Abstract
Schistosomiasis, a parasitic disease caused by trematode flatworms of the genus Schistosoma, affects more than 200 million people worldwide, and its control is dependent on a single drug, praziquantel. Tanacetum vulgare (Asteraceae) is used in folk medicine as a vermifuge. This study aimed to investigate the in vitro schistosomicidal activity of the crude extract (TV) and the essential oil (TV-EO) from the aerial parts of T. vulgare. TV-EO was obtained by hydrodistillation and analyzed by GC/MS, which allowed the identification of β-thujone (84.13%) as the major constituent. TV and TV-EO, at 200 μg/mL, decreased motor activity and caused 100% mortality of all adult worms. At 100 and 50 μg/mL, only TV caused death of all adult worms, while TV-EO was inactive. TV (200 μg/mL) was also able to reduce viability and decrease production of developed eggs. Confocal laser scanning microscopy showed morphological alterations in the tegument of the S. mansoni surface after incubation with TV (50 and 100 μg/mL). Quantitative analysis on the schistosomes tegument showed that TV caused changes in the numbers of tubercles of S. mansoni male worms in a dose-dependent manner. The findings suggest that T. vulgare is a potential source of schistosomicidal compounds.
1. Introduction
Schistosomiasis, a parasitic disease caused by trematode flatworms of the genus Schistosoma, is the second major neglected tropical disease, with major economic and public-health consequences. The major etiological agent of human schistosomiasis is Schistosoma mansoni, and it is estimated that more than 200 million people are infected and 779 million people are at risk of infection worldwide [1, 2].
The treatment of schistosomiasis is based on the control of adult worms in infected patients, with praziquantel (PZQ) being the most widely used drug. However, PZQ does not prevent reinfection, is inactive against juvenile schistosomes, and has only a limited effect on already developed liver and spleen lesions [3, 4]. These limitations, in combination with a considerable concern about the development of PZQ resistance, have motivated the scientific community to develop novel and inexpensive drugs against schistosomiasis [5, 6]. In this regard, there has been intensification in the search for new schistosomicidal compounds from natural sources, mainly from plants, which continue to be a major source of biologically active metabolites that may provide lead structures for the development of new drugs [6, 7].
A number of recent studies have investigated the schistosomicidal activity of plants and their isolated compounds [6–8]. A number of extracts, essential oils, and isolated natural compounds displaying in vitro schistosomicidal activity have been identified [9–13]. Among tested plants, several members of the family Asteraceae have shown promising in vitro schistosomicidal activity [14, 15].
Plants of the genus Tanacetum (Asteraceae) are used over the years in folk medicines all over the world for many medicinal purposes, including anti-inflammatory and anthelmintic [16, 17]. Tanacetum extracts and its isolated compounds have been also reported to exhibit antiviral activity [17] and trypanocidal [18] and leishmanicidal [19] activities.
Tanacetum vulgare L., known as “Tansy” in Europe and “catinga-de-mulata” in Brazil, is widely used in folk medicine as a vermifuge and anti-inflammatory [16]. Also, the aerial parts of this plant are popularly used to treat migraine, neuralgia, and rheumatism, and as an anthelmintic and insect repellent [17]. Phytochemical studies have shown that T. vulgare contain several biologically active metabolites, mainly sesquiterpene lactones [16–19].
On the basis of the folkloric uses of T. vulgare as an anthelmintic and a vermifuge, as well as in continuation of our search for active natural sources against S. mansoni [20–24], the present study evaluated the in vitro schistosomicidal effects of the extract and the essential oil of T. vulgare, which have not yet been described.
2. Materials and Methods
2.1. Plant Material and Extraction
Aerial parts of T. vulgare L. were collected from the Horto Medicinal da Faculdade de Fármacia, in Juiz de Fora, MG, Brazil. A voucher specimen was deposited in the herbarium of the Botany Department of the Universidade Federal de Juiz de Fora, MG, Brazil. The plant material (229 g) was dried, powdered, and exhaustively extracted, by maceration at room temperature, using EtOH/H2O 8 : 2 (v/v). After filtration, the solvent was removed under reduced pressure to yield 5.3 g of the crude hydroalcoholic extract of T. vulgare (TV).
2.2. Essential Oil Extraction
The essential oil of T. vulgare (TV-EO) was obtained from aerial parts by hydrodistillation using a Clevenger-type apparatus for 4 h. After manual collection of the essential oil, traces of water were removed by freezing the sample below 4°C, followed by transfer of the unfrozen essential oil to a new vial. The yield was calculated as % (w/w) of the fresh aerial parts.
2.3. CG/MS Analysis
The TV-EO was analyzed by GC/MS analysis using a Shimadzu QP2010 Plus (Shimadzu Corporation, Kyoto, Japan) system equipped with an AOC-20i autosampler and a Restek Rtx-5MS fused-silica capillary column (5% phenyl-, 95% methylpolysiloxane; 30 m × 0.25 mm i.d., film thickness 0.25 μm). The oven temperature was programmed to increase from 60 to 240°C at 3°C/min; injector temperature, 240°C; ion-source temperature, 280°C; carrier gas, He (1.0 mL/min); split ratio, 1 : 10; injection volume, 0.1 μL. The mass spectrometer was operated in the electron ionization mode (70 eV), and the spectra were taken with a scan interval of 0.5 s over the mass range 40–600 Da. The quantification of each TV-EO constituent was done by internal normalization (%). The identification of the TV-EO components was based on the comparison of their retention indices (RI; determined in relation to the RT of n-alkanes (C8–C26)) and mass spectra with those of the Wiley 7, NIST 08, and FFNSC 1.2 spectral libraries, as well as with those reported in the literature [25].
2.4. Parasite. S. mansoni
(BH strain Belo Horizonte, Brazil) worms have been maintained in Biomphalaria glabrata snails and Mesocricetus auratus hamsters hosts at the Adolfo Lutz Institute (São Paulo, Brazil). Female hamsters, weighting 20–22 g, were infected by subcutaneous injection of 150 cercariae. After 9 weeks, adults S. mansoni specimens were recovered from the hamster by perfusion with RPMI 1640 medium supplemented with heparin [26]. The worms were washed in RPMI 1640 medium (Gibco) supplemented with 200 μg/mL of streptomycin, 200 UI/mL of penicillin (Invitrogen), and 25 mM of Hepes. Pairs of adult worms (male and female) were incubated in a 24-well culture plate (Techno Plastic Products, TPP) containing 2 mL of the same medium supplemented with 10% heat-inactivated calf serum at 37°C in a 5% CO2 atmosphere [27, 28]. All experiments were authorized by the Committee for Ethics in Animal Care of Adolfo Lutz Institute, in accordance with nationally and internationally accepted principles for laboratory animal use and care.
2.5. In Vitro Studies with S. mansoni
For the in vitro test with S. mansoni, TV and TV-EO were evaluated at concentrations of 10, 50, 100, and 200 μg/mL, according to works previously described [29, 30]. Samples were dissolved in 0.5% DMSO and added to RPMI 1640 medium containing one pair of adult worms that had been allowed for 24 h to adapt to the culture medium. Worm motor activity, tegumental alterations, changes in the pairing, and survival of the parasites were monitored on daily basis for 3 days using an inverted microscope and a stereomicroscope (SMZ 1000, Nikon). All experiments were carried out in triplicate and repeated at least three times, using 10 μM praziquantel (PZQ) as positive control group, and RPMI 1640 medium and RPMI 1640 with 0.5% DMSO as negative control groups.
2.6. Tegumental Changes
The quantification of the number of tubercles was performed for TV (the most active sample in tegument) using a confocal microscope. After the established times or in the occurrence of death, the parasites were fixed in formalin-acetic-alcohol solution (FAA) and analyzed under a confocal microscope (Laser Scanning Microscopy, LSM 510 META, Zeiss) at 488 nm (exciting) and 505 nm (emission) as described by [28, 29]. A minimum of three areas of the tegument of each parasite were assessed. The numbers of tubercles were counted in 20,000 μm2 of area calculated with the Zeiss LSM Image Browser software. A blind analysis was performed by observer with experience and training in parasitology.
2.7. Viability Assay
For the viability assay against S. mansoni, pairs of adult worms were incubated for 120 h with TV (the most active sample) at concentration of 200 μg/mL; the viability assay was performed according to MTT assay [31, 32]. After incubation, each pair of adult worms was placed individually into wells (96-well plates) containing 100 μL of phosphate-buffered saline (PBS) with 5 mg MTT/mL for 30 min at 37°C. The solution was carefully removed and replaced with 200 μL of DMSO and the worms were allowed to stand in DMSO at room temperature for 1 h. The absorbance was read at 550 nm using as negative control groups RPMI 1640 medium and RPMI 1640 with 0.5% DMSO. Heat-killed worms at 56°C were used as positive controls groups.
2.8. Statistical Analysis
The statistical tests were performed with the GRAPHPAD PRISM (version 4.0) software. Significant differences were determined by one-way analysis of variance (ANOVA) and applying Tukey's test for multiple comparisons with a level of significance set at P < 0.05.
3. Results and Discussion
Many plants have been used throughout the world in traditional medicine for the treatment of parasite diseases [6]. In this scenario, several in vitro studies against Schistosoma species have been performed with crude plant extracts and essential oils, especially from species from the Asteraceae family [6, 14, 15]. T. vulgare is popularly used as anthelmintic and vermifuge, which encouraged us to evaluate the in vitro effect of its crude extract and essential oil against adult worms of S. mansoni, which have not been performed to date.
As shown in Table 1, positive control (PZQ, 10 μM) resulted in the death of all parasites within 24 hours, whereas no mortality was observed in the worms of the negative (RPMI medium) and solvent control (RPMI medium plus 0.5% DMSO) groups. On the other hand, after 24 h of incubation, both TV and TV-EO, at 200 μg/mL, cause 100% mortality of all adult worms. However, at 100 and 50 μg/mL, only TV causes 100% mortality, while TV-EO was inactive at concentrations of 100 to 10 μg/mL after 72 h of incubation (Table 1).
Table 1.
In vitro effects of crude hydroalcoholic extract (TV) and essential oil of T. vulgare (TV-EO) against S. mansoni adult worms.
| Groups | Incubation period (h) | Separated worms (%)a | Dead worms (%)a | Decrease in motor activity (%)a | Worms with tegumental alteration (%)a | ||
|---|---|---|---|---|---|---|---|
| Slight | Significant | Partial | Extensive | ||||
| Controlb | 24 | 0 | 0 | 0 | 0 | 0 | 0 |
| 72 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 0.5% DMSO | 24 | 0 | 0 | 0 | 0 | 0 | 0 |
| 72 | 0 | 0 | 0 | 0 | 0 | 0 | |
| PZQc | 24 | 0 | 100 | 0 | 100 | 0 | 100 |
| 72 | 0 | 100 | 0 | 100 | 0 | 100 | |
| TV | |||||||
| 10 μg/mL | 24 | 0 | 0 | 0 | 0 | 0 | 0 |
| 72 | 0 | 0 | 0 | 0 | 0 | 0 | |
| 50 μg/mL | 24 | 0 | 0 | 0 | 0 | 0 | 0 |
| 72 | 0 | 100 | 0 | 100 | 0 | 100 | |
| 100 μg/mL | 24 | 0 | 0 | 0 | 0 | 0 | 0 |
| 72 | 100 | 100 | 0 | 100 | 100 | 100 | |
| 200 μg/mL | 24 | 0 | 100 | 0 | 100 | 0 | 100 |
| 72 | 100 | 100 | 0 | 100 | 0 | 100 | |
| TV-EO | |||||||
| 10 μg/mL | 24 | 0 | 0 | 0 | 0 | 0 | 0 |
| 72 | 50 | 0 | 0 | 100 | 0 | 0 | |
| 50 μg/mL | 24 | 0 | 0 | 0 | 0 | 0 | 0 |
| 72 | 100 | 0 | 0 | 100 | 0 | 0 | |
| 100 μg/mL | 24 | 100 | 0 | 0 | 0 | 0 | 0 |
| 72 | 100 | 0 | 0 | 100 | 0 | 0 | |
| 200 μg/mL | 24 | 100 | 100 | 0 | 100 | 0 | 0 |
| 72 | 100 | 100 | 0 | 100 | 0 | 0 | |
aPercentages relative to the 20 worms investigated. bRPMI 1640. cTested at concentration of 10 μM.
Also, all pairs of worms separated into individual male and female when exposed to 100 and 200 μg/mL of both TV and TV-EO after 24 h of incubation. However, at 50 μg/mL, 100% worm pairs were separated by TV-EO only after 72 h of incubation. Moreover, male and female adults showed a significant decrease in motor activity after 24 h of incubation with 200 μg/mL of TV and TV-EO. However, motor activity was significantly decreased by the action of TV and TV-EO (at concentrations ranging from 50 to 100 μg/mL) only after 72 h of incubation. PZQ (10 μM) caused 100% mortality but no separation of worm pairs, whereas no effects were observed in worms in the negative (RPMI 1640 medium) and control (RPMI medium plus 0.5% DMSO) groups. Additionally, only TV (50, 100, and 200 μg/mL) caused significant tegumental alterations in adult worms after 72 h of incubation (Table 1).
In relation to the decrease in motor activity, studies revealed that the musculature of S. mansoni can serve as a therapeutic target, because the motility of worms is associated with important neurotransmitters or neuromodulators [33]. Nematode neuropeptide systems comprise an exceptionally complex array of ~250 peptidic signaling molecules that operate within a structurally simple nervous system of ~300 neurons. Thus, these signaling systems can provide tools for the discovery of more amenable targets such as neuropeptide receptors or neuropeptide processing enzymes [34].
In recent years, some essential oils have been reported as promising schistosomicidal agents [12–15, 33]. In this study, the in vitro effects of different concentrations of the essential oil from the T. vulgare (TV-EO) on S. mansoni adult worms were assessed.
The essential oil of T. vulgare (TV-EO) was obtained by hydrodistillation of the aerial parts, yielding 0.05% (w/w). The chemical composition of this oil is shown in Table 2 and Figure 1. A total of 7 compounds were identified, being six monoterpenes (89.58%) and one sesquiterpene (6.81%). β-Thujone (3, 84.15%) was identified as the major constituent (Figure 1) of TV-EO. β-Thujone has also been previously described as major constituent of the essential oils obtained from specimens of T. vulgare collected in Eurasian and North America [35]. Considering the chemical composition of TV-EO, its schistosomicidal in vitro activity may be related to β-thujone, its major constituent. Studies have associated the anthelmintic activity of the essential oils obtained from specimens of T. vulgare with β-thujone [36]. It has been reported that thujone may act on GABAA receptor, similarly to some anthelmintic drugs, such as ivermectin [37, 38]. Also, considering the mechanism by which TV-EO exerts its in vitro schistosomicidal effect, essential oils may have no specific cellular target, because lipophilic compounds, typically present in essential oils, may pass through the cell wall, tegument, and cytoplasmic membrane, damaging their structures, which may lead to the cellular lysis [39].
Table 2.
Composition of the essential oil of T. vulgare (TV-EO) identified by CG/MS analysis.
| Compounds | RIexp a | RIlit b | Content (%)c | Identificationd |
|---|---|---|---|---|
| 1,8-Cineol (1) | 1036 | 1031 | 0.55 | RI, MS |
| α-Thujone (2) | 927 | 930 | 1.64 | RI, MS |
| β-Thujone (3) | 1117 | 1114 | 84.13 | RI, MS |
| α-Pinene (4) | 935 | 939 | 1.04 | RI, MS |
| p-Cimene (5) | 1025 | 1026 | 0.68 | RI, MS |
| Terpinen-4-ol (6) | 1167 | 1171 | 1.54 | RI, MS |
| Germacrene-D (7) | 1480 | 1480 | 6.81 | RI, MS |
| Not identified | 1499 | — | 1.12 | RI, MS |
| Not identified | 1965 | — | 1.68 | RI, MS |
| Total | 96.39 | |||
|
| ||||
| Monoterpenes: 89.58% | ||||
| Sesquiterpenes: 6.81% | ||||
aRIexp: retention index determined relative to n-alkanes (C8–C26) on the Rtx-5MS column. bRIlit: retention index from [25]. cCalculated from the peak area relative to the total peak area. dCompound identification: RI, comparison of the RI with those of [25]; MS: comparison of the mass spectra with those of the Wiley 7, NIST 08, and FFNSC 1.2 spectral libraries as well as with those of [25].
Figure 1.
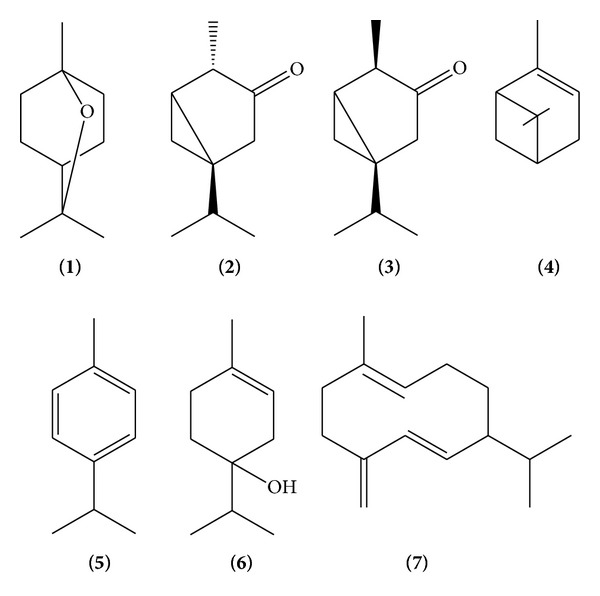
Chemical constituents identified by GC-MS in the essential oil of T. vulgare (TV-EO).
According to results observed in preliminary survival of 56-day-old adult worms of S. mansoni test (Table 1), TV was more active than TV-EO, due to its ability to cause 100% mortality of all parasites at lower concentrations, as well as to cause significant tegumental alterations. Because of that, TV was further tested to assess viability, oviposition, and morphological alterations in the parasite's tegument.
The viability of adult S. mansoni worms was evaluated during their in vitro incubation with TV (200 μg/mL) after a period of 120 hours (Figure 2). A 50% reduction was observed in the worms viability incubated with TV when compared with the negative control groups. However, the viability of these worms was significantly higher than that of the group of worms dead by heating.
Figure 2.
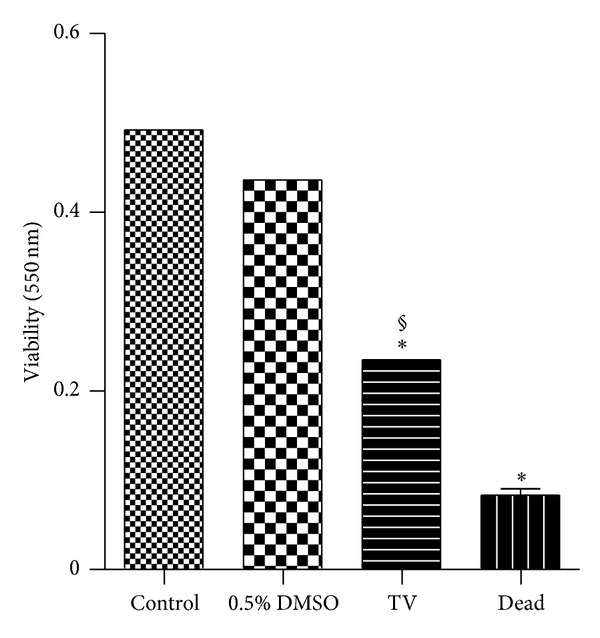
In vitro effect of the crude hydroalcoholic extract of T. vulgare (TV) on the viability of the S. mansoni adult worms. Pairs of adult worms were treated with TV at 200 μg/mL during 120 h and the viability was measured by MTT assay at 550 nm. RPMI 1640 medium and 0.5% DMSO in RPMI 1640 medium were used as negative control groups. Heat-killed worms at 56°C (dead) were used as positive control group. The viability was expressed as mean of the absorbance values from three experiments. *Significantly different from control (P < 0.05); §significantly different from dead group (P < 0.05).
Regarding production of developed eggs by adult worms of S. mansoni (Figure 3), TV (200 μg/mL) showed a significant decrease in the number of developed eggs, by 29.8%, after 120 h of incubation, in comparison with the negative control group treated with RPMI 1640 medium. However, the effect of TV on egg production may be correlated with its ability to separate adult worms into male and female.
Figure 3.
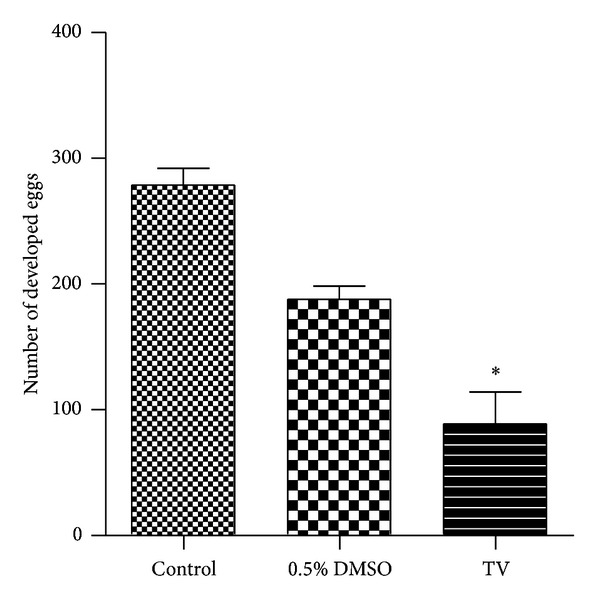
In vitro effect of the crude hydroalcoholic extract of T. vulgare (TV) at 200 μg/mL on egg development (quantitative analysis of the development phenotype). After treatment, the eggs were microscopically examined and scored as developed or undeveloped based on the presence or absence of the miracidium. Data are presented as the mean of developed eggs from two separate experiments. *Significantly different from control (P < 0.05).
The presence of S. mansoni eggs in the host tissues has been reported to be closely related to the pathology of human schistosomiasis [12, 20]. The permanent pairing of the schistosomes couples in the blood system of their hosts vertebrates throughout their lifespan causes high rate of oviposition, which is responsible for the resulting immunopathological lesions, characterized by inflammation and fibrosis in the target [13].
Moreover, light microscopic investigations (Table 1) showed that TV caused morphological alterations in the parasite's tegument, with no distinction between male and female worms (data not shown). No tegumental changes in adult worms were observed for the negative control group, while the positive control (PZQ) had tegumental alteration in 100% of the worms. In addition, the effect of TV on the parasite's tegument was monitored microscopically using confocal laser scanning microscopy. As shown in Figure 4, morphological alterations of the tegument on the S. mansoni surface were detected with TV at 50 μg/mL (Figure 4(d)) and 100 μg/mL (Figure 4(e)). Meanwhile, no abnormality was seen in the worms maintained in the negative control group. Thus, a pattern consisting of a combination of changes in the surface morphology was detected and correlated to the death of the adult worms. These pronounced changes in the aspect of the tubercles, which often appeared collapsed and disrupted, were similar to those reported in studies with some isolated natural compounds, such as piplartine, epiisopiloturine, and (+)-limonene epoxide [29, 30, 40].
Figure 4.
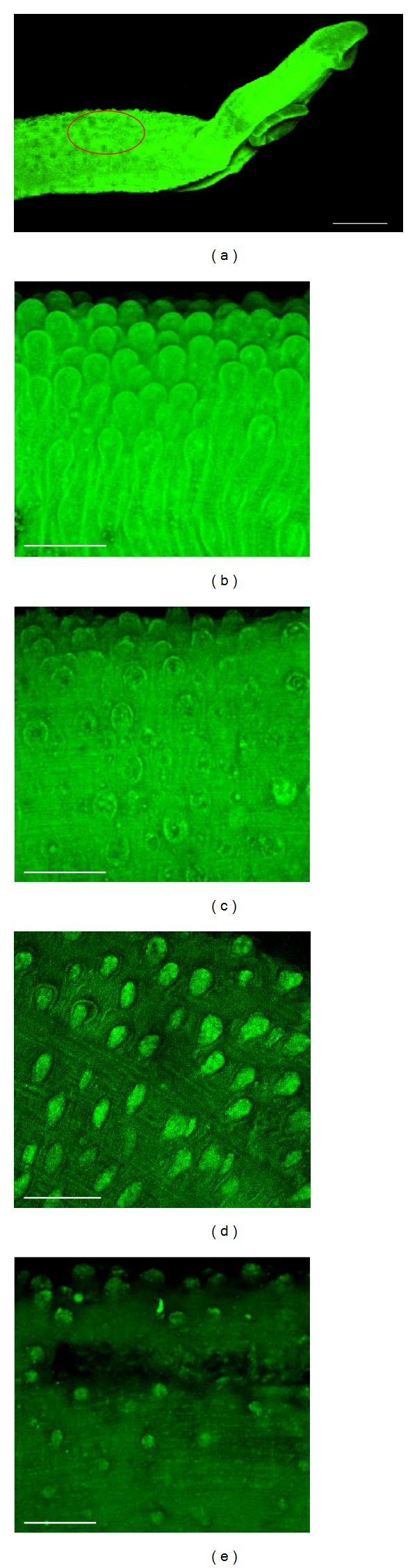
Confocal laser scanning microscopy investigation of the crude hydroalcoholic extract of T. vulgare (TV) in vitro schistosomicidal effect. In these experiments, pairs of adult worms were incubated in 24-well culture plates containing RPMI 1640 medium and treated with different concentrations of TV. After 120 h of incubation or in the case of death, adult male worms were fixed in FAA solution and fluorescent images were obtained using confocal microscopy (Carl Zeiss LSM 510 META). General view of the anterior worm region showing, in red, the location where tegument was analysed (a). Control worms in RPMI 1640 with 0.5% DMSO (b), and 10 μM praziquantel (c), compared to worms treated with 50 μg/mL of TV (d), and 100 μg/mL of TV (e). Scale bars, 500 μm (a) and 50 μm (b–e).
Additionally, morphological alterations on S. mansoni tegument were evaluated quantitatively after exposure to different concentrations of TV. In this quantitative analysis, areas of tegument of male worms were assessed, and the numbers of tubercles on the dorsal surface of parasites were counted [23, 27–29]. As shown in Figure 5, TV caused changes on tubercles of S. mansoni male worms in a dose-dependent manner. For example, the number of intact tubercles in an area of 20 000 μm2 on male worms of the negative control was 46, while in the groups exposed to 10, 25, 50, and 100 μg/mL of TV the number was, respectively, 39, 32, 23, and 16. Similar results, with the pattern of tubercle destruction in a dose-dependent manner, were obtained from the paired schistosomes exposed to some natural products, such as antimicrobial peptide dermaseptin and (+)-limonene epoxide [23, 29].
Figure 5.
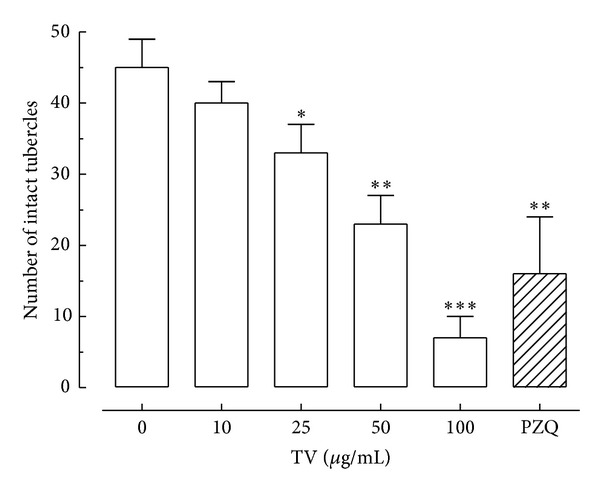
Quantitative analysis of morphological alterations on the tegument of S. mansoni after exposure to different concentrations of the crude hydroalcoholic extract of T. vulgare (TV). The quantification of the number of tubercles was performed using three-dimensional images obtained from laser scanning confocal microscopy. Indicated are numbers of intact tubercles, and these numbers were measured in a 20 000 μm2 area in a dorsal region of Schistosoma mansoni adult male worm (see Figure 4(a)) and calculated with the Zeiss LSM Image Browser software. Praziquantel (PZQ, 10 μM) was used as a positive control. A minimum of three tegument areas of each parasite were assessed. Values are means ± SD (bars) of ten male adult worms. *P < 0.05, **P < 0.01, and ***P < 0.001 compared with untreated group.
The tegument is extremely important to the infection success and survival in the host, and it has been a major target for the development of antischistosomal drugs [28], since most of the currently used drugs against schistosomes, such as PZQ [41], mefloquine [42], and artemether [43], act by damaging the schistosome tegument.
Several compounds, mainly sesquiterpene lactones (STL), have been identified as active constituents in previous phytochemical studies of the aerial parts of T. vulgare [17–19]. Some STL from T. vulgare, mainly parthenolide, proved to be active against some parasites, such as Trypanosoma cruzi [18] and Leishmania amazonensis [19]. Also, T. vulgare shows remarkable antioxidant properties, mainly due to its phenolic compounds content, especially flavonoids and caffeoylquinic acid derivatives [44]. The wide spectrum of T. vulgare activities can be mainly ascribed to the occurrence of STL [16]. Some STL have been reported as molluscicidal compounds, showing activity against adult Biomphalaria sp., a snail directly implicated in the transmission of schistosomiasis [45].
The mechanism by which TV exerts its in vitro schistosomicidal effect is unclear. However, adult worms of S. mansoni died accompanied by destruction of the worm body, and a relationship between tegumental damage and the death of worms was observed in the in vitro assays. Furthermore, in vivo investigations in mice infected by S. mansoni are necessary to determine the clinical potential of TV to treat schistosomiasis. In this case, mice may be treated orally using single or multiple oral doses at different life-cycle stages (e.g., schistosomula, juvenile, and adult worms). In addition, toxicological studies (e.g., acute oral LD50 of TV) should be examined.
4. Conclusion
In this study, we have reported an investigation on the in vitro schistosomicidal effects of the crude hydroalcoholic extract (TV) and the essential oil from T. vulgare (TV-EO) for the first time. It was demonstrated that both TV and TV-EO possess significant activity against adult worms of S. mansoni. The activity of TV-EO may be related, at least in part, to monoterpene thujones, which were detected as major constituents in TV-EO. Our study reinforced the traditional use of T. vulgare as a vermifuge and anthelmintic. Considering the obtained results, further biological studies, as well as phytochemical investigations, are in progress with TV in order to identify its major active compound and elucidate its mechanism(s) of schistosomicidal action.
Acknowledgments
The authors are grateful to FAPEMIG (Grant no. 0171/11) and FAPESP for financial support and CAPES, PIBIC/CNPq/UFJF and CNPq for fellowships. They are grateful to Mr. Jefferson S. Rodrigues for excellent technical assistance with S. mansoni life cycle maintenance at the Adolfo Lutz Institute (São Paulo, SP, Brazil). They also thank Dr. Henrique K. Roffato and Dr. Ronaldo Z. Mendonça (Butantan Institute, São Paulo, SP, Brazil) for expert help with confocal microscope studies (FAPESP, Project 00/11624-5).
Conflict of Interests
The authors declare that there is no conflict of interests.
References
- 1.de Moraes J, Nacimento C, Yamagushi LF, Kato MJ, Nakano E. Schistosoma mansoni: in vitro schistosomicidal activity schistosomicidal activity and tegumental alterations induced by piplartine on schistosomula. Experimental Parasitology. 2012;132(2):222–227. doi: 10.1016/j.exppara.2012.07.004. [DOI] [PubMed] [Google Scholar]
- 2.WHO. Weekly Epidemiological Record. Vol. 86. Geneva, Switzerland: World Health Organization; 2011. Schistosomiasis: number of people treated worldwide in 2009. [Google Scholar]
- 3.Stelma FF, Talla I, Sow S, et al. Efficacy and side effects of praziquantel in an epidemic focus of Schistosoma mansoni . American Journal of Tropical Medicine and Hygiene. 1995;53(2):167–170. doi: 10.4269/ajtmh.1995.53.167. [DOI] [PubMed] [Google Scholar]
- 4.Pica-Mattoccia L, Cioli D. Sex- and stage-related sensitivity of Schistosoma mansoni to in vivo and in vitro praziquantel treatment. International Journal for Parasitology. 2004;34(4):527–533. doi: 10.1016/j.ijpara.2003.12.003. [DOI] [PubMed] [Google Scholar]
- 5.Braguine CG, Costa ES, Magalhães LG, et al. Schistosomicidal evaluation of Zanthoxylum naranjillo and its isolated compounds against Schistosoma mansoni adult worms. Journal of Biosciences C. 2010;64(11-12):793–797. doi: 10.1515/znc-2009-11-1207. [DOI] [PubMed] [Google Scholar]
- 6.de Castro CC, Dias MM, Pessoa de Rezende T, Magalhães LG, da Silva Filho AA. Fighting Multidrug Resistance with Herbal Extracts, Essential Oils and Their Components. Londres, UK: Elsevier; 2013. Natural products with activity against Schistosoma species; pp. 109–134. [Google Scholar]
- 7.Ramalhete C, Magalhães LG, Rodrigues V, et al. In vitro schistosomicidal activity of Balsaminol F and Karavilagenin C. Planta Medica. 2012;78(18):1912–1917. doi: 10.1055/s-0032-1327832. [DOI] [PubMed] [Google Scholar]
- 8.Pereira AC, Magalhães LG, Gonalves UO, et al. Schistosomicidal and trypanocidal structure-activity relationships for (±)-licarin A and its (-)- and (+)-enantiomers. Phytochemistry. 2011;72(11-12):1424–1430. doi: 10.1016/j.phytochem.2011.04.007. [DOI] [PubMed] [Google Scholar]
- 9.Cunha NL, Uchôa CJM, Cintra LS, et al. In vitro schistosomicidal activity of some brazilian cerrado species and their isolated compounds. Evidence-Based Complementary and Alternative Medicine. 2012;2012:8 pages. doi: 10.1155/2012/173614.173614 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Aguiar GP, Melo NI, Wakabayashi KAL, et al. Chemical composition and in vitro schistosomicidal activity of the essential oil from the flowers of Bidens sulphurea (Asteraceae) Natural Product Research. 2013;27(10):920–924. doi: 10.1080/14786419.2012.671314. [DOI] [PubMed] [Google Scholar]
- 11.Porto TS, da Silva Filho AA, Magalhães LG, et al. Fungal transformation and schistosomicidal effects of pimaradienoic acid. Chemistry and Biodiversity. 2012;9(8):1465–1479. doi: 10.1002/cbdv.201100336. [DOI] [PubMed] [Google Scholar]
- 12.Caixeta SC, Magalhães LG, Demelo NI, et al. Chemical composition and in vitro schistosomicidal activity of the essential oil of Plectranthus neochilus grown in Southeast Brazil. Chemistry and Biodiversity. 2011;8(11):2149–2157. doi: 10.1002/cbdv.201100167. [DOI] [PubMed] [Google Scholar]
- 13.Tonuci LRS, de Melo NI, Dias HJ, et al. In vitro schistosomicidal effects of the essential oil of Tagetes erecta . Brazilian Journal of Pharmacognosy. 2011;22(1):88–93. [Google Scholar]
- 14.de Melo NI, Magalhães LG, de Carvalho CE, et al. Schistosomicidal activity of the essential oil of Ageratum conyzoides L. (Asteraceae) against adult Schistosoma mansoni worms. Molecules. 2011;16(1):762–773. doi: 10.3390/molecules16010762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Parreira NA, Magalhães LG, Morais DR, et al. Antiprotozoal, schistosomicidal, and antimicrobial activities of the essential oil from the leaves of Baccharis dracunculifolia . Chemistry and Biodiversity. 2010;7(4):993–1001. doi: 10.1002/cbdv.200900292. [DOI] [PubMed] [Google Scholar]
- 16.Rosselli S, Bruno M, Raimondo FM, et al. Cytotoxic effect of eudesmanolides isolated from flowers of Tanacetum vulgare ssp. Siculum . Molecules. 2012;17:8186–8195. doi: 10.3390/molecules17078186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Onozato T, Nakamura CV, Garcia Cortez DA, Dias Filho BP, Ueda-Nakamura T. Tanacetum vulgare: antiherpes virus activity of crude extract and the purified compound parthenolide. Phytotherapy Research. 2009;23(6):791–796. doi: 10.1002/ptr.2638. [DOI] [PubMed] [Google Scholar]
- 18.Pelizzaro-Rocha KJ, Tiuman TS, Izumi E, Ueda-Nakamura T, Filho BPD, Nakamura CV. Synergistic effects of parthenolide and benznidazole on Trypanosoma cruzi . Phytomedicine. 2010;18(1):36–39. doi: 10.1016/j.phymed.2010.09.005. [DOI] [PubMed] [Google Scholar]
- 19.Tiuman TS, Ueda-Nakamura T, Garcia Cortez DA, et al. Antileishmanial activity of parthenolide, a sesquiterpene lactone isolated from Tanacetum parthenium . Antimicrobial Agents and Chemotherapy. 2005;49(1):176–182. doi: 10.1128/AAC.49.11.176-182.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fernandes FS, Rezende Júnior CO, Fernandes TS, et al. Anthelmintic effects of alkylated diamines and amino alcohols against Schistosoma mansoni . BioMed Research International. 2013;2013:9 pages. doi: 10.1155/2013/783490.783490 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.de Moraes J, Keiser J, Ingram K, et al. In vitro synergistic interaction between amide piplartine and antimicrobial peptide dermaseptin against Schistosoma mansoni schistosomula and adult worms. Current Medicinal Chemistry. 2013;20(2):301–309. doi: 10.2174/092986713804806694. [DOI] [PubMed] [Google Scholar]
- 22.de Moraes J, Carvalho AAL, Nakano E, et al. Anthelmintic activity of carvacryl acetate against Schistosoma mansoni . Parasitology Research. 2013;112(2):603–607. doi: 10.1007/s00436-012-3172-7. [DOI] [PubMed] [Google Scholar]
- 23.de Moraes J, Nascimento C, Miura LMCV, Leite JRSA, Nakano E, Kawano T. Evaluation of the in vitro activity of dermaseptin 01, a cationic antimicrobial peptide, against Schistosoma mansoni . Chemistry and Biodiversity. 2011;8(3):548–558. doi: 10.1002/cbdv.201000163. [DOI] [PubMed] [Google Scholar]
- 24.Carrara VS, Vieira SCH, Paula RG, et al. In vitro schistosomicidal effects of aqueous and dichloromethane fractions from leaves and stems of Piper species and the isolation of an active amide from P. amalago L., (Piperaceae) Journal of Helminthology. 2013;8:1–6. doi: 10.1017/S0022149X13000205. [DOI] [PubMed] [Google Scholar]
- 25.Adams RP. Identification of Essential Oil Components by Chomatography/Mass Spectrometry. 4th edition. Carol Stream, Ill, USA: Allured; 2007. [Google Scholar]
- 26.Smithers SR, Terry RJ. The infection of laboratory hosts with cercariae of Schistosoma mansoni and the recovery of the adult worms. Parasitology. 1965;55(4):695–700. doi: 10.1017/s0031182000086248. [DOI] [PubMed] [Google Scholar]
- 27.Moraes J. Antischistosomal natural compounds: present challenges for new drug screens. In: Rodriguez-Morales AJ, editor. Current Topics in Tropical Medicine. Rijeka, Croatia: InTech; 2012. pp. 333–358. [Google Scholar]
- 28.Moraes JD, Nascimento C, Lopes POMV, et al. Schistosoma mansoni: in vitro schistosomicidal activity of piplartine. Experimental Parasitology. 2011;127(2):357–364. doi: 10.1016/j.exppara.2010.08.021. [DOI] [PubMed] [Google Scholar]
- 29.de Moraes J, Nunes LCC, Almeida AAC, et al. Anthelmintic activity of a natural compound (+)-limonene epoxide against Schistosoma mansoni . Planta Medica. 2013;79:253–258. doi: 10.1055/s-0032-1328173. [DOI] [PubMed] [Google Scholar]
- 30.de Moraes J, Nascimento da Silva MP, Ohlweiler FP, Kawano T. Schistosoma mansoni and other larval trematodes in Biomphalaria tenagophila (planorbidae) from Guarulhos, São Paulo State, Brazil. Revista do Instituto de Medicina Tropical de São Paulo. 2009;51(2):77–82. doi: 10.1590/s0036-46652009000200004. [DOI] [PubMed] [Google Scholar]
- 31.Magalhães LG, Kapadia GJ, da Silva Tonuci LR, et al. In vitro schistosomicidal effects of some phloroglucinol derivatives from Dryopteris species against Schistosoma mansoni adult worms. Parasitology Research. 2010;106(2):395–401. doi: 10.1007/s00436-009-1674-8. [DOI] [PubMed] [Google Scholar]
- 32.Magalhães LG, Machado CB, Morais ER, et al. In vitro schistosomicidal activity of curcumin against Schistosoma mansoni adult worms. Parasitology Research. 2009;104(5):1197–1201. doi: 10.1007/s00436-008-1311-y. [DOI] [PubMed] [Google Scholar]
- 33.de Oliveira RN, Rehder VLG, Santos Oliveira AS, et al. Schistosoma mansoni: in vitro schistosomicidal activity of essential oil of Baccharis trimera (less) DC. Experimental Parasitology. 2012;132:135–143. doi: 10.1016/j.exppara.2012.06.005. [DOI] [PubMed] [Google Scholar]
- 34.Marks NJ, Maule AG. Neuropeptides in helminths: occurrence and distribution. Advances in Experimental Medicine and Biology. 2010;692:49–77. doi: 10.1007/978-1-4419-6902-6_4. [DOI] [PubMed] [Google Scholar]
- 35.Wolf VC, Gassmann A, Clasen BM, Smith AG, Müller C. Genetic and chemical variation of Tanacetum vulgare in plants of native and invasive origin. Biological Control. 2012;61(3):240–245. [Google Scholar]
- 36.Albert-Puleo M. Mythobotany, pharmacology, and chemistry of thujone-containing plants and derivatives. Economic Botany. 1978;32(1):65–74. [Google Scholar]
- 37.Czyzewska MM, Mozrzymas JW. Monoterpene α-thujone exerts a differential inhibitory action on GABAAreceptors implicated in phasic and tonic GABAergic inhibition. European Journal of Pharmacology. 2013;702:38–43. doi: 10.1016/j.ejphar.2013.01.032. [DOI] [PubMed] [Google Scholar]
- 38.Gatti FR, de Oliveira CM, Servilha TR, Sanchez APG. Tungiase disseminada tratada com ivermectina. Anais Brasileiros de Dermatologia. 2008;83(4):339–342. [Google Scholar]
- 39.Bakkali F, Averbeck S, Averbeck D, Idaomar M. Biological effects of essential oils—a review. Food and Chemical Toxicology. 2008;46(2):446–475. doi: 10.1016/j.fct.2007.09.106. [DOI] [PubMed] [Google Scholar]
- 40.Veras LM, Guimarães MA, Campelo YD, et al. Activity of epiisopiloturine against Schistosoma mansoni . Current Medicinal Chemistry. 2012;19(13):2051–2058. doi: 10.2174/092986712800167347. [DOI] [PubMed] [Google Scholar]
- 41.Shuhua X, Binggui S, Chollet J, Tanner M. Tegumental changes in adult Schistosoma mansoni harboured in mice treated with praziquantel enantiomers. Acta Tropica. 2000;76(2):107–117. doi: 10.1016/s0001-706x(00)00076-0. [DOI] [PubMed] [Google Scholar]
- 42.Manneck T, Haggenmüller Y, Keiser J. Morphological effects and tegumental alterations induced by mefloquine on schistosomula and adult flukes of Schistosoma mansoni . Parasitology. 2010;137(1):85–98. doi: 10.1017/S0031182009990965. [DOI] [PubMed] [Google Scholar]
- 43.Xiao S, Shen B, Chollet J, Utzinger J, Tanner M. Tegumental changes in adult Schistosoma mansoni harbored in mice treated with artemether. Journal of Parasitology. 2000;86(5):1125–1132. doi: 10.1645/0022-3395(2000)086[1125:TCIASM]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 44.Álvarez AL, Habtemariam S, Juan-Badaturuge M, Jackson C, Parra F. In vitro anti HSV-1 and HSV-2 activity of Tanacetum vulgare extracts and isolated compounds: an approach to their mechanisms of action. Phytotherapy Research. 2011;25(2):296–301. doi: 10.1002/ptr.3382. [DOI] [PubMed] [Google Scholar]
- 45.Borkosky S, Ponce de Leòn S, Juárez G, González Sierra M, Bardón A. Molluscicidal sesquiterpene lactones from species of the tribe Vernonieae (Compositae) Chemistry and Biodiversity. 2009;6(4):513–519. doi: 10.1002/cbdv.200800156. [DOI] [PubMed] [Google Scholar]


