Abstract
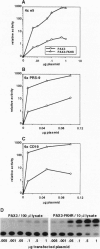
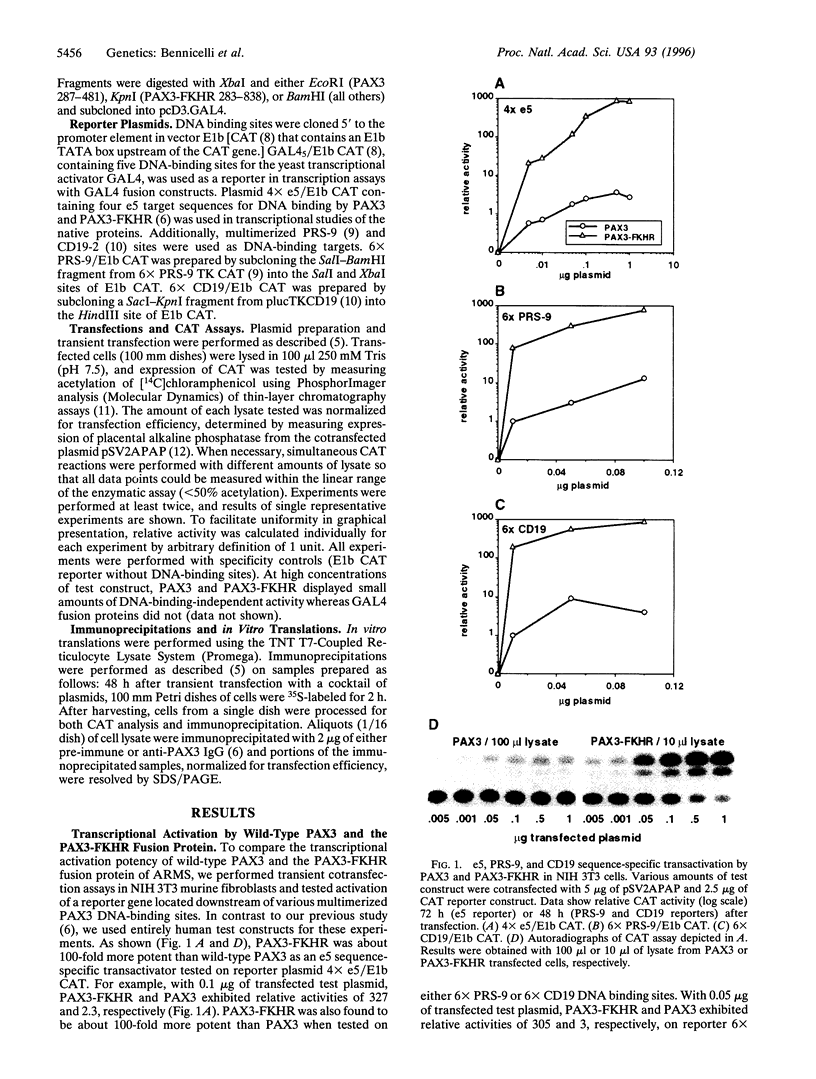
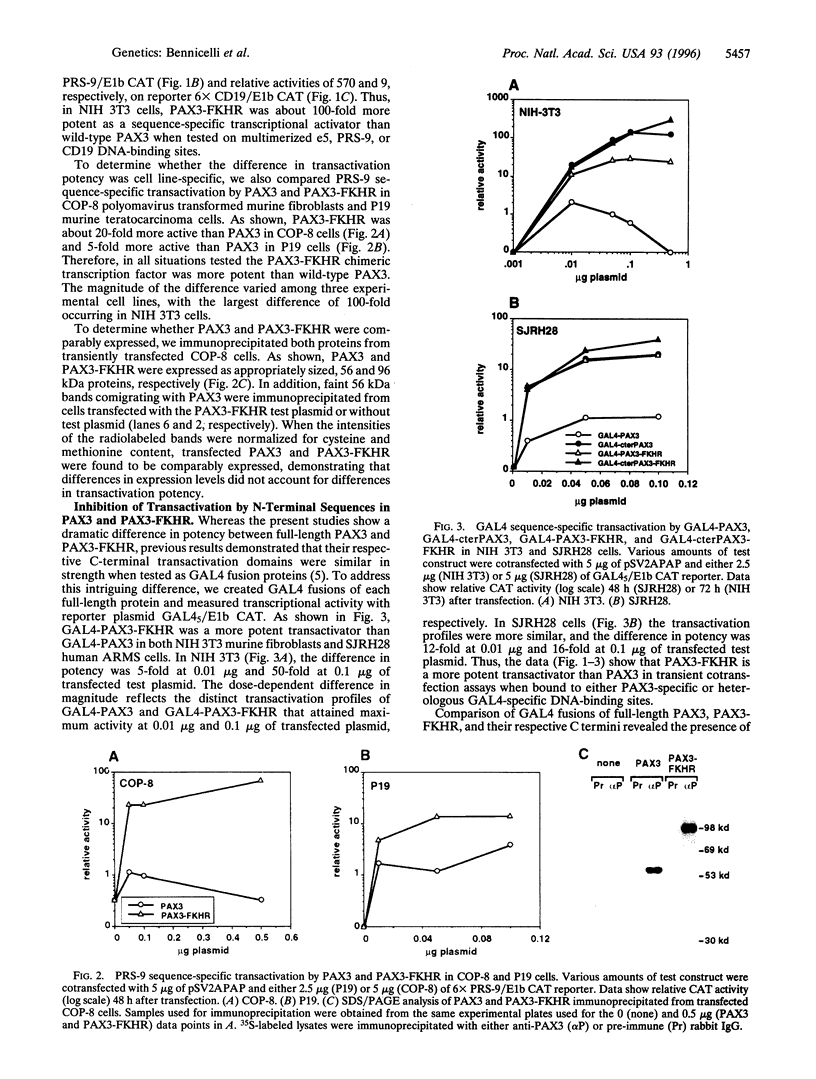
The t(2;13) translocation of alveolar rhabdomyosarcoma results in tumor-specific expression of a chimeric transcription factor containing the N-terminal DNA-binding domain of PAX3 and the C-terminal transactivation domain of FKHR. Here we have tested the hypothesis that PAX3-FKHR gains function relative to PAX3 as a consequence of switching PAX3 and FKHR transactivation domains, which were previously shown to have similar potency but distinct structural motifs. In transient cotransfection assays with human expression constructs, we have demonstrated the increased ability of PAX3-FKHR to activate transcription of a reporter gene located downstream of multimerized e5, PRS-9, or CD19 DNA-binding sites in three cell lines. For example, PAX3-FKHR was 100-fold more potent than PAX3 as an activator binding to e5 sites in NIH 3T3 cells. To compare transactivation potency independent of PAX3-specific DNA binding, we tested GAL4 fusions of full-length PAX3 and PAX3-FKHR or their respective C-terminal transactivation domains on a reporter with GAL4 DNA-binding sites. In this context, full-length PAX3-FKHR was also much more potent than PAX3. Additionally, the activity of each full-length protein was decreased relative to its C-terminal domain, demonstrating that N-terminal sequences are inhibitory. By deletion analysis, we mapped a bipartite cis-acting inhibitory domain to the same subregions within the DNA-binding domains of both PAX3 and PAX3-FKHR. We have shown, however, that the structurally distinct transactivation domains of PAX3 and PAX3-FKHR differ 10- to 100-fold in their susceptibility to inhibition, thus elucidating a mechanism by which PAX3 gains enhanced function during oncogenesis.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baichwal V. R., Tjian R. Control of c-Jun activity by interaction of a cell-specific inhibitor with regulatory domain delta: differences between v- and c-Jun. Cell. 1990 Nov 16;63(4):815–825. doi: 10.1016/0092-8674(90)90147-7. [DOI] [PubMed] [Google Scholar]
- Barr F. G., Galili N., Holick J., Biegel J. A., Rovera G., Emanuel B. S. Rearrangement of the PAX3 paired box gene in the paediatric solid tumour alveolar rhabdomyosarcoma. Nat Genet. 1993 Feb;3(2):113–117. doi: 10.1038/ng0293-113. [DOI] [PubMed] [Google Scholar]
- Bennicelli J. L., Fredericks W. J., Wilson R. B., Rauscher F. J., 3rd, Barr F. G. Wild type PAX3 protein and the PAX3-FKHR fusion protein of alveolar rhabdomyosarcoma contain potent, structurally distinct transcriptional activation domains. Oncogene. 1995 Jul 6;11(1):119–130. [PubMed] [Google Scholar]
- Brown H. J., Sutherland J. A., Cook A., Bannister A. J., Kouzarides T. An inhibitor domain in c-Fos regulates activation domains containing the HOB1 motif. EMBO J. 1995 Jan 3;14(1):124–131. doi: 10.1002/j.1460-2075.1995.tb06982.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chalepakis G., Fritsch R., Fickenscher H., Deutsch U., Goulding M., Gruss P. The molecular basis of the undulated/Pax-1 mutation. Cell. 1991 Sep 6;66(5):873–884. doi: 10.1016/0092-8674(91)90434-z. [DOI] [PubMed] [Google Scholar]
- Chalepakis G., Jones F. S., Edelman G. M., Gruss P. Pax-3 contains domains for transcription activation and transcription inhibition. Proc Natl Acad Sci U S A. 1994 Dec 20;91(26):12745–12749. doi: 10.1073/pnas.91.26.12745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis R. J., D'Cruz C. M., Lovell M. A., Biegel J. A., Barr F. G. Fusion of PAX7 to FKHR by the variant t(1;13)(p36;q14) translocation in alveolar rhabdomyosarcoma. Cancer Res. 1994 Jun 1;54(11):2869–2872. [PubMed] [Google Scholar]
- Dubendorff J. W., Whittaker L. J., Eltman J. T., Lipsick J. S. Carboxy-terminal elements of c-Myb negatively regulate transcriptional activation in cis and in trans. Genes Dev. 1992 Dec;6(12B):2524–2535. doi: 10.1101/gad.6.12b.2524. [DOI] [PubMed] [Google Scholar]
- Epstein J. A., Lam P., Jepeal L., Maas R. L., Shapiro D. N. Pax3 inhibits myogenic differentiation of cultured myoblast cells. J Biol Chem. 1995 May 19;270(20):11719–11722. doi: 10.1074/jbc.270.20.11719. [DOI] [PubMed] [Google Scholar]
- Fredericks W. J., Galili N., Mukhopadhyay S., Rovera G., Bennicelli J., Barr F. G., Rauscher F. J., 3rd The PAX3-FKHR fusion protein created by the t(2;13) translocation in alveolar rhabdomyosarcomas is a more potent transcriptional activator than PAX3. Mol Cell Biol. 1995 Mar;15(3):1522–1535. doi: 10.1128/mcb.15.3.1522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galili N., Davis R. J., Fredericks W. J., Mukhopadhyay S., Rauscher F. J., 3rd, Emanuel B. S., Rovera G., Barr F. G. Fusion of a fork head domain gene to PAX3 in the solid tumour alveolar rhabdomyosarcoma. Nat Genet. 1993 Nov;5(3):230–235. doi: 10.1038/ng1193-230. [DOI] [PubMed] [Google Scholar]
- Gorman C. M., Moffat L. F., Howard B. H. Recombinant genomes which express chloramphenicol acetyltransferase in mammalian cells. Mol Cell Biol. 1982 Sep;2(9):1044–1051. doi: 10.1128/mcb.2.9.1044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goulding M. D., Chalepakis G., Deutsch U., Erselius J. R., Gruss P. Pax-3, a novel murine DNA binding protein expressed during early neurogenesis. EMBO J. 1991 May;10(5):1135–1147. doi: 10.1002/j.1460-2075.1991.tb08054.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henthorn P., Zervos P., Raducha M., Harris H., Kadesch T. Expression of a human placental alkaline phosphatase gene in transfected cells: use as a reporter for studies of gene expression. Proc Natl Acad Sci U S A. 1988 Sep;85(17):6342–6346. doi: 10.1073/pnas.85.17.6342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johansen F. E., Prywes R. Identification of transcriptional activation and inhibitory domains in serum response factor (SRF) by using GAL4-SRF constructs. Mol Cell Biol. 1993 Aug;13(8):4640–4647. doi: 10.1128/mcb.13.8.4640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lillie J. W., Green M. R. Transcription activation by the adenovirus E1a protein. Nature. 1989 Mar 2;338(6210):39–44. doi: 10.1038/338039a0. [DOI] [PubMed] [Google Scholar]
- Pruitt S. C. Expression of Pax-3- and neuroectoderm-inducing activities during differentiation of P19 embryonal carcinoma cells. Development. 1992 Nov;116(3):573–583. doi: 10.1242/dev.116.3.573. [DOI] [PubMed] [Google Scholar]
- Sadowski I., Ptashne M. A vector for expressing GAL4(1-147) fusions in mammalian cells. Nucleic Acids Res. 1989 Sep 25;17(18):7539–7539. doi: 10.1093/nar/17.18.7539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schäfer B. W., Czerny T., Bernasconi M., Genini M., Busslinger M. Molecular cloning and characterization of a human PAX-7 cDNA expressed in normal and neoplastic myocytes. Nucleic Acids Res. 1994 Nov 11;22(22):4574–4582. doi: 10.1093/nar/22.22.4574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tremblay P., Gruss P. Pax: genes for mice and men. Pharmacol Ther. 1994;61(1-2):205–226. doi: 10.1016/0163-7258(94)90063-9. [DOI] [PubMed] [Google Scholar]
- Turc-Carel C., Lizard-Nacol S., Justrabo E., Favrot M., Philip T., Tabone E. Consistent chromosomal translocation in alveolar rhabdomyosarcoma. Cancer Genet Cytogenet. 1986 Jan 15;19(3-4):361–362. doi: 10.1016/0165-4608(86)90069-5. [DOI] [PubMed] [Google Scholar]
- Zappavigna V., Sartori D., Mavilio F. Specificity of HOX protein function depends on DNA-protein and protein-protein interactions, both mediated by the homeo domain. Genes Dev. 1994 Mar 15;8(6):732–744. doi: 10.1101/gad.8.6.732. [DOI] [PubMed] [Google Scholar]