension. 19 A possible side effect of any diuretic is vascular volume contraction, which may paradoxically cause further elevations of BP in preeclamptic women. Women with preeclampsia have lower plasma volumes compared to those with normal pregnancies, and volume contraction may stimulate the renin-angiotensin-aldosterone axis, causing further increases in peripheral vascular resistance, thus worsening hypertension. 24,61 However, the 2000 NHBPEP Working Group report on High BP in Pregnancy recognized that the major concern for diuretic use in pregnancy is mainly theoretical, as supporting evidence for their adverse effects is lacking. Therefore, if a woman is on a diuretic prior to pregnancy, this can be continued during pregnancy, with the exception of spironolactone, which may have fetal anti-androgen effects.
Hydralazine
Hydralazine is a direct vasodilator that can be administered orally or intravenously, and is often used in the setting of hypertensive urgency, given its fast onset of action. However, hydralazine has been associated with hypotension, oliguria and fetal distress. 62 It is also associated with a lupus-like syndrome and peripheral neuropathy. The lupus-like syndrome is usually seen with higher oral daily doses (> 200 mg daily)63, though has been seen with doses as low as 50 mg daily when the exposure is prolonged (months to years)64. Due to these potential side effects, labetalol is increasingly viewed as a safer agent for inpatient management of hypertension. Hydralazine remains, however, commonly used when other treatment regimens have failed to achieve adequate BP control, as most obstetricians are quite familiar with its pharmacological actions and find its side-effect profile acceptable.
Renin-Angiotensin-Aldosterone System Blockade
Renin-Angiotensin-Aldosterone System (RAAS) blockers, including most commonly ACE inhibitors and ARBs, are extremely effective in lowering BP and have significant benefit in proteinuric diseases. Initially, these medications were considered to be relatively safe during the first trimester, and only associated with defects when taken in the second trimester, potentially leading to oligohydramnios, anuria, and fetal renal failure. However, a study published in 2006 by Cooper and colleagues demonstrated that exposure to ACE inhibitors in the first trimester can be associated with significant congenital malformations affecting both the cardiovascular and central nervous systems. 65 The relative risk of major congenital malformations after a first trimester exposure was 2.71 (95% CI 1.72–4.27) as compared to infants with no exposure to anti-hypertensives. A 2011 study of over 400,000 women-infant pairs in the Northern California Kaiser Permanente region did find an increased risk of congenital heart defects in the offspring after exposure to ACE inhibitors in the first trimester as compared to healthy controls (OR 1.54, CI 0.9–2.62), but not as compared to women on other anti-hypertensive agents (OR 1.14, CI 0.65–1.98).66 However, ACE inhibitors are still considered contraindicated in pregnancy, despite the controversy regarding the magnitude of the risk associated with exposure. If a woman becomes pregnant while on one of these agents, she should be switched immediately to an alternative therapy and offered an ultrasound and fetal echocardiography at 18 weeks gestation, and providers should discuss the possible risks for congenital malformations with the patient. There is little data regarding direct renin inhibitors, such as aliskerin, in pregnancy, but they are also considered category D by the US Federal Drug Administration as they block the RAAS system (see Table 3). As mentioned previously, spironolactone is contraindicated in pregnancy as it can cross the placenta and have anti-androgen effects on the fetus. There is a case report of eplerenone being used successfully for blood pressure control in a women with primary hyperaldosteronism during pregnancy.67
Future Directions
Over the last several years, evidence has emerged suggesting that women with hypertensive pregnancy disorders are at an increased risk for both immediate and long-term cardiovascular disease. On the other hand, current recommendations for the treatment of hypertension in pregnancy, unlike those for the general population, have remained the same for many years, mainly due to a lack of studies that address the safety and benefits of lowering BP in pregnancy. At present, several interventional studies for the treatment of hypertension in pregnancy are underway (ClinicalTrials.gov – NCT01192412, NCT01351428, NCT00293735, NCT00194974, NCT01361425) that may provide long needed information, with respect to therapeutic targets and preferred agents in pregnant hypertensive patients. 24 While awaiting the results of these trials, a recent editorial by Moser and colleagues suggested using a new approach to the treatment of hypertension in pregnancy, whereby women with a history of chronic hypertension should continue their anti-hypertensive medications throughout pregnancy, provided that they are safe, with careful monitoring of BP and dose adjustments, as necessary. 24 The authors suggest that a new target BP in previously normotensive women should be 140/90 mm Hg, with beta-blockers, diuretics or calcium-channel blockers as first line therapies. The emphases of this proposal are to simplify the classification of hypertensive pregnancy disorders into chronic hypertension and de novo hypertension after 20 weeks, simplify the target BP goal to less than 140/90 mm Hg, and recommend specific pharmacologic agents. The authors suggest that the currently available methods for monitoring fetal well-being and safety should be instrumental in this approach, and applied during both the introduction and titration of anti-hypertensive medications. The studies currently underway may help address what the appropriate targets for BP should be in pregnancy, and which anti-hypertensive agents are most effective.
Current guidelines for the prevention of cardiovascular disease in women recommend referral of patients with a history of hypertensive pregnancy to primary care or cardiology, in order to facilitate monitoring and control of risk factors, 68 and though there are currently no guidelines, routine screening for renal disease with blood urea nitrogen, serum creatinine and urinalysis may be indicated in affected women. Future research should address the question as to whether more aggressive BP control during hypertensive pregnancies may result in the reduction of the future cardiovascular disease outcomes and renal disease in the affected women.
Conclusion
Hypertension in pregnancy is a common complication of pregnancy and one associated with significant maternal and fetal morbidity and mortality. The central issue in the management of hypertension in pregnancy is achieving a balance between the maternal benefits derived from improved BP control, and the fetal risks resulting from intrauterine medication toxicity and possible uteroplacental hypoperfusion. For the severe forms of hypertensive pregnancy disorders, including eclampsia, severe preeclampsia and HELLP syndrome, delivery remains the standard of care. Women with mild preeclampsia prior to 32 weeks gestation may be candidates for expectant management, but after 37 weeks, current evidence supports induction of labor to prevent adverse maternal and fetal outcomes. Women with chronic hypertension should undergo a pre-pregnancy evaluation, with a focus on end-organ damage, medication profile, potential secondary causes of hypertension, and counseling on the risks of pregnancy, including the development of superimposed preeclampsia. Women must be followed carefully during pregnancy and in the intra- and post-partum settings. There is ongoing research focusing on the appropriate management of hypertension in pregnancy and the long-term consequences for the mother that may influence future recommendations in this field.
Clinical Summary.
Hypertensive pregnancy disorders complicate 6–8% of pregnancies in the United States.
The strategy for the treatment of hypertension in pregnancy is to prevent maternal cerebrovascular and cardiac complications, while preserving uteroplacental blood flow and limiting medication toxicity to the fetus.
The definitive therapy for acute hypertensive syndromes of pregnancy is delivery, although anti-hypertensive medications are often necessary to lower the blood pressure in the mother to a) prevent maternal complications, and b) decrease neonatal complications related to prematurity by allowing for the continuation of pregnancy
Women with chronic hypertension should have a prenatal visit that includes an evaluation for secondary causes of hypertension, medication adjustments, and counseling regarding the high risk of developing preeclampsia.
Acknowledgments
Grant support - The project described was supported by Award Number K08 HD051714 (Vesna D. Garovic) from the Eunice Kennedy Shriver National Institute of Child Health & Human Development and by Award Number P-50 AG44170 (Vesna D. Garovic) from the National Institute on Aging. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. The writing of the manuscript and the decision to submit it for publication were solely the authors’ responsibilities.
Footnotes
Financial disclosures – The authors have no relevant financial disclosures.
References
- 1.Report of the National High Blood Pressure Education Program Working Group on High Blood Pressure in Pregnancy. Am J Obstet Gynecol. 2000 Jul;183(1):S1–S22. [PubMed] [Google Scholar]
- 2.Berg CJ, Callaghan WM, Syverson C, Henderson Z. Pregnancy-related mortality in the United States, 1998 to 2005. Obstet Gynecol. 2010 Dec;116(6):1302–1309. doi: 10.1097/AOG.0b013e3181fdfb11. [DOI] [PubMed] [Google Scholar]
- 3.Ayala DE, Hermida RC. Ambulatory Blood Pressure Monitoring for the Early Identification of Hypertension in Pregnancy. Chronobiol Int. 2012 Sep 24; doi: 10.3109/07420528.2012.714687. [DOI] [PubMed] [Google Scholar]
- 4.Hermida RC, Ayala DE, Mojon A, et al. Blood pressure excess for the early identification of gestational hypertension and preeclampsia. Hypertension. 1998 Jan;31(1):83–89. doi: 10.1161/01.hyp.31.1.83. [DOI] [PubMed] [Google Scholar]
- 5.Vollebregt KC, Gisolf J, Guelen I, Boer K, van Montfrans G, Wolf H. Limited accuracy of the hyperbaric index, ambulatory blood pressure and sphygmomanometry measurements in predicting gestational hypertension and preeclampsia. J Hypertens. 2010 Jan;28(1):127–134. doi: 10.1097/HJH.0b013e32833266fc. [DOI] [PubMed] [Google Scholar]
- 6.Brown MA, Bowyer L, McHugh L, Davis GK, Mangos GJ, Jones M. Twenty-four-hour automated blood pressure monitoring as a predictor of preeclampsia. Am J Obstet Gynecol. 2001 Sep;185(3):618–622. doi: 10.1067/mob.2001.117664. [DOI] [PubMed] [Google Scholar]
- 7.Bergel E, Carroli G, Althabe F. Ambulatory versus conventional methods for monitoring blood pressure during pregnancy. Cochrane Database Syst Rev. 2002;(2):CD001231. doi: 10.1002/14651858.CD001231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Natarajan P, Shennan AH, Penny J, Halligan AW, de Swiet M, Anthony J. Comparison of auscultatory and oscillometric automated blood pressure monitors in the setting of preeclampsia. Am J Obstet Gynecol. 1999 Nov;181(5 Pt 1):1203–1210. doi: 10.1016/s0002-9378(99)70109-2. [DOI] [PubMed] [Google Scholar]
- 9.Lo C, Taylor RS, Gamble G, McCowan L, North RA. Use of automated home blood pressure monitoring in pregnancy: is it safe? Am J Obstet Gynecol. 2002 Nov;187(5):1321–1328. doi: 10.1067/mob.2002.126847. [DOI] [PubMed] [Google Scholar]
- 10.August P. Preeclampsia: New Thoughts on an Ancient Problem. J Clin Hypertens (Greenwich) 2000 Mar;2(2):115–123. [PubMed] [Google Scholar]
- 11.Sibai BM, Anderson GD. Pregnancy outcome of intensive therapy in severe hypertension in first trimester. Obstet Gynecol. 1986 Apr;67(4):517–522. [PubMed] [Google Scholar]
- 12.Noraihan MN, Sharda P, Jammal AB. Report of 50 cases of eclampsia. J Obstet Gynaecol Res. 2005 Aug;31(4):302–309. doi: 10.1111/j.1447-0756.2005.00292.x. [DOI] [PubMed] [Google Scholar]
- 13.Altman D, Carroli G, Duley L, et al. Do women with pre-eclampsia, and their babies, benefit from magnesium sulphate? The Magpie Trial: a randomised placebo-controlled trial. Lancet. 2002 Jun 1;359(9321):1877–1890. doi: 10.1016/s0140-6736(02)08778-0. [DOI] [PubMed] [Google Scholar]
- 14.Odendaal HJ, Pattinson RC, Bam R, Grove D, Kotze TJ. Aggressive or expectant management for patients with severe preeclampsia between 28–34 weeks’ gestation: a randomized controlled trial. Obstet Gynecol. 1990 Dec;76(6):1070–1075. [PubMed] [Google Scholar]
- 15.Sibai BM, Mercer BM, Schiff E, Friedman SA. Aggressive versus expectant management of severe preeclampsia at 28 to 32 weeks’ gestation: a randomized controlled trial. Am J Obstet Gynecol. 1994 Sep;171(3):818–822. doi: 10.1016/0002-9378(94)90104-x. [DOI] [PubMed] [Google Scholar]
- 16.Churchill D, Duley L. Interventionist versus expectant care for severe pre-eclampsia before term. Cochrane Database Syst Rev. 2002;(3):CD003106. doi: 10.1002/14651858.CD003106. [DOI] [PubMed] [Google Scholar]
- 17.Sibai BM. Evaluation and management of severe preeclampsia before 34 weeks’ gestation. Am J Obstet Gynecol. 2011 Sep;205(3):191–198. doi: 10.1016/j.ajog.2011.07.017. [DOI] [PubMed] [Google Scholar]
- 18.Koopmans CM, Bijlenga D, Groen H, et al. Induction of labour versus expectant monitoring for gestational hypertension or mild pre-eclampsia after 36 weeks’ gestation (HYPITAT): a multicentre, open-label randomised controlled trial. Lancet. 2009 Sep 19;374(9694):979–988. doi: 10.1016/S0140-6736(09)60736-4. [DOI] [PubMed] [Google Scholar]
- 19.Bateman BT, Shaw KM, Kuklina EV, Callaghan WM, Seely EW, Hernandez-Diaz S. Hypertension in women of reproductive age in the United States: NHANES 1999–2008. PLoS One. 2012;7(4):e36171. doi: 10.1371/journal.pone.0036171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.von Dadelszen P, Ornstein MP, Bull SB, Logan AG, Koren G, Magee LA. Fall in mean arterial pressure and fetal growth restriction in pregnancy hypertension: a meta-analysis. Lancet. 2000 Jan 8;355(9198):87–92. doi: 10.1016/s0140-6736(98)08049-0. [DOI] [PubMed] [Google Scholar]
- 21.Rothberger S, Carr D, Brateng D, Hebert M, Easterling TR. Pharmacodynamics of clonidine therapy in pregnancy: a heterogeneous maternal response impacts fetal growth. Am J Hypertens. 2010 Nov;23(11):1234–1240. doi: 10.1038/ajh.2010.159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Abalos E, Duley L, Steyn DW, Henderson-Smart DJ. Antihypertensive drug therapy for mild to moderate hypertension during pregnancy. Cochrane Database Syst Rev. 2001;(2):CD002252. doi: 10.1002/14651858.CD002252. [DOI] [PubMed] [Google Scholar]
- 23.Magee LA, Helewa M, Moutquin JM, von Dadelszen P. Diagnosis, evaluation, and management of the hypertensive disorders of pregnancy. J Obstet Gynaecol Can. 2008 Mar;30(3 Suppl):S1–48. [Google Scholar]
- 24.Moser M, Brown CM, Rose CH, Garovic VD. Hypertension in pregnancy: is it time for a new approach to treatment? J Hypertens. 2012 Jun;30(6):1092–1100. doi: 10.1097/HJH.0b013e3283536319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ferrer RL, Sibai BM, Mulrow CD, Chiquette E, Stevens KR, Cornell J. Management of mild chronic hypertension during pregnancy: a review. Obstet Gynecol. 2000 Nov;96(5 Pt 2):849–860. doi: 10.1016/s0029-7844(00)00938-8. [DOI] [PubMed] [Google Scholar]
- 26.Thorsteinsdottir B, Kane GC, Hogan MJ, Watson WJ, Grande JP, Garovic VD. Adverse outcomes of renovascular hypertension during pregnancy. Nat Clin Pract Nephrol. 2006 Nov;2(11):651–656. doi: 10.1038/ncpneph0310. [DOI] [PubMed] [Google Scholar]
- 27.Smith CM, Wigent PJ. Pheochromocytoma in pregnancy: considerations for the advanced practice nurse. J Perinat Neonatal Nurs. 1998 Sep;12(2):11–25. doi: 10.1097/00005237-199809000-00003. [DOI] [PubMed] [Google Scholar]
- 28.Oliva R, Angelos P, Kaplan E, Bakris G. Pheochromocytoma in pregnancy: a case series and review. Hypertension. 2010 Mar;55(3):600–606. doi: 10.1161/HYPERTENSIONAHA.109.147579. [DOI] [PubMed] [Google Scholar]
- 29.Stowasser M, Ahmed AH, Pimenta E, Taylor PJ, Gordon RD. Factors affecting the aldosterone/renin ratio. Horm Metab Res. 2012 Mar;44(3):170–176. doi: 10.1055/s-0031-1295460. [DOI] [PubMed] [Google Scholar]
- 30.Sibai BM, Lindheimer M, Hauth J, et al. Risk factors for preeclampsia, abruptio placentae, and adverse neonatal outcomes among women with chronic hypertension. National Institute of Child Health and Human Development Network of Maternal-Fetal Medicine Units. N Engl J Med. 1998 Sep 3;339(10):667–671. doi: 10.1056/NEJM199809033391004. [DOI] [PubMed] [Google Scholar]
- 31.van Rijn BB, Hoeks LB, Bots ML, Franx A, Bruinse HW. Outcomes of subsequent pregnancy after first pregnancy with early-onset preeclampsia. Am J Obstet Gynecol. 2006 Sep;195(3):723–728. doi: 10.1016/j.ajog.2006.06.044. [DOI] [PubMed] [Google Scholar]
- 32.Sibai BM, Ramadan MK, Usta I, Salama M, Mercer BM, Friedman SA. Maternal morbidity and mortality in 442 pregnancies with hemolysis, elevated liver enzymes, and low platelets (HELLP syndrome) Am J Obstet Gynecol. 1993 Oct;169(4):1000–1006. doi: 10.1016/0002-9378(93)90043-i. [DOI] [PubMed] [Google Scholar]
- 33.Nwoko R, Plecas D, Garovic VD. Acute kidney injury in the pregnant patient. Clin Nephrol. 2012 Dec;78(6):478–486. doi: 10.5414/cn107323. [DOI] [PubMed] [Google Scholar]
- 34.Weinstein L. Syndrome of hemolysis, elevated liver enzymes, and low platelet count: a severe consequence of hypertension in pregnancy. Am J Obstet Gynecol. 1982 Jan 15;142(2):159–167. doi: 10.1016/s0002-9378(16)32330-4. [DOI] [PubMed] [Google Scholar]
- 35.Sibai BM, Barton JR. Expectant management of severe preeclampsia remote from term: patient selection, treatment, and delivery indications. Am J Obstet Gynecol. 2007 Jun;196(6):514, e511–519. doi: 10.1016/j.ajog.2007.02.021. [DOI] [PubMed] [Google Scholar]
- 36.ACOG technical bulletin Hypertension in pregnancy. Number 219--January 1996 (replaces no. 91, February 1986) Committee on Technical Bulletins of the American College of Obstetricians and Gynecologists. Int J Gynaecol Obstet. 1996 May;53(2):175–183. [PubMed] [Google Scholar]
- 37.Lindheimer MD, Taler SJ, Cunningham FG. ASH position paper: hypertension in pregnancy. J Clin Hypertens (Greenwich) 2009 Apr;11(4):214–225. doi: 10.1111/j.1751-7176.2009.00085.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Okanloma KA, Moodley J. Neurological complications associated with the pre-eclampsia/eclampsia syndrome. Int J Gynaecol Obstet. 2000 Dec;71(3):223–225. doi: 10.1016/s0020-7292(00)00295-2. [DOI] [PubMed] [Google Scholar]
- 39.Sharshar T, Lamy C, Mas JL. Incidence and causes of strokes associated with pregnancy and puerperium. A study in public hospitals of Ile de France. Stroke in Pregnancy Study Group. Stroke. 1995 Jun;26(6):930–936. doi: 10.1161/01.str.26.6.930. [DOI] [PubMed] [Google Scholar]
- 40.Martin JN, Jr, Thigpen BD, Moore RC, Rose CH, Cushman J, May W. Stroke and severe preeclampsia and eclampsia: a paradigm shift focusing on systolic blood pressure. Obstet Gynecol. 2005 Feb;105(2):246–254. doi: 10.1097/01.AOG.0000151116.84113.56. [DOI] [PubMed] [Google Scholar]
- 41.Hinchey J, Chaves C, Appignani B, et al. A reversible posterior leukoencephalopathy syndrome. N Engl J Med. 1996 Feb 22;334(8):494–500. doi: 10.1056/NEJM199602223340803. [DOI] [PubMed] [Google Scholar]
- 42.Wagner SJ, Acquah LA, Lindell EP, et al. Posterior reversible encephalopathy syndrome and eclampsia: pressing the case for more aggressive blood pressure control. Mayo Clin Proc. 2011 Sep;86(9):851–856. doi: 10.4065/mcp.2011.0090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wilson BJ, Watson MS, Prescott GJ, et al. Hypertensive diseases of pregnancy and risk of hypertension and stroke in later life: results from cohort study. Bmj. 2003 Apr 19;326(7394):845. doi: 10.1136/bmj.326.7394.845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Garovic VD, Bailey KR, Boerwinkle E, et al. Hypertension in pregnancy as a risk factor for cardiovascular disease later in life. J Hypertens. 2010 Apr;28(4):826–833. doi: 10.1097/HJH.0b013e328335c29a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ray JG, Vermeulen MJ, Schull MJ, Redelmeier DA. Cardiovascular health after maternal placental syndromes (CHAMPS): population-based retrospective cohort study. Lancet. 2005 Nov 19;366(9499):1797–1803. doi: 10.1016/S0140-6736(05)67726-4. [DOI] [PubMed] [Google Scholar]
- 46.Bar J, Kaplan B, Wittenberg C, et al. Microalbuminuria after pregnancy complicated by pre-eclampsia. Nephrol Dial Transplant. 1999 May;14(5):1129–1132. doi: 10.1093/ndt/14.5.1129. [DOI] [PubMed] [Google Scholar]
- 47.Vikse BE, Irgens LM, Leivestad T, Skjaerven R, Iversen BM. Preeclampsia and the risk of end-stage renal disease. N Engl J Med. 2008 Aug 21;359(8):800–809. doi: 10.1056/NEJMoa0706790. [DOI] [PubMed] [Google Scholar]
- 48.Artal R, O’Toole M. Guidelines of the American College of Obstetricians and Gynecologists for exercise during pregnancy and the postpartum period. Br J Sports Med. 2003 Feb;37(1):6–12. doi: 10.1136/bjsm.37.1.6. discussion 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Falcao S, Bisotto S, Michel C, et al. Exercise training can attenuate preeclampsia-like features in an animal model. J Hypertens. 2010 Dec;28(12):2446–2453. doi: 10.1097/HJH.0b013e32833e97d0. [DOI] [PubMed] [Google Scholar]
- 50.Saftlas A, Wang W, Risch H, Woolson R, Hsu C, Bracken M. Prepregnancy body mass index and gestational weight gain as risk factors for preeclampsia and transient hypertension. Ann Epidemiol. 2000 Oct 1;10(7):475. doi: 10.1016/s1047-2797(00)00167-8. [DOI] [PubMed] [Google Scholar]
- 51.Siega-Riz AM, Viswanathan M, Moos MK, et al. A systematic review of outcomes of maternal weight gain according to the Institute of Medicine recommendations: birthweight, fetal growth, and postpartum weight retention. Am J Obstet Gynecol. 2009 Oct;201(4):339 e331–314. doi: 10.1016/j.ajog.2009.07.002. [DOI] [PubMed] [Google Scholar]
- 52.Papiernik E, Kaminski M. Multifactorial study of the risk of prematurity at 32 weeks of gestation. I. A study of the frequency of 30 predictive characteristics. J Perinat Med. 1974;2(1):30–36. doi: 10.1515/jpme.1974.2.1.30. [DOI] [PubMed] [Google Scholar]
- 53.Abenhaim HA, Bujold E, Benjamin A, Kinch RA. Evaluating the role of bedrest on the prevention of hypertensive diseases of pregnancy and growth restriction. Hypertens Pregnancy. 2008;27(2):197–205. doi: 10.1080/10641950701826273. [DOI] [PubMed] [Google Scholar]
- 54.Cockburn J, Moar VA, Ounsted M, Redman CW. Final report of study on hypertension during pregnancy: the effects of specific treatment on the growth and development of the children. Lancet. 1982 Mar 20;1(8273):647–649. doi: 10.1016/s0140-6736(82)92202-4. [DOI] [PubMed] [Google Scholar]
- 55.Sibai BM, Mabie WC, Shamsa F, Villar MA, Anderson GD. A comparison of no medication versus methyldopa or labetalol in chronic hypertension during pregnancy. Am J Obstet Gynecol. 1990 Apr;162(4):960–966. doi: 10.1016/0002-9378(90)91297-p. discussion 966–967. [DOI] [PubMed] [Google Scholar]
- 56.Butters L, Kennedy S, Rubin PC. Atenolol in essential hypertension during pregnancy. Bmj. 1990 Sep 22;301(6752):587–589. doi: 10.1136/bmj.301.6752.587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ismail AA, Medhat I, Tawfic TA, Kholeif A. Evaluation of calcium-antagonist (Nifedipine) in the treatment of pre-eclampsia. Int J Gynaecol Obstet. 1993 Jan;40(1):39–43. doi: 10.1016/0020-7292(93)90770-w. [DOI] [PubMed] [Google Scholar]
- 58.Magee LA, Schick B, Donnenfeld AE, et al. The safety of calcium channel blockers in human pregnancy: a prospective, multicenter cohort study. Am J Obstet Gynecol. 1996 Mar;174(3):823–828. doi: 10.1016/s0002-9378(96)70307-1. [DOI] [PubMed] [Google Scholar]
- 59.Gowda RM, Khan IA, Mehta NJ, Vasavada BC, Sacchi TJ. Cardiac arrhythmias in pregnancy: clinical and therapeutic considerations. Int J Cardiol. 2003 Apr;88(2–3):129–133. doi: 10.1016/s0167-5273(02)00601-0. [DOI] [PubMed] [Google Scholar]
- 60.Khandelwal M, Kumanova M, Gaughan JP, Reece EA. Role of diltiazem in pregnant women with chronic renal disease. J Matern Fetal Neona. 2002 Dec;12(6):408–412. doi: 10.1080/jmf.12.6.408.412. [DOI] [PubMed] [Google Scholar]
- 61.Frishman WH, Schlocker SJ, Awad K, Tejani N. Pathophysiology and medical management of systemic hypertension in pregnancy. Cardiol Rev. 2005 Nov-Dec;13(6):274–284. doi: 10.1097/01.crd.0000137738.16166.cc. [DOI] [PubMed] [Google Scholar]
- 62.Magee LA, Cham C, Waterman EJ, Ohlsson A, von Dadelszen P. Hydralazine for treatment of severe hypertension in pregnancy: meta-analysis. Bmj. 2003 Oct 25;327(7421):955–960. doi: 10.1136/bmj.327.7421.955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Cameron HA, Ramsay LE. The lupus syndrome induced by hydralazine: a common complication with low dose treatment. Br Med J (Clin Res Ed) 1984 Aug 18;289(6442):410–412. doi: 10.1136/bmj.289.6442.410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Handler J. Hydralazine-induced lupus erythematosis. J Clin Hypertens (Greenwich) 2012 Feb;14(2):133–136. doi: 10.1111/j.1751-7176.2011.00573.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Cooper WO, Hernandez-Diaz S, Arbogast PG, et al. Major congenital malformations after first-trimester exposure to ACE inhibitors. N Engl J Med. 2006 Jun 8;354(23):2443–2451. doi: 10.1056/NEJMoa055202. [DOI] [PubMed] [Google Scholar]
- 66.Li DK, Yang C, Andrade S, Tavares V, Ferber JR. Maternal exposure to angiotensin converting enzyme inhibitors in the first trimester and risk of malformations in offspring: a retrospective cohort study. Bmj. 2011;343:d5931. doi: 10.1136/bmj.d5931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Cabassi A, Rocco R, Berretta R, Regolisti G, Bacchi-Modena A. Eplerenone use in primary aldosteronism during pregnancy. Hypertension. 2012 Feb;59(2):e18–19. doi: 10.1161/HYPERTENSIONAHA.111.187237. [DOI] [PubMed] [Google Scholar]
- 68.Mosca L, Benjamin EJ, Berra K, et al. Effectiveness-based guidelines for the prevention of cardiovascular disease in women--2011 update: a guideline from the American Heart Association. J Am Coll Cardiol. 2011 Mar 22;57(12):1404–1423. doi: 10.1016/j.jacc.2011.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]


