Abstract
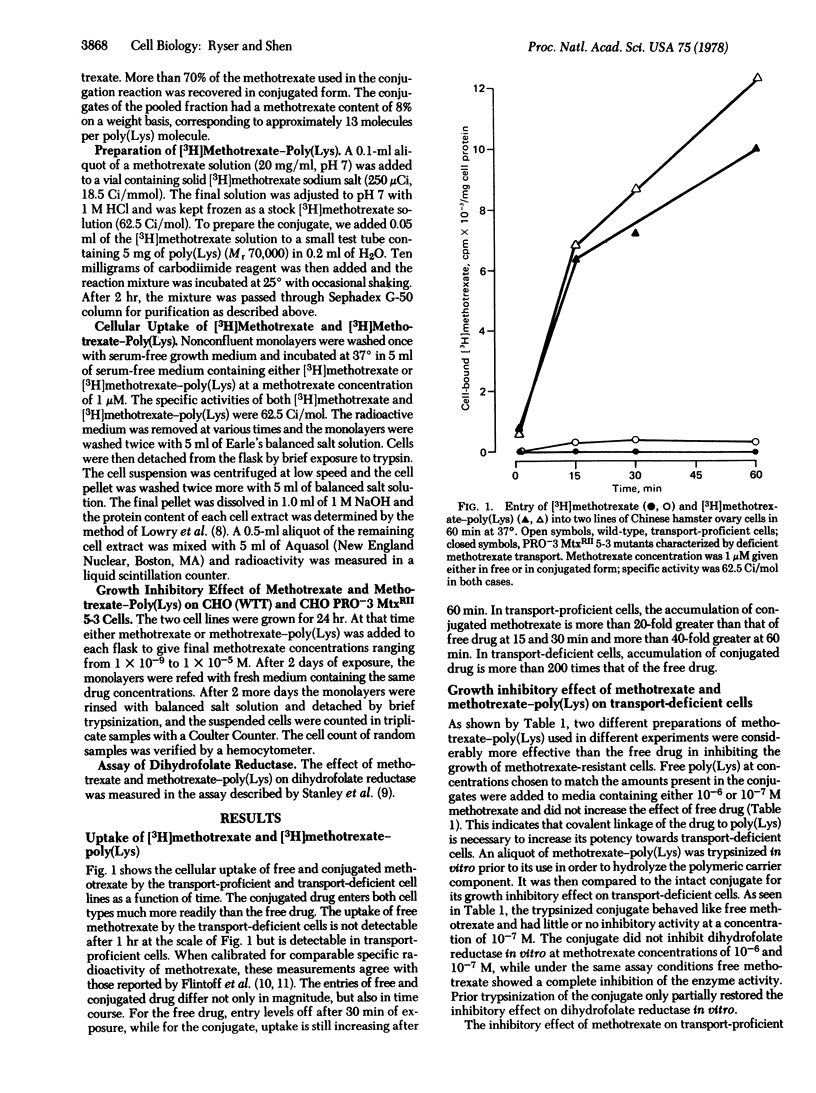
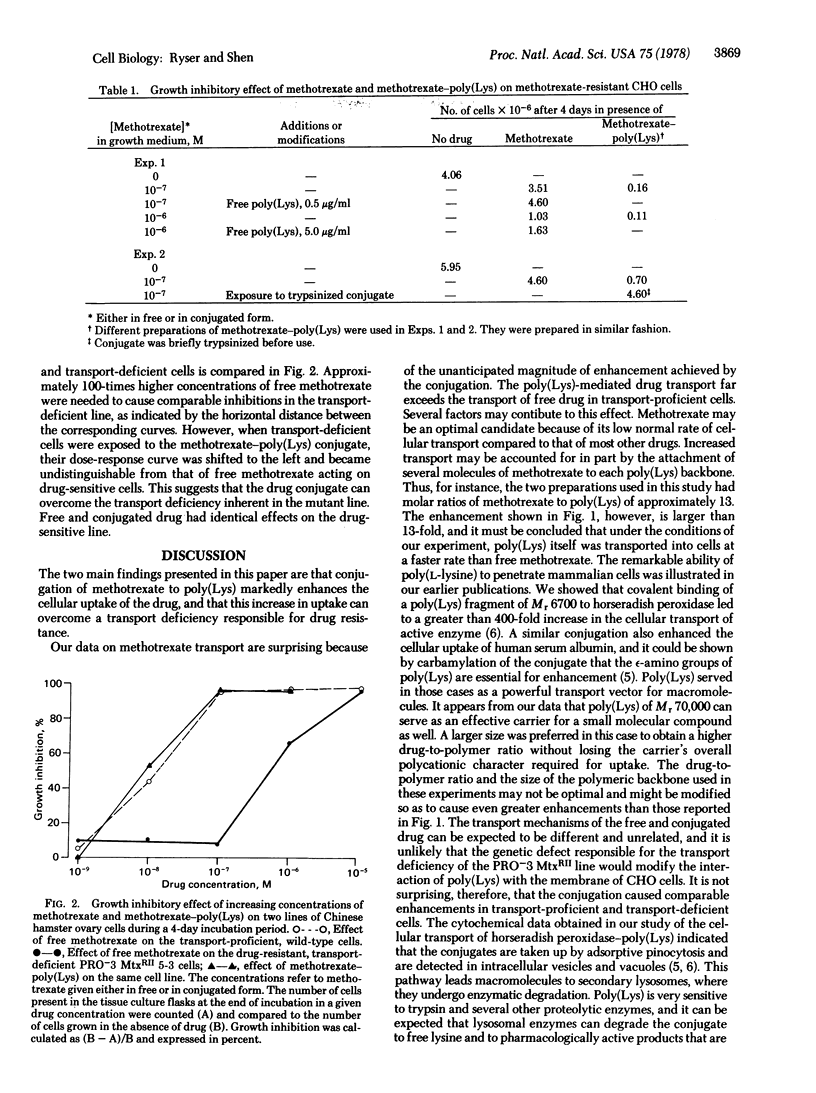
Methotrexate and [3H]methotrexate were conjugated through a carbodiimide-catalyzed reaction to a 70,000 molecular weight poly(L-lysine) in molar ratios of approximately 13 to 1. The cellular uptake of labeled conjugate was far in excess of the uptake of free drug in cells that were either proficient or deficient in methotrexate transport. The conjugate markedly inhibited the growth of PRO-3 MtxRII 5-3 Chinese hamster ovary cells, which are known to be drug resistant by virtue of a deficient methotrexate transport. The cells, however, were not inhibited by the same concentrations of free poly(Lys) and free drug. The 100-fold difference in drug concentration needed to inhibit the mutant cells and their corresponding wild type was totally abolished by exposing the methotrexate-resistant cells to methotrexate-poly(Lys). That the drug is carried into the resistant cells as intact drug-poly(Lys) is evident also from the fact that the conjugate is rendered inactive by brief trypsinization in vitro. Because the conjugate fails to inhibit dihydrofolate reductase (5,6,7,8-tetrahydrofolate: NADP+ oxidoreductase; EC 1.5.1.3) in vitro, it must be concluded that the strong growth inhibitory effect of the conjugate is due to the intracellular hydrolysis of its polymeric backbone, followed by the release inside the cell of a pharmacologically active form of methotrexate. Our date show that in methotrexate-resistant cells the intracellular release of active drug after uptake of conjugate is of the same order of magnitude as the uptake of free drug by transport-proficient cells and, hence, that the drug resistance due to deficient transport can be totally overcome.
Keywords: polymer-bound drug, piggyback pinocytosis, membrane transport of macromolecules, polycationic carrier
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- EYBL V., RYSER H. THE ACUTE TOXICITY OF CRYSTALLIZED FERRITIN, CADMIUM-FREE FERRITIN AND CDCL2 ON EHRLICH ASCITES CARCINOMA CELLS. Naunyn Schmiedebergs Arch Exp Pathol Pharmakol. 1964 May 11;248:153–165. doi: 10.1007/BF00246670. [DOI] [PubMed] [Google Scholar]
- Flintoff W. F., Davidson S. V., Siminovitch L. Isolation and partial characterization of three methotrexate-resistant phenotypes from Chinese hamster ovary cells. Somatic Cell Genet. 1976 May;2(3):245–261. doi: 10.1007/BF01538963. [DOI] [PubMed] [Google Scholar]
- Flintoff W., Saya L. The selection of wild-type revertants from methotrexate permeability mutants. Somatic Cell Genet. 1978 Mar;4(2):143–156. doi: 10.1007/BF01538980. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Levy R., Hurwitz E., Maron R., Arnon R., Sela M. The specific cytotoxic effects of daunomycin conjugated to antitumor antibodies. Cancer Res. 1975 May;35(5):1182–1186. [PubMed] [Google Scholar]
- MATHE G., TRAN BA L. O. C., BERNARD J. Effet sur la leucémie 1210 de la souris d'un combinaison par diazotation d'A-méthoptèrine et de gamma-globulines de hamsters porteurs de cette leucèmie par hétérogreffe. C R Hebd Seances Acad Sci. 1958 Mar 10;246(10):1626–1628. [PubMed] [Google Scholar]
- Marks T. A., Venditti J. M. Potentiation of actinomycin D or adriamycin antitumor activity with DNA. Cancer Res. 1976 Feb;36(2 Pt 1):496–504. [PubMed] [Google Scholar]
- Ryser H. J., Hancock R. Histones and basic polyamino acids stimulate the uptake of albumin by tumor cells in culture. Science. 1965 Oct 22;150(3695):501–503. doi: 10.1126/science.150.3695.501. [DOI] [PubMed] [Google Scholar]
- Ryser H. J., Shen W. C., Merk F. B. Membrane transport of macromolecules: new carrier functions of proteins and poly(amino acids). Life Sci. 1978 Apr 3;22(13-15):1253–1260. doi: 10.1016/0024-3205(78)90094-2. [DOI] [PubMed] [Google Scholar]
- Shen W. C., Ryser H. J. Conjugation of poly-L-lysine to albumin and horseradish peroxidase: a novel method of enhancing the cellular uptake of proteins. Proc Natl Acad Sci U S A. 1978 Apr;75(4):1872–1876. doi: 10.1073/pnas.75.4.1872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trouet A., Deprez-de Campeneere D., De Duve C. Chemotherapy through lysosomes with a DNA-daunorubicin complex. Nat New Biol. 1972 Sep 27;239(91):110–112. doi: 10.1038/newbio239110a0. [DOI] [PubMed] [Google Scholar]
- de Duve C., de Barsy T., Poole B., Trouet A., Tulkens P., Van Hoof F. Commentary. Lysosomotropic agents. Biochem Pharmacol. 1974 Sep 15;23(18):2495–2531. doi: 10.1016/0006-2952(74)90174-9. [DOI] [PubMed] [Google Scholar]



