Abstract
Human pluripotent stem cells (hPSCs), including human embryonic stem cells and human induced pluripotent stem cells, are promising sources for hematopoietic cells due to their unlimited growth capacity and the pluripotency. Dendritic cells (DCs), the unique immune cells in the hematopoietic system, can be loaded with tumor specific antigen and used as vaccine for cancer immunotherapy. While autologous DCs from peripheral blood are limited in cell number, hPSC-derived DCs provide a novel alternative cell source which has the potential for large scale production. This review summarizes recent advances in differentiating hPSCs to DCs through the intermediate stage of hematopoietic stem cells. Step-wise growth factor induction has been used to derive DCs from hPSCs either in suspension culture of embryoid bodies (EBs) or in co-culture with stromal cells. To fulfill the clinical potential of the DCs derived from hPSCs, the bioprocess needs to be scaled up to produce a large number of cells economically under tight quality control. This requires the development of novel bioreactor systems combining guided EB-based differentiation with engineered culture environment. Hence, recent progress in using bioreactors for hPSC lineage-specific differentiation is reviewed. In particular, the potential scale up strategies for the multistage DC differentiation and the effect of shear stress on hPSC differentiation in bioreactors are discussed in detail.
Keywords: Pluripotent stem cells, Dendritic cells, Bioreactor, Hematopoietic differentiation, Large scale production
Core tip: Human embryonic stem cells (hESCs) and human induced pluripotent stem cells (hiPSCs) are promising sources for hematopoietic cells. This review summarizes recent advances in differentiating hESCs and hiPSCs to dendritic cells (DCs), which are unique immune cells in the hematopoietic system and can be loaded with tumor specific antigen and used as vaccine for cancer immunotherapy. While autologous DCs from peripheral blood are limited in number, human PSC (hPSC)-derived DCs provide a novel alternative cell source for clinical application. Different strategies and effects of shear stress on large-scale production of hPSC-derived DCs in bioreactors are also discussed.
INTRODUCTION
Human pluripotent stem cells (hPSCs), including human embryonic stem cells (hESCs) and human induced PSCs (hiPSCs), have unlimited self-renewal ability and can give rise to all cell types from three germ layers[1-5]. While the applications of hESCs are limited by their origin, hiPSCs can be derived from individual patients by reprogramming the somatic cells using pluripotent genes or even small molecules[6,7]. The derived patient-specific hiPSCs can be indefinitely expanded in culture and differentiated into hematopoietic cells, providing an universal cell source for autologous cell replacement without immune rejection and the models to recapitulate genetic hematological diseases for pathological studies[8]. Therefore, hiPSCs have emerged as a broad platform to develop medicines for cell therapy, to establish disease models, and to screen compounds for drug discovery, not only for research but also for commercialization[9,10].
Various blood components have been generated from hPSCs, including red blood cells, platelets, leukocytes, natural killer cells, erythroblasts, T cells, and B cells[11-13]. Dendritic cells (DCs) are potent antigen presenting cells which can be produced as vaccines for cancer immunotherapy[14]. Based on the unique ability to mediate immunity, about 200 DC trials have been reported in treating various types of cancer such as lung cancer and breast cancer[15]. However, autologous DCs are limited in cell number (-108) and subject to large donor-to-donor variability[16]. In addition, DC defects have been observed in circulating peripheral blood mononuclear cells (PBMCs) for some cancer patients[17]. DCs derived from hPSCs can potentially overcome these drawbacks. An unlimited number of DCs with little variability can be derived from hPSCs, which are independent of circulating blood. Thus, hPSC-derived DCs are especially useful for cancer patients who have functional defects in PBMC-derived DCs. Functional DCs have been derived from both hESCs and hiPSCs recently, providing novel alternative sources to autologous DCs[18-20]. Given the unique proliferative capacity, hPSC-based process has the potential for mass production of DCs at a scale of more than 1010 cells[9].
For large-scale production, bioreactor provides a powerful tool to fulfill the unlimited proliferation capacity of hPSCs to derive hematopoietic cells. Bioreactors not only allow the scale up of the process, but may also regulate the differentiation pathway due to the unique hydrodynamic environment, especially the presence of shear stress[21,22]. Several types of bioreactors have been used for hPSC expansion and differentiation, including spinner flasks, rotating wall vessels, and perfusion bioreactors[23-25]. These bioreactors provide a closed and homogenous culture environment along with the capability for online monitoring and control of culture parameters that fulfill regulatory requirements[26]. For example, spinner flask has been tested for hematopoietic differentiation from hESC through embryoid body (EB) formation[27]. EBs in suspension bioreactors have more uniform size distribution, less agglomeration, and similar percentage of the differentiated hematopoietic cells compared to static culture. To provide cell adhesion surfaces, microcarriers have also been applied in hPSC expansion and differentiation due to the scale up potential in stirred bioreactors[28].
In this review article, we discuss the potential of hPSCs as the cell source to generate DCs [through the intermediate stage of hematopoietic stem cells (HSCs)] (Figure 1) and the current progress in DC differentiation from hPSCs. The scale up potential of the differentiation procedure in bioreactors and the potential effect of hydrodynamic environment on hPSC differentiation are also discussed. Combining the guided biological differentiation following the developmental pathway with the engineering principle in scalable bioreactors, the potential of hPSCs in producing DCs to treat cancers can be better fulfilled.
Figure 1.
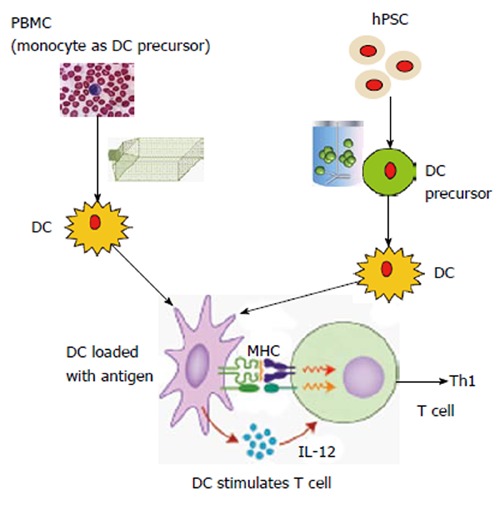
Schematic comparison between conventional dendritic cell vaccine production and human pluripotent stem cell-based dendritic cell C vaccine production. Conventional dendritic cell (DC) vaccine production is usually from peripheral blood mononuclear cells (PBMCs) in patients. T-flasks, Cell Factories or bags are used for DC maturation. From human pluripotent stem cells (hPSCs), DC precursors (similar to monocytes isolated from PBMCs) can be generated in large scale in bioreactors and then mature into DCs. In theory, this approach can generate an unlimited number of DCs. To stimulate T cell response, the activated DCs can release cytokines such as interleukin (IL)-12 to trigger T helper type 1 (Th1) immune response. DCs are also able to capture and process antigens, converting proteins to peptides that are presented on major histocompatibility complex (MHC) molecules and recognized by T cells. The induced Th1 immune response can target on the cancer cells, which express tumor-specific antigens.
WHY AN ALTERNATIVE SOURCE OF HEMATOPOIETIC CELLS IS IMPORTANT
Limitation of autologous HSCs
HSCs are critical sources for various blood cells including DCs. Therefore, HSC transplantation of human leukocyte antigen-matched bone marrow, cord blood, or mobilized peripheral blood CD34+ cells has been the standard medical treatment for cancer patients to repopulate hematopoietic system. However, current allogeneic HSC transplantation is accompanied with high frequency of graft-versus-host diseases due to immune response and high risk of infection[8]. Autologous HSC transplantation has a lower rate of immune rejection, but the cells are not available for patients with genetic defects. Both allogeneic and autologous HSCs from somatic sources are limited in cell number, which significantly affects the therapeutic outcomes. To avoid transplantation-related mortality due to the delayed neutrophil engraftment, a large number of HSCs are required for infusion to patients. For example, a doubled cord blood unit (3.5 × 107 nucleated cells/kg) was applied to increase cell number, which however, did not show a significant impact on neutrophil recovery[29]. Ex vivo expansion strategies have been extensively studied using soluble cytokines, but only 2 to 5-fold increase in long-term repopulating cells was achieved and modest effect on neutrophil recovery was observed[30]. New approaches are being explored to provide niche factors that target on molecular pathways such as Notch or Wnt but still resulted in limited long-term engraftment[29,31]. Besides repopulating bone marrow, HSCs have been used to produce mature blood cell types such as red blood cells and DCs for transfusion or immune therapy[14,32]. However, the number of mature blood cells needed is enormous and current cell expansion technology is not efficient or economical. For example, for DC therapy, which requires a cell number of 108 per patient, the cost of generating sufficient PBMC-derived DCs for each patient is as high as $93000[20]. In contrast to the current HSC sources, hPSC is an ideal cell source that has the potential to generate a large number of immune-compatible hematopoietic cells in a scalable bioreactor system.
Limitation of autologous DC vaccine
Among the mature blood cells, DCs are the most potent immune cells for antigen presentation and the only cells with the ability to induce a primary immune response in resting naïve T lymphocytes[33]. Numerous DC trials have been reported, and the trials in cancer immunotherapy showed encouraging results[34,35]. One such trial in breast cancer was performed in 27 patients with human epidermal growth factor receptor 2 (HER2)/neu overexpressing ductal carcinoma. Sensitization of T-helper cells was observed in 22 of 25 patients. In addition, responses of anti-HER2/neu peptides were observed up to 52-mo post-immunization[34]. Some other DC trials also achieved positive outcomes, including the trials using Dendreon’s lapuleucel-T[36-38]. These promising results encourage further study with multiple doses of DC vaccines in various cancer patients.
DCs for clinical trials can be generated from CD34+ cells in bone marrow using granulocyte-macrophage colony stimulating factor (GM-CSF) and tumor necrosis factor (TNF)-α, or CD14+ monocytes derived from PBMCs using GM-CSF and interleukin (IL)-4[14]. Comparison of CD34+ cells-derived DCs and monocyte-derived DCs from the same patients demonstrated similar morphology and performance in mixed lymphocyte reaction (MLR), while differences in some surface markers such as CD86 and human lymphocyte antigen-DR were observed[39]. Difference in the capacity to activate CD8+ T cells was also reported in these two populations[40]. Because DC functions are affected by patient health, cytokine selection, and isolation procedures, DCs from both sources have been used in clinical trials[41]. However, CD34+ cells are usually found in small numbers and a longer time is needed to generate sufficient DCs (14 d vs 7 d), thus monocyte-derived DCs have been used more often in clinical trials[42]. Production of autologous DCs involves the purification of monocytes from PBMCs, generating immature DCs from monocytes, and differentiating immature DCs into mature DCs[42,43]. For clinical use, a scalable culture system that meets current Good Manufacturing Practices (cGMP) guidelines is required. Such large scale closed-systems have been developed based on Elutra™ cell separation device and cell culture in Teflon bags, which can produce an order of 108 mature DCs[16]. This number generally provides one dose for one patient and multiple preparations are required for multiple doses. Importantly, circulating PBMC-derived DCs from some cancer patients have been shown to have poor ability in stimulating T cell proliferation and reduced capacity of capturing antigens[17]. Both the number limitation and DC defects in some cancer patients have motivated the demand for an alternative DC source.
hPSCs: A NEW SOURCE FOR HSCs
Hematopoietic differentiation from hPSCs has been investigated recently[44,45]. Although both hiPSC and hESC can be differentiated to hematopoietic cells with comparable efficiency, generating hematopoietic cells from the patient-specific iPSCs has the advantage of immunologic compatibility[46]. Methods for hematopoietic differentiation from hPSCs have been performed either by co-culturing on stromal feeder layers or in the form of EBs, an aggregate-like structure mimicking embryonic development[44]. Currently, the most commonly used stromal feeder is murine bone marrow stromal line OP9, which augments the hematopoiesis by promoting the survival of hematopoietic precursors and progenitors[47]. It has been suggested that mKirre, Notch ligand, or other unidentified factors may contribute to the observed effects of OP9 in hPSC differentiation[48]. However, OP9 cells cannot be used to produce hematopoietic cells for clinical application due to their animal origin. EB-based differentiation is more suitable for clinical use due to the absence of murine stroma and the adaptability in suspension culture. However, it is difficult to control the differentiation efficiency and the derived cell phenotype for the EB-based differentiation because of the heterogeneity of EB size and morphology. Therefore, novel methods that can make homogenous EBs such as forced aggregation have been developed recently[49].
To date, CD34+ cells generated from hPSCs using the existing methods are less clonogenic and less proliferative than CD34+ cells isolated from somatic sources[44]. Furthermore, the engraftment capacity of hPSC-derived HSCs was poor compared to cells from somatic source[50]. Despite the poor engraftment capacity, the erythroid, myeloid, natural killer cells, and DCs from hPSC-derived CD34+ cells were qualitatively similar to their somatic counterparts[51-53]. It has been suggested that transfusion and immune therapy may be the immediate clinical applications for hPSC-derived HSCs, which require high derivation efficiency and a clinical-scale culture system suitable for mass production of these cells[11].
hPSCs: A NEW SOURCE FOR DC
DC differentiation from hPSCs: promise and progress
Similar to deriving HSCs, there are two methods for DC differentiation from hPSCs: (1) through the formation of EBs in suspension culture; and (2) by co-culture with mouse OP9 stromal cell line[54]. The differentiation into DCs starts from mesoderm specification induced by growth factors including vascular endothelial growth factor (VEGF) and bone morphogenetic protein (BMP)-4 (Figure 2)[20,52]. Mesoderm cells further develop into HSCs (characterized by HOXB4+ and CD34+ cells) and then become common myeloid progenitors (CMPs) (characterized by CD34+ and CD45+ cells). CMPs are differentiated into monocyte-like DC precursors (CD14+, CD11c+) indicated by single cells (i.e., DC precursors) released from the aggregates. After reaching the DC precursor stage, the differentiation procedure is almost identical to PBMC-derived DCs.
Figure 2.
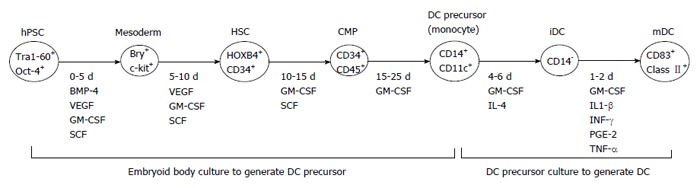
Schematic differentiation process from human pluripotent stem cells to dendritic cells. The differentiation into dendritic cells (DCs) was initiated from mesoderm specification. Then, the cells become hematopoietic stem cells (HSCs) after treatment with growth factors, followed by the stage of more committed common myeloid progenitors (CMP). CMP will then become monocyte-like cells as DC precursors. DC precursors will become immature DCs (iDCs), which further differentiate into mature DCs (mDCs). Definite markers can be used to identify each stage over the course of differentiation. Bry: Mesoderm marker brachyury; BMP-4: Bone morphogenetic protein-4; VEGF: Vascular endothelial growth factor; GM-CSF: Granulocyte-macrophage colony-stimulating factor; SCF: Stem cell factor; IL-4: Interleukin 4; IL1-β: Interleukin-1 beta; IFN-γ: Interferon gamma; PGE-2: Prostaglandin E2; TNF-α: Tumor necrosis factor alpha.
For hESC-derived DCs based on EB formation, only about 25% DCs with low CD83 expression, which indicates the relative immature DC stage, were obtained from spontaneous differentiation from EBs[18]. A similar method was tested in the presence of BMP-4 to enhance mesoderm differentiation[55]. The purity of DCs was improved to more than 80% CD11c+ cells, but CD83 was still less than 50%. A serum-free EB-based protocol was assessed using four growth factors including VEGF, stem cell factor (SCF), BMP-4, and GM-CSF[52,56]. BMP-4 and VEGF are critical growth factors which synergistically enhanced hematopoietic differentiation by activating the Nodal/Activin pathway[57]. About 75% of cells expressed DC markers CD86 and CD83, and the derived DCs had similar stimulatory function compared to PBMC-DCs. This promising protocol serves as the baseline process for Geron’s potential product GRNVAC2, i.e., DCs derived from hESCs, which is the second generation of DCs following GRNVAC1, i.e., DCs derived from PBMC. The four-growth factor (VEGF, SCF, BMP-4, and GM-CSF) protocol was also evaluated for DC differentiation from hiPSCs. High-purity DCs (> 70% CD83+ cells) were generated, but a subset of cell population co-expressed CD141 and XC chemokine receptor 1 (XCR1), which was phenotypically different from CD141+ XCR1- hESC-derived DCs[58]. The expression of XCR1 in DCs may be better suited to the induction of antitumor responses due to the augmentation of antigen-driven expansion of CD8+ cytotoxic T lymphocytes[59]. For the OP9 co-culture protocol, hESCs were plated onto OP9 feeder layer to facilitate hematopoietic differentiation and about 50% of DCs was achieved[60]. However, this protocol only achieved 20% purity of hiPSC-derived DCs[19]. Apparently, the OP9 protocol needs to be improved for efficient DC differentiation from hiPSCs.
Functional assessments were performed for hPSC-derived DCs in comparison with PBMC-derived DCs, including specific cytokine secretion, allogeneic T-cell response by MLR assay, endocytosis ability, and antigen-specific T cell response[19,52,55]. For example, hESC-derived DCs produced similar level of IL-6 and IL-12p70 compared to PBMC-derived DCs[52]. Strong allogeneic T-cell response and phagocytosis ability were also demonstrated for both hESC-DCs and hiPSC-DCs[19,55]. Human telomerase reverse transcriptase antigen-specific T-cell stimulation was observed with significant IFN-γ production which was greater than the response stimulated by PBMC-DCs[52]. Further enhancement of stimulatory function was also demonstrated for hESC-DCs after transfection with mRNA encoding IL-12p70[56]. Despite these successes, a fully understanding of the functions of hPSC-derived DCs, especially DCs derived from hiPSCs, is still required for potential clinical applications.
Scalability of DC differentiation from hPSCs
Compared to the OP9 co-culture system, the EB-based protocol (free of feeder cells) is more suitable for large-scale production in stirred bioreactors for potential clinical applications[61]. Because the DC precursors are released from EBs as single cells, the DC precursors are also able to be expanded and differentiated in suspension. Hence, the process of DC differentiation from hPSCs through EB formation is scalable in bioreactors. Different from traditional static tissue culture vessels, hPSCs in bioreactors are exposed to dynamic physiochemical environments. Especially, the presence of shear stress affects not only cell growth and viability, but also the cell phenotype and lineage commitment[23,62]. Understanding the impact of hydrodynamic environment on hPSC differentiation is a critical step to scale up the process for producing DCs from hPSCs.
POTENTIAL SCALABLE PRODUCTION OF hPSC-DERIVED DCs IN BIOREACTORS
Bioreactors for hPSC-derived cells
Dynamic bioreactors can be used to enhance mass transfer coefficient, alter the kinetics of receptor-ligand binding, and control the aggregate collision[21,62,63]. Thus, stem cell aggregation, metabolism, and cell phenotype can be modulated in the hydrodynamic environment of bioreactors. Several scalable EB-based culture systems, including those for hematopoietic differentiation, have been developed recently (Table 1)[61]. The initial bioreactor culture was performed in slow turning lateral vessel (STLV) for spontaneous EB formation[64]. The STLV with a low shear stress supported better cell expansion than static culture although massive EB agglomeration was observed. Later, STLV was compared with the spinner bioreactor, which gave better cell proliferation[65]. Bioreactor configuration has also been shown to affect hematopoietic differentiation from PSCs. For example, EB formation and the subsequent hematopoietic differentiation in spinner flasks generated more c-kit+ progenitors while more Sca-1+ cells were observed in the STLV, possibly due to different hydrodynamic environments in these two types of bioreactors[25]. Pre-formed EBs were also seeded in spinner bioreactors for hematopoietic differentiation and 5%-6% CD34+ cells were detected[27]. The spinner bioreactor was further improved with oxygen control and size-controlled cell clumps as the inoculum[66]. In general, compared to static cultures, dynamic bioreactors promoted cell expansion with a similar or higher differentiation efficiency. However, the differentiation efficiency is still low (< 30%), and needs significant improvements for clinical applications. This can be done by using a specific lineage differentiation protocol rather than spontaneous differentiation.
Table 1.
Bioreactor systems for hematopoietic differentiation from pluripotent stem cells
| Bioreactor type | PSC line | Performance | Ref. |
| Spinner flask | hESC lines: H9 and H1 | 15-fold expansion in cell number compared to 4-fold in static culture; 5%-6% CD34+ CD31+ cells | Cameron et al[27] |
| RWV | Mouse ESC line: R1 | Hematopoietic differentiation in RWV generated more Sca-1+ cells; while spinner flasks generated more c-kit+ progenitors | Fridley et al[25] |
| Microcarrier-based spinner flask | hESC lines: WA01 (H1), WA09 (H9), HuES3, MA09; hiPSC line: IMR90-1 | DE-53 microcarriers were used. EBs were formed 10 times more efficiently compared to 2-D culture; CFUs were similar to static culture | Lu et al[67] |
| Perfusion bioreactor | Mouse ESC line: D3 Mouse ESC line: CCE | Perfusion promoted hematopoietic differentiation; Perfusion in 3-D fibrous scaffolds supported 2-3 fold higher cell density compared to static culture; comparable CFU percentage | Wolfe et al[77]; Li et al[24] |
hESC: Human embryonic stem cell; ESC: Embryonic stem cell; EBs: Embryoid bodies; hiPSC: Human induced pluripotent stem cell; RWV: Rotary wall vessel; CFU: Colony-forming units.
Recently, microcarriers have been used to support hematopoietic differentiation of hPSCs to improve EB-forming efficiency[67]. Microcarrier culture provides a high surface-to-volume ratio, leading to a high cell density. The process is also suitable for scale up in stirred bioreactors. The hPSCs grown on DE-53 microcarriers were able to form EBs with 10-fold higher efficiency compared to hPSCs grown in 2-D cultures[67]. After replating, these EBs developed into hemangioblasts which can differentiate into hematopoietic and endothelial cells. This system can be further developed into a cGMP-compatible scalable system to generate blood cells for clinical applications. However, there are several challenges for microcarrier cultures, including microcarrier clumping, cell damage caused by shear stress, and difficult operation for cell-carrier separation[68].
Microencapsulation of hPSCs in hydrogel is another approach of suspension culture, which can avoid EB aggregation or microcarrier clumping while protecting cells from shear stress[69,70]. Using specific biomaterials such as alginate, agarose, and hyaluronic acid, microencapsulation can preserve 3D cell-cell and cell-matrix contacts, which simulate in vivo stem cell niches for efficient hPSC differentiation[70,71]. However, there are some limitations in microencapsulation cultures. For example, gas and mass diffusion inside the hydrogel could be limited; monitoring and observing the culture are difficult; and the additional cell releasing process is required[68]. For all these suspension cultures (i.e., EBs, microcarriers, and microencapsulation), the effect of shear stress on hPSC differentiation needs to be further studied for efficient differentiation in bioreactors.
Effects of shear stress in bioreactors
Shear stress is an important parameter that can be used to regulate hPSC expansion and differentiation (Table 2)[68,72]. During the differentiation of hPSCs into cardiomyocytes or osteoblasts, the PSC aggregates cultured in stirred bioreactors preserved the Oct-4 expressing pluripotent cells, which were absent in the cell population differentiated in static culture[73,74]. It was postulated that shear stress modulated gene expression through mechano-transduction and that the non-canonical Wnt pathway might play an important role in bioreactor-induced pluripotency. Mechanical strain also suppressed spontaneous differentiation and induced autocrine or paracrine signaling through transforming growth factor-β superfamily ligand to activate Smad 2/3[75]. However, in both the microcarrier-based stirred bioreactors and the EB-based rotary orbital bioreactors, shear stress in the hydrodynamic environment was found to promote spontaneous differentiation of PSCs[63,76]. Perfusion-induced shear stress (1.5-15 dyne/cm2) was also shown to promote hematopoietic differentiation from PSCs by up-regulating fetal liver kinase-1 (VEGF receptor) expression[77]. The effect of shear stress on fate decision of hPSCs has not been fully understood and requires future investigation.
Table 2.
Effects of shear stress in bioreactors on pluripotent stem cells expansion and differentiation
| Shear stress | Effect on pluripotency | Effect on differentiation | Ref. |
| Agitation in stirred bioreactors | Bioreactor-differentiated ESCs retained the ability to express pluripotent markers | EBs in bioreactors differentiated into cardiomyocytes | Shafa et al[73] |
| Agitation in stirred bioreactors (3 dyne/cm2 vs 6 dyne/cm2) | Shear stress maintained certain pluripotent markers (e.g., Nanog, Rex-1) | Reduced spontaneous differentiation | Gareau et al[72] |
| Agitation in stirred bioreactors | A subpopulation of bioreactor-differentiated ESCs expressed the pluripotent markers | Differentiation into osteogenic and chondrogenic cell types | Taiani et al[74] |
| Agitation in stirred bioreactors (glass ball impeller, < 1.52 dyne/cm2) | Homogeneous aggregate size distribution | Cells maintained the differentiation potential into hematopoietic cells | Wang et al[79] |
| Rotary orbital shaking (< 2.5 dyne/cm2) | Shear stress up-regulated genes specific for endoderm and mesoderm differentiation | Spontaneous three-germ layer differentiation | Sargent et al[63] |
| Perfusion flow (1.5-15 dyne/cm2) | Shear stress promoted early differentiation of ESCs | Shear stress promoted hematopoietic and endothelial differentiation | Wolfe et al[77] |
| Agitation in microcarrier-based bioreactors | HES-2 line and hiPSC line IMR-90 were shear sensitive, showing the down-regulation of pluripotent markers | Shear stress induced spontaneous differentiation | Leung et al[76] |
ESC: Embryonic stem cell; EBs: Embryoid bodies; hiPSC: Human induced pluripotent stem cell.
The shear stress exerted on the cells during initial seeding may affect EB-forming efficiency in the dynamic culture. For EB formation, a low shear stress could lead to inadequate nutrient diffusion and massive EB agglomeration, while a high shear stress could lead to the dissociation of receptor-ligand binding and may also cause significant cell death[78]. A few studies tried to avoid direct EB formation in spinner bioreactors by seeding pre-formed EBs[27,65]. However, this approach is not practical for large-scale production. The rotary speed (20-60 rpm) in a rotary orbital suspension culture has been found to affect EB size distribution, where a mild shear stress (< 2.5 dyne/cm2) led to homogenous EBs[63]. A mild shear stress (< 1.5 dyne/cm2) in the spinner flask equipped with a pendulum-shape impeller at 40-75 rpm also produced homogeneous hiPSC aggregates[79]. To minimize the initial cell death and EB aggregation, novel agitation scheme based on intermittent mixing may also need to be applied.
Scale up challenges for producing DCs from hPSCs
DC differentiation from hPSCs, which lasts about 32 d, has multiple stages of differentiation with a different cocktail of growth factors at each stage (Figure 2). Specifically, the differentiation procedure comprises of EB stage and single-cell stage, each requiring a different agitation rate as the EBs and the single cells might have different sensitivities to shear stress. Although hPSC-derived EBs have been expanded in bioreactors, the lineage specification of DC differentiation from hPSCs in bioreactors has not been demonstrated. For conventional production of DC vaccines, isolated monocytes from PBMCs are cultured as DC precursors to generate DCs. To integrate with monocyte isolation from PBMCs, autologous DC production for clinical use is usually performed in large T-flasks, roller bottles, and bags[80-82]. The bags are widely used because they are easy to be connected with cell separation system, enabling DC production in a complete closed-system[16,81]. However, to integrate hPSC-DC precursor culture (single cell stage) with dynamic hPSC-EB culture (aggregate stage), stirred spinner bioreactors may be required. The concept of process integration, which includes reprogramming, expansion, and differentiation in one fully integrated process, has been demonstrated for iPSC-derived cardiac cells[83]. For DCs, it would be difficult and inconvenient to transfer the EB-derived DC precursors from spinner flasks to roller bottles or bags. Continuous differentiation of hPSC-derived DC precursors in the same culture vessel offers obvious advantages for large scale production. Due to different sensitivities to shear stress at various differentiation stages, different agitation programs may be required for day 0-2 (EB formation stage), day 2-25 (EB culture stage), and day 25-32 (single cell stage). Therefore, an integrated bioreactor system can significantly enhance the process efficiency and scalability for hPSC-derived DCs.
Online monitoring the differentiation status of hPSCs is also a crucial element in bioreactor-based DC production. Given that hPSC differentiation usually generates a heterogeneous cell population, including the residue undifferentiated cells that can form tumor in vivo, novel reliable technologies for downstream cell separation in clinical scale also need to be developed[84]. Establishing the correlation of cell-secreted molecules with the differentiation outcome can facilitate the in-process monitoring. For DC differentiation, DC precursors are released from EBs and the frequency of undifferentiated cells is low. More importantly, DCs loaded with tumor-specific antigen can be irradiated for vaccine injection, thus there is minimal risk of tumor formation. Instead, cell irradiation will be a part of the production process and the procedure needs to be optimized to better preserve DC function.
CONCLUSION
Pluripotent stem cells have emerged as new cell sources for HSC-derived mature blood cells, especially DCs. PSC-derived DCs can overcome the limitations of autologous DCs from cancer patients, including the limited cell number and possible functional defects. Although the engraftment capacity of hPSC-derived HSCs has been poor compared to cells from somatic sources, mature blood cells, including DCs from hPSC-derived CD34+ cells, are qualitatively similar to their somatic counterparts. Efficient DC differentiation from hPSCs has been achieved through EB formation with high purity. To fulfill the potential of hPSC-derived DCs, large-scale production in bioreactors is a critical step toward clinical applications. The hydrodynamic environment in bioreactors, especially shear stress, is a potent regulator for hPSC expansion and differentiation, while the effect of shear stress on fate decision of hPSCs has not been fully understood. For DC differentiation in bioreactors, an integrated process from EB formation to DC maturation will offer significant advantages in process efficiency and scalability. However, it is challenging to integrate the multiple stages of DC differentiation from hPSCs, which needs a better understanding of the stage-specific responses to the hydrodynamic environment.
Footnotes
P- Reviewers: Chapel A, Li GR, Yao CL S- Editor: Ma YJ L- Editor: A E- Editor: Liu SQ
References
- 1.Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, Jones JM. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282:1145–1147. doi: 10.1126/science.282.5391.1145. [DOI] [PubMed] [Google Scholar]
- 2.Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861–872. doi: 10.1016/j.cell.2007.11.019. [DOI] [PubMed] [Google Scholar]
- 3.Li Y, Gautam A, Yang J, Qiu L, Melkoumian Z, Weber J, Telukuntla L, Srivastava R, Whiteley EM, Brandenberger R. Differentiation of oligodendrocyte progenitor cells from human embryonic stem cells on vitronectin-derived synthetic Peptide acrylate surface. Stem Cells Dev. 2013;22:1497–1505. doi: 10.1089/scd.2012.0508. [DOI] [PubMed] [Google Scholar]
- 4.Li Y, Powell S, Brunette E, Lebkowski J, Mandalam R. Expansion of human embryonic stem cells in defined serum-free medium devoid of animal-derived products. Biotechnol Bioeng. 2005;91:688–698. doi: 10.1002/bit.20536. [DOI] [PubMed] [Google Scholar]
- 5.Xu C. Differentiation and enrichment of cardiomyocytes from human pluripotent stem cells. J Mol Cell Cardiol. 2012;52:1203–1212. doi: 10.1016/j.yjmcc.2012.03.012. [DOI] [PubMed] [Google Scholar]
- 6.Wu SM, Hochedlinger K. Harnessing the potential of induced pluripotent stem cells for regenerative medicine. Nat Cell Biol. 2011;13:497–505. doi: 10.1038/ncb0511-497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hou P, Li Y, Zhang X, Liu C, Guan J, Li H, Zhao T, Ye J, Yang W, Liu K, et al. Pluripotent stem cells induced from mouse somatic cells by small-molecule compounds. Science. 2013;341:651–654. doi: 10.1126/science.1239278. [DOI] [PubMed] [Google Scholar]
- 8.Lengerke C, Daley GQ. Autologous blood cell therapies from pluripotent stem cells. Blood Rev. 2010;24:27–37. doi: 10.1016/j.blre.2009.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Abbasalizadeh S, Baharvand H. Technological progress and challenges towards cGMP manufacturing of human pluripotent stem cells based therapeutic products for allogeneic and autologous cell therapies. Biotechnol Adv. 2013;31:1600–1623. doi: 10.1016/j.biotechadv.2013.08.009. [DOI] [PubMed] [Google Scholar]
- 10.Schnittger S, Bacher U, Alpermann T, Reiter A, Ulke M, Dicker F, Eder C, Kohlmann A, Grossmann V, Kowarsch A, et al. Use of CBL exon 8 and 9 mutations in diagnosis of myeloproliferative neoplasms and myelodysplastic/myeloproliferative disorders: an analysis of 636 cases. Haematologica. 2012;97:1890–1894. doi: 10.3324/haematol.2012.065375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kaufman DS. Toward clinical therapies using hematopoietic cells derived from human pluripotent stem cells. Blood. 2009;114:3513–3523. doi: 10.1182/blood-2009-03-191304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kimbrel EA, Lu SJ. Potential clinical applications for human pluripotent stem cell-derived blood components. Stem Cells Int. 2011;2011:273076. doi: 10.4061/2011/273076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Larbi A, Gombert JM, Auvray C, l’Homme B, Magniez A, Féraud O, Coulombel L, Chapel A, Mitjavila-Garcia MT, Turhan AG, et al. The HOXB4 homeoprotein promotes the ex vivo enrichment of functional human embryonic stem cell-derived NK cells. PLoS One. 2012;7:e39514. doi: 10.1371/journal.pone.0039514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nestle FO, Banchereau J, Hart D. Dendritic cells: On the move from bench to bedside. Nat Med. 2001;7:761–765. doi: 10.1038/89863. [DOI] [PubMed] [Google Scholar]
- 15.Cranmer LD, Trevor KT, Hersh EM. Clinical applications of dendritic cell vaccination in the treatment of cancer. Cancer Immunol Immunother. 2004;53:275–306. doi: 10.1007/s00262-003-0432-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jarnjak-Jankovic S, Hammerstad H, Saebøe-Larssen S, Kvalheim G, Gaudernack G. A full scale comparative study of methods for generation of functional Dendritic cells for use as cancer vaccines. BMC Cancer. 2007;7:119. doi: 10.1186/1471-2407-7-119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pinzon-Charry A, Ho CS, Laherty R, Maxwell T, Walker D, Gardiner RA, O’Connor L, Pyke C, Schmidt C, Furnival C, et al. A population of HLA-DR+ immature cells accumulates in the blood dendritic cell compartment of patients with different types of cancer. Neoplasia. 2005;7:1112–1122. doi: 10.1593/neo.05442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhan X, Dravid G, Ye Z, Hammond H, Shamblott M, Gearhart J, Cheng L. Functional antigen-presenting leucocytes derived from human embryonic stem cells in vitro. Lancet. 2004;364:163–171. doi: 10.1016/S0140-6736(04)16629-4. [DOI] [PubMed] [Google Scholar]
- 19.Senju S, Haruta M, Matsumura K, Matsunaga Y, Fukushima S, Ikeda T, Takamatsu K, Irie A, Nishimura Y. Generation of dendritic cells and macrophages from human induced pluripotent stem cells aiming at cell therapy. Gene Ther. 2011;18:874–883. doi: 10.1038/gt.2011.22. [DOI] [PubMed] [Google Scholar]
- 20.Li Y, Ma T. Stem cell-based dendritic cell vaccine development: A review with emphasis on lung cancer treatment. J Hematol Malig. 2011;1:35–48. [Google Scholar]
- 21.Liu M, Liu N, Zang R, Li Y, Yang ST. Engineering stem cell niches in bioreactors. World J Stem Cells. 2013;5:124–135. doi: 10.4252/wjsc.v5.i4.124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Liu N, Zang R, Yang ST, Li Y. Stem cell engineering in bioreactors for large scale bioprocessing. Eng Life Sci. 2014:14. [Google Scholar]
- 23.Rodrigues CA, Fernandes TG, Diogo MM, da Silva CL, Cabral JM. Stem cell cultivation in bioreactors. Biotechnol Adv. 2011;29:815–829. doi: 10.1016/j.biotechadv.2011.06.009. [DOI] [PubMed] [Google Scholar]
- 24.Li Y, Kniss DA, Lasky LC, Yang ST. Culturing and differentiation of murine embryonic stem cells in a three-dimensional fibrous matrix. Cytotechnology. 2003;41:23–35. doi: 10.1023/A:1024283521966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fridley KM, Fernandez I, Li MT, Kettlewell RB, Roy K. Unique differentiation profile of mouse embryonic stem cells in rotary and stirred tank bioreactors. Tissue Eng Part A. 2010;16:3285–3298. doi: 10.1089/ten.tea.2010.0166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sharma S, Raju R, Sui S, Hu WS. Stem cell culture engineering - process scale up and beyond. Biotechnol J. 2011;6:1317–1329. doi: 10.1002/biot.201000435. [DOI] [PubMed] [Google Scholar]
- 27.Cameron CM, Hu WS, Kaufman DS. Improved development of human embryonic stem cell-derived embryoid bodies by stirred vessel cultivation. Biotechnol Bioeng. 2006;94:938–948. doi: 10.1002/bit.20919. [DOI] [PubMed] [Google Scholar]
- 28.Sart S, Agathos SN, Li Y. Engineering stem cell fate with biochemical and biomechanical properties of microcarriers. Biotechnol Prog. 2013;29:1354–1366. doi: 10.1002/btpr.1825. [DOI] [PubMed] [Google Scholar]
- 29.Dahlberg A, Delaney C, Bernstein ID. Ex vivo expansion of human hematopoietic stem and progenitor cells. Blood. 2011;117:6083–6090. doi: 10.1182/blood-2011-01-283606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Boiron JM, Dazey B, Cailliot C, Launay B, Attal M, Mazurier F, McNiece IK, Ivanovic Z, Caraux J, Marit G, et al. Large-scale expansion and transplantation of CD34(+) hematopoietic cells: in vitro and in vivo confirmation of neutropenia abrogation related to the expansion process without impairment of the long-term engraftment capacity. Transfusion. 2006;46:1934–1942. doi: 10.1111/j.1537-2995.2006.01001.x. [DOI] [PubMed] [Google Scholar]
- 31.Delaney C, Heimfeld S, Brashem-Stein C, Voorhies H, Manger RL, Bernstein ID. Notch-mediated expansion of human cord blood progenitor cells capable of rapid myeloid reconstitution. Nat Med. 2010;16:232–236. doi: 10.1038/nm.2080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Giarratana MC, Kobari L, Lapillonne H, Chalmers D, Kiger L, Cynober T, Marden MC, Wajcman H, Douay L. Ex vivo generation of fully mature human red blood cells from hematopoietic stem cells. Nat Biotechnol. 2005;23:69–74. doi: 10.1038/nbt1047. [DOI] [PubMed] [Google Scholar]
- 33.Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998;392:245–252. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- 34.Koski GK, Koldovsky U, Xu S, Mick R, Sharma A, Fitzpatrick E, Weinstein S, Nisenbaum H, Levine BL, Fox K, et al. A novel dendritic cell-based immunization approach for the induction of durable Th1-polarized anti-HER-2/neu responses in women with early breast cancer. J Immunother. 2012;35:54–65. doi: 10.1097/CJI.0b013e318235f512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Baek S, Kim CS, Kim SB, Kim YM, Kwon SW, Kim Y, Kim H, Lee H. Combination therapy of renal cell carcinoma or breast cancer patients with dendritic cell vaccine and IL-2: results from a phase I/II trial. J Transl Med. 2011;9:178. doi: 10.1186/1479-5876-9-178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Park JW, Melisko ME, Esserman LJ, Jones LA, Wollan JB, Sims R. Treatment with autologous antigen-presenting cells activated with the HER-2 based antigen Lapuleucel-T: results of a phase I study in immunologic and clinical activity in HER-2 overexpressing breast cancer. J Clin Oncol. 2007;25:3680–3687. doi: 10.1200/JCO.2006.10.5718. [DOI] [PubMed] [Google Scholar]
- 37.Koski GK, Cohen PA, Roses RE, Xu S, Czerniecki BJ. Reengineering dendritic cell-based anti-cancer vaccines. Immunol Rev. 2008;222:256–276. doi: 10.1111/j.1600-065X.2008.00617.x. [DOI] [PubMed] [Google Scholar]
- 38.Peethambaram PP, Melisko ME, Rinn KJ, Alberts SR, Provost NM, Jones LA, Sims RB, Lin LR, Frohlich MW, Park JW. A phase I trial of immunotherapy with lapuleucel-T (APC8024) in patients with refractory metastatic tumors that express HER-2/neu. Clin Cancer Res. 2009;15:5937–5944. doi: 10.1158/1078-0432.CCR-08-3282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Syme R, Bajwa R, Robertson L, Stewart D, Glück S. Comparison of CD34 and monocyte-derived dendritic cells from mobilized peripheral blood from cancer patients. Stem Cells. 2005;23:74–81. doi: 10.1634/stemcells.2004-0070. [DOI] [PubMed] [Google Scholar]
- 40.Ferlazzo G, Wesa A, Wei WZ, Galy A. Dendritic cells generated either from CD34+ progenitor cells or from monocytes differ in their ability to activate antigen-specific CD8+ T cells. J Immunol. 1999;163:3597–3604. [PubMed] [Google Scholar]
- 41.Ballestrero A, Boy D, Moran E, Cirmena G, Brossart P, Nencioni A. Immunotherapy with dendritic cells for cancer. Adv Drug Deliv Rev. 2008;60:173–183. doi: 10.1016/j.addr.2007.08.026. [DOI] [PubMed] [Google Scholar]
- 42.Castiello L, Sabatino M, Jin P, Clayberger C, Marincola FM, Krensky AM, Stroncek DF. Monocyte-derived DC maturation strategies and related pathways: a transcriptional view. Cancer Immunol Immunother. 2011;60:457–466. doi: 10.1007/s00262-010-0954-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tuyaerts S, Aerts JL, Corthals J, Neyns B, Heirman C, Breckpot K, Thielemans K, Bonehill A. Current approaches in dendritic cell generation and future implications for cancer immunotherapy. Cancer Immunol Immunother. 2007;56:1513–1537. doi: 10.1007/s00262-007-0334-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dravid GG, Crooks GM. The challenges and promises of blood engineered from human pluripotent stem cells. Adv Drug Deliv Rev. 2011;63:331–341. doi: 10.1016/j.addr.2010.12.006. [DOI] [PubMed] [Google Scholar]
- 45.Wang L, Menendez P, Cerdan C, Bhatia M. Hematopoietic development from human embryonic stem cell lines. Exp Hematol. 2005;33:987–996. doi: 10.1016/j.exphem.2005.06.002. [DOI] [PubMed] [Google Scholar]
- 46.Choi KD, Yu J, Smuga-Otto K, Salvagiotto G, Rehrauer W, Vodyanik M, Thomson J, Slukvin I. Hematopoietic and endothelial differentiation of human induced pluripotent stem cells. Stem Cells. 2009;27:559–567. doi: 10.1634/stemcells.2008-0922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ji J, Vijayaragavan K, Bosse M, Menendez P, Weisel K, Bhatia M. OP9 stroma augments survival of hematopoietic precursors and progenitors during hematopoietic differentiation from human embryonic stem cells. Stem Cells. 2008;26:2485–2495. doi: 10.1634/stemcells.2008-0642. [DOI] [PubMed] [Google Scholar]
- 48.Vodyanik MA, Bork JA, Thomson JA, Slukvin II. Human embryonic stem cell-derived CD34+ cells: efficient production in the coculture with OP9 stromal cells and analysis of lymphohematopoietic potential. Blood. 2005;105:617–626. doi: 10.1182/blood-2004-04-1649. [DOI] [PubMed] [Google Scholar]
- 49.Ng ES, Davis RP, Azzola L, Stanley EG, Elefanty AG. Forced aggregation of defined numbers of human embryonic stem cells into embryoid bodies fosters robust, reproducible hematopoietic differentiation. Blood. 2005;106:1601–1603. doi: 10.1182/blood-2005-03-0987. [DOI] [PubMed] [Google Scholar]
- 50.Wang L, Menendez P, Shojaei F, Li L, Mazurier F, Dick JE, Cerdan C, Levac K, Bhatia M. Generation of hematopoietic repopulating cells from human embryonic stem cells independent of ectopic HOXB4 expression. J Exp Med. 2005;201:1603–1614. doi: 10.1084/jem.20041888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Woll PS, Grzywacz B, Tian X, Marcus RK, Knorr DA, Verneris MR, Kaufman DS. Human embryonic stem cells differentiate into a homogeneous population of natural killer cells with potent in vivo antitumor activity. Blood. 2009;113:6094–6101. doi: 10.1182/blood-2008-06-165225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tseng SY, Nishimoto KP, Silk KM, Majumdar AS, Dawes GN, Waldmann H, Fairchild PJ, Lebkowski JS, Reddy A. Generation of immunogenic dendritic cells from human embryonic stem cells without serum and feeder cells. Regen Med. 2009;4:513–526. doi: 10.2217/rme.09.25. [DOI] [PubMed] [Google Scholar]
- 53.Olivier EN, Qiu C, Velho M, Hirsch RE, Bouhassira EE. Large-scale production of embryonic red blood cells from human embryonic stem cells. Exp Hematol. 2006;34:1635–1642. doi: 10.1016/j.exphem.2006.07.003. [DOI] [PubMed] [Google Scholar]
- 54.Senju S, Hirata S, Motomura Y, Fukuma D, Matsunaga Y, Fukushima S, Matsuyoshi H, Nishimura Y. Pluripotent stem cells as source of dendritic cells for immune therapy. Int J Hematol. 2010;91:392–400. doi: 10.1007/s12185-010-0520-1. [DOI] [PubMed] [Google Scholar]
- 55.Su Z, Frye C, Bae KM, Kelley V, Vieweg J. Differentiation of human embryonic stem cells into immunostimulatory dendritic cells under feeder-free culture conditions. Clin Cancer Res. 2008;14:6207–6217. doi: 10.1158/1078-0432.CCR-08-0309. [DOI] [PubMed] [Google Scholar]
- 56.Nishimoto KP, Tseng SY, Lebkowski JS, Reddy A. Modification of human embryonic stem cell-derived dendritic cells with mRNA for efficient antigen presentation and enhanced potency. Regen Med. 2011;6:303–318. doi: 10.2217/rme.11.19. [DOI] [PubMed] [Google Scholar]
- 57.Pick M, Azzola L, Mossman A, Stanley EG, Elefanty AG. Differentiation of human embryonic stem cells in serum-free medium reveals distinct roles for bone morphogenetic protein 4, vascular endothelial growth factor, stem cell factor, and fibroblast growth factor 2 in hematopoiesis. Stem Cells. 2007;25:2206–2214. doi: 10.1634/stemcells.2006-0713. [DOI] [PubMed] [Google Scholar]
- 58.Silk KM, Silk JD, Ichiryu N, Davies TJ, Nolan KF, Leishman AJ, Carpenter L, Watt SM, Cerundolo V, Fairchild PJ. Cross-presentation of tumour antigens by human induced pluripotent stem cell-derived CD141(+)XCR1+ dendritic cells. Gene Ther. 2012;19:1035–1040. doi: 10.1038/gt.2011.177. [DOI] [PubMed] [Google Scholar]
- 59.Kroczek RA, Henn V. The Role of XCR1 and its Ligand XCL1 in Antigen Cross-Presentation by Murine and Human Dendritic Cells. Front Immunol. 2012;3:14. doi: 10.3389/fimmu.2012.00014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Slukvin II, Vodyanik MA, Thomson JA, Gumenyuk ME, Choi KD. Directed differentiation of human embryonic stem cells into functional dendritic cells through the myeloid pathway. J Immunol. 2006;176:2924–2932. doi: 10.4049/jimmunol.176.5.2924. [DOI] [PubMed] [Google Scholar]
- 61.Rungarunlert S, Techakumphu M, Pirity MK, Dinnyes A. Embryoid body formation from embryonic and induced pluripotent stem cells: Benefits of bioreactors. World J Stem Cells. 2009;1:11–21. doi: 10.4252/wjsc.v1.i1.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kinney MA, Sargent CY, McDevitt TC. The multiparametric effects of hydrodynamic environments on stem cell culture. Tissue Eng Part B Rev. 2011;17:249–262. doi: 10.1089/ten.teb.2011.0040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Sargent CY, Berguig GY, Kinney MA, Hiatt LA, Carpenedo RL, Berson RE, McDevitt TC. Hydrodynamic modulation of embryonic stem cell differentiation by rotary orbital suspension culture. Biotechnol Bioeng. 2010;105:611–626. doi: 10.1002/bit.22578. [DOI] [PubMed] [Google Scholar]
- 64.Gerecht-Nir S, Cohen S, Itskovitz-Eldor J. Bioreactor cultivation enhances the efficiency of human embryoid body (hEB) formation and differentiation. Biotechnol Bioeng. 2004;86:493–502. doi: 10.1002/bit.20045. [DOI] [PubMed] [Google Scholar]
- 65.Yirme G, Amit M, Laevsky I, Osenberg S, Itskovitz-Eldor J. Establishing a dynamic process for the formation, propagation, and differentiation of human embryoid bodies. Stem Cells Dev. 2008;17:1227–1241. doi: 10.1089/scd.2007.0272. [DOI] [PubMed] [Google Scholar]
- 66.Niebruegge S, Bauwens CL, Peerani R, Thavandiran N, Masse S, Sevaptisidis E, Nanthakumar K, Woodhouse K, Husain M, Kumacheva E, et al. Generation of human embryonic stem cell-derived mesoderm and cardiac cells using size-specified aggregates in an oxygen-controlled bioreactor. Biotechnol Bioeng. 2009;102:493–507. doi: 10.1002/bit.22065. [DOI] [PubMed] [Google Scholar]
- 67.Lu SJ, Kelley T, Feng Q, Chen A, Reuveny S, Lanza R, Oh SK. 3D microcarrier system for efficient differentiation of human pluripotent stem cells into hematopoietic cells without feeders and serum [corrected] Regen Med. 2013;8:413–424. doi: 10.2217/rme.13.36. [DOI] [PubMed] [Google Scholar]
- 68.Serra M, Brito C, Correia C, Alves PM. Process engineering of human pluripotent stem cells for clinical application. Trends Biotechnol. 2012;30:350–359. doi: 10.1016/j.tibtech.2012.03.003. [DOI] [PubMed] [Google Scholar]
- 69.Serra M, Correia C, Malpique R, Brito C, Jensen J, Bjorquist P, Carrondo MJ, Alves PM. Microencapsulation technology: a powerful tool for integrating expansion and cryopreservation of human embryonic stem cells. PLoS One. 2011;6:e23212. doi: 10.1371/journal.pone.0023212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Siti-Ismail N, Bishop AE, Polak JM, Mantalaris A. The benefit of human embryonic stem cell encapsulation for prolonged feeder-free maintenance. Biomaterials. 2008;29:3946–3952. doi: 10.1016/j.biomaterials.2008.04.027. [DOI] [PubMed] [Google Scholar]
- 71.Gerecht S, Burdick JA, Ferreira LS, Townsend SA, Langer R, Vunjak-Novakovic G. Hyaluronic acid hydrogel for controlled self-renewal and differentiation of human embryonic stem cells. Proc Natl Acad Sci USA. 2007;104:11298–11303. doi: 10.1073/pnas.0703723104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Gareau T, Lara GG, Shepherd RD, Krawetz R, Rancourt DE, Rinker KD, Kallos MS. Shear stress influences the pluripotency of murine embryonic stem cells in stirred suspension bioreactors. J Tissue Eng Regen Med. 2012:Jun 1; Epub ahead of print. doi: 10.1002/term.1518. [DOI] [PubMed] [Google Scholar]
- 73.Shafa M, Krawetz R, Zhang Y, Rattner JB, Godollei A, Duff HJ, Rancourt DE. Impact of stirred suspension bioreactor culture on the differentiation of murine embryonic stem cells into cardiomyocytes. BMC Cell Biol. 2011;12:53. doi: 10.1186/1471-2121-12-53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Taiani JT, Krawetz RJ, Zur Nieden NI, Elizabeth Wu Y, Kallos MS, Matyas JR, Rancourt DE. Reduced differentiation efficiency of murine embryonic stem cells in stirred suspension bioreactors. Stem Cells Dev. 2010;19:989–998. doi: 10.1089/scd.2009.0297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Saha S, Ji L, de Pablo JJ, Palecek SP. TGFbeta/Activin/Nodal pathway in inhibition of human embryonic stem cell differentiation by mechanical strain. Biophys J. 2008;94:4123–4133. doi: 10.1529/biophysj.107.119891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Leung HW, Chen A, Choo AB, Reuveny S, Oh SK. Agitation can induce differentiation of human pluripotent stem cells in microcarrier cultures. Tissue Eng Part C Methods. 2011;17:165–172. doi: 10.1089/ten.TEC.2010.0320. [DOI] [PubMed] [Google Scholar]
- 77.Wolfe RP, Ahsan T. Shear stress during early embryonic stem cell differentiation promotes hematopoietic and endothelial phenotypes. Biotechnol Bioeng. 2013;110:1231–1242. doi: 10.1002/bit.24782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Kehoe DE, Jing D, Lock LT, Tzanakakis ES. Scalable stirred-suspension bioreactor culture of human pluripotent stem cells. Tissue Eng Part A. 2010;16:405–421. doi: 10.1089/ten.tea.2009.0454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Wang Y, Chou BK, Dowey S, He C, Gerecht S, Cheng L. Scalable expansion of human induced pluripotent stem cells in the defined xeno-free E8 medium under adherent and suspension culture conditions. Stem Cell Res. 2013;11:1103–1116. doi: 10.1016/j.scr.2013.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Campbell-Anson RE, Kentor D, Wang YJ, Bushnell KM, Li Y, Vence LM, Radvanyi LG. A new approach for the large-scale generation of mature dendritic cells from adherent PBMC using roller bottle technology. J Immune Based Ther Vaccines. 2008;6:1. doi: 10.1186/1476-8518-6-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Büchler T, Kovárová L, Musilová R, Bourková L, Ocadlíková D, Buliková A, Hanák L, Michálek J, Hájek R. Generation of dendritic cells using cell culture bags--description of a method and review of literature. Hematology. 2004;9:199–205. doi: 10.1080/10245330410001701486. [DOI] [PubMed] [Google Scholar]
- 82.Schmitt A, Reinhardt P, Hus I, Tabarkiewicz J, Roliñski J, Barth T, Giannopoulos K, Dmoszyñska A, Wiesneth M, Schmitt M. Large-scale generation of autologous dendritic cells for immunotherapy in patients with acute myeloid leukemia. Transfusion. 2007;47:1588–1594. doi: 10.1111/j.1537-2995.2007.01328.x. [DOI] [PubMed] [Google Scholar]
- 83.Fluri DA, Tonge PD, Song H, Baptista RP, Shakiba N, Shukla S, Clarke G, Nagy A, Zandstra PW. Derivation, expansion and differentiation of induced pluripotent stem cells in continuous suspension cultures. Nat Methods. 2012;9:509–516. doi: 10.1038/nmeth.1939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Diogo MM, da Silva CL, Cabral JM. Separation technologies for stem cell bioprocessing. Biotechnol Bioeng. 2012;109:2699–2709. doi: 10.1002/bit.24706. [DOI] [PubMed] [Google Scholar]


