Abstract
Plexiform angiomyxoid myofibroblastic tumor (PAMT) is a rare mesenchymal tumor of the stomach. We report herein a case with CT findings, which illustrate the characteristic growth pattern of PAMT. A 27-year-old female patient visited our hospital because of epigastric pain and anemia. The CT scan showed a heterogeneous tumor in the gastric antrum, which was drastically enhanced with contrast medium, and consisted of a number of highly stained small nodules around the tumor rim. The resected tumor, 4.6 cm in size, was c-kit negative and SMA-positive by immunohistochemistry, and composed of bland spindle cells which were separated by abundant myxomatous stroma. The tumor showed plexiform growth in the entire stomach wall, with multiple nodules protruding outward within the serosa. The CT findings in this case reflect the characteristic PAMT growth pattern, and are distinct enough to differentiate it from gastrointestinal stromal tumor (GIST).
Keywords: PAMT, GIST, diagnosis, CT
Introduction
Plexiform angiomyxoid myofibroblastic tumor (PAMT) is a recently identified tumor of the stomach, which was first described by Takahashi et al. in 2007 [1]. PAMT is also called “plexiform angiomyxoma” or “plexiform angiomyxoid tumor”. Although the exact name is disputed, the term “plexiform fibromyxoma” was added as a mesenchymal tumor of the stomach in the 2010 WHO classification of tumors of the digestive system. However, there have been only 26 cases of PAMT reported in English-language publications [1-12]. Histopathology of PAMT is distinct from gastrointestinal stromal tumor (GIST), a representative mesenchymal tumor of the stomach, and their clinical management is also different. This report presents a case of PAMT that showed characteristic radiological images which reflect the growth pattern of this particular mesenchymal tumor of the stomach.
Case report
Clinical presentation
A 27-year-old female patient visited the hospital with a complaint of epigastric pain and tarry stool. The patient was hospitalized for further examinations because of her anemia (Hb=4.7 g/dL). In an upper gastrointestinal endoscopy examination, a submucosal tumor was found in the pyloric antrum with partial ulceration on the surface.
A noncontrast CT scan indicated a low-attenuation area in a mass, approximately 3 cm in size, along the posterior wall of the lower body of the stomach, which showed a strong enhancement pattern in the late phase of contrast imaging (Figure 1). Although the mass was observed in the lesser curvature, arborization of the left gastric artery was observed immediately under the mass, indicating a hypervascular tumor. Inside of the tumor was heterogeneous, with a number of strongly enhanced spots in the periphery. Radiological imaging suggested differential diagnoses of angioma and paraganglioma because of the hypervascular nature of the tumor. However, because the tumor did not protrude into the lumen of the stomach, these diagnoses were unlikely. Furthermore, the imaging findings ruled out the possibility of GIST.
Figure 1.
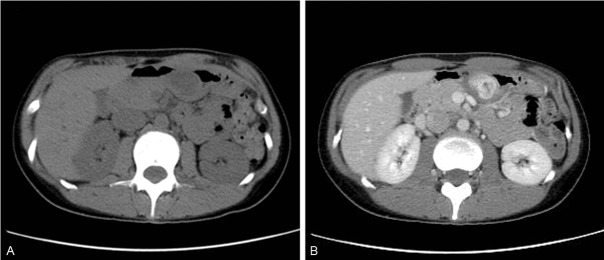
CT scan of the tumor. A. Unenhanced CT image shows inhomogeneous tumor mass in the lower body of the stomach. B. Contrast-enhanced CT image revealed a tumor mass with an inhomogeneous, prominent enhancement in the late phase of contrast imaging.
A partial gastrectomy revealed the tumor at the antrum of the lesser curvature side of the stomach. Aggregation of small polypoid nodules, red-colored and angiomatous, was characteristically seen on the serous membrane surface (Figure 2). Seeding nodules were not found. There were no recurrences or metastases at the 40-month follow-up.
Figure 2.
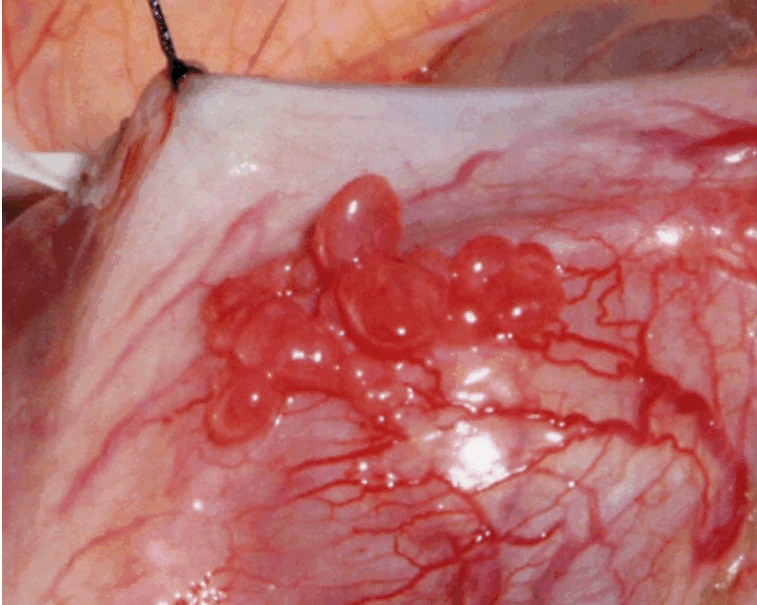
Laparotomy finding at the surgery.
Pathological findings
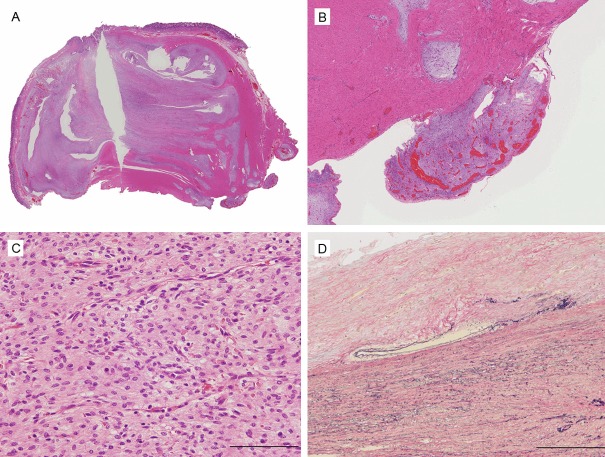
On gross examination, a 4.6 × 3.0 × 2.8-cm lobulated tumor with focal ulceration of the mucosa was found (Figure 3A), while the serosal surface was granular in appearance. On the cut surface, the tumor was not encapsulated, and involved all layers of the gastric wall, protruding to the surface. The tumor showed a mixture of white-fibrous, myxomatous and hemorrhagic regions (Figure 3B).
Figure 3.
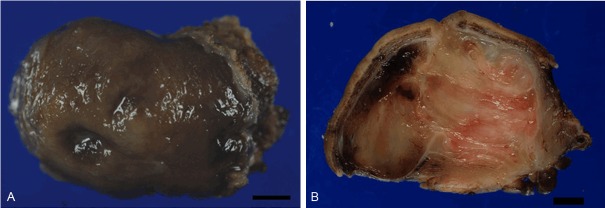
Gross appearance of the tumor. A. A submucosal tumor with focal ulceration is observed. Bar: 5 mm. B. The cut section of the tumor. Lobulated tumor is found in all layers of the gastric wall.
Histologically, the tumor mainly showed a small nodular pattern as it pushed into proper muscular layers, extending to the subserosa and partially to the mucosa (Figure 4A and 4B). A proliferation of bland spindle- or star-shaped tumor cells was accompanied by abundant myxomatous stroma or fibrous tissue. Regions of low tumor cell density with abundant stroma were intermingled with regions of comparatively high cell density with a storiform growth (Figure 4C). Tumor cells had relatively uniform-shaped oval nuclei; cellular atypia and mitosis were both rare. The lesion was rich in small vessels that partially involved irregular anastomosis and thrombus formation. Although the tumor occasionally disrupted vessel walls, no lesion showed intravascular invasion (Figure 4D). The surgical margin was negative.
Figure 4.
Histological appearance of the tumor. A. The tumor shows a multinodular plexiform growth pattern. B. Fibromyxoid nodules of tumor extend into serosal surface. C. The tumor contains spindle-shaped bland tumor cells in a fibromyxoid stroma, which is rich in small caliber blood vessels. D. Although the tumor disrupted the vessel wall, there was no evidence of invasion of the intravascular lumen.
Immunohistochemically, the tumor cells showed smooth muscle actin (SMA) (+) and desmin (+) in only small number of tumor cells. The results of other markers were CD10(+), c-kit(-), CD34(-), ER(-), PgR(-) and Claudin-1(-). Only a small percentage of cells were MIB-1+. Based on the above results, the tumor was diagnosed as PAMT.
Discussion
Plexiform angiomyxoid myofibroblastic tumor (PAMT) is a rare mesenchymal tumor of the stomach, which consists of spindle cells with myofibroblastic characteristics. PAMT is also called as plexiform angiomyxoma, angiomyxoid tumor or myxofibroma. Miettinen et al. estimated that the frequency of PAMT is less than 1/150 that of GIST [4], but only 27 cases of PAMT have been reported, including the present case [1-12]. The prevalence of PAMT does not vary by sex (M:F=13:14) and it can develop at any age (range: 7-75 years). Most cases present certain symptoms, such as anemia due to chronic gastric bleeding from tumor with mucosal ulcer in the present case, although this PAMT was incidentally detected at a medical checkup. The tumor size varies (range: 1.5-14 cm), but most are a few cm in size (mean: 5.5 cm).
PAMT consists of a proliferation of bland spindle cells with myofibroblastic differentiation at ultrastructural level [1]. PAMT cells show immunohistochemical and molecular characteristics different from those of GIST: PAMT cells show c-kit(-) and CD34(-), and lack of mutations in c-kit and PDGFA genes. Plexiform growth pattern is also characteristic of PAMT, and absence of this pattern simply excludes myxoid variant of GIST, inflammatory fibroid polyp and solitary fibrous tumor. Other mesenchemal tumors that share a similar growth pattern are as follows. Plexiform leiomyoma, a distinctive but rare variant, has been reported in the esophagus [13], but it can be distinguished by its distinct differentiation of smooth muscle, such as diffuse expression of desmin and actin Nerve sheath tumors (schwannoma, neurofibroma and perineuroma) can also show a plexiform growth pattern. PAMT cells show none of the neural and perineural characteristics, such as S-100 and EMA/Claudin-1, respectively. In female patients, low-grade endometrial stromal sarcoma (ESS) should be included in the differential diagnosis [3,7]. In the present case, whereas PAMT cells were CD10(+), hormone receptors (ER and PgR) were totally negative, which eliminated the possibility of ESS.
Only few reports have described CT findings for PAMT [6-8,12]. In this case, contrast-enhanced CT image revealed a mass with a heterogenous, prominent enhancement in the late phase of contrast imaging. Moreover, small nodules were observed around the rim that showed strong enhancement, which suggests a possibility of reflecting the plexiform growth of PAMT in the rim. The CT findings of GIST vary depending on tumor size [14]. Small GIST (≤5 cm) are usually seen as smooth, sharply defined intramural masses with homogenous attenuation in contrasted-enhanced CT. Characterization of CT images for PAMT would require accumulation of many cases. However, the following CT findings can be used for radiologic diagnosis of PAMT: (1) relatively small tumor found in the gastric antrum, (2) strong and heterogeneous internal enhancement effect, and (3) small nodules that show a strong enhancement effect in the rim.
PAMT is considered to have a good prognosis with no recurrence or metastasis reported so far. However, as vascular invasion has been reported in one case [5], we cannot completely exclude the possibility that PAMT can metastasize. The good prognosis might be attributed to the fact that the most of the PAMT cases have been detected early and had shorter follow-up periods.
In the present report, we reported a case of PAMT with characteristic CT findings, which show the growth pattern of PAMT. Accurate diagnosis of PAMT is important; in particular PAMT should not be misdiagnosed as GIST or other plexiform lesions, which require different follow-up and treatment approaches. We proposed three signs in CT findings to aid diagnosis of PAMT, as described above, and hope that these will be useful for treatment of the patients with gastric mesenchymal tumors.
Disclosure of conflict of interest
The authors declare that they have no competing interests.
References
- 1.Takahashi Y, Shimizu S, Ishida T, Aita K, Toida S, Fukusato T, Mori S. Plexiform angiomyxoid myofibroblastic tumor of the stomach. Am J Surg Pathol. 2007 May;31:724–728. doi: 10.1097/01.pas.0000213448.54643.2f. [DOI] [PubMed] [Google Scholar]
- 2.Galant C, Rousseau E, Ho Minh Duc DK, Pauwels P. Plexiform angiomyxoid myofibroblastic tumor of the stomach. Am J Surg Pathol. 2008 Dec;32:1910. doi: 10.1097/PAS.0b013e3181838fa9. [DOI] [PubMed] [Google Scholar]
- 3.Yoshida A, Klimstra DS, Antonescu CR. Plexiform angiomysoid tumor of the stomach. Am J Surg Pathol. 2008 Dec;32:1910–1912. doi: 10.1097/PAS.0b013e3181838fd1. [DOI] [PubMed] [Google Scholar]
- 4.Miettinen M, Makhlouf HR, Sobin LH, Lasota J. Plexiform Fibromyxoma. Am J Surg Pathol. 2009 Nov;33:1624–1632. doi: 10.1097/PAS.0b013e3181ae666a. [DOI] [PubMed] [Google Scholar]
- 5.Rau TT, Hartmann A, Dietmaier W, Schmitz J, Hohenberger W, Hofstaedter F, Katenkamp K. Plexiform angiomyxoid myofibroblastic tumour: differential diagnosis of gastrointestinal stromal tumour in the stomach. J Clin Pathol. 2008;61:1136–1137. doi: 10.1136/jcp.2008.059162. [DOI] [PubMed] [Google Scholar]
- 6.Pailoor J, Mun KS, Chen CT, Pillay B. Plexiform angiomyxoid myofibroblastic tumour of the stomach. Pathology. 2009 Dec;41:698–699. doi: 10.3109/00313020903305753. [DOI] [PubMed] [Google Scholar]
- 7.Sing Y, Subrayan S, Mqadi B, Ramdial PK, Reddy J, Moodley MS, Bux S. Gastric plexiform angiomyxoid myofibroblastic tumor. Pathol Int. 2010;60:621–625. doi: 10.1111/j.1440-1827.2010.02569.x. [DOI] [PubMed] [Google Scholar]
- 8.Takahashi Y, Suzuki M, Fukusato T. Plexiform angiomyxoid myofibroblastic tumor of the stomach. World J Gastroenterol. 2010 Jun 21;16:2835–2840. doi: 10.3748/wjg.v16.i23.2835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tan CY, Santos LD, Biankin A. Plexiform angiomyxoid myofibroblastic tumour of the stomach: a case report. Pathology. 2010 Oct;42:581–583. doi: 10.3109/00313025.2010.508739. [DOI] [PubMed] [Google Scholar]
- 10.Wang WY, Li JN, Li GD. Plexiform angiomyxoid myofibroblastic tumour of the gastric fundus: successful diagnosis and treatment by endoscopy. J Clin Pathol. 2010 Jun;63:569–570. doi: 10.1136/jcp.2010.076646. [DOI] [PubMed] [Google Scholar]
- 11.Kim A, Bae YK, Shin HC, Choi JH. Plexiform angiomyxoid myofibroblastic tumor of the stomach: A case report. J Korean Med Sci. 2011;26:1508–1511. doi: 10.3346/jkms.2011.26.11.1508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kang Y, Jung W, Do IG, Lee EJ, Lee MH, Kim KM, Choi J. Plexiform angiomyxoid myofibroblastic tumor of the stomach: Report of two cases and review of the literature. Korean J Pathol. 2012;46:292–296. doi: 10.4132/KoreanJPathol.2012.46.3.292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jaroszewski DE, Lam-Himlin D, Gruden J, Lidner TK, Etxebarria AA, Petris GD. Plexiform leiomyoma of the esophagus: a complex radiographic, pathologic and endoscopic diagnosis. Ann Diagn Pathol. 2011;15:342–346. doi: 10.1016/j.anndiagpath.2011.04.006. [DOI] [PubMed] [Google Scholar]
- 14.Ghanem N, Altehoefer C, Furtwängler A, Winterer J, Schäfer O, Springer O, Kotter E, Langer M. Computed tomography in gastrointestinal stroma tumors. Eur Radiol. 2003;13:1669–1678. doi: 10.1007/s00330-002-1803-6. [DOI] [PubMed] [Google Scholar]