Abstract
Background:
Laparoscopic radical cystectomy (LRC) is increasingly being used for muscle-invasive bladder cancer. However, high levels of clinical evidence comparing laparoscopic vs open radical cystectomy (ORC) are lacking.
Methods:
A prospective randomised controlled clinical trial comparing LRC vs ORC in patients undergoing radical cystectomy for bladder cancer. Thirty-five patients were eligible for final analysis in each group.
Results:
The median follow-up was 26 months (range, 4–59 months) for laparoscopic vs 32 months (range, 6–60 months) for ORC. Significant differences were noted in operative time, estimated blood loss (EBL), blood transfusion rate, analgesic requirement, and time to resumption of oral intake. No significant differences were noted in the length of hospital stay, complication rate, lymph node yield (14.1±6.3 for LRC and 15.2±5.9 for ORC), positive surgical margin rate, postoperative pathology, or recurrence rate (7 for LRC and 8 for ORC). The 5-year recurrence-free survival with laparoscopic vs ORC was 78.5% vs 70.9%, respectively (P=0.773). The overall survival with laparoscopic vs ORC was 73.8% vs 67.4%, respectively (P=0.511).
Conclusion:
Our study demonstrated that LRC is superior to ORC in perioperative outcomes, including EBL, blood transfusion rate, and analgesic requirement. We found no major difference in oncologic outcomes. The number of patients is too small to allow for a final conclusion.
Keywords: bladder cancer, laparoscopic radical cystectomy, open radical cystectomy, orthotopic ileal neobladder, randomised controlled study
Although open radical cystectomy (ORC) and pelvic lymphadenectomy remain the gold standard treatment for muscle-invasive bladder cancer and high-risk recurrent noninvasive bladder cancer, there is increasing interest in laparoscopic radical cystectomy (LRC) and robot-assisted radical cystectomy. LRC may provide certain advantages in terms of blood loss, analgesic requirements, reduced scarring, and recovery. Studies of robot-assisted radical cystectomy also demonstrated its feasiblity and potential benefits of decreased blood loss (Nix et al, 2010; Parekh et al, 2012). However, because of the lack of adequate comparative data and sufficient follow-up to demonstrate similar oncologic outcomes compared with traditional ORC, there remains significant scepticism about the true benefits of MIRC (Challacombe et al, 2011).
Currently, high levels of clinical evidence with respect to the long-term oncologic results of MIRC are limited in the peer-reviewed literature. Most reports are retrospective comparative studies (Hemal and Kolla, 2007; Porpiglia et al, 2007; Ha et al, 2010; Yu et al, 2012) or case series studies (Haber and Gill, 2007; Gillion et al, 2011), which represent significant selection bias. Herein, we present the perioperative complications, ileal neobladder function, and medium-term survival of a prospective randomised controlled clinical trial (RCT) comparing LRC vs ORC.
Patients and Methods
All patients provided written informed consent, and were eligible for random assignment to either laparoscopic or open-surgery groups. This trial was approved by our local institutional ethical committee, and was conducted in accordance with good clinical practices and the Declaration of Helsinki. This study was funded by Sun Yat-Sen University Clinical Research 5010 Program, and was registered in Chinese Clinical Trial Registry (http://www.chictr.org/cn/, No: ChiCTR-TRC-10001003).
Patient selection
The inclusion criteria were (1) muscle-invasive bladder cancer T2–4a, N0–Nx, M0; (2) high-risk and recurrent non-–muscle-invasive tumours; (3) T1G3; and (4) extensive non-muscle-invasive disease that could not be controlled by transurethral resection and intravesical therapy. The exclusion criteria were (1) patient elected not to participate; (2) the presence of contraindications to radical cystectomy, including distant metastasis, an American Society of Anaesthesiologists score >3, and severe cardiac or pulmonary disease; (3) the presence of contraindications to neobladder, including urethral tumour, urethral stricture, abnormal abdominal straining, and decompensated renal function.
The preoperative evaluation included complete blood count, blood biochemistry, urine cytology, and computed tomography. Diagnostic transurethral resection and pathologic examination were performed in patients with invasive bladder cancer.
Randomisation
Patients were randomly assigned 1 : 1 at the preoperative assessment to receive either laparoscopic or open surgery at the time of preoperative assessment. Random numbers were computer generated by a statistician (J Zhang), and patients were stratified according to sex, age, and clinical stage.
Surgical technique
The surgical procedure was the same in the two groups except for the surgical approach. Our surgical techniques for LRC, bilateral PLND, and extracorporeal diversion have been previously described in detail (Huang et al, 2008). A five-port, fan-shaped transperitoneal approach was used, and bilateral PLND was performed first. The boundaries of a standard PLND were the bifurcation of the common iliac artery proximally, the genitofemoral nerve laterally, the circumflex iliac vein and lymph node of Cloquet distally, and the hypogastric vessels posteriorly, including the obturator fossa. Patients with a preoperative evaluation of T3 to T4a disease underwent extended PLND, in which the proximal extent of the dissection was the aortic bifurcation.
The peritoneum of the rectovesical pouch in men (Douglas pouch in women) was incised transversely, and the ampullae of the vas deferens were transected bilaterally. The dissection was along the dorsal side of the seminal vesicles. The seminal vesicles were not dissected but were left en bloc with the bladder. Denonvilliers' fascia was incised, and Denonvilliers' space between the rectum and the prostate was developed. An inverted U-shaped peritoneotomy was made to expose the anterior bladder wall and Retzius' space, and the endopelvic fascia was incised bilaterally. The puboprostatic ligament was dissected, and the dorsal vein complex was suture ligated.
Both ureters were ligated and transected with a LigaSure (Covidien, Boulder, CO, USA) just outside the bladder. The lateral pedicles of the bladder and the prostate were bilaterally divided with the LigaSure, and the apex of the prostate was dissected using scissors. The specimen was placed in an endobag and immediately extracted through a 5-cm midline incision in the lower abdomen. The construction of an M-shaped ileal neobladder and ureteral reimplantation using a ureteral split-nipple technique was performed extracorporeally. Vesicourethral anastomosis was performed intracorporeally with a running-suture technique under laparoscopic visualisation.
Prophylactic antibiotic was given 30 min preoperatively. An additional dose was given when the operative time exceeded 3 h. Postoperative antibiotic administration was discontinued unless evidence of infection persisted. All surgeries were performed by a single surgeon (J. Huang), experienced in more than 100 cases of LRC and also experienced in ORC before this trial (Huang et al, 2008, 2010).
Pathological analysis and adjuvant therapy
All specimens were analysed according to the TNM classification (2002 Union Internationale Contre le Cancer) and graded by the 1973 World Health Organization classification. Pathologic subgroups were defined as organ confined (⩽pT2) and non-organ confined (⩾pT3) as well as lymph node negative and positive. Adjuvant chemotherapy was administered to patients with non-organ-confined or lymph node-positive disease with a gemcitabine and cisplatin combination chemotherapy regimen (Pectasides et al, 2005).
Follow-up scheme
Postoperative follow-up was conducted at 3-month intervals during the first year, at 6-month intervals during the second year, and annually thereafter. Follow-up visits consisted of a history, physical examination, and routine biochemical profile. Ultrasonography of the abdomen, urography, and chest X-rays was performed at 3, 6, and 12 months postoperatively, then annually unless otherwise clinically indicated. Abdominal/pelvic computed tomography scans were performed 6 months postoperatively and annually thereafter. Bone scans were performed when clinically indicated. We updated the survival status of all patients through telephone contact in February 2013.
Outcomes
Several outcomes were used when comparing the two groups. We assessed the following perioperative outcomes: operative time, estimated blood loss (EBL), analgesic requirement, time to resumption of oral intake, postoperative length of stay (LOS), and complication rate. All surgical complications were classified according to the Clavien classification system (Clavien et al, 2009). We also assessed neobladder functional outcomes; and oncologic outcomes such as LN yield, positive surgical margin rate (PSM), recurrence-free survival (RFS), and overall survival.
Statistical analysis
This trial was designed as a randomised non-inferiority single-centre study (Fueglistaler et al, 2007). The primary end point by which the sample size was determined was LN yield. Assuming the mean difference of LN yield between the two groups was 2 with a standard deviation of 4, a lost to follow-up proportion of 5%, and with a power of 80% at a significance level of 5% (two-tailed Student's t-test), 70 patients were enrolled in this trail.
Analyses were conducted following the intention-to-treat principle, with all randomised patients analysed according to their assigned treatment group. Categorical variables were analysed with the χ2 test or Fisher's exact test as appropriate. Continuous variables were compared with the Student's t-test or the Mann–Whitney U-test as appropriate. Survival curves were estimated using the Kaplan–Meier method, and differences were compared with the log-rank test. Univariate and multivariable analysis of survival outcomes were conducted using Cox regression analysis, taking into account randomised procedure, age, LN status, and tumour pT stage. All P-values were two-sided and P<0.05 was considered statistically significant. All analyses were conducted using SPSS v.19.0 (SPSS Inc., Chicago, IL, USA).
Results
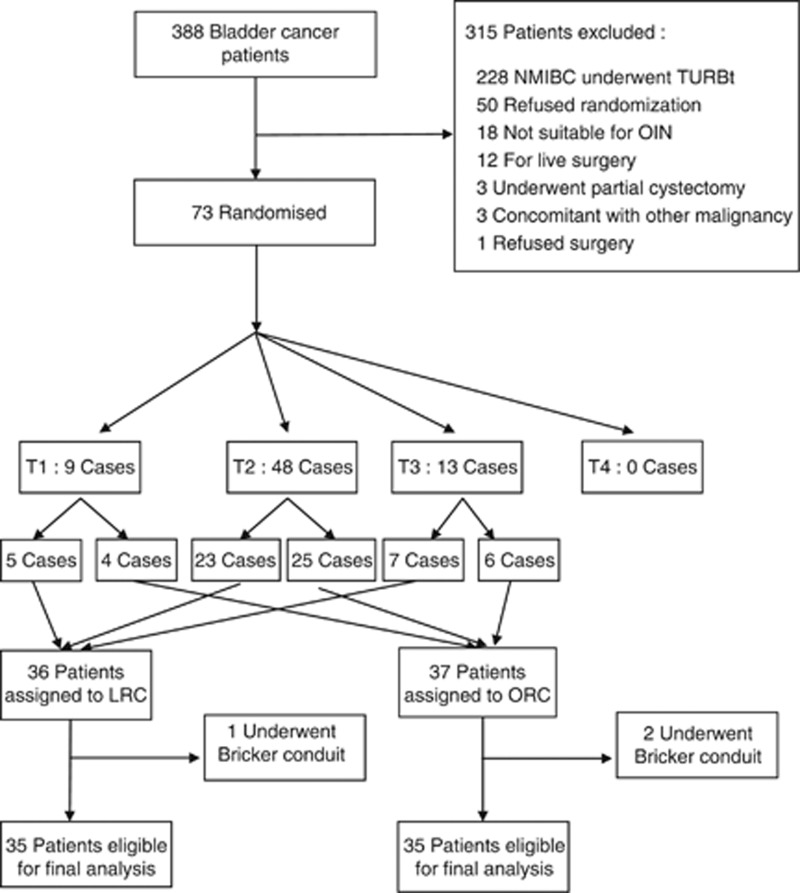
Between 16 January 2008 and 30 November 2011, 73 patients were randomly assigned to either laparoscopic or open surgery. Three patients were excluded for tumour invasion of the urethra, and subsequently underwent the Bricker conduit procedure. A total of 35 patients in each group who underwent radical cystectomy and M-shaped ileal neobladder were included in the final analysis. Figure 1 presents the flowchart. The two treatment groups were homogeneous in terms of preoperative characteristics and clinical stages, as shown in Table 1. The median follow-up was 26 (4–59) months in the laparoscopic group, and 32 (6–60) months in the open-surgery group (P=0.972). Three patients in each group were lost to follow-up, and they were included in the analysis but censored at the point of last follow-up.
Figure 1.
Flow chart of patients. Abbreviations: LRC=laparoscopic radical cystectomy; NMIBC=non-muscle-invasive bladder cancer; OIN=orthotopic ileal neobladder; ORC=open radical cystectomy; TURBt=transurethral resection of bladder tumour.
Table 1. Baseline characteristics of the patients and tumours.
| Category | LRC (n=35) | ORC (n=35) | P value |
|---|---|---|---|
| Mean age, mean±s.d., years |
63.2±9.1 |
63.6±8.9 |
0.851a |
|
Sex, No. | |||
| Male | 32 | 32 | 1.0b |
| Female | 3 | 3 | — |
| BMI, mean±s.d., kg m−2 | 22.0±2.7 | 22.9±3.1 | 0.414a |
| Previous abdominal surgery, No. |
3 |
5 |
0.707b |
|
ASA grade, No. | |||
| II | 28 | 25 | 0.403b |
| III |
7 |
10 |
— |
|
Clinical stage, No. | |||
| cT1 | 5 | 4 | 0.873b |
| cT2 | 23 | 25 | — |
| cT3 | 7 | 6 | — |
| Concomitant CIS, No. |
1 |
2 |
1.0b |
|
Tumour history, No. | |||
| Primary tumour | 25 | 28 | 0.403b |
| Recurrent after TURBT and/or partial cystectomy | 10 | 7 | — |
| Follow-up, median (range), months | 26 (4–59) | 32 (6–60) | 0.972c |
| Lost to follow-up, No. | 3 | 3 | 1.0b |
Abbreviations: ASA=American Society of Anesthesiologists; BMI=body mass index; CIS=carcinoma in situ; LRC=laparoscopic radical cystectomy; ORC=open radical cystectomy; TURBT=transurethral resection of bladder tumour.
Student's t-test.
χ2 test or Fisher's exact test.
Mann–Whitney U-test.
Perioperative outcomes
The results of the perioperative outcomes are listed in Table 2. Operative time was significantly longer (282 vs 235 min; P<0.001); blood loss was significantly lower (median 215 vs 510 ml; P<0.001); analgesic requirement was significantly less (morphine equivalents, 12.6 vs 19.3 mg; P<0.001), and the mean time to resumption of oral intake was significantly shorter (3.8 vs 4.8 d; P=0.001) comparing LRC with ORC. Blood transfusions were given to 2 patients (both are intraoperative ) in LRC and 12 patients (10 intraoperative, 2 postoperative) in ORC ( P=0.003).The mean postoperative length of stay was 15.8 days for LRC compared with 16.4 days in ORC, and this difference was not statistically significant (P=0.667). No patient was converted to open surgery in the laparoscopic group and no perioperative mortality was observed in either group.
Table 2. Perioperative outcomes.
| Category | LRC (n=35) | ORC (n=35) | P value |
|---|---|---|---|
| Operative time, min |
282±51 |
235±34 |
<0.001a |
| PLND |
84±24 |
51±17 |
<0.001a |
| Cystectomy |
67±18 |
56±16 |
0.013a |
| OIN making |
77±22 |
74±22 |
0.627a |
| OIN-urethra anastomosis |
34±9 |
25±8 |
<0.001a |
| Estimated blood loss, ml |
215 (55–810) |
510 (105–1700) |
<0.001b |
| Blood transfusions, No. |
2 |
12 |
0.003c |
| Intraoperative |
2 |
10 |
|
| Postoperative |
0 |
2 |
|
| Analgesic requirement, mg (morphine equivalents) |
12.6±5.2 |
19.3±6.3 |
<0.001a |
| Resumption of oral intake, d |
3.8±1.1 |
4.8±1.2 |
0.001a |
| Postoperative LOS, d |
15.8±5.5 |
16.4±6.1 |
0.667a |
| Conversion to ORC, No. |
0 |
— |
— |
| Perioperative mortality, No. | 0 | 0 | 1.0c |
Abbreviations: LOS=length of hospital stay; LRC=laparoscopic radical cystectomy; OIN=orthotopic ileal neobladder; ORC=open radical cystectomy; PLND=pelvic lymph node dissection.
Student's t-test.
Mann–Whitney U-test.
χ2 test or Fisher's exact test.
Complications
Complications graded by the Clavien classification system are summarised in Table 3. There was no significant difference in grade II (P=0.322) complications between LRC and ORC. There was also no significant difference between LRC and ORC (P=0.607 and P=0.669, respectively) for grade IIIa and IIIb complications requiring surgical intervention.
Table 3. Surgical complications stratified by the Clavien classification system.
| Complications (Clavien units) | LRC (n=35) | ORC (n=35) | P value |
|---|---|---|---|
| Grade II, No. (%) |
20 (57.1%) |
24 (68.6%) |
0.322a |
| Urinary tract infection |
1 |
2 |
— |
| Blood transfusion |
2 |
12 |
— |
| Ileus |
4 |
4 |
— |
| Lymphatic leakage |
6 |
2 |
— |
| Delirium |
2 |
0 |
— |
| Infectious peritonitis |
0 |
1 |
— |
| Vesico-urethral anastomotic leakage |
1 |
2 |
— |
| Uretero-pouch anastomotic leakage |
2 |
0 |
— |
| Neobladder leakage |
2 |
1 |
— |
| Grade IIIa, No. (%) |
1 (2.9%) |
3 (8.6%) |
0.607b |
| Incisional haematoma |
0 |
2 |
— |
| Rupture of incision required II stage suture |
1 |
1 |
— |
| Grade IIIb, No. (%) |
2 (5.7%) |
4 (11.4%) |
0.669b |
| Intraoperative damage of external iliac vessels |
1 |
1 |
— |
| Abdominal-pouch fistula |
0 |
1 |
— |
| Colon-pouch fistula |
1 |
1 |
— |
| Ileo-pouch fistula | 0 | 1 | — |
Abbreviations: LRC=laparoscopic radical cystectomy; ORC=open radical cystectomy.
χ2 test.
Fisher's exact test.
Pathologic outcomes
Postoperative pathologic outcomes are presented in Table 4. There was no significant difference when comparing all pathologic categories between LRC and ORC. PSM was observed in one patient in each group. The mean numbers of LN yield were 14.1 in LRC and 15.2 in ORC (P=0.467) and the positive LN rate was 22.9% (8/35) in both groups. The most common histologic type was transitional cell carcinoma (TCC, 97.1% in both groups), but seven patients were diagnosed with TCC with squamous differentiation (5.7%, 2/35 in LRC), glandular differentiation (2.9%, 1/35 in LRC), or coexisting prostate cancer (5.7%, 2/35 in each group). Bladder cancer in six patients in LRC (17.1%) and eight (22.9%) patients in ORC was associated with non-organ-confined diseases.
Table 4. Pathologic outcomes and sites of metastases.
| Category | LRC (n=35) | ORC (n=35) | P value |
|---|---|---|---|
| Positive margins, No. (%) |
0 |
1 (2.9%) |
1.0a |
|
Lymph node yield, No. | |||
| Mean±s.d. | 14.1±6.3 | 15.2±5.9 | 0.467b |
| Median (range) |
12 (4–32) |
14 (5–25) |
0.261c |
|
LN status, No. (%) | |||
| Positive | 8 (22.9%) | 8 (22.9%) | 1.0a |
| Negative |
27 (77.1%) |
27 (77.1%) |
— |
|
Histologic type, No. | |||
| TCC | 29 | 32 | 0.273a |
| TCC with squamous differentiation | 2 | — | — |
| TCC with glandular differentiation | 1 | — | — |
| TCC coexisting prostate cancer | 2 | 2 | |
| Squamous cell carcinoma | — | 1 | — |
| Carcinosarcoma |
1 |
— |
— |
|
pT stage, No. | |||
| pT1 | 8 | 7 | 0.935a |
| pT2 | 21 | 20 | — |
| pT2a | 14 | 12 | — |
| pT2b | 7 | 8 | — |
| pT3 | 5 | 7 | — |
| pT3a | 4 | 2 | — |
| pT3b | 1 | 5 | — |
| pT4a |
1 |
1 |
— |
|
Grade, No. | |||
| Grade 1 | 4 | 3 | 0.511a |
| Grade 2 | 15 | 16 | — |
| Grade 3 | 15 | 12 | — |
| Unclassified |
1 |
4 |
— |
| Concomitant CIS, No. |
2 |
3 |
1.0a |
| Sites of recurrence |
7 (20%) |
8 (22.9%) |
0.937a |
| Local recurrence | 3 (8.6%) | 3 (8.6%) | — |
| Distant metastases | 4 (11.4%) | 5 (14.3%) | — |
| Lung | 2 | 3 | — |
| Brain | — | 1 | — |
| Bone | 2 | 1 | — |
Abbreviations: CIS=carcinoma in situ. LRC=laparoscopic radical cystectomy; ORC=open radical cystectomy; TCC=transitional cell carcinoma.
χ2 test or Fisher's exact test.
Student's t-test.
Mann-Whitney U-test.
Neobladder function
Table 5 summarises the results of the ileal neobladders function 1 year after surgery, and no statistically significant difference was found between the two groups. The daytime continence rate was 89.3% in LRC and 86.7% in ORC (P=1.0). The nighttime continence rate was 78.6% in LRC and 70% in ORC (P=0.456). The median functional capacity was 305 ml in LRC and 325 ml in ORC (P=0.489). There was no statistically significant difference regarding the number of patients with residual urine, their amount of residual urine, and the incidence of infected urine. Two patients in LRC and one patient in ORC could not void, and they required clean intermittent catheterisation four to six times a day to empty their ileal neobladders.
Table 5. Neobladder functional outcomes 12 months after surgery.
| Category | LRC (n=28) | ORC (n=30) | P value |
|---|---|---|---|
| Daytime continent, No. (%) |
25 (89.3) |
26 (86.7) |
1.00a |
| Nighttime continent, No. (%) |
22 (78.6) |
21 (70) |
0.456a |
| Functional capacity, ml |
305 (155–660) |
325 (170–730) |
0.489b |
| Patients with residual urine, No. (%) |
11 (39.3%) |
8 (26.7%) |
0.306a |
| Residual urine volume, ml |
60 (10–145) |
45 (15–115) |
0.310b |
| Infected urine, No. (%) |
7 (25%) |
5 (16.7%) |
0.434a |
| Patients on clean intermittent catheterization, No. (%) | 2 (7.1%) | 1 (3.3%) | 0.951a |
Abbreviations: LRC=laparoscopic radical cystectomy; ORC=open radical cystectomy.
χ2 test or Fisher's exact test.
Mann–Whitney U-test.
Survival outcomes
Local recurrence and distant metastases occurred in seven patients in LRC compared with eight patients in ORC (P=0.937; Table 4).
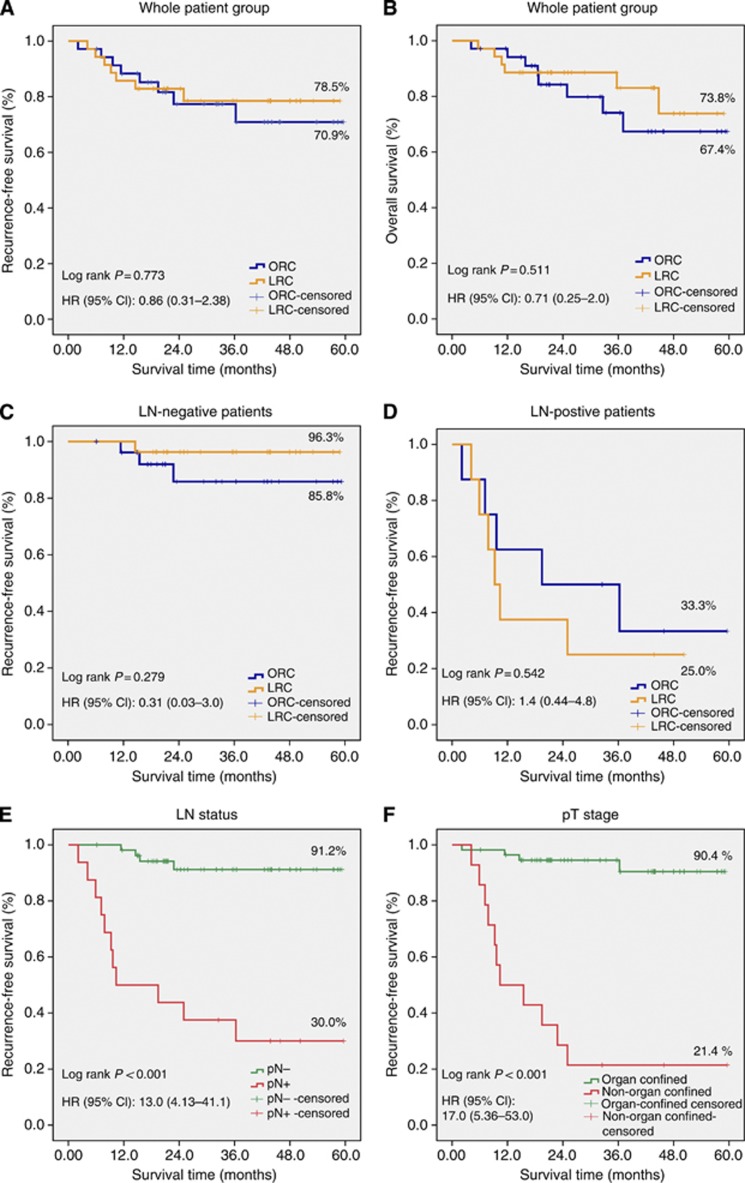
As shown in Figures 2A and B, there was no significant difference in RFS (P=0.773) and overall survival (P=0.551) between LRC and ORC. The difference in RFS remained not statistically significant when restricting analyses to LN-negative patients (P=0.279; Figure 2C) and LN-positive patients (P=0.542; Figure 2D). We also analysed RFS according to LN status and pT stage and found that LN-negative patients were associated with a statistically significantly better RFS than LN-positive patients (P<0.001; Figure 2E). Organ-confined patients were associated with a statistically significantly better RFS than non-organ-confined patients (P<0.001; Figure 2F).
Figure 2.
Kaplan–Meier curves for comparison of (A) recurrence-free survival (RFS) between laparoscopic radical cystectomy (LRC) and open radical cystectomy (ORC), (B) overall survival between LRC and ORC, (C) RFS in lymph node (LN)-negative patients, (D) RFS in LN-positive patients, (E) RFS according to LN status, (F) RFS according to pT stage. Abbreviations: CI=confidence interval; HR=hazards ratio.
In univariate analyses, surgical technique (P=0.773) was not associated with RFS, but age group (P=0.043), LN status (P<0.001), and pT stage (P<0.001) were associated with RFS. The results of multivariate analyses were similar to the results of the univariate analyses except that LN status (P=0.133) was not associated with RFS.
Discussion
The primary aim of radical cystectomy for bladder cancer is to remove the primary tumour safely and completely, to perform adequate PLND, to achieve negative margins, and to provide optimal long-term survival and quality of life (Hemal and Kolla, 2007). Although ORC remains the standard treatment for bladder cancer, it is associated with significant morbidity even when performed by experienced surgeons, in part, because of the long incision, prolonged abdominal wall retraction, prolonged exposure of the peritoneal surface with major fluid shifts and poor vision, particularly in the deep pelvis and retrovesical area (Chang et al, 2001). Since the first laparoscopic simple cystectomy reported by Parra et al (1992) 20 years ago, the minimal invasiveness of LRC in terms of reduced blood loss have made this procedure increasingly popular in the field of urology. However, what remains paramount is the long-term oncologic efficacy of LRC. Although the oncologic safety of laparoscopic surgery has not been proven inferior to open surgery in the treatment of other malignancies such as colon cancer and rectal cancer (Group TCOoSTS, 2004; Buunen et al, 2009; Lujan et al, 2009), the oncologic efficacy evidence of LRC for bladder cancer remains limited.
Several retrospective comparative studies (Hemal and Kolla, 2007; Porpiglia et al, 2007; Ha et al, 2010) and case series (Haber and Gill, 2007; Gillion et al, 2011) from major institutions have shown good early oncologic results over the initial 2–3 years (and in some patients up to 5 years) with low morbidity. Our institution reported the largest series of LRC (Huang et al, 2010; Challacombe et al, 2011), where 171 patients had an orthotopic ileal neobladder reconstruction with a median follow-up of 37 months (maximum: 7 years), with encouraging oncologic outcomes. Nevertheless, these reports are susceptible to significant selection biases in which only the most favorable candidates underwent the laparoscopic procedure. Higher levels of evidence such as those with RCT are still needed. Therefore, we conducted this prospective, randomised trial of LRC vs ORC beginning in January 2008, with a maximum follow-up of 5 years. The baseline characteristics of the patients and tumours were well matched. Also, all patients underwent radical cystectomy by a single surgeon, who was experienced in both LRC and ORC. Therefore, we minimised the bias introduced by patient selection and surgeon, and provided sound evidence comparing LRC and ORC.
The perioperative outcomes of this study showed that the operative time was significantly longer, but EBL, analgesic requirement, and time to oral intake were significantly lower in LRC compared with ORC. These findings are similar to those of previous studies (Hemal and Kolla, 2007; Guillotreau et al, 2009). In our institution, the gastrostomy tube was removed after the patient passed flatus or stool and oral intake was then restarted with a liquid diet. Therefore, the time to resumption of oral intake seems longer compared with other centres, where cystectomy patients are allowed to drink on the day of open surgery and have a liquid diet on the first postoperative day (fast track surgery; Roth et al, 2013). As a result, the difference observed in the resumption of oral intake in our study should not be overemphasised.
Despite the longer operative time in LRC, its complication rate was not higher than ORC. According to Challacombe et al (2011), major (Clavien grade III-IV) complication rates ranged from 10 to 13% in LRC. Shabsigh et al (2009) analysed complications in 1142 ORC patients, and found a major complication rate of 13%. In our trial, we did not find a high rate of complications specific to the laparoscopic approach, such as uretero-pouch anastomotic leakage and neobladder leakage. This may be because our surgeon was very experienced in LRC. Also, ileal neobladders were constructed extracorporeally, which is a safe and effective way to decrease operative time and surgical complications (Hemal et al, 2008).
Bilateral PLND is important for both accurate staging and adequate local and regional control (Abol-Enein et al, 2011). Several studies have taken LN yield as an indicator of surgical quality with cystectomy (Huang and Stein, 2007; Nix et al, 2010). LRC must be able to replicate the standardised PLND possible with open surgery to be established as an alternative technique (Challacombe et al, 2011). Based on this premise, we used LN yield as the basis for sample size calculation and to serve as the primary end point for statistical comparison between laparoscopic vs open approaches. Our result showed that LN yield in LRC was not inferior to ORC. In previous LRC series studies (Haber et al, 2008), the mean number of lymph nodes retrieved was 15.5 (range, 2–49), which was consistent with our study. Retrospective comparative studies (Hemal and Kolla, 2007; Porpiglia et al, 2007) also showed no statistically significant difference in LN yield between LRC and ORC.
PSM is an independent predictor of metastatic progression in patients undergoing radical cystectomy, which reportedly increased the risk of metastatic progression at 5 years from 32 to 74% (Dotan et al, 2007). According to Chade et al (2010), the incidence of PSM ranged from 4 to 5% of the ORC cases, and from 0 to 5% of the LRC cases. In our trial, the PSM rate was 2.9% (1/35) in both groups. These two patients' pathologic stage was pT3b. Dotan et al (2007) reported that none of the patients with organ-confined disease had evidence of PSM, suggesting that tumour infiltration may be the main limitation in achieving a negative margin.
Port-site metastasis is another debated issue for LRC. In our previous LRC series study including 171 patients (Huang et al, 2010), only 1 patient with grade 3 pT3N1M0 TCC developed port-site seeding. In the present trial, no port-site metastasis occurred. No port-site metastasis was found in other studies of LRC (Haber and Gill, 2007; Hemal and Kolla, 2007; Porpiglia et al, 2007; Guillotreau et al, 2009; Ha et al, 2010). Generally, port-site metastasis is a rare event in LRC. Improved techniques, including gentle clamping and entrapping of LNs and specimens into a secured endobag before extraction, could minimise the risk. Our results showed that LRC can achieve a low risk of tumour dissemination, and our results are reproducible by following the principles of oncologic surgery: do not transgress the tumour boundaries, ensure adequate margins during the resection, immediately close the bladder neck and prostate apex once opened, and avoid any bladder wall perforation to prevent potential spillage of cancer cells (Cathelineau et al, 2005).
Long-term survival is the final touchstone for the oncologic efficiency of LRC. A large ORC series by Stein et al (2001) demonstrated a 5-year RFS of 68%. RFS results varied most widely at 2–3 years in previous LRC reports with rates of 60–85% (Chade et al, 2010). Two retrospective comparative studies (Hemal and Kolla, 2007; Ha et al, 2010) showed no significant difference in 3-year RFS and overall survival between LRC and ORC. In the present study, we also observed similar RFS and overall survival in two treatment groups. However, a final conclusion is not possible because both groups were having mainly low-risk patients as expressed and reflected by the small number of events.
Our study has the following limitations: first, both the patients and the surgeons could not be blinded because of the surgical nature of the trial. Second, our patient numbers were relatively small and our study was a single-institution study. The basis for the statistical analysis (non-inferiority) was a possible difference in the LN yield. However, most of the other end points did not have enough events to conclude non-inferiority. Another limitation is that we primarily included patients with organ-confined disease. Indeed, only six patients were stage pT3b, and five of them were in the open group. Therefore, it is not surprising that the PSM as well as the oncological outcome, especially for the laparoscopic group, showed favourable results with respect to PSM or survival. The only patient with stage pT3b in the laparoscopic group did have PSM. It is unknown whether the outcome would be less favourable for the laparoscopic group if more high-risk patients for PSM were included. Also, despite the maximum follow-up of this trial of nearly 5 years, the median follow-up was only approximately 2 years. Therefore, future RCTs with large sample sizes and longer follow-ups are necessary to provide more convincing survival outcomes.
One of the strong points of this study is that the same surgeon (J Huang) performed LRC and ORC. Before running this randomised trial, he performed more than 100 LRCs. Thus, the results achieved by this single surgeon should not be over-interpreted. Rather it shows that the dedicated front runner for laparoscopic surgery is able—after a long run in phase—to achieve results comparable to ORC. However, this does not mean that laparoscopic surgery in general is equivalent to open surgery. It requires more skill, more training, and a long learning curve. Therefore, the interpretation of the results must be more careful.
In conclusion, our study demonstrated that LRC is superior to ORC in perioperative outcomes including EBL, blood transfusion rate, and analgesic requirement. We found no major difference in oncologic outcomes. However, the number of patients is too small to allow for a final conclusion. Future large sample-size RCTs with longer follow-up are needed to provided more convincing survival outcomes.
Acknowledgments
This study was funded by Sun Yat-Sen University Clinical Research 5010 Program (Grant No. 2007018), the National Natural Science Foundation of China (Grant No. U1301221, 81372729, 81001138, 81071688, 81172431, 81272808, 81101519, 81101935), and Guangdong Province Natural Scientific Foundation (Grant No. 07117336, 10151008901000024).
The authors declare no conflict of interest.
Footnotes
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License.
References
- Abol-Enein H, Tilki D, Mosbah A, El-Baz M, Shokeir A, Nabeeh A, Ghoneim MA. Does the extent of lymphadenectomy in radical cystectomy for bladder cancer influence disease-free survival? A prospective single-center study. Eu Urol. 2011;60 (3:572–577. doi: 10.1016/j.eururo.2011.05.062. [DOI] [PubMed] [Google Scholar]
- Buunen M, Veldkamp R, Hop WC, Kuhry E, Jeekel J, Haglind E, Pahlman L, Cuesta MA, Msika S, Morino M, Lacy A, Bonjer HJ. Survival after laparoscopic surgery vs open surgery for colon cancer: long-term outcome of a randomised clinical trial. Lancet Oncol. 2009;10 (1:44–52. doi: 10.1016/S1470-2045(08)70310-3. [DOI] [PubMed] [Google Scholar]
- Cathelineau X, Arroyo C, Rozet F, Barret E, Vallancien G. Laparoscopic assisted radical cystectomy: the montsouris experience after 84 cases. Eur Urol. 2005;47 (6:780–784. doi: 10.1016/j.eururo.2005.04.001. [DOI] [PubMed] [Google Scholar]
- Chade DC, Laudone VP, Bochner BH, Parra RO. Oncological outcomes after radical cystectomy for bladder cancer: open vs minimally invasive approaches. J Urol. 2010;183 (3:862–869. doi: 10.1016/j.juro.2009.11.019. [DOI] [PubMed] [Google Scholar]
- Challacombe BJ, Bochner BH, Dasgupta P, Gill I, Guru K, Herr H, Mottrie A, Pruthi R, Redorta JP, Wiklund P. The role of laparoscopic and robotic cystectomy in the management of muscle-invasive bladder cancer with special emphasis on cancer control and complications. Eur Urol. 2011;60 (4:767–775. doi: 10.1016/j.eururo.2011.05.012. [DOI] [PubMed] [Google Scholar]
- Chang SS, Smith JA, Jr, Wells N, Peterson M, Kovach B, Cookson MS. Estimated blood loss and transfusion requirements of radical cystectomy. J Urol. 2001;166 (6:2151–2154. [PubMed] [Google Scholar]
- Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, Schulick RD, de Santibanes E, Pekolj J, Slankamenac K, Bassi C, Graf R, Vonlanthen R, Padbury R, Cameron JL, Makuuchi M. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009;250 (2:187–196. doi: 10.1097/SLA.0b013e3181b13ca2. [DOI] [PubMed] [Google Scholar]
- Dotan ZA, Kavanagh K, Yossepowitch O, Kaag M, Olgac S, Donat M, Herr HW.2007Positive surgical margins in soft tissue following radical cystectomy for bladder cancer and cancer specific survival J Urol 178(62308–2312.discussion 2313). [DOI] [PubMed] [Google Scholar]
- Fueglistaler P, Adamina M, Guller U. Non-inferiority trials in surgical oncology. Ann Surg Oncol. 2007;14 (5:1532–1539. doi: 10.1245/s10434-006-9295-2. [DOI] [PubMed] [Google Scholar]
- Gillion N, Xylinas E, Durand X, Ploussard G, Vordos D, Allory Y, Hoznek A, de la Taille A, Abbou CC, Salomon L. Mid-term oncological control after laparoscopic radical cystectomy in men: a single-centre experience. BJU Int. 2011;108 (7:1180–1184. doi: 10.1111/j.1464-410X.2010.10054.x. [DOI] [PubMed] [Google Scholar]
- Group TCOoSTS A comparison of laparoscopically assisted and open colectomy for colon cancer. N Engl J Med. 2004;350 (20:2050–2059. doi: 10.1056/NEJMoa032651. [DOI] [PubMed] [Google Scholar]
- Guillotreau J, Game X, Mouzin M, Doumerc N, Mallet R, Sallusto F, Malavaud B, Rischmann P.2009Radical cystectomy for bladder cancer: morbidity of laparoscopic vs open surgery J Urol 181(2554–559.discussion 559. [DOI] [PubMed] [Google Scholar]
- Ha US, Kim SI, Kim SJ, Cho HJ, Hong SH, Lee JY, Kim JC, Kim SW, Hwang TK. Laparoscopic vs open radical cystectomy for the management of bladder cancer: mid-term oncological outcome. Inte J Urol. 2010;17 (1:55–61. doi: 10.1111/j.1442-2042.2009.02425.x. [DOI] [PubMed] [Google Scholar]
- Haber GP, Crouzet S, Gill IS. Laparoscopic and Robotic Assisted Radical Cystectomy for Bladder Cancer: A Critical Analysis. Eur Urol. 2008;54 (1:54–64. doi: 10.1016/j.eururo.2008.03.076. [DOI] [PubMed] [Google Scholar]
- Haber GP, Gill IS. Laparoscopic radical cystectomy for cancer: oncological outcomes at up to 5 years. BJU Int. 2007;100 (1:137–142. doi: 10.1111/j.1464-410X.2007.06865.x. [DOI] [PubMed] [Google Scholar]
- Hemal AK, Kolla SB. Comparison of laparoscopic and open radical cystoprostatectomy for localized bladder cancer with 3-year oncological followup: a single surgeon experience. J Urol. 2007;178 (6:2340–2343. doi: 10.1016/j.juro.2007.08.020. [DOI] [PubMed] [Google Scholar]
- Hemal AK, Kolla SB, Wadhwa P, Dogra PN, Gupta NP. Laparoscopic radical cystectomy and extracorporeal urinary diversion: a single center experience of 48 cases with three years of follow-up. Urology. 2008;71 (1:41–46. doi: 10.1016/j.urology.2007.08.056. [DOI] [PubMed] [Google Scholar]
- Huang GJ, Stein JP. Open radical cystectomy with lymphadenectomy remains the treatment of choice for invasive bladder cancer. Curr Opin Urol. 2007;17 (5:369–375. doi: 10.1097/MOU.0b013e3282dc95b5. [DOI] [PubMed] [Google Scholar]
- Huang J, Lin T, Liu H, Xu K, Zhang C, Jiang C, Huang H, Yao Y, Guo Z, Xie W. Laparoscopic radical cystectomy with orthotopic ileal neobladder for bladder cancer: oncologic results of 171 cases with a median 3-year follow-up. Eur Urol. 2010;58 (3:442–449. doi: 10.1016/j.eururo.2010.05.046. [DOI] [PubMed] [Google Scholar]
- Huang J, Lin T, Xu K, Huang H, Jiang C, Han J, Yao Y, Guo Z, Xie W, Yin X, Zhang C. Laparoscopic radical cystectomy with orthotopic ileal neobladder: a report of 85 cases. J Endourol/Endourol Soc. 2008;22 (5:939–946. doi: 10.1089/end.2007.0298. [DOI] [PubMed] [Google Scholar]
- Lujan J, Valero G, Hernandez Q, Sanchez A, Frutos MD, Parrilla P. Randomized clinical trial comparing laparoscopic and open surgery in patients with rectal cancer. Br J Surg. 2009;96 (9:982–989. doi: 10.1002/bjs.6662. [DOI] [PubMed] [Google Scholar]
- Nix J, Smith A, Kurpad R, Nielsen ME, Wallen EM, Pruthi RS. Prospective randomized controlled trial of robotic vs open radical cystectomy for bladder cancer: perioperative and pathologic results. Eur Urol. 2010;57 (2:196–201. doi: 10.1016/j.eururo.2009.10.024. [DOI] [PubMed] [Google Scholar]
- Parekh DJ, Messer J, Fitzgerald J, Ercole B, Svatek R. Perioperative outcomes and oncologic efficacy from a pilot prospective randomized clinical trial of open vs robotic assisted radical cystectomy. J Urol. 2012;189 (2:474–479. doi: 10.1016/j.juro.2012.09.077. [DOI] [PubMed] [Google Scholar]
- Parra RO, Andrus CH, Jones JP, Boullier JA. Laparoscopic cystectomy: initial report on a new treatment for the retained bladder. J Urol. 1992;148 (4:1140–1144. doi: 10.1016/s0022-5347(17)36843-x. [DOI] [PubMed] [Google Scholar]
- Pectasides D, Pectasides M, Nikolaou M.2005Adjuvant and neoadjuvant chemotherapy in muscle invasive bladder cancer: literature review Eur Urol 48(160–67.discussion 67-8. [DOI] [PubMed] [Google Scholar]
- Porpiglia F, Renard J, Billia M, Scoffone C, Cracco C, Terrone C, Scarpa RM. Open vs laparoscopy-assisted radical cystectomy: results of a prospective study. J Endourol Endourol Soc. 2007;21 (3:325–329. doi: 10.1089/end.2006.0224. [DOI] [PubMed] [Google Scholar]
- Roth B, Birkhauser FD, Zehnder P, Thalmann GN, Huwyler M, Burkhard FC, Studer UE. Parenteral nutrition does not improve postoperative recovery from radical cystectomy: results of a prospective randomised trial. Eur Urol. 2013;63 (3:475–482. doi: 10.1016/j.eururo.2012.05.052. [DOI] [PubMed] [Google Scholar]
- Shabsigh A, Korets R, Vora KC, Brooks CM, Cronin AM, Savage C, Raj G, Bochner BH, Dalbagni G, Herr HW, Donat SM. Defining early morbidity of radical cystectomy for patients with bladder cancer using a standardized reporting methodology. Eur Urol. 2009;55 (1:164–174. doi: 10.1016/j.eururo.2008.07.031. [DOI] [PubMed] [Google Scholar]
- Stein JP, Lieskovsky G, Cote R, Groshen S, Feng AC, Boyd S, Skinner E, Bochner B, Thangathurai D, Mikhail M, Raghavan D, Skinner DG. Radical cystectomy in the treatment of invasive bladder cancer: long-term results in 1,054 patients. J Clin Oncol. 2001;19 (3:666–675. doi: 10.1200/JCO.2001.19.3.666. [DOI] [PubMed] [Google Scholar]
- Yu HY, Hevelone ND, Lipsitz SR, Kowalczyk KJ, Nguyen PL, Choueiri TK, Kibel AS, Hu JC. Comparative analysis of outcomes and costs following open radical cystectomy vs robot-assisted laparoscopic radical cystectomy: results from the US Nationwide Inpatient Sample. Eur Urol. 2012;61 (6:1239–1244. doi: 10.1016/j.eururo.2012.03.032. [DOI] [PubMed] [Google Scholar]