Abstract
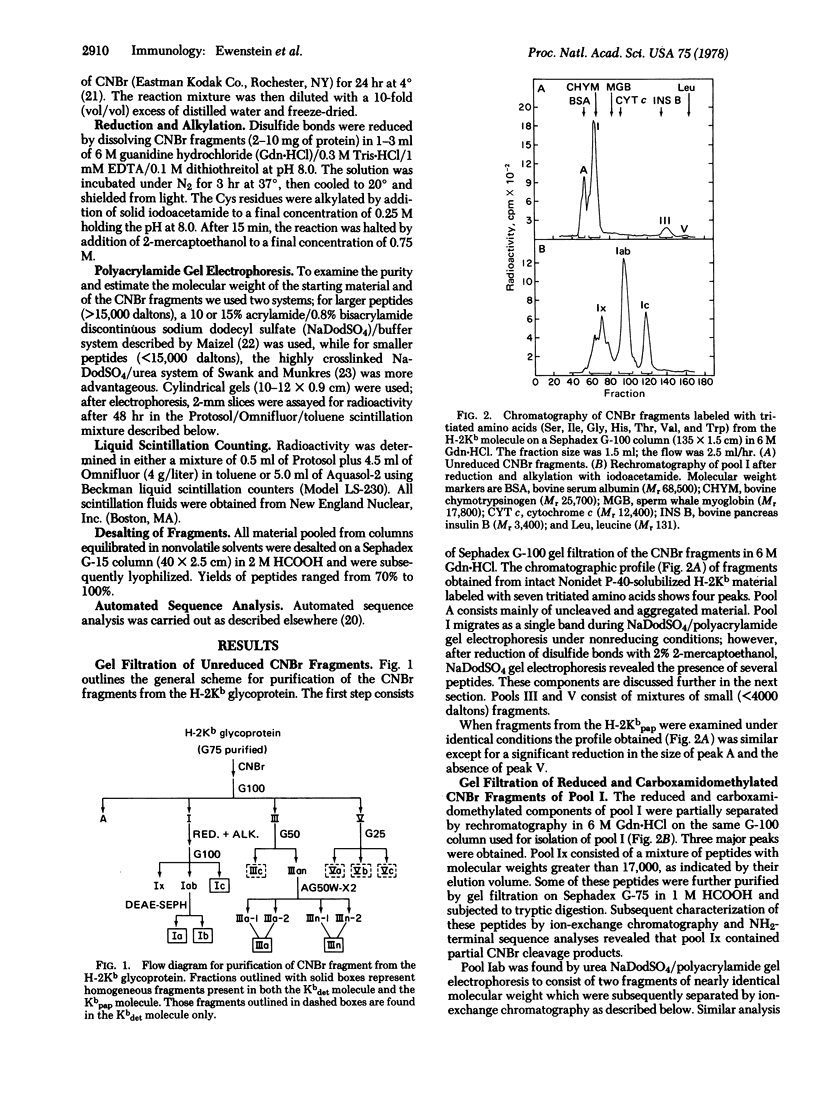
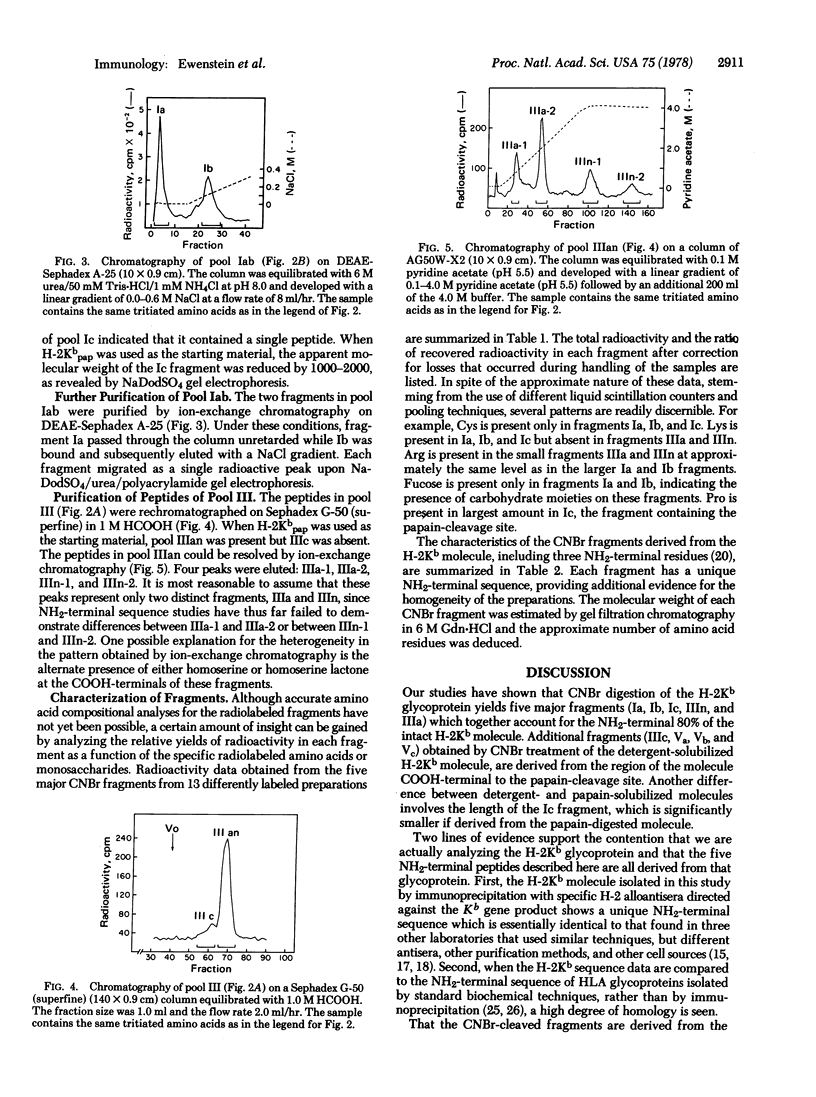
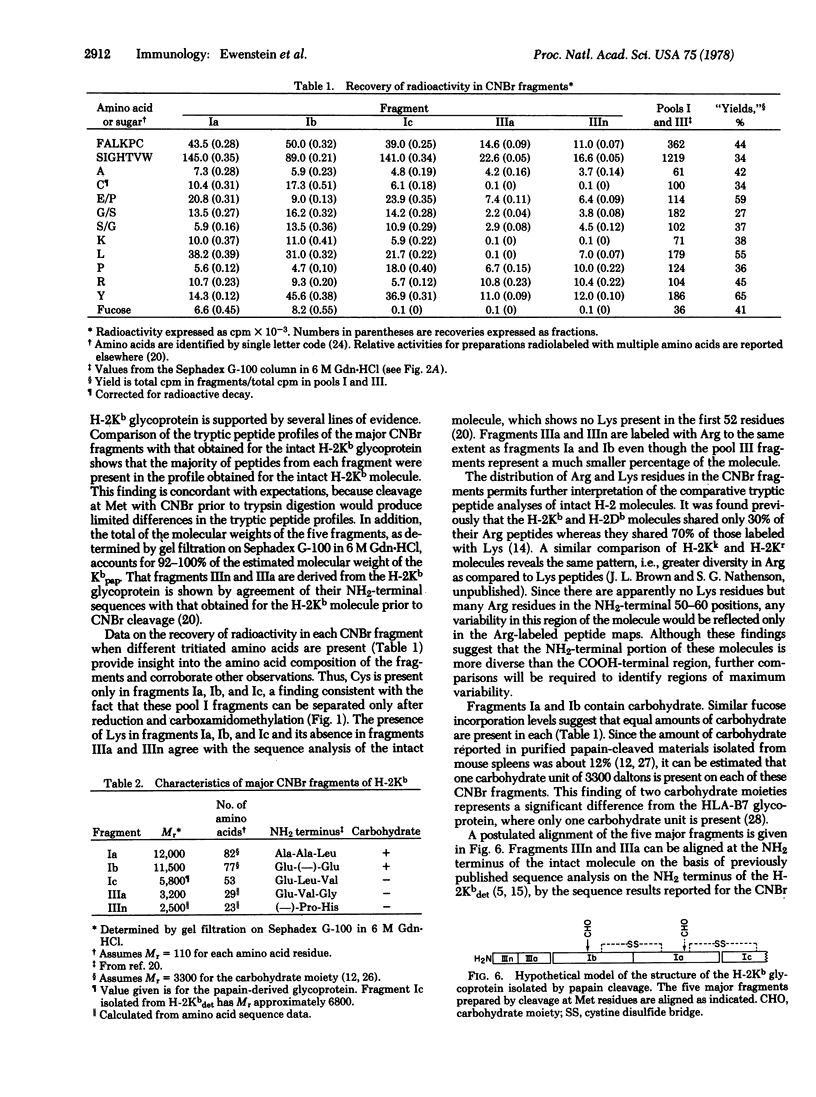
Radiochemical methodology has been applied in studies directed toward the determination of the amino acid sequence of the murine H-2Kb gene product. Five major CNBr fragments, which comprise the NH2-terminal 80% of the intact glycoprotein and include the H-2Kb fragment isolated by papain cleavage, were isolated and characterized. By means of amino-terminal sequence analysis, homology to other products of the major histocompatibility complex, identification of partial CNBr cleavage products, and localization of the site of papain cleavage, it was possible to align the fragments obtained from the molecule in the following order: the NH2-terminal fragment, designated IIIn, contains 23 residues and is followed by fragment IIIa, which contains 29 residues. Two peptides Ib (Mr 11,500) and Ia (Mr 12,000), each of which contains a carbohydrate moiety, are followed by fragment Ic (Mr 5800), which includes the site of papain cleavage. The data indicate the presence of disulfide bonds between fragments Ib and Ia and between Ia and Ic.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bailey D. W., Kohn H. I. Inherited histocompatibility changes in progeny of irradiated and unirradiated inbred mice. Genet Res. 1965 Nov;6(3):330–340. doi: 10.1017/s0016672300004225. [DOI] [PubMed] [Google Scholar]
- Brown J. L., Kato K., Silver J., Nathenson S. G. Notable diversity in peptide composition of murine H-2K and H-2D alloantigens. Biochemistry. 1974 Jul 16;13(15):3174–3178. doi: 10.1021/bi00712a027. [DOI] [PubMed] [Google Scholar]
- Brown J. L., Nathenson S. G. Structural differences between parent and mutant H-2K glycoproteins from two H-2K gene mutants: b6.c-h-2ba (Hzl) and B6-H-2bd (M505). J Immunol. 1977 Jan;118(1):98–102. [PubMed] [Google Scholar]
- Doherty P. C., Blanden R. V., Zinkernagel R. M. Specificity of virus-immune effector T cells for H-2K or H-2D compatible interactions: implications for H-antigen diversity. Transplant Rev. 1976;29:89–124. doi: 10.1111/j.1600-065x.1976.tb00198.x. [DOI] [PubMed] [Google Scholar]
- Ewenstein B. M., Freed J. H., Mole L. E., Nathenson S. G. Localization of the papain cleavage site of H-2 glycoproteins. Proc Natl Acad Sci U S A. 1976 Mar;73(3):915–918. doi: 10.1073/pnas.73.3.915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henning R., Milner R. J., Reske K., Cunningham B. A., Edelman G. M. Subunit structure, cell surface orientation, and partial amino-acid sequences of murine histocompatibility antigens. Proc Natl Acad Sci U S A. 1976 Jan;73(1):118–122. doi: 10.1073/pnas.73.1.118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muramatsu T., Nathenson S. G. Studies on the carbohydrate portion of membrane-located mouse H-2 alloantigens. Biochemistry. 1970 Dec 8;9(25):4875–4883. doi: 10.1021/bi00827a008. [DOI] [PubMed] [Google Scholar]
- Natori T., Tanigaki N., Appella E., Pressman D. Amino acid composition and physicochemical properties of mouse beta2-microglobulin. Biochem Biophys Res Commun. 1975 Jul 22;65(2):611–617. doi: 10.1016/s0006-291x(75)80190-2. [DOI] [PubMed] [Google Scholar]
- Parham P., Alpert B. N., Orr H. T., Strominger J. L. Carbohydrate moiety of HLA antigens. Antigenic properties and amino acid sequences around the site of glycosylation. J Biol Chem. 1977 Nov 10;252(21):7555–7567. [PubMed] [Google Scholar]
- Paul W. E., Benacerraf B. Functional specificity of thymus- dependent lymphocytes. Science. 1977 Mar 25;195(4284):1293–1300. doi: 10.1126/science.320663. [DOI] [PubMed] [Google Scholar]
- Rask L., Lindblom J. B., Peterson P. A. Subunit structure of H-2 alloantigens. Nature. 1974 Jun 28;249(460):833–834. doi: 10.1038/249833a0. [DOI] [PubMed] [Google Scholar]
- Schwartz B. D., Kato K., Cullen S. E., Nathenson S. G. H-2 histocompatibility alloantigens. Some biochemical properties of the molecules solubilized by NP-40 detergent. Biochemistry. 1973 May 22;12(11):2157–2164. doi: 10.1021/bi00735a023. [DOI] [PubMed] [Google Scholar]
- Shearer G. M., Rehn T. G., Garbarino C. A. Cell-mediated lympholysis of trinitrophenyl-modified autologous lymphocytes. Effector cell specificity to modified cell surface components controlled by H-2K and H-2D serological regions of the murine major histocompatibility complex. J Exp Med. 1975 Jun 1;141(6):1348–1364. doi: 10.1084/jem.141.6.1348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimada A., Nathenson S. G. Murine histocompatibility-2 (H-2) alloantigens. Purification and some chemical properties of soluble products from H-2b and H-2d genotypes released by papain digestion of membrane fractions. Biochemistry. 1969 Oct;8(10):4048–4062. doi: 10.1021/bi00838a023. [DOI] [PubMed] [Google Scholar]
- Shreffler D. C., David C. S. The H-2 major histocompatibility complex and the I immune response region: genetic variation, function, and organization. Adv Immunol. 1975;20:125–195. doi: 10.1016/s0065-2776(08)60208-4. [DOI] [PubMed] [Google Scholar]
- Silver J., Hood L. Detergent-solubilised H-2 alloantigen is associated with a small molecular weight polypeptide. Nature. 1974 Jun 21;249(459):764–765. doi: 10.1038/249764a0. [DOI] [PubMed] [Google Scholar]
- Silver J., Hood L. Structure and evolution of transplantation antigens: partial amino-acid sequences of H-2K and H-2D alloantigens. Proc Natl Acad Sci U S A. 1976 Feb;73(2):599–603. doi: 10.1073/pnas.73.2.599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swank R. T., Munkres K. D. Molecular weight analysis of oligopeptides by electrophoresis in polyacrylamide gel with sodium dodecyl sulfate. Anal Biochem. 1971 Feb;39(2):462–477. doi: 10.1016/0003-2697(71)90436-2. [DOI] [PubMed] [Google Scholar]
- Terhorst C., Robb R., Jones C., Strominger J. L. Further structural studies of the heavy chain of HLA antigens and its similarity to immunoglobulins. Proc Natl Acad Sci U S A. 1977 Sep;74(9):4002–4006. doi: 10.1073/pnas.74.9.4002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vitetta E. S., Capra J. D., Klapper D. G., Klein J., Uhr J. W. The partial amino-acid sequence of an H-2K molecule. Proc Natl Acad Sci U S A. 1976 Mar;73(3):905–909. doi: 10.1073/pnas.73.3.905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vitetta E. S., Capra J. D. The protein products of the murine 17th chromosome: genetics and structure. Adv Immunol. 1978;26:147–193. doi: 10.1016/s0065-2776(08)60230-8. [DOI] [PubMed] [Google Scholar]


