Significance
Cancer stem cells (CSCs) are thought to be responsible for growth and dissemination of many malignant tumors and for relapse after therapy. Therefore methods for the noninvasive imaging of CSCs could have profound consequences for diagnosis and therapy monitoring in oncology. However, clinically applicable methods for noninvasive CSC imaging are still lacking. The AC133 epitope of CD133 is one of the most intensely investigated CSC markers and is particularly important for aggressive brain tumors. Here we describe the development of clinically relevant tracers that permit high-sensitivity and high-resolution monitoring of AC133+ glioblastoma stem cells in both subcutaneous and intracerebral xenograft tumors using positron emission tomography and near-infrared fluorescence imaging, two clinically highly relevant imaging modalities.
Keywords: cancer stem cells, CSCs, glioblastoma
Abstract
A technology that visualizes tumor stem cells with clinically relevant tracers could have a broad impact on cancer diagnosis and treatment. The AC133 epitope of CD133 currently is one of the best-characterized tumor stem cell markers for many intra- and extracranial tumor entities. Here we demonstrate the successful noninvasive detection of AC133+ tumor stem cells by PET and near-infrared fluorescence molecular tomography in subcutaneous and orthotopic glioma xenografts using antibody-based tracers. Particularly, microPET with 64Cu-NOTA-AC133 mAb yielded high-quality images with outstanding tumor-to-background contrast, clearly delineating subcutaneous tumor stem cell-derived xenografts from surrounding tissues. Intracerebral tumors as small as 2–3 mm also were clearly discernible, and the microPET images reflected the invasive growth pattern of orthotopic cancer stem cell-derived tumors with low density of AC133+ cells. These data provide a basis for further preclinical and clinical use of the developed tracers for high-sensitivity and high-resolution monitoring of AC133+ tumor stem cells.
Cancer stem cells (CSCs) are highly undifferentiated tumor cells with characteristics similar to normal stem cells. These characteristics include long-term replication, self-renewal, and aberrant differentiation (1, 2). Based on these characteristics, it has been hypothesized that only CSCs are able to propagate tumors for long periods of time and to initiate relapses or metastases. Furthermore, CSCs are considered to be more resistant to conventional radio- and chemotherapy than more differentiated tumor cells (3–5). Hence, elimination of CSCs is challenging but necessary for successful tumor eradication. The stem cell hypothesis of cancer development and progression is conceptually attractive and is supported by many preclinical (1, 2, 5–7) and some clinical studies (4, 8). However, larger clinical trials investigating the role of CSCs in patients have been hampered by the lack of techniques to detect, localize, and quantify the presence of CSCs noninvasively. Specifically, successful noninvasive imaging of unmanipulated CSCs with clinically relevant imaging probes (e.g., antibodies or other ligands binding CSC-specific cell-surface proteins) has not yet been reported (9–11).
AC133 is an N-glycosylation–dependent epitope of the second extracellular loop of CD133/prominin-1, a cholesterol-binding protein of unknown function that locates to plasma membrane protrusions (12–14). Postnatally, the CD133 protein is expressed by certain epithelial and nonepithelial cells, by stem and progenitor cells of various organs, and by CSCs of many different types of malignant tumors (15). With a few exceptions, recognition of the AC133 epitope by the AC133 mAb appears to be limited to cells harboring stem cell properties, and the AC133 epitope—but not necessarily the CD133 protein—is down-regulated upon differentiation, presumably because of changes in glycosylation (12, 13, 15).
AC133+ tumor stem cells have been described for glioblastoma multiforme (the most common and most aggressive primary brain tumor in adults), various pediatric brain and central nervous system tumors (medulloblastoma, ependymoma, pineoblastoma, teratoid/rhabdoid tumors, and retinoblastoma), brain metastases, many different types of carcinomas including colon, pancreatic, lung, liver, and ovarian cancer, melanoma, sarcomas, and different types of leukemia. Although AC133− tumor stem cells also exist (16–18), AC133+ cells found in these and other tumor types have been shown to be able to self-renew, to differentiate, and to recreate the original tumors when injected into immunocompromised mice (8, 17, 19–22). Both, stemness and highly agressive malignant tumors often are associated with hypoxia (23), and hypoxia can promote the expansion of CD133+ cells (24). Therefore the frequent expression of AC133 on CSCs may reflect, in part, their common localization in a hypoxic environment (25).
We previously reported the successful noninvasive detection of the AC133 epitope by antibody-based near-infrared fluorescence molecular tomography (NIR FMT) in mice with s.c. xenografts of CD133-overexpressing tumor cells or traditional tumor cell lines naturally displaying AC133 (26). However, we did not investigate patient-derived CSCs with the above-mentioned stem cell characteristics in that study, and NIR fluorescence, although penetrating tissues more deeply (2–4 cm) than visible-light fluorescence, has limited importance for clinical whole-body imaging (27).
We report here the successful noninvasive detection of tumor-associated AC133 by PET, using a radiolabeled AC133-specific mAb in mice xenografted with tumor cell lines overexpressing CD133 or with patient-derived AC133+ CSCs. PET is highly sensitive and is widely used for clinical whole-body diagnostic imaging. As a PET nuclide, we used 64Cu (t1/2 = 12.7 h), which allows long-term tracking for at least 48 h, to follow the tumoral accumulation of relatively large molecules such as antibodies, that exhibit relatively slow tumor penetration (28). We chose S-2-(4-isothiocyanatobenzyl)-1,4,7-triazacyclononane-1,4,7-triacetic acid (p-SCN-Bn-NOTA, hereafter abbreviated as NOTA) as the 64Cu chelator, because high labeling efficiencies and high in vivo stability have been reported for 64Cu-NOTA-antibody conjugates (29). In addition, we report the successful noninvasive detection of AC133+ CSCs with fluorescently labeled AC133 mAb and NIR imaging, a modality that is important for whole-body small-animal imaging and for intraoperative and endoscopic imaging and imaging of superficial tumors in humans (27, 30). In addition to imaging s.c. growing tumors, we report the successful antibody-mediated imaging of orthotopic xenografts initiated from AC133+ glioblastoma stem cells in the brain of immunocompromised mice, emphasizing the feasibility of noninvasive antibody-mediated imaging of brain tumors.
Results
Tracer Development and Characterization.
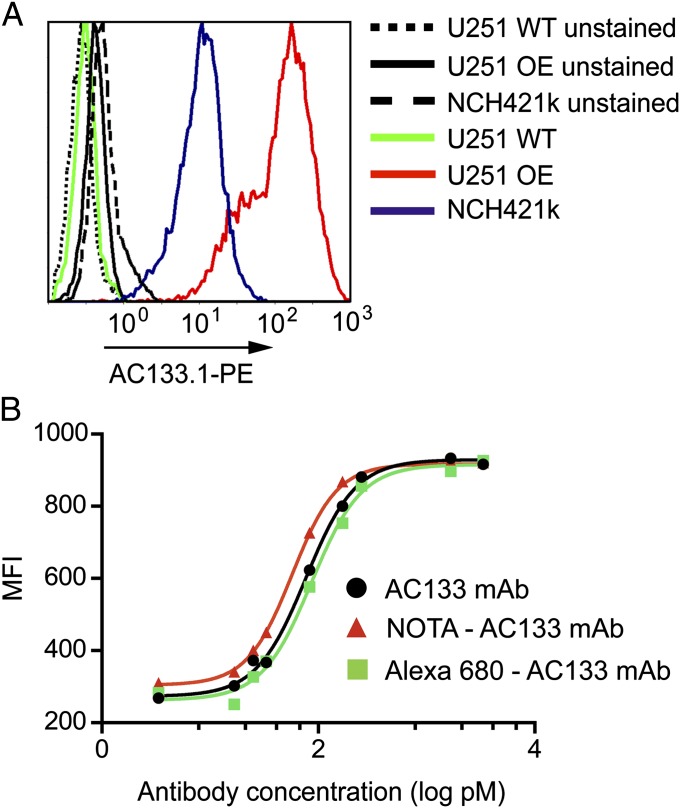
After conjugation of the chelator NOTA, we radiolabeled the AC133 mAb, which recognizes the AC133 epitope on CD133-overexpressing cells and CSCs (Fig. 1A), and an isotype control antibody with 64Cu. The NOTA-AC133 and NOTA-isotype control mAb conjugates were functionalized with an average of 5.1 ± 0.8 and 2.3 ± 0.3 chelators per molecule of antibody, respectively, and in all radiolabeling experiments the radiochemical purity was >95%, with specific activities of 48.4 ± 9.5 MBq/nmol and 41.4 ± 5.6 MBq/nmol for 64Cu-NOTA-AC133 and the isotype control mAb, respectively. Conjugation of the AC133 mAb or the isotype control antibody to the NIR dye Alexa 680 resulted in 2.3 ± 0.2 and 1.5 ± 0.2 Alexa 680 molecules conjugated to the two mAbs, respectively. Neither conjugation of NOTA nor that of Alexa 680 impaired binding to the AC133 epitope, as revealed by flow cytometric titration of NOTA-AC133 and Alexa 680-AC133 against the unmodified AC133 antibody (Fig. 1B).
Fig. 1.
Characterization of AC133+ cell lines and the modified AC133 mAbs. (A) Flow-cytometric detection of AC133 epitope expression on in vitro-cultured CD133-overexpressing U251 glioma cells and NCH421k glioblastoma stem cells compared with CD133− U251 wild-type cells. Results shown are representative of more than 10 independent experiments. (B) Flow-cytometric analysis of the binding specificity of the NOTA-AC133 and Alexa 680-AC133 mAbs compared with unmodified AC133 mAb. The analysis was performed as described in Methods. Data are representative of three independent experiments. MFI, mean fluorescence intensity; OE, overexpressing.
PET Imaging of s.c. CD133-Overexpressing Glioma Xenografts.
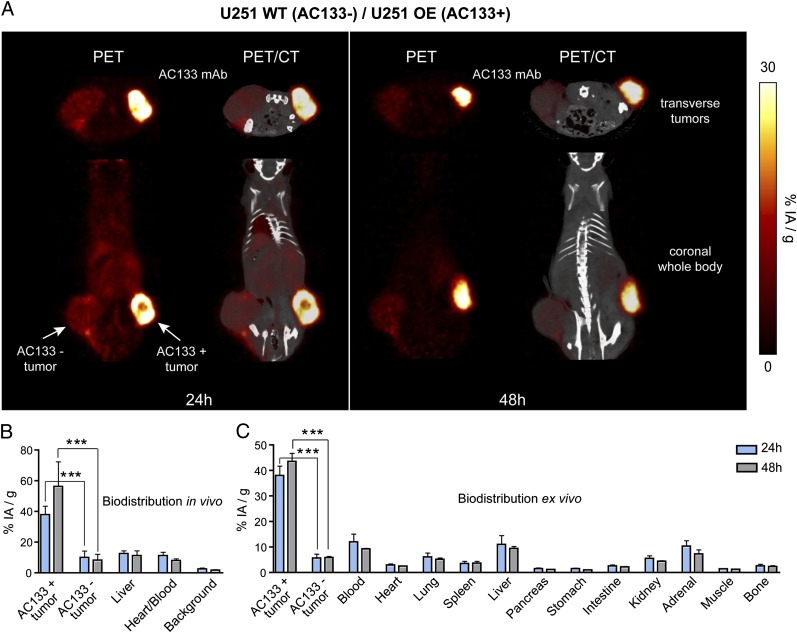
We first established small-animal PET with the 64Cu-NOTA-AC133 mAb in a robust nude mouse model with s.c. xenografts of U251 glioma cells transduced with a CD133-encoding lentivirus resulting in high expression of CD133 associated with high cell-surface expression of the AC133 epitope (Fig. 1A). We s.c. injected these AC133-high cells into the right flank and, as an internal control, injected CD133− U251 wild-type cells into the left flank of each animal. The uptake of the 64Cu-NOTA-AC133 mAb in the CD133-overexpressing xenografts was very high (Fig. 2A). As quantified from PET images, the uptake reached 37.9 ± 5.6% of the injected activity (IA)/g at 24 h and increased to values as high as 56.3 ± 16.2% 48 h after i.v. injection of the radiolabeled antibody. In contrast, the uptake of antigen-negative tumors decreased from 10.1 to 8.3% at 24 and 48 h, respectively, and was similar to or slightly lower than that of liver and blood (Fig. 2B). At 48 h, the uptake ratios of AC133+ tumor to AC133− tumor, liver, blood, and background were 7.3 ± 1.9, 5.0 ± 0.6, 6.9 ± 1.2, and 32.0 ± 8.8, respectively. Ex vivo biodistribution studies at 48 h after administration of the 64Cu-NOTA-AC133 mAb corroborated the PET imaging data (Fig. 2C). In contrast to the highly different uptake of the 64Cu-NOTA-AC133 mAb, both the AC133+ and the AC133− tumors showed a similar uptake of 18F-fluorodeoxyglucose (FDG) (Fig. S1).
Fig. 2.
PET/CT imaging and biodistribution of 64Cu-NOTA-AC133 mAb in mice bearing s.c. implanted U251 gliomas overexpressing CD133. Nude mice received ∼8.0 ± 0.5 MBq 64Cu-NOTA-AC133 mAb via tail vein injection, and PET/CT images were acquired. The mice carried AC133− U251 wild-type and AC133/CD133-overexpressing U251 gliomas in the left and right flanks, respectively. (A) Representative transverse tumor and coronal whole-body PET and fused PET/CT sections at 24 and 48 h p.i. (B) Uptake of 64Cu-NOTA-AC133 mAb as measured by microPET in various organs and AC133− and AC133-overexpressing tumors at 24 and 48 h p.i. Values are the mean %IA/g of tissue. (C) Ex vivo biodistribution at 24 and 48 h p.i. Values are the mean %IA/g of tissue. n = 7–8 mice per group. ***P < 0.001, t test; values represent means ± SD.
FMT Imaging of Glioblastoma Stem Cell-Derived s.c. Xenografts.
To develop animal models for imaging of AC133+ CSCs, which usually display much lower levels of AC133 than cell lines overexpressing CD133, we chose the well-characterized glioma stem cell line NCH421k. This cell line has been derived from a primary glioblastoma [World Health Organization (WHO) grade IV] under stem cell culture conditions, and its CSC characteristics have been reported repeatedly (Fig. S2 and refs. 24, 31–33). NCH421k cells display 10- to 15-fold lower surface AC133 than CD133-overexpressing U251 cells both in vitro (Fig. 1A) and in vivo (Fig. S3A). We first established in vivo imaging of CSC-containing tumors with NIR FMT, a sensitive and inexpensive imaging technique permitting quantitative 3D detection of NIR fluorophores in mice. Because CD133− derivatives of the NCH421k line do not exist, we used an Alexa 680-labeled IgG1 isotype antibody as specificity control.
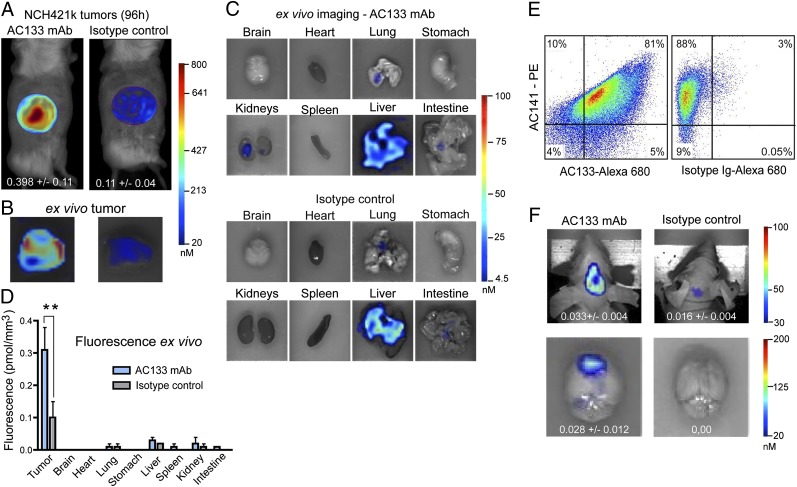
The Alexa 680-labeled AC133 mAb yielded a significantly higher in vivo fluorescence signal in the tumor region than did the Alexa 680-labeled isotype control antibody (Fig. 3A). The ex vivo biodistribution analysis confirmed that the fluorescently labeled AC133 mAb accumulated in the tumor (Fig. 3 B–D). In addition, flow-cytometric analyses of tumor single-cell suspensions directly after the scans showed that the i.v.-injected Alexa 680-AC133 mAb had bound to almost all CSC marker-positive cells in the tumor, verifying that the injected Alexa 680-AC133 mAb had penetrated the tumor tissue efficiently (Fig. 3E, Left). To identify the CSCs ex vivo, the tumor single-cell suspensions were costained with the AC141 antibody, which is specific for a second stem cell-specific epitope of CD133. As expected, binding of the injected Alexa 680-labeled isotype control antibody to CSCs could not be detected (Fig. 3E, Right).
Fig. 3.
NIR FMT imaging of xenografts containing AC133+ glioblastoma stem cells. Mice with s.c. growing NCH421k xenografts were injected i.v. with Alexa 680-AC133 mAb or Alexa 680-isotype control antibody. After 1, 2, 3, and 4 d, the mice were imaged using the FMT-1500 system. The pictures presented correspond to the last measurement acquired 96 h p.i. (A–D) 3D whole-body images (A), 3D images of the excised tumors (B) and organs (C), and quantification of their fluorescence (D). (E) Flow-cytometric detection of i.v. injected Alexa 680-labeled AC133 mAb on AC133+ CSCs in single-cell suspensions of excised s.c. tumors. AC133+ CSCs were counterstained in single-cell suspensions with the AC141 mAb. Only cells falling in the CSC (FSC/SSC) gate are shown. Results shown are representative of four independent experiments. (F) 3D whole-body images and 3D images of excised brains of mice with intracerebral NCH421k xenografts 72 h after i.v. injection of Alexa 680-AC133 or Alexa 680-isotype control antibodies. For A–D and F, n = 5 mice per group. **P < 0.05; t test; values represent means ± SD.
FMT Imaging of Intracerebral Xenograft Tumors.
Antibodies have only limited access to the brain because the undisturbed blood–brain barrier (BBB) is impermeable to macromolecules, and whether systemically administered antibodies can reach extravascular targets in brain tumors with a disturbed BBB is of great interest (34, 35). We therefore wanted to find out whether the AC133 mAb is suitable for imaging orthotopically growing AC133+ glioma xenografts. We indeed could detect orthotopically growing NCH421k gliomas noninvasively by NIR FMT imaging upon i.v. injection of the Alexa 680-labeled AC133 mAb (Fig. 3F, Upper Left), and the signal caused by the Alexa 680-AC133 mAb also could be detected directly postmortem on the excised tumor-bearing brains (Fig. 3F, Lower Left).
PET Imaging of Tumor Stem Cell-Derived s.c. Xenografts.
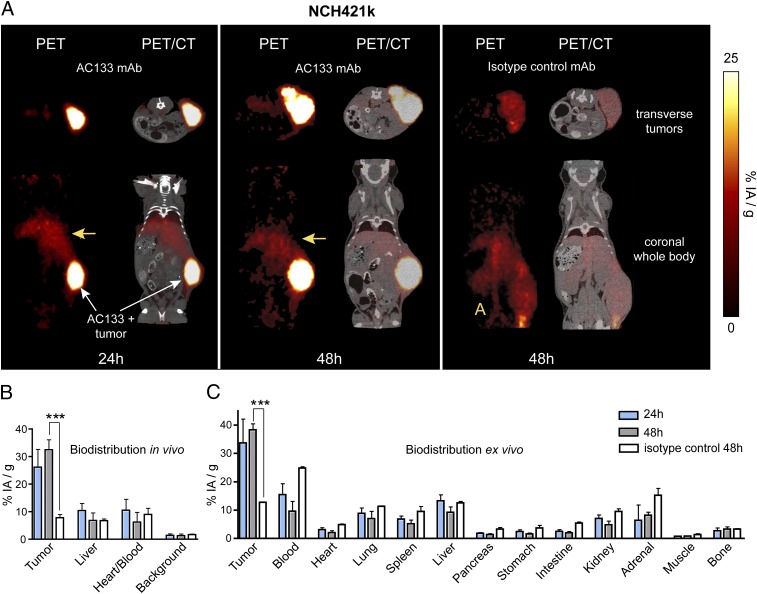
After the FMT studies had demonstrated that noninvasive mAb-mediated visualization of AC133+ CSCs was possible in principle, we explored immuno-PET detection of AC133+ CSCs. The 64Cu-NOTA-AC133 mAb strongly marked s.c. growing NCH421k gliomas at 24 and 48 h postinjection (p.i.) (Fig. 4A, Left and Center), despite the considerably lower expression of AC133 on NCH421k cells as compared with CD133-overexpressing U251 cells (see Fig. 1A and Fig. S3A). Particularly remarkable was the much higher tracer uptake liver [an organ exhibiting relatively high unspecific activity because of high blood perfusion, antibody metabolism, and potential transchelation of 64Cu (28, 36)]. The tumor-to-contralateral background, tumor-to-blood pool, and tumor-to-liver contrasts were 21.4 ± 8.2, 2.7 ± 0.9, and 2.6 ± 0.8 at 24 h and 32.8 ± 19, 6.4 ± 2.5, and 5.4 ± 1.8 at 48 h p.i., respectively. The 64Cu-NOTA-isotype control antibody caused only a very weak tumor signal. As judged by visual inspection (Fig. 4A, Right) and according to in vivo and in vitro quantification (Fig. 4 B and C), this signal was in the range of or slightly lower than that of the blood pool (heart and blood vessels), presumably reflecting a high vascularization of the aggressively growing tumors and unspecific accumulation of a proportion of the antibody in the interstitial space via the enhanced permeation and retention effect (37).
Fig. 4.
PET/CT imaging and biodistribution of 64Cu-NOTA-AC133 and isotype control mAbs in mice bearing s.c. implanted xenografts containing AC133+ glioblastoma stem cells. NOD/SCID mice bearing NCH421k xenografts in the right flank were given ∼6.4 ± 1.7 MBq of either 64Cu-NOTA-AC133 or 64Cu-NOTA-isotype control mAb via tail vein injection, and PET/CT images were acquired. (A) Representative transverse tumor and coronal whole-body PET and fused PET/CT sections at 24 and 48 h p.i. The yellow arrows indicate the liver; “A” indicates aorta branching into the two common iliac arteries. (B) Uptake of both 64Cu-NOTA-AC133 and 64Cu-NOTA-isotype control mAb as determined by microPET in various organs and in the tumor. Values are the mean %IA/g of tissue. (C) Ex vivo biodistribution. Values are the mean %IA/g of tissue. n = 5 mice per group. ***P < 0.001, t test; values represent means ± SD.
PET Imaging of Intracerebral Xenograft Tumors.
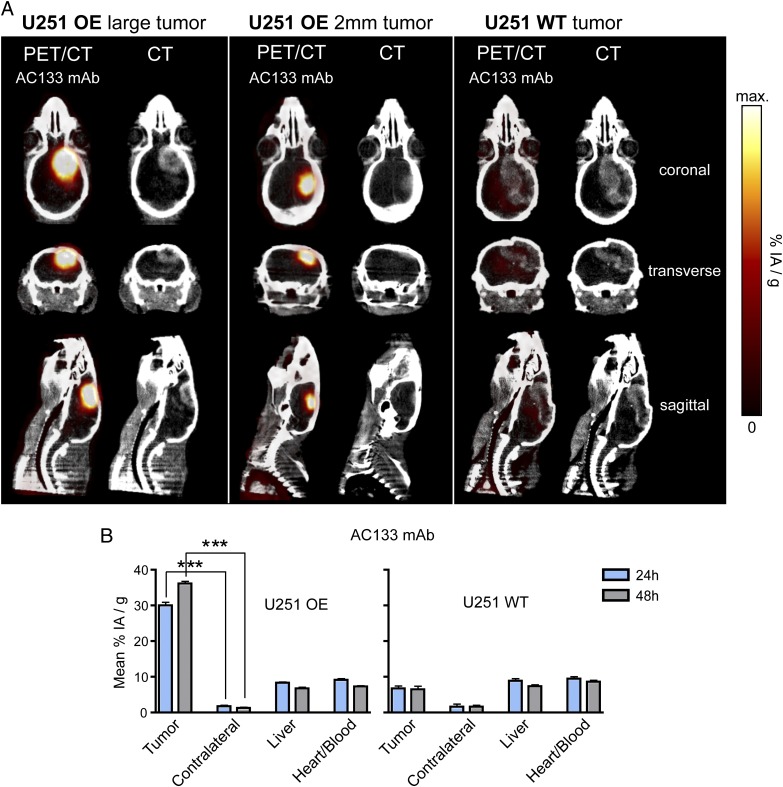
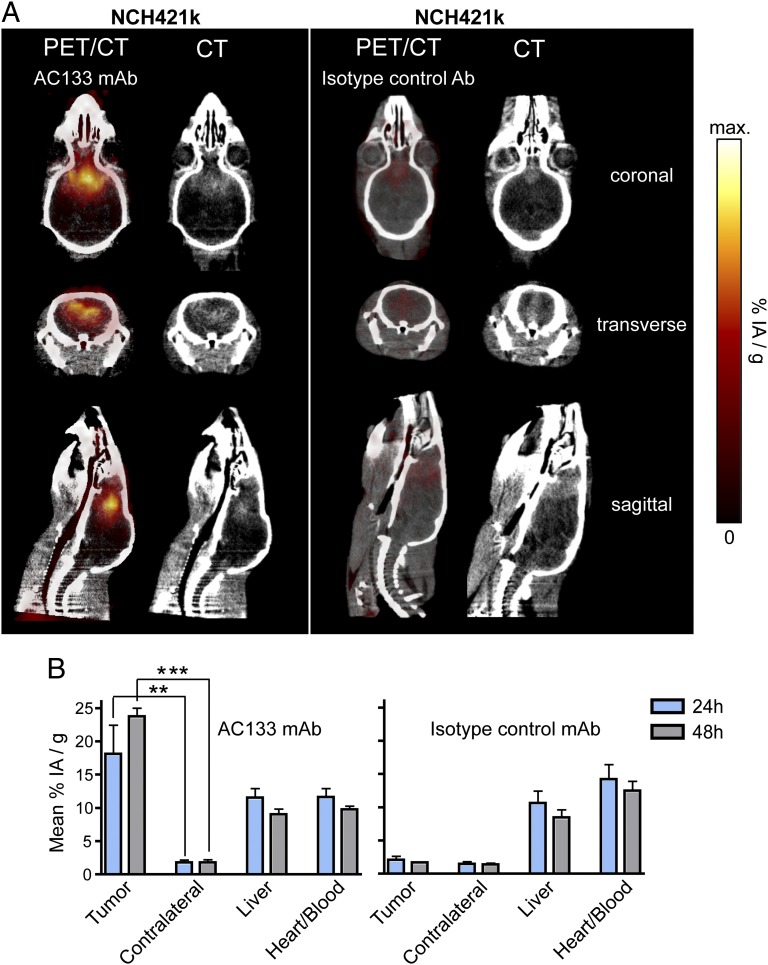
We first performed microPET of intracerebrally growing tumor lesions in mice bearing CD133-overexpressing gliomas. MicroPET after i.v. injection of the 64Cu-NOTA-AC133 mAb permitted the detection not only of relatively large intracerebral U251 gliomas overexpressing CD133 but also of very small ones (Fig. 5A, Left and Center). These small lesions were imaged as soon as 12 d after tumor cell implantation, when they had reached a size of only 2–3 mm in diameter, as determined by contrast-enhanced CT. In contrast, the 64Cu-NOTA-AC133 mAb caused only a very weak signal in AC133− U251 wild-type tumors (Fig. 5A, Right, and Fig. 5B). We then asked whether intracerebral tumors initiated from AC133+ glioma stem cells could be detected also. To this end, NCH421k glioblastoma stem cells were injected into the forebrains of immunodeficient mice. MicroPET after i.v. injection of the 64Cu-NOTA-AC133 mAb clearly allowed the detection of intracerebral xenografts initiated from AC133+ glioblastoma stem cells (Fig. 6A, Left, and Fig. 6B). At 48 h, the uptake ratio of AC133+ NCH421k stem cell-containing tumors to background in the brain was 14 ± 3.4. In contrast, the brain tumor signal caused by the 64Cu-NOTA-isotype control antibody was not significantly above background (Fig. 6A, Right, and Fig. 6B).
Fig. 5.
PET/CT imaging and biodistribution of 64Cu-NOTA-AC133 mAb in mice bearing orthotopic U251 glioma xenografts. Nude mice bearing orthotopic xenografts of U251 glioma cells overexpressing CD133 or orthotopic xenografts of CD133− U251 wild-type cells received 7.5 ± 0.8 MBq 64Cu-NOTA-AC133 mAb via tail vein injection, and PET/CT images were acquired 24 and 48 h p.i. (A) Representative contrast-enhanced microCT and fused microPET/CT sections from mice bearing CD133-overexpressing U251 gliomas and from mice bearing U251 wild-type tumors. For the PET/CT images, an upper threshold corresponding to the maximum tracer uptake of the CD133-overexpressing U251 tumors (43%IA/g) was chosen. (B) Uptake of 64Cu-NOTA-AC133 mAb as determined by microPET in brain tumor, normal brain tissue in the contralateral hemisphere, liver, and in the heart/blood pool. Values are the mean %IA/g of tissue. n = 5–6 mice. ***P < 0.001, t test; values represent means ± SD.
Fig. 6.
PET/CT imaging and biodistribution of 64Cu-NOTA-AC133 and isotype control mAbs in mice bearing orthotopic xenografts initiated from AC133+ glioblastoma stem cells. Mice bearing orthotopic xenografts containing patient-derived NCH421k glioblastoma stem cells were given 7.3 ± 1.9 MBq of either 64Cu-NOTA-AC133 or 64Cu-NOTA-isotype control mAb via tail vein injection, and PET/CT images were acquired. (A) Representative contrast-enhanced microCT and fused microPET/CT sections are shown. For the PET/CT images, an upper threshold corresponding to the maximum 64Cu-NOTA-AC133 mAb uptake (34.6%IA/g) in the NCH421 tumor was chosen. (B) Uptake of both 64Cu-NOTA-AC133 and 64Cu-NOTA-isotype control mAbs as determined by microPET in brain tumor, normal brain tissue in the contralateral hemisphere, liver, and in the heart/blood pool. Values are the mean %IA/g of tissue; n = 5 mice per group. ***P < 0.001, **P < 0.05, t test; values represent means ± SD.
Correlation of MicroPET Images with Histopathologic Tumor Appearance.
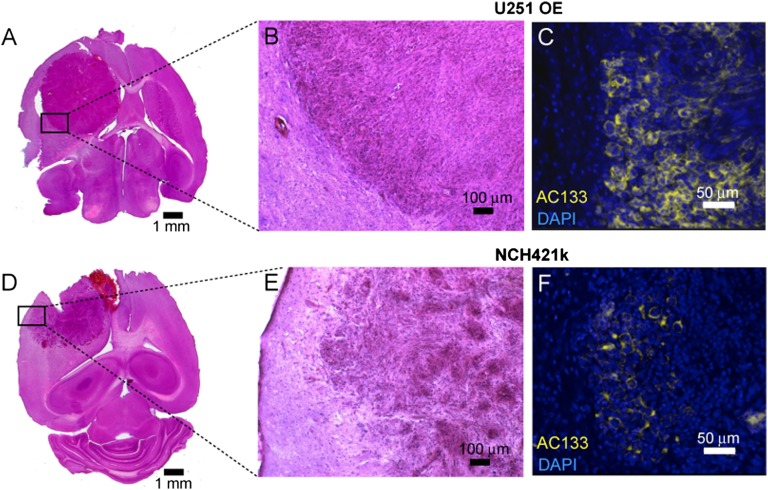
The PET signal of orthotopic CD133-overexpressing U251 gliomas was homogeneous and sharply delineated from the surrounding brain tissue, whereas that of the NCH421k tumors was more heterogeneous. Particularly striking was the reduced and diffuse signal in the periphery of these CSC-derived tumors (compare Fig. 5A, Left and Center and Fig. 6A, Left). These differences in microPET signal intensity correlated with histopathological differences. On H&E-stained brain sections, the U251 tumors appeared as very compact, homogeneous, and well-delineated tumor masses (Fig. 7 A and B), reflecting noninfiltrative growth behavior. This appearance is typical of orthotopic xenografts of conventional glioma cell lines, which generally do not recapitulate the invasive growth pattern that is a major feature (17, 31) of the highly malignant tumor entity human glioblastoma multiforme (38) and certainly contributes to the failure of current therapies. In contrast, NCH421k xenografts exhibited a very different cyto-architecture. The H&E staining appeared more heterogeneous, with a lower intensity in the 1- to 2-mm-thick periphery of the tumors than in the center (Fig. 7D). Higher magnifications revealed tumor cells migrating along blood vessels in the tumor periphery, forming irregular tumor margins (Fig. 7E). Together, these histopathological features reflect the highly infiltrative growth behavior characteristic of human glioblastoma multiforme. AC133 staining of brain tissue sections also revealed a high density (72 ± 20%) of intensely stained AC133-expressing cells in CD133-overexpressing U251 gliomas (Fig. 7C and Fig. S3 B and C). In contrast, the AC133+ cell density (20.0 ± 11%) and cellular AC133 expression levels were much lower in the NCH421k tumors, and the AC133+ tumor cells tended to occur in clusters (Fig. 7F and Fig. S3 B and C). The reduction in the percentage of AC133+ cells compared with the initial 90–100% in the NCH421k implant suspension may be the result mainly of differentiation and the invasive growth behavior. Nonetheless, microPET using the 64Cu-NOTA-AC133 mAb clearly enabled the detection of the highly invasive, CSC-derived NCH421k tumors.
Fig. 7.
Histopathological examination of orthotopically growing brain tumors. Brain sections of mice bearing U251 gliomas overexpressing CD133 or gliomas derived from NCH421k glioblastoma stem cells. (A and D) Brain sections were stained with H&E and scanned by a whole-slide scanner. (Scale bars: 1 mm.) (B and E) Higher-magnification views of the areas boxed in the tumor periphery in A and D including the tumor margins. (C and F) Confocal microscopic images of brain sections encompassing the tumor periphery. The sections were stained with DAPI to visualize nuclei and with PE-labeled AC133 mAb. Note the plasma membrane staining for AC133. Pictures shown are representative of three independent experiments.
Discussion
The AC133 epitope of CD133/prominin is perhaps the most intensely investigated of the known CSC markers (8, 13, 17, 19, 20, 22). Although of great interest and importance, noninvasive imaging of AC133+ CSCs or other types of CSCs with clinically relevant tracers and imaging modalities has not yet been reported (9–11). Here, we demonstrate the successful noninvasive antibody-mediated imaging of AC133+ glioblastoma stem cells by PET and NIR fluorescence imaging in xenograft tumor models in mice. Despite the relatively low AC133 expression on CSCs, we obtained high-quality PET images with the 64Cu-loaded NOTA-conjugated AC133-specific antibody. The AC133 epitope is particularly important as a CSC marker of brain tumors (3, 8, 17, 19, 31), and we demonstrate here the successful noninvasive imaging of intracerebral xenografts with a low density of AC133+ glioblastoma stem cells by both the Alexa 680-labeled AC133 mAb (using FMT) and the 64Cu-NOTA-AC133 mAb (using microPET).
Current imaging techniques used for standard treatment monitoring in patients with solid tumors assess the shrinkage of the tumor as a response criterion. However, when non-CSCs outnumber the CSCs, tumor shrinkage may reflect largely the elimination of treatment-sensitive bulk tumor cells, and whether long-term self-renewing CSCs also are being eliminated can be answered only with CSC-specific tracers and sensitive imaging modalities. Because CSCs also may be responsible for the initiation of local recurrences and distant metastases, specific and sensitive CSC imaging techniques could enable the early detection of these events as well.
PET and NIR fluorescence imaging are sensitive imaging modalities that are becoming increasingly important in clinical practice (27, 30, 39–41). We show here that particularly 64Cu-NOTA-AC133 mAb-mediated microPET yields high-quality, high-resolution images with outstanding tumor-to-background contrast. 64Cu-NOTA-AC133 mAb-mediated microPET permitted the detection of very small brain tumor lesions (2–3 mm in size) and also reflected differences in invasive behavior between orthotopically growing U251 (noninvasive) and NCH421k (invasive) gliomas. Whereas sharply delineated PET signals reflected the compact and spherical microscopic appearance of U251 tumors, more diffuse PET signals seemed to reflect the lower density of AC133+ cells and the chaotic and infiltrative growth pattern detected microscopically in the orthotopic NCH421k tumors. In addition, the 64Cu-NOTA-AC133 mAb could image s.c. xenografts containing AC133+ CSCs clearly. The tracer uptake was significantly higher in these flank tumors than in liver and blood, which cause the highest background signals in immuno-PET with intact antibodies. However, future studies are needed to determine whether intraabdominal tumors with AC133+ CSCs within or close to the liver also can be detected readily using this tracer. AC133− tumors or the 64Cu-labeled isotype control antibody caused only signals as low as background, not exceeding that of the blood pool. Because CSCs show rather low AC133 expression, these data considered together suggest that the 64Cu-NOTA-AC133 mAb is a very specific and highly sensitive tool for the noninvasive detection of AC133+ CSCs.
A proportion of the WHO grade IV glioblastomas and of other tumor entities contains high frequencies of AC133+ cells. The AC133+ fraction among the highly aggressive glioblastomas was reported to range from 19 to 29% (19) or from ≤1–50% (8). In the latter study, a considerable proportion of tumors contained >10% or even >25% AC133+ cells, and the AC133+ cells tended to organize in clusters. For medulloblastomas, 6–21% AC133+ cells have been reported (19), and for teratoid/rhabdoid brain tumors 1–36% have been reported at diagnosis and 30–70% at relapse (42). For colon cancer, 2–19% AC133+ cells were reported, compared with 0.4–2% in the surrounding tissue (20), and for ovarian cancer the reported range is 0.3–35% (43). Other tumor entities (e.g., lower-grade gliomas) usually contain only smaller populations of AC133+ tumor cells (8). For pancreatic cancers the mean is 1.8% (21); and for lung cancers the mean is 5% and that of surrounding tissue is <1% (44). We were able to image diffusely growing glioblastoma xenografts with a low density (mean: 20%) of AC133+ CSCs with high tumor-to-background contrast ratio (mean: 14 at 48 h p.i.). Because specific tumor detection would be possible at a tumor-to-background contrast ratio of 2 or even less, it is likely that much lower frequencies of AC133+ tumor cells could be detectable by 64Cu-NOTA-AC133 PET. Nevertheless, future studies are needed to define more precisely the lower limit of detection in terms of the percentage of tumor cells that can be noninvasively detected using AC133 mAb-based tracers.
High-density areas of normal stem cells also may pose a challenge in CSC imaging, because the currently known CSC markers are shared by subsets of normal tissue stem cells. However, human hematopoietic stem cells, a subset of which is AC133+, constitute only a very minor fraction (<0.2%) of blood cells (45) and express considerably less surface AC133 than NCH421k glioblastoma stem cells. Prostate, liver, kidney, skin, and retina also contain AC133+ stem and progenitor cells (15). The AC133 epitope also is a known marker of fetal human neural stem cells. However, postnatally, the typical stem cell regions in the brain (the subventricular zone and the hippocampus) seem not to contain CD133+ stem cells (46). This absence of stem cells is a good prerequisite for AC133 mAb-based brain tumor imaging. The AC133 epitope can be detected on differentiated human luminal epithelia by immunohistochemistry of tissue fixed in paraformaldehyde (16). However, because fixation strongly affects the recognition of the epitope (13), its accessibility in these epithelia to the AC133 mAb in noninvasive imaging is difficult to predict.
Current imaging techniques used in patients with primary or metastatic brain tumors quantify the tumor burden indirectly through edema, vascular integrity (i.e., contrast enhancement using low-molecular-weight contrast agents), or metabolic activity (mainly using amino acid PET). Although it is known that, in highly malignant brain tumors, the BBB is disturbed and that its permeability is increased for macromolecules (34), reports on antibody-mediated PET of brain tumor lesions are scarce (40). Our microPET imaging and biodistribution analyses show that the 64Cu-NOTA-AC133 mAb is exquisitely suited for imaging orthotopically growing glioblastoma xenografts, indicating that this antibody tracer penetrates these tumors quite efficiently. Antibody-mediated brain tumor CSC imaging also may have advantages over other currently used brain tumor imaging techniques in distinguishing true tumor relapse from pseudoprogression, because it is based on a molecular antigen/antibody interaction that is more tumor-specific than imaging based on vascular integrity or metabolic activity. In addition, our data suggest that noninvasive antibody-mediated imaging could be very useful in general for assessing receptor expression and evaluating antibody therapeutics in neurooncology.
In conclusion, we have developed antibody-based PET and NIR fluorescence tracers that enable the specific and highly sensitive detection of AC133+ CSCs. FMT and PET detection of AC133+ CSCs is important for preclinical studies in human tumor xenograft models. The present study also sets the foundation for the application of fluorescent and positron emitter-labeled AC133 mAbs, or humanized versions thereof, in human studies with clinical NIR fluorescence and PET scanners. In addition, our study confirms that the AC133 mAb also may be useful for therapeutic targeting, including the treatment of brain tumors such as the highly aggressive and presently incurable glioblastoma multiforme (32) as well as for the development of theranostic probes (47).
Methods
Cell Lines.
The U251 cell line was obtained from American Type Culture Collection (ATCC). NCH421k is a glioblastoma stem cell line that was derived directly from a primary glioblastoma tumor sample. The establishment of the CD133-overexpressing U251 glioma cell line and of the NCH421k glioblastoma stem cell line have been described previously (26, 31). NCH421k cells were cultured under normoxic conditions (21% O2) in Neurobasal-A medium (Invitrogen) supplemented with 20 ng/mL EGF and 20 ng/mL FGF-2 (Prospec), 1× penicillin/streptomycin (PAA Laboratories), 0.5× minimum essential medium (MEM) nonessential amino acids, 1× Glutamax-I, and B27 supplement (all from Invitrogen) on normal plastic, where they grew as spheres. CD133-overexpressing U251 cells and wild-type U251 cells were cultured in medium containing 10% (vol/vol) FBS. All three cell lines were transduced with a lentivirus coding for a fusion protein consisting of the latest generation of firefly luciferase and a neomycin resistance cassette (L1-mCherry-IRES-FFneo or L1-FF-IRESneo) constructed in the G.N. laboratory according to standard procedures.
Antibodies.
AC133.1 hybridoma cells (14) (ATCC HB-12346) were cultured in DMEM with Ultra Low IgG FBS (Invitrogen), and the AC133 mAb was purified from the hybridoma supernatant according to standard methods. The IgG1 isotype control antibody was purchased from BioXcell.
Antibody Conjugation with Alexa 680.
Conjugation of Alexa Fluor 680 (Alexa 680) to the AC133 and IgG1 isotype control antibodies was conducted with the Alexa Fluor-680 SAIVI Rapid Antibody Labeling Kit from Invitrogen according to the manufacturer’s instructions. Protein concentrations of the Alexa Fluor 680-conjugated mAbs and the degree of labeling were determined with a Nanodrop 1000 spectrophotometer (Thermo Fisher Scientific).
Antibody Conjugation with NOTA.
To AC133 or the isotype control antibody (in metal-free PBS), p-SCN-Bn-NOTA (Macrocyclics) dissolved in DMSO was added at a ratio of 1:32 (final volume 500 μL, 0.1 M bicarbonate buffer, pH 9.5). Conjugation was allowed to proceed at room temperature for 5 h. Excess chelator was removed by G25-Sephadex size-exclusion chromatography on PD-10 columns. The columns were washed with PBS supplied with 1 μM EDTA to remove metal ions, followed by washing with only PBS. Fractions containing the immunoconjugate were collected in PBS and concentrated to 1 mg/mL with Amicon Ultra 4 centrifugation tubes (Millipore), followed by buffer exchange to ammonium acetate buffer, pH 5.2.
Labeling of NOTA-AC133 mAb with 64Cu.
64CuCl2 was obtained from the Department of Preclinical Imaging and Radiopharmacy, Eberhard Karls University, Tübingen Germany. Labeling was performed in 250-μL ammonium acetate buffer (0.1 M, pH 8.2) with 240 μg NOTA-AC133 or 240 μg isotype control mAb. About 100 MBq 64CuCl2 was added and incubated for 40 min at 37 °C. The labeling reaction was stopped by adding 100 μL 0.1 M EDTA solution to chelate-free 64CuCl2. Quality control was performed by an isocratic HPLC run (Ramona Star HPLC system; Raytest GmbH) on a BioSilect SEC 250–5 size-exclusion column (Bio-Rad Laboratories) with PBS as eluent at a flow rate of 1 mL/min. The retention times for the labeled compounds were (mean ± SD) 6:48 ± 0:11 min and 9:44 ± 0:08 min for the labeled mAb and free 64Cu(EDTA)2−, respectively. Free 64Cu(EDTA)2− was separated by filtrating through an Amicon 10-kDa cutoff filter. Together with this purification step, the labeled compound was buffer exchanged into 0.9% NaCl solution for injection and adjusted to the final volume.
Determination of NOTA Chelators per Antibody.
The number of NOTA chelators per antibody molecule was determined by isotope dilution and confirmed by LC-MS using an Agilent 1260 nano/capillary LC system coupled to an Agilent 6530 QTOF equipped with a Chip-Cube.
Titration of Labeled Antibodies.
Preservation of binding affinity upon labeling with either Alexa 680 or NOTA was ascertained through side-by-side flow cytometric titration analysis of labeled and unlabeled antibody, respectively. Serially diluted antibody was incubated with 5 × 105 HCT116 colon carcinoma cells (which are AC133+) and 5 × 105 p53-deficient HCT116 cells (which are AC133−) suspended in 100 μL FACS buffer (0.5% BSA/2 mM EDTA in PBS) for 15 min. After washing, the cells were incubated with 1.5 μg anti-mouse IgG phycoerythrin (PE)-conjugated F(ab’)2 fragment (Dianova) in 100 μL FACS buffer for 20 min. Samples then were washed twice and analyzed on a FACSVerse (BD Biosciences).
Animal Experiments.
All animal experiments were performed in accordance with the German Animal License Regulations and were approved by the animal care committee of the Regierungspräsidium Freiburg (registration number: G-10/64).
Tumor cell Implantation and Tumor Growth Monitoring.
For s.c. tumor models, 5 × 106 U251 wild-type, CD133-overexpressing U251 or NCH421k cells were implanted into the flanks of 6- to 8-wk-old immunodeficient mice. BALB/c nude and the more immunodeficient NOD/SCID mice (Charles River) were used for U251 and NCH421k cells, respectively. The growth of the xenografts was monitored by caliper measurement. When the s.c. growing tumors reached a size of 500–1,000 mm3, they were used for in vivo imaging experiments. For orthotopic brain tumor models, 2.5 × 105 wild-type or CD133-overexpressing U251 or NCH421k cells stably transduced with luciferase were implanted manually 3 mm anterior and 3 mm to the right of the bregma in the brains of 6- to 8-wk-old nude mice. The cells, which were suspended in 4 μL PBS, were injected at a depth of 3 mm with a Hamilton syringe, which was held in position for 5 min. After the injection, the surface was cleaned with a sterile cotton swab, and the burr hole was filled with bone wax. Thereafter, tumor growth was monitored noninvasively using in vivo bioluminescence imaging on an IVIS spectrum imaging system (PerkinElmer) three times per week (Fig. S2D). Mice with orthotopic brain tumors were imaged with FMT or PET when the bioluminescence signal reached an intensity between 5 × 107 and 1 × 108 photons/s. When the NCH421k tumor-bearing mice met the above criteria, they were randomized into groups and were injected with either AC133 or isotype control antibodies.
FMT in Vivo Imaging.
Alexa 680-labeled AC133 or isotype control mAb (70 μg) was injected i.v. The fluorescence signals were visualized using an FMT1500 system (PerkinElmer) at several time points p.i. The mice were anesthetized by gas anesthesia (isoflurane/oxygen mixture) and then were placed into an imaging cassette. After the cassette was positioned in the FMT1500 imaging system, reflectance images were captured in white light and fluorescence (2D planar). For 3D imaging, a field enclosing the tumor was selected, and the tomographic scan was carried out. The scan data were analyzed using reconstruction software provided by the manufacturer (PerkinElmer). For tomographic data analysis, 3D regions of interest were drawn around the tumor, and the total amount of fluorochrome (in picomoles) was calculated by TrueQuant software (PerkinElmer), using calibrated standards of the labeled mAbs.
PET and CT Imaging.
Mice were injected i.v. with either 30 μg 64Cu-NOTA-AC133 or isotype control mAb. PET imaging was performed with a microPET Focus 120 (Concorde). To account for the physical decay of 64Cu, the acquisition time was 20 min at 24 h p.i. and 45 min at 48 h p.i. PET acquisition was followed immediately by CT imaging (CT Imaging microCT scanner). For studies of intracranial tumors, mice were injected i.v. with 100 μL Imeron 350 as contrast agent immediately before the start of the CT scan. The head region of each mouse was scanned in one bed position for 90 s using a 360° rotation step, a tube voltage of 40 keV, and a tube current of 1 mA.
PET and CT Image Analysis.
PET images were reconstructed by the routine 2D ordered subset expectation maximization (OSEM2D) algorithm provided by the scanner software, as previously described (48). The resolution of the reconstructed images ranged between 1.5 and 1.7 mm. Quantitative analysis of radiotracer uptake was performed with AMIDE software, and the reported values represent the mean activity concentration expressed as percent of injected activity per gram of tissue (%IA/g), assuming a tissue density of 1 g/mL. Image contrast was assessed by calculating the ratio of radiotracer uptake to contralateral brain, left ventricular cavity, and liver. Images of the CT scans were reconstructed with a voxel size of 0.12 × 0.12 × 0.12 mm and a T30 kernel, using the software provided by the manufacturer. Fusion of the PET and CT images was performed by ROVER software (ABX).
Ex Vivo Biodistribution.
After the imaging studies, the animals were killed by cervical dislocation, and tumor and other organs were sampled. In animals injected with fluorescent antibody, either ex vivo FMT imaging or FACS analysis of tumor single-cell suspensions (as described below) was performed. After PET/CT imaging, radioactivity within the tumor and the normal organs was measured using a gamma counter Packard Cobra II (PerkinElmer). All values were decay-corrected and expressed as %IA/g ± SD, by measuring a standard of known activity from the respective injected probe.
Histopathology.
Paraformaldehyde (4%)-fixed brains were cut in a Vibratome (Leica VT-1000S; Leica) in horizontal sections 60 μm apart. Brain sections were mounted and stained with H&E. For analysis of AC133 expression in tumor tissue, 60-μm sections were stained using a PE-labeled antibody against human AC133.1 (Miltenyi Biotec). For negative control, slices were stained with PE-labeled IgG1 isotype control antibody. After washings and nuclear staining with DAPI, sections were mounted and analyzed with a krypton-argon laser scanning confocal imaging system (TCSNT; Leica Microsystems AG).
FACS Analysis of Tumor Single-Cell Suspensions.
At 96 h after injection of Alexa 680-labeled mAbs, pieces of s.c. grown tumors were digested with 0.7 U/mL Liberase-Blendzyme (Roche)/Accutase (eBioscience)/100 U DNase I (Invitrogen)/10 mM MgCl2 for 30 min at 37 °C. Nondigested pieces were digested further with Accutase for 30 min and then were pressed through a cell strainer. Red blood cells were removed using ice-cold RBC Lysis Buffer (eBioscience). Cells then were washed with PBS, resuspended in FACS buffer, and transferred through a preseparation filter. The cells were incubated with FcR blocking reagent (Miltenyi) for 10 min, followed by incubation with 5 μg/mL anti-AC141-PE (Miltenyi), and then were analyzed by flow cytometry.
FACS Analysis of AC133 Expression on Cultured Tumor Cells.
Tumor cells (CD133-overexpressing U251 and wild-type U251 as well as NCH421k cells) were collected, incubated with FcR blocking reagent, stained with anti-AC133.1-PE antibody from Miltenyi Biotec, and then subjected to FACS analysis.
Statistical Analysis.
Results are presented as means ± SD. Data were compared using the unpaired two-tailed Student t test. A P value <0.05 was considered significant. Analyses were performed using GraphPad Prism software version 6.0 (GraphPad Software Inc.).
Supplementary Material
Acknowledgments
We thank Dr. Felix Heinemann and Dr. Ursula Nestle for discussions. This work was supported by a grant from the Clotten Foundation.
Footnotes
The authors declare no conflict of interest.
This article is a PNAS Direct Submission.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1314189111/-/DCSupplemental.
References
- 1.Nguyen LV, Vanner R, Dirks P, Eaves CJ. Cancer stem cells: An evolving concept. Nat Rev Cancer. 2012;12(2):133–143. doi: 10.1038/nrc3184. [DOI] [PubMed] [Google Scholar]
- 2.Medema JP. Cancer stem cells: The challenges ahead. Nat Cell Biol. 2013;15(4):338–344. doi: 10.1038/ncb2717. [DOI] [PubMed] [Google Scholar]
- 3.Eyler CE, et al. Brain cancer stem cells display preferential sensitivity to Akt inhibition. Stem Cells. 2008;26(12):3027–3036. doi: 10.1634/stemcells.2007-1073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Maugeri-Saccà M, Di Martino S, De Maria R. Biological and clinical implications of cancer stem cells in primary brain tumors. Front Oncol. 2013;3:6. doi: 10.3389/fonc.2013.00006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chen J, et al. A restricted cell population propagates glioblastoma growth after chemotherapy. Nature. 2012;488(7412):522–526. doi: 10.1038/nature11287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Driessens G, Beck B, Caauwe A, Simons BD, Blanpain C. Defining the mode of tumour growth by clonal analysis. Nature. 2012;488(7412):527–530. doi: 10.1038/nature11344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schepers AG, et al. Lineage tracing reveals Lgr5+ stem cell activity in mouse intestinal adenomas. Science. 2012;337(6095):730–735. doi: 10.1126/science.1224676. [DOI] [PubMed] [Google Scholar]
- 8.Zeppernick F, et al. Stem cell marker CD133 affects clinical outcome in glioma patients. Clin Cancer Res. 2008;14(1):123–129. doi: 10.1158/1078-0432.CCR-07-0932. [DOI] [PubMed] [Google Scholar]
- 9.Shigdar S, et al. Cancer stem cell targeting: The next generation of cancer therapy and molecular imaging. Ther Deliv. 2012;3(2):227–244. doi: 10.4155/tde.11.148. [DOI] [PubMed] [Google Scholar]
- 10.Xia T, Jiang H, Li C, Tian M, Zhang H. Molecular imaging in tracking tumor stem-like cells. J Biomed Biotechnol. 2012;2012:420364. doi: 10.1155/2012/420364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Baker M. Cancer stem cells tracked. Nature. 2012;488(7409):13–14. doi: 10.1038/488013a. [DOI] [PubMed] [Google Scholar]
- 12.Corbeil D, et al. The human AC133 hematopoietic stem cell antigen is also expressed in epithelial cells and targeted to plasma membrane protrusions. J Biol Chem. 2000;275(8):5512–5520. doi: 10.1074/jbc.275.8.5512. [DOI] [PubMed] [Google Scholar]
- 13.Kemper K, et al. The AC133 epitope, but not the CD133 protein, is lost upon cancer stem cell differentiation. Cancer Res. 2010;70(2):719–729. doi: 10.1158/0008-5472.CAN-09-1820. [DOI] [PubMed] [Google Scholar]
- 14.Yin AH, et al. AC133, a novel marker for human hematopoietic stem and progenitor cells. Blood. 1997;90(12):5002–5012. [PubMed] [Google Scholar]
- 15.Grosse-Gehling P, et al. CD133 as a biomarker for putative cancer stem cells in solid tumours: Limitations, problems and challenges. J Pathol. 2013;229(3):355–378. doi: 10.1002/path.4086. [DOI] [PubMed] [Google Scholar]
- 16.Shmelkov SV, et al. CD133 expression is not restricted to stem cells, and both CD133+ and CD133- metastatic colon cancer cells initiate tumors. J Clin Invest. 2008;118(6):2111–2120. doi: 10.1172/JCI34401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chen R, et al. A hierarchy of self-renewing tumor-initiating cell types in glioblastoma. Cancer Cell. 2010;17(4):362–375. doi: 10.1016/j.ccr.2009.12.049. [DOI] [PubMed] [Google Scholar]
- 18.Beier D, et al. CD133(+) and CD133(-) glioblastoma-derived cancer stem cells show differential growth characteristics and molecular profiles. Cancer Res. 2007;67(9):4010–4015. doi: 10.1158/0008-5472.CAN-06-4180. [DOI] [PubMed] [Google Scholar]
- 19.Singh SK, et al. Identification of human brain tumour initiating cells. Nature. 2004;432(7015):396–401. doi: 10.1038/nature03128. [DOI] [PubMed] [Google Scholar]
- 20.O’Brien CA, Pollett A, Gallinger S, Dick JE. A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature. 2007;445(7123):106–110. doi: 10.1038/nature05372. [DOI] [PubMed] [Google Scholar]
- 21.Hermann PC, et al. Distinct populations of cancer stem cells determine tumor growth and metastatic activity in human pancreatic cancer. Cell Stem Cell. 2007;1(3):313–323. doi: 10.1016/j.stem.2007.06.002. [DOI] [PubMed] [Google Scholar]
- 22.Mizrak D, Brittan M, Alison M. CD133: Molecule of the moment. J Pathol. 2008;214(1):3–9. doi: 10.1002/path.2283. [DOI] [PubMed] [Google Scholar]
- 23.Mohyeldin A, Garzón-Muvdi T, Quiñones-Hinojosa A. Oxygen in stem cell biology: A critical component of the stem cell niche. Cell Stem Cell. 2010;7(2):150–161. doi: 10.1016/j.stem.2010.07.007. [DOI] [PubMed] [Google Scholar]
- 24.Kahlert UD, et al. Resistance to hypoxia-induced, BNIP3-mediated cell death contributes to an increase in a CD133-positive cell population in human glioblastomas in vitro. J Neuropathol Exp Neurol. 2012;71(12):1086–1099. doi: 10.1097/NEN.0b013e3182772d83. [DOI] [PubMed] [Google Scholar]
- 25.Beck B, Blanpain C. Unravelling cancer stem cell potential. Nat Rev Cancer. 2013;13(10):727–738. doi: 10.1038/nrc3597. [DOI] [PubMed] [Google Scholar]
- 26.Tsurumi C, et al. Non-invasive in vivo imaging of tumor-associated CD133/prominin. PLoS ONE. 2010;5(12):e15605. doi: 10.1371/journal.pone.0015605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kosaka N, Ogawa M, Choyke PL, Kobayashi H. Clinical implications of near-infrared fluorescence imaging in cancer. Future Oncol. 2009;5(9):1501–1511. doi: 10.2217/fon.09.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Anderson CJ, Ferdani R. Copper-64 radiopharmaceuticals for PET imaging of cancer: Advances in preclinical and clinical research. Cancer Biother Radiopharm. 2009;24(4):379–393. doi: 10.1089/cbr.2009.0674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cooper MS, et al. Comparison of (64)Cu-Complexing Bifunctional Chelators for Radioimmunoconjugation: Labeling Efficiency, Specific Activity, and in Vitro/in Vivo Stability. Bioconjug Chem. 2012;23:1029–1039. doi: 10.1021/bc300037w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.van Dam GM, et al. Intraoperative tumor-specific fluorescence imaging in ovarian cancer by folate receptor-α targeting: First in-human results. Nat Med. 2011;17(10):1315–1319. doi: 10.1038/nm.2472. [DOI] [PubMed] [Google Scholar]
- 31.Campos B, et al. Differentiation therapy exerts antitumor effects on stem-like glioma cells. Clin Cancer Res. 2010;16(10):2715–2728. doi: 10.1158/1078-0432.CCR-09-1800. [DOI] [PubMed] [Google Scholar]
- 32.Bach P, et al. Specific elimination of CD133+ tumor cells with targeted oncolytic measles virus. Cancer Res. 2013;73(2):865–874. doi: 10.1158/0008-5472.CAN-12-2221. [DOI] [PubMed] [Google Scholar]
- 33.Nakata S, et al. LGR5 is a marker of poor prognosis in glioblastoma and is required for survival of brain cancer stem-like cells. Brain Pathol. 2013;23(1):60–72. doi: 10.1111/j.1750-3639.2012.00618.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lampson LA. Monoclonal antibodies in neuro-oncology: Getting past the blood-brain barrier. MAbs. 2011;3(2):153–160. doi: 10.4161/mabs.3.2.14239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Chen KS, Mitchell DA. Monoclonal antibody therapy for malignant glioma. Adv Exp Med Biol. 2012;746:121–141. doi: 10.1007/978-1-4614-3146-6_10. [DOI] [PubMed] [Google Scholar]
- 36.Knowles SM, Wu AM. Advances in immuno-positron emission tomography: Antibodies for molecular imaging in oncology. J Clin Oncol. 2012;30(31):3884–3892. doi: 10.1200/JCO.2012.42.4887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Prabhakar U, et al. Challenges and key considerations of the enhanced permeability and retention effect for nanomedicine drug delivery in oncology. Cancer Res. 2013;73(8):2412–2417. doi: 10.1158/0008-5472.CAN-12-4561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Yi D, Hua TX, Lin HY. EGFR gene overexpression retained in an invasive xenograft model by solid orthotopic transplantation of human glioblastoma multiforme into nude mice. Cancer Invest. 2011;29(3):229–239. doi: 10.3109/07357907.2010.550665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Marshall MV, et al. Near-infrared fluorescence imaging in humans with indocyanine green: A review and update. Open Surg Oncol J. 2010;2(2):12–25. doi: 10.2174/1876504101002010012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Dijkers EC, et al. Biodistribution of 89Zr-trastuzumab and PET imaging of HER2-positive lesions in patients with metastatic breast cancer. Clin Pharmacol Ther. 2010;87(5):586–592. doi: 10.1038/clpt.2010.12. [DOI] [PubMed] [Google Scholar]
- 41.Vach W, Høilund-Carlsen PF, Gerke O, Weber WA. Generating evidence for clinical benefit of PET/CT in diagnosing cancer patients. J Nucl Med. 2011;52(Suppl 2):77S–85S. doi: 10.2967/jnumed.110.085704. [DOI] [PubMed] [Google Scholar]
- 42.Chiou SH, et al. Identification of CD133-positive radioresistant cells in atypical teratoid/rhabdoid tumor. PLoS ONE. 2008;3(5):e2090. doi: 10.1371/journal.pone.0002090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Curley MD, et al. CD133 expression defines a tumor initiating cell population in primary human ovarian cancer. Stem Cells. 2009;27(12):2875–2883. doi: 10.1002/stem.236. [DOI] [PubMed] [Google Scholar]
- 44.Bertolini G, et al. Highly tumorigenic lung cancer CD133+ cells display stem-like features and are spared by cisplatin treatment. Proc Natl Acad Sci USA. 2009;106(38):16281–16286. doi: 10.1073/pnas.0905653106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ebener U, Brinkmann A, Zotova V, Niegemann E, Wehner S. [Expression of AC133 vs. CD34 in acute childhood leukemias] Klin Padiatr. 2000;212(3):90–98. doi: 10.1055/s-2000-9659. [DOI] [PubMed] [Google Scholar]
- 46.Pfenninger CV, et al. CD133 is not present on neurogenic astrocytes in the adult subventricular zone, but on embryonic neural stem cells, ependymal cells, and glioblastoma cells. Cancer Res. 2007;67(12):5727–5736. doi: 10.1158/0008-5472.CAN-07-0183. [DOI] [PubMed] [Google Scholar]
- 47.Mitsunaga M, et al. Cancer cell-selective in vivo near infrared photoimmunotherapy targeting specific membrane molecules. Nat Med. 2011;17(12):1685–1691. doi: 10.1038/nm.2554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Dumont RA, et al. Novel (64)Cu- and (68)Ga-labeled RGD conjugates show improved PET imaging of α(ν)β(3) integrin expression and facile radiosynthesis. J Nucl Med. 2011;52(8):1276–1284. doi: 10.2967/jnumed.111.087700. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.