Abstract
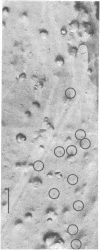
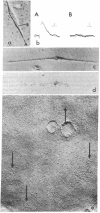
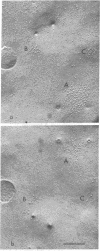
Electrophysiological and autoradiographic studies have shown that the distribution of acetylcholine (AcCho) receptors on uninnervated cultured chicken muscle cells is not uniform. Regions of high receptor density (hot spots 10--40 times more sensitive than surrounding areas are localized as discrete patches or clusters about 10 micrometer in diameter o myotube muscle membranes. Hot spots were also found on fusion-arrested mononucleated myoblasts. We have developed a method for freeze-fracturing monolayer cultures that allows the unambiguous reidentification of membrane patches previously assayed for ACCho sensitivity. The freeze-fractured membranes at physiologically defined hot spots contain aggregates of many (10--20) small clusters of large (10--19 nm in diameter) intramembranous particles. Clusters are found on both fracture faces, but the particle density is much greater on the protoplasmic (P) face than on the extracellular (E) face (about 2000/micrometer2 vs. 700/micrometer2). Some of the particles appear to be composed of five or six "subunits" arranged cylindrically around a central dark dot. Because the aggregates are present at sites of high AcCho sensitivity, it is likely that the intramembranous particles are in some way related to the AcCho receptor molecule.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Albertini D. F., Fawcett D. W., Olds P. J. Morphological variations in gap junctions of ovarian granulosa cells. Tissue Cell. 1975;7(2):389–405. doi: 10.1016/0040-8166(75)90014-2. [DOI] [PubMed] [Google Scholar]
- Anderson M. J., Cohen M. W. Nerve-induced and spontaneous redistribution of acetylcholine receptors on cultured muscle cells. J Physiol. 1977 Jul;268(3):757–773. doi: 10.1113/jphysiol.1977.sp011880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson M. J., Cohen M. W., Zorychta E. Effects of innervation on the distribution of acetylcholine receptors on cultured muscle cells. J Physiol. 1977 Jul;268(3):731–756. doi: 10.1113/jphysiol.1977.sp011879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Axelrod D., Ravdin P., Koppel D. E., Schlessinger J., Webb W. W., Elson E. L., Podleski T. R. Lateral motion of fluorescently labeled acetylcholine receptors in membranes of developing muscle fibers. Proc Natl Acad Sci U S A. 1976 Dec;73(12):4594–4598. doi: 10.1073/pnas.73.12.4594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cartaud J., Benedetti E. L., Cohen J. B., Meunier J. C., Changeux J. P. Presence of a lattice structure in membrane fragments rich in nicotinic receptor protein from the electric organ of Torpedo marmorata. FEBS Lett. 1973 Jun 15;33(1):109–113. doi: 10.1016/0014-5793(73)80171-1. [DOI] [PubMed] [Google Scholar]
- Changeux J. P., Benedetti L., Bourgeois J. P., Brisson A., Cartaud J., Devaux P., Grünhagen H., Moreau M., Popot J. L., Sobel A. Some structural properties of the cholinergic receptor protein in its membrane environmental relevant to its function as a pharmacological receptor. Cold Spring Harb Symp Quant Biol. 1976;40:211–230. doi: 10.1101/sqb.1976.040.01.023. [DOI] [PubMed] [Google Scholar]
- Cohen S. A., Fischbach G. D. Clusters of acetylcholine receptors located at identified nerve-muscle synapses in vitro. Dev Biol. 1977 Aug;59(1):24–38. doi: 10.1016/0012-1606(77)90237-8. [DOI] [PubMed] [Google Scholar]
- Fertuck H. C., Salpeter M. M. Localization of acetylcholine receptor by 125I-labeled alpha-bungarotoxin binding at mouse motor endplates. Proc Natl Acad Sci U S A. 1974 Apr;71(4):1376–1378. doi: 10.1073/pnas.71.4.1376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fertuck H. C., Salpeter M. M. Quantitation of junctional and extrajunctional acetylcholine receptors by electron microscope autoradiography after 125I-alpha-bungarotoxin binding at mouse neuromuscular junctions. J Cell Biol. 1976 Apr;69(1):144–158. doi: 10.1083/jcb.69.1.144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fischbach G. D., Cohen S. A. The distribution of acetylcholine sensitivity over uninnervated and innervated muscle fibers grown in cell culture. Dev Biol. 1973 Mar;31(1):147–162. doi: 10.1016/0012-1606(73)90326-6. [DOI] [PubMed] [Google Scholar]
- Fischbach G. D. Synapse formation between dissociated nerve and muscle cells in low density cell cultures. Dev Biol. 1972 Jun;28(2):407–429. doi: 10.1016/0012-1606(72)90023-1. [DOI] [PubMed] [Google Scholar]
- Heuser J. E., Reese T. S., Landis D. M. Functional changes in frog neuromuscular junctions studied with freeze-fracture. J Neurocytol. 1974 Mar;3(1):109–131. doi: 10.1007/BF01111936. [DOI] [PubMed] [Google Scholar]
- Hourani B. T., Torain B. F., Henkart M. P., Carter R. L., Marchesi V. T., Fischbach G. D. Acetylcholine receptors of cultured muscle cells demonstrated with ferritin-alpha-bungarotoxin conjugates. J Cell Sci. 1974 Nov;16(2):473–479. doi: 10.1242/jcs.16.2.473. [DOI] [PubMed] [Google Scholar]
- Johnson R., Hammer M., Sheridan J., Revel J. P. Gap junction formation between reaggregated Novikoff hepatoma cells. Proc Natl Acad Sci U S A. 1974 Nov;71(11):4536–4540. doi: 10.1073/pnas.71.11.4536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuffler S. W., Yoshikami D. The distribution of acetylcholine sensitivity at the post-synaptic membrane of vertebrate skeletal twitch muscles: iontophoretic mapping in the micron range. J Physiol. 1975 Jan;244(3):703–730. doi: 10.1113/jphysiol.1975.sp010821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Land B. R., Podleski T. R., Salpeter E. E., Salpeter M. M. Acetylcholine receptor distribution on myotubes in culture correlated to acetylcholine sensitivity. J Physiol. 1977 Jul;269(1):155–176. doi: 10.1113/jphysiol.1977.sp011897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meunier J. C., Sealock R., Olsen R., Changeux J. P. Purification and properties of the cholinergic receptor protein from Electrophorus electricus electric tissue. Eur J Biochem. 1974 Jun 15;45(2):371–394. doi: 10.1111/j.1432-1033.1974.tb03563.x. [DOI] [PubMed] [Google Scholar]
- Nickel E., Potter L. T. Ultrastructure of isolated membranes of Torpedo electric tissue. Brain Res. 1973 Jul 27;57(2):508–517. doi: 10.1016/0006-8993(73)90158-3. [DOI] [PubMed] [Google Scholar]
- Paterson B., Strohman R. C. Myosin synthesis in cultures of differentiating chicken embryo skeletal muscle. Dev Biol. 1972 Oct;29(2):113–138. doi: 10.1016/0012-1606(72)90050-4. [DOI] [PubMed] [Google Scholar]
- Peng H. B., Nakajima Y. Membrane particle aggregates in innervated and noninnervated cultures of Xenopus embryonic muscle cells. Proc Natl Acad Sci U S A. 1978 Jan;75(1):500–504. doi: 10.1073/pnas.75.1.500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peper K., Dreyer F., Sandri C., Akert K., Moor H. Structure and ultrastructure of the frog motor endplate. A freeze-etching study. Cell Tissue Res. 1974 Jun 24;149(4):437–455. doi: 10.1007/BF00223024. [DOI] [PubMed] [Google Scholar]
- Rash J. E., Ellisman M. H. Studies of excitable membranes. I. Macromolecular specializations of the neuromuscular junction and the nonjunctional sarcolemma. J Cell Biol. 1974 Nov;63(2 Pt 1):567–586. doi: 10.1083/jcb.63.2.567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Revel J. P., Yee A. G., Hudspeth A. J. Gap junctions between electrotonically coupled cells in tissue culture and in brown fat. Proc Natl Acad Sci U S A. 1971 Dec;68(12):2924–2927. doi: 10.1073/pnas.68.12.2924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenbluth J. Synaptic membrane structure in Torpedo electric organ. J Neurocytol. 1975 Dec;4(6):697–712. doi: 10.1007/BF01181631. [DOI] [PubMed] [Google Scholar]
- Shainberg A., Yagil G., Yaffe D. Control of myogenesis in vitro by Ca 2 + concentration in nutritional medium. Exp Cell Res. 1969 Nov;58(1):163–167. doi: 10.1016/0014-4827(69)90127-x. [DOI] [PubMed] [Google Scholar]
- Sommer J. R., Waugh R. A. The ultrastructure of the mammalian cardiac muscle cell--with special emphasis on the tubular membrane systems. A review. Am J Pathol. 1976 Jan;82(1):192–232. [PMC free article] [PubMed] [Google Scholar]
- Sytkowski A. J., Vogel Z., Nirenberg M. W. Development of acetylcholine receptor clusters on cultured muscle cells. Proc Natl Acad Sci U S A. 1973 Jan;70(1):270–274. doi: 10.1073/pnas.70.1.270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogel Z., Sytkowski A. J., Nirenberg M. W. Acetylcholine receptors of muscle grown in vitro. Proc Natl Acad Sci U S A. 1972 Nov;69(11):3180–3184. doi: 10.1073/pnas.69.11.3180. [DOI] [PMC free article] [PubMed] [Google Scholar]