Abstract
There is an accumulation of evidence in the literature demonstrating the integral role of vimentin intermediate filaments (IFs) in the progression of lung cancers. Vimentin IF proteins have been implicated in many aspects of cancer initiation and progression, including tumorigenesis, epithelial-to-mesenchymal transition (EMT), and the metastatic spread of cancer. Specifically, vimentin IFs have been recognized as an essential component regulating EMT, major signal transduction pathways involved in EMT and tumor progression, cell migration and invasion, the positioning and anchorage of organelles, such as mitochondria, and cell–cell and cell–substrate adhesion. In tumorgenesis, vimentin forms a complex with 14-3-3 and beclin 1 to inhibit autophagy via an AKT-dependent mechanism. Vimentin is a canonical marker of EMT, and recent evidence has shown it to be an important regulator of cellular motility. Transcriptional regulation of vimentin through hypoxia-inducible factor-1 may be a potential driver of EMT. Finally, vimentin regulates 14-3-3 complexes and controls various intracellular signaling and cell cycle control pathways by depleting the availability of free 14-3-3. There are many exciting advances in our understanding of the complex role of vimentin IFs in cancer, pointing to the key role vimentin IFs may play in tumor progression.
Keywords: epithelial-to-mesenchymal transition, invadopodia, lung cancer, metastatic cascade, vimentin
Clinical Relevance
The focus of this Translational Review is on vimentin, a type III intermediate filament (IF) protein, and the role it plays in the progression of lung cancer. Vimentin IF proteins have been implicated in many aspects of cancer initiation and progression, including tumorigenesis, epithelial-to-mesenchymal transition, and the metastatic spread of cancer.
Historically, intermediate filament (IF) proteins served as markers of the tissue origin of poorly differentiated tumors (keratin IFs define epithelial cells, whereas vimentin IFs define cells of mesenchymal origin), tumor markers in serum, and a means of detecting micrometastases. More recently, IFs are being recognized as essential signaling proteins involved in key cancer biological functions. These include epithelial-to-mesenchymal transition (EMT), regulation of major signal transduction pathways, cell migration and invasion, the positioning and anchorage of organelles, such as mitochondria (1), and cell–cell and cell–substrate adhesion (2). The focus of this Review is the role played by vimentin, a type III IF protein, in the progression of lung cancer. Vimentin is a widely expressed and highly conserved 57-kD protein that is constitutively expressed in mesenchymal cells, including endothelial cells lining blood vessels, renal tubular cells, macrophages, neutrophils, fibroblasts, and leukocytes (3–8). We discuss the role of vimentin in tumorigensis and the progression of lung cancer, and how this important cytoskeletal protein regulates a number of key stages in the metastatic cascade.
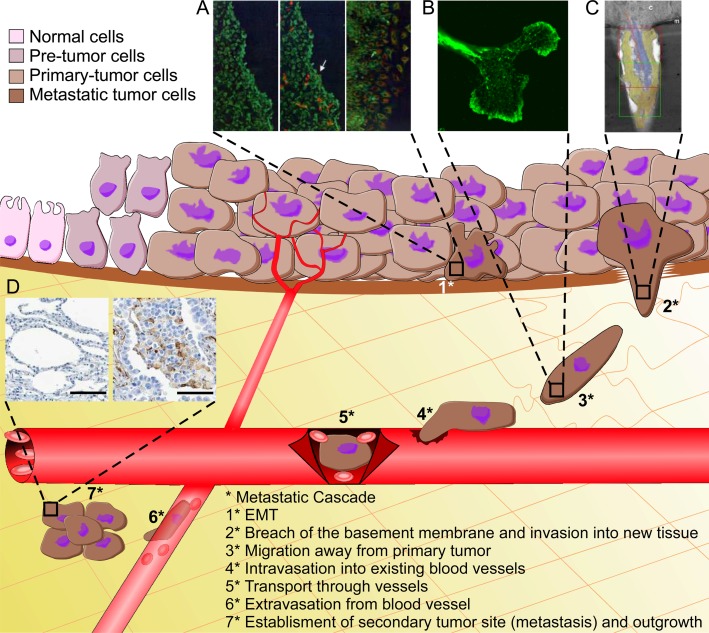
The regulation of the metastatic cascade is vital to our understanding of lung cancer. This type of cancer has the worst 5-year survival rate of all cancers worldwide (9, 10). Approximately 80% of lung cancers are non–small cell lung cancer (NSCLC) and 20% are small cell lung cancer. NSCLC tumors are further subdivided based on similarities to treatment approach and overall prognosis; the three most common types of NSCLC are squamous cell carcinoma, adenocarcinoma, and large cell carcinoma. Despite advancements in treatment and improved methods of monitoring disease progression, lung cancer patients have a bleak outlook, because there are few early stage symptoms (11). Between 40 and 50% of patients with NSCLC are diagnosed with stage IV lung cancer, and have a 5-year survival rate of less than 1% (11, 12). In addition, the majority of patients who die of lung cancer have an extensive array of secondary tumor sites, which are established through the metastatic cascade (13). There are several steps that define the metastatic cascade, many of which are directly or indirectly regulated by vimentin: EMT, breach of the basement membrane, dissociation of cells from the original tumor, invasion into new tissue, and establishment at secondary site (Figure 1). With such high mortality rates, a better understanding of the cellular and molecular mechanisms regulating lung cancer and the metastatic cascade are required.
Figure 1.
Vimentin’s role in the metastatic cascade. (A) When an epithelial-derived tumor cell undergoes epithelial-to-mesenchymal transition (EMT), vimentin expression is extensively expressed. EMT is characterized by down-regulation of epithelial markers (such as E-cadherin and keratins), up-regulation of mesenchymal markers (such as vimentin), and an increase in cellular motility. Epithelial cells are indicated in green (keratin), and cells that have undergone EMT are shown in red (vimentin). The left panel shows epithelial cells before EMT, the middle panel shows epithelial cells after EMT (white arrow indicating cells expressing vimentin), and the right panel showing vimentin-expressing cells (cells that have undergone EMT) migrating. (B) The migration of metastatic tumor cells away from the primary tumor is mediated by the formation of lamellipodia. The formation of lamellipodia has classically been seen as regulated by F-actin and actin-associated proteins, but there is growing evidence that indicates that vimentin intermediate filament (IFs) also play an integral role in lamellipodia formation and maintenance of cell polarity in migrating cells. In cellular migration, vimentin disassembly in the lamellipodia has been shown to be necessary for formation of cellular polarity, leading to an increase in migration. Vimentin disassembly in lamellipodia is regulated by Rac1, a small G protein in the Rho family that is known to drive actin polymerization and mediate lamellipodia formation. Rac1 was shown to mediate vimentin disassembly in lamellipodia by phosphorylating vimentin at serine 38 (32). Vimentin (shown in green) can be seen to be disassembled in the lamellipodia of a migrating cell. (C) Vimentin is required for invadopodia maturation. Vimentin has been found at the leading edge of mature invadopodia (shown in blue). When vimentin expression was reduced using small interfering RNA (siRNA) or when vimentin filaments were disrupted using a dominant-negative vimentin probe, there was a significant decrease in formation of mature invadopodia (31). Therefore, vimentin is required for invadopodia maturation and the subsequent invasive spread of cancer cells. Reproduced by permission from Reference 31. (D) Vimentin expression is highly up-regulated in metastatic lung cancer. Left: normal human lung tissue. Right: metastatic adenocarcinoma of the lung. Vimentin is shown in brown.
Vimentin: Regulation of Signaling Pathways and Tumorigenesis
Traditionally, vimentin IFs, found in the cytoplasm of mesenchymal cells, were thought to play a role in the maintenance of the cytoarchitecture and tissue integrity (14). However, our understanding of the role of vimentin has evolved, and it is now widely accepted that vimentin is also involved in the formation of signaling complexes with cell signaling molecules and other adaptor proteins (14). Tumorigenesis is a multistep process that transforms a noncancerous cell into a tumor cell, and this process can be influenced by the serine/threonine kinase, AKT (15). Residues phosphorylated by AKT may create a binding motif (RSx[pS/pT]xP or Rxxx[pS/pT]xP) that interacts with 14-3-3 proteins (2). The 14-3-3 proteins are involved in signal transduction pathways, adhesion, cellular proliferation, and inhibition of tumorigenesis (16). There are seven variants of the 14-3-3 proteins, and they mainly act as modulators of protein function to alter signaling pathways. The functional unit of 14-3-3 proteins is either a homodimer or a heterodimer (17). An example of 14-3-3 function is the negative regulation of FoxO genes that promotes tumorigenesis. FoxO3, a member of the forkhead family of transcription factors, coordinates many diverse cellular functions, such as cell proliferation, apoptosis, and reactive oxygen species response (18). When phosphorylated by AKT, FoxO is transported out of the nucleus, where it binds to 14-3-3 (19). Furthermore, phosphorylated vimentin was shown to interact with 14-3-3 proteins and prevent the assembly of Raf/14-3-3 and similar complexes (2). These findings suggest that vimentin regulates 14-3-3 complexes and modulates various intracellular signaling and cell cycle control pathways. We propose that, in addition to vimentin’s role in EMT, invasion, and motility, vimentin has an additional role in tumorigenesis.
The PI3K/AKT signaling pathway is often up-regulated in many cancers. AKT1 kinase binds to and phosphorylates vimentin at serine 39, which prevents vimentin from caspase-induced proteolysis, leading to increased motility and invasiveness of soft-tissue sarcoma cells. Moreover, vimentin phosphorylation was shown to enhance tumor and metastasis growth in vivo (20). Vimentin has also been reported to be a downstream target of PI3Kγ signaling, the activation of which results in increased vimentin phosphorylation that is required for efficient cellular migration (1). Furthermore, protein kinase C ζ was shown to promote the interaction of IFs with 14-3-3 (21). Scrib, a protein involved in cell migration, is protected from proteasomal degradation upon interaction with vimentin, suggesting that vimentin up-regulation during EMT leads to stabilization of Scrib, thereby promoting directed cell migration and increasing the invasive capacity of cells (22). In addition, Slug- and Ras-induced EMT changes were shown to be dependent on the up-regulation of vimentin (23). From these studies, it is evident that vimentin not only acts as a scaffolding protein, but also mediates several signaling pathways and cellular processes important in EMT and tumor progression.
We hypothesize that, after AKT phosphorylation, vimentin acts like a scaffold to sequester 14-3-3 protein complexes and enhance the effect of activated AKT. When Cos-7 cells were treated with calyculin A, a phosphatase inhibitor, there was an increased association between 14-3-3 proteins and phosphorylated vimentin (2), suggesting a functional interaction between AKT, 14-3-3, and vimentin. An additional implication that there is an interaction between AKT, 14-3-3, and vimentin came from a study on multidrug-resistant, diffuse, large B cell lymphoma (24). Two-dimensional differential in-gel analysis indicated that AKT, 14-3-3, and vimentin were up-regulated in multidrug-resistant diffuse, large B cell lymphoma cells. Based on the known interactions between AKT and 14-3-3, as well as AKT and vimentin, the authors proposed that there was a pathway involving AKT, 14-3-3, and vimentin (24). This possibility was uncovered when the role of AKT in inhibiting beclin 1–dependent autophagy was investigated. A coimmunoprecipitation experiment demonstrated that beclin 1 interacted both with 14-3-3 proteins when a constitutively active form of AKT was present, as well as with activated AKT (25). Phosphorylation of beclin 1 by AKT was correlated with reduced autophagy and increased tumorigenesis. Furthermore, in the presence of constitutively active AKT, vimentin coimmunoprecipitates with beclin 1 (25). These findings suggest that AKT phosphorylates both beclin 1 and vimentin, then a complex forms between beclin 1, 14-3-3, and vimentin forms which promotes tumorigenesis.
Another tumorigenesis pathway involving vimentin is the extracellular signal–regulated kinase (Erk) pathway, which is often up-regulated in many cancers and is involved in many cellular functions, including survival, proliferation, and motility. The second coiled-coil domain of vimentin was shown to interact with phosphorylated Erk, a mitogen-activated protein kinase, in a calcium-dependent mechanism, and to protect it from dephosphorylation (26). One mechanism by which Erk promotes tumorigenesis is through phosphorylation of FoxO3, leading to FoxO3 being polyubiquinated and then degraded (27). These results suggest that vimentin stabilizes Erk, leading to degradation of FoxO3. Interestingly, Erk is activated by 14-3-3 (28), which suggests that vimentin is involved both in sequestering FoxO3 as well as promoting its degradation. There are likely many other proteins the function of which could be modulated after AKT activation and sequestration on vimentin filaments through 14-3-3. Uncovering these interactions will likely provide significant insights into the regulation of signaling pathways and tumorigenesis.
Vimentin: Canonical Marker of EMT
Vimentin is a widely expressed and highly conserved 57-kD protein that is constitutively expressed in mesenchymal cells, including endothelial cells lining blood vessels, renal tubular cells, macrophages, neutrophils, fibroblasts, and leukocytes (3–8). Vimentin is a canonical marker of EMT (reviewed in Ref. 29), a cellular reprogramming process in which epithelial cells acquire a mesenchymal phenotype that causes them to dramatically alter their shape and exhibit increased motility (Figure 1A). EMT is a process characterized by a loss of cell polarity, down-regulation of epithelial markers (such as E-cadherin and keratins), and up-regulation of mesenchymal markers (such as vimentin [13, 30]). In normal lung tissue, vimentin expression in the bronchial epithelium is restricted to the basal and columnar cells (31). However, in lung cancer, increased vimentin expression is associated with epithelial-derived tumor cells (32), and is used as a diagnostic marker for the initial progression of epithelial cells from a localized lesion to invasive, metastatic tumor cells (13, 33). Increased vimentin expression has also been reported in various tumor cell lines and tissues, including prostate cancer, breast cancer, endometrial cancer, tumors of the central nervous system, malignant melanoma, and gastrointestinal tract tumors that include pancreatic, colorectal, and hepatic cancers (32). Hepatocellular carcinoma metastasis has been characterized by the increased expression of vimentin (34). In a study comparing human primary hepatocellular carcinoma tissue samples with their matched metastatic tumors, vimentin expression was significantly increased in matched metastatic tumors compared with the primary tumor (34). Furthermore, we have found vimentin expression to be up-regulated in human metastatic lung adenocarcinoma compared with normal lung tissue (unpublished data), showing that vimentin expression is also associated with metastatic lung tumors (Figure 1D). These data suggest vimentin expression to be a potential marker for the occurrence of metastasis in epithelial-derived tumors.
EMT: Transcriptional Regulation of Vimentin
Vimentin is encoded by a single-copy gene and is located on the short arm of chromosome 10, specifically at chromosome 10p13 (22). The vimentin promoter is composed of many elements, including a TATA box, eight putative GC boxes (35), Activator protein 1 (AP-1) binding sites (36), the NF-κB–binding site (37), and a Smad binding element (38, 39). Hypoxia-inducible factor-1 (HIF-1) is a major regulator of the cellular response to hypoxia, which has been shown to regulate vimentin gene expression (40). Hypoxia is one of the key drivers of metastatic progression, is a driver of EMT, is clinically associated with the occurrence of metastasis, and is present in all solid tumors with dimensions greater than 1 cm3 (41). The expression of vimentin is associated with increased tumor cell invasiveness (42). Therefore, vimentin transcriptional regulation by HIF-1 may be a potential driver of EMT. In addition, the Smad binding element was found within the activated protein complex-1 region of the vimentin promoter, and was shown to be involved in the regulation of vimentin expression in alveolar epithelial cells and to induce phenotypic change toward EMT (2, 21, 38).
Assembly Dynamics of Vimentin
Vimentin IFs are dynamic polypeptides that have a highly conserved α-helical “rod” domain that is flanked by non–α-helical N- and C-terminal domains (3). The assembly dynamics of vimentin IFs are controlled by phosphorylation status (43), as seen by fluorescent imaging of live cells, and are capable of assembling and disassembling rapidly in response to external stimuli, such as tumor-associated hypoxia (44). In addition, vimentin has been shown to be phosphorylated by protein kinase A at Serines 38 and 72, which leads to decreased filament formation in vivo. However, site-directed mutagenesis of these sites showed no significant effect on filament assembly, indicating that phosphorylation primarily regulates disassembly of vimentin IFs (43). The p21-activated kinase was shown to phosphorylate vimentin at several sites and to be involved in the regulation of vimentin structural reorganization (45). These dynamic changes in vimentin assembly state have been shown to play a critical role in cell attachment, migration, and cell signaling (reviewed in Ref. 46).
Vimentin: Invasion and Migration
Vimentin organization within the cell has important effects on the formation and function of invadopodia and lamellipodia during cellular invasion and migration. To invade into the surrounding tissue, an invasive tumor cell will first form invadopodia and degrade the surrounding basement membrane. Rather than digesting the entire basement membrane, cells will create small perforations where invadopodia will form (47, 48). After this initial stage, the invadopodia will elongate or mature and allow the cell to invade through these perforations into the surrounding tissue. It is during this crucial step of invadopodia elongation that vimentin is required (31). This requirement was demonstrated in MDA-MB-231 cells, a breast cancer cell line, in which vimentin expression was reduced using small interfering RNA (siRNA) or vimetin filaments were disrupted using a dominant-negative vimentin probe. In both cases, cells with reduced or disrupted vimentin expression showed a significant decrease in formation of mature invadopodia (31). Therefore, vimentin is required for invadopodia maturation and the subsequent invasive spread of cancer cells (Figure 1C). Furthermore, inhibition of vimentin expression by RNA interference has been shown to reduce metastatic cell invasiveness and decrease tumor volume (49). Nodal metastatic squamous cell carcinomas, which express high levels of vimentin, are highly proliferative and motile in vitro. Vimentin knockdown using RNA interference in these cells showed a threefold reduction in cellular invasion and migration (49), demonstrating the importance of vimentin expression in tumor cell motility.
After the formation and maturation of invadopodia, metastatic tumor cells will migrate away from the primary tumor site (Figure 1B). During migration, a cell will form a lamellipodia, which is the leading edge of the cell and is often characterized by ruffled membranes. The formation of a lamellipodium causes a migrating cell to become polarized, with a leading edge in the direction of migration. The formation of lamellipodia has classically been seen as regulated by F-actin and actin-associated proteins (50); however, there is growing evidence indicating that vimentin IFs also play an integral role in lamellipodia formation and maintenance of cell polarity in migrating cells. Specifically, vimentin assembly dynamics have been shown to regulate cell migration. In migratory fibroblasts, vimentin IFs were shown to extend throughout the cell, from the tail to the perinuclear region, but they were disassembled at the lamellipodia (32). This disassembly at the leading edge was not observed in serum-starved cells or in nonmigratory fibroblasts. Furthermore, it was shown that vimentin disassembly in lamellipodia was regulated by Rac1, a small G protein in the Rho family that is known to drive actin polymerization and mediate lamellipodia formation. Rac1 was shown to mediate vimentin disassembly in lamellipodia by phosphorylating vimentin at Serine 38 (32). Finally, vimentin assembly dynamics were shown to maintain the polarity of migrating cells. Vimentin filaments were disrupted in migratory fibroblasts with a dominant-negative mutant. In the dominant-negative mutant cells, a loss of polarity (loss of leading edge) and a reduction in migration was observed (32). These studies demonstrate how vimentin assembly dynamics appear to be essential in lamellipodia formation and maintenance of cell polarity in migrating cells.
Future Directions
IFs are an attractive potential therapeutic target for lung cancer, due to their involvement in cellular motility, transcriptional regulation, and association with EMT and tumor metastasis. Vimentin may be a key regulator of several tumorigenic pathways, as it forms a complex with 14-3-3 that may prevent the dephosphorylation of proteins in the complex, thereby inhibiting antitumor activity within cells. Vimentin has been shown to be required for maturation of invadopodia and to mediate cellular migration and formation of lamellipodia. Furthermore, increased vimentin expression has been seen in metastatic human adenocarcinoma of the lung as compared with normal lung tissue. These results lend evidence to the role of vimentin as a key regulator of the metastatic spread of lung cancers, and show vimentin to be an attractive potential therapeutic target for future cancer therapies. There are many key questions still to be answered to fully understand the role of vimentin in tumor metastasis. These include understanding the mechanism by which HIF-1 regulates vimentin expression during EMT, understanding how vimentin assembly state, as well as vimentin expression, can alter a cell’s metastatic potential, and elucidating the effect of vimentin expression on metastasis using in vivo models.
Acknowledgments
Acknowledgments
The authors thank Ernst Fattakhov and Jennifer Marie Davis, who provided valuable comments and edits to the final manuscript.
Footnotes
This work was supported by National Institutes of Health/National Heart, Lung, and Blood Institute grants NIH-P01 HL71643 and HL-07190, a Department of Veterans Affairs MERIT Award, and Training Program in Lung Science grant NIH-T32HL076139 (M.E.K.).
Originally Published in Press as DOI: 10.1165/rcmb.2013-0314TR on August 27, 2013
Author disclosures are available with the text of this article at www.atsjournals.org.
References
- 1.Barberis L, Pasquali C, Bertschy-Meier D, Cuccurullo A, Costa C, Ambrogio C, Vilbois F, Chiarle R, Wymann M, Altruda F, et al. Leukocyte transmigration is modulated by chemokine-mediated PI3Kγ-dependent phosphorylation of vimentin. Eur J Immunol. 2009;39:1136–1146. doi: 10.1002/eji.200838884. [DOI] [PubMed] [Google Scholar]
- 2.Tzivion G, Luo Z-J, Avruch J. Calyculin A–induced vimentin phosphorylation sequesters 14-3-3 and displaces other 14-3-3 partners in vivo. J Biol Chem. 2000;275:29772–29778. doi: 10.1074/jbc.M001207200. [DOI] [PubMed] [Google Scholar]
- 3.Satelli A, Li S. Vimentin in cancer and its potential as a molecular target for cancer therapy. Cell Mol Life Sci. 2011;68:3033–3046. doi: 10.1007/s00018-011-0735-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fuchs E, Weber K. Intermediate filaments: structure, dynamics, function and disease. Annu Rev Biochem. 1994;63:345–382. doi: 10.1146/annurev.bi.63.070194.002021. [DOI] [PubMed] [Google Scholar]
- 5.Steinert PM, Marekov LN, Fraser RD, Parry DA. Keratin intermediate filament structure: crosslinking studies yield quantitative information on molecular dimensions and mechanism of assembly. J Mol Biol. 1993;230:436–452. doi: 10.1006/jmbi.1993.1161. [DOI] [PubMed] [Google Scholar]
- 6.McKeon FD, Kirschner MW, Caput D. Homologies in both primary and secondary structure between nuclear envelope and intermediate filament proteins. Nature. 1986;319:463–468. doi: 10.1038/319463a0. [DOI] [PubMed] [Google Scholar]
- 7.Steinert PM. Structure, function, and dynamics of keratin intermediate filaments. J Invest Dermatol. 1993;100:729–734. doi: 10.1111/1523-1747.ep12475665. [DOI] [PubMed] [Google Scholar]
- 8.Crystal RG, Randell SH, Engelhardt JF, Voynow J, Sunday ME. Airway epithelial cells: current concepts and challenges. Proc Am Thorac Soc. 2008;5:772–777. doi: 10.1513/pats.200805-041HR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dubey S, Powell CA. Update in lung cancer 2008. Am J Respir Crit Care Med. 2009;179:860–868. doi: 10.1164/rccm.200902-0289UP. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sutherland KD, Berns A. Cell of origin of lung cancer. Mol Oncol. 2010;4:397–403. doi: 10.1016/j.molonc.2010.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hammerschmidt S, Wirtz H. Lung cancer: current diagnosis and treatment. Dtsch Arztebl Int. 2009;106:809–818. doi: 10.3238/arztebl.2009.0809. quiz 819–820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Non–small cell lung cancer survival rates by stage [Internet]. Atlanta: American Cancer Society, Inc.; 2012[updated 2013 July 12; accessed 2013 July 28]. Available from: http://www.cancer.org/cancer/lungcancer-non-smallcell/detailedguide/non-small-cell-lung-cancer-survival-rates
- 13.Geiger TR, Peeper DS. Metastasis mechanisms. Biochim Biophys Acta. 2009;1796:293–308. doi: 10.1016/j.bbcan.2009.07.006. [DOI] [PubMed] [Google Scholar]
- 14.Eriksson JE, Dechat T, Grin B, Helfand B, Mendez M, Pallari HM, Goldman RD. Introducing intermediate filaments: from discovery to disease. J Clin Invest. 2009;119:1763–1771. doi: 10.1172/JCI38339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Heron-Milhavet L, Khouya N, Fernandez A, Lamb NJ. Akt1 and Akt2: differentiating the aktion. Histol Histopathol. 2011;26:651–662. doi: 10.14670/HH-26.651. [DOI] [PubMed] [Google Scholar]
- 16.Gardino AK, Yaffe MB. 14-3-3 proteins as signaling integration points for cell cycle control and apoptosis. Semin Cell Dev Biol. 2011;22:688–695. doi: 10.1016/j.semcdb.2011.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Aitken A. Post-translational modification of 14-3-3 isoforms and regulation of cellular function. Semin Cell Dev Biol. 2011;22:673–680. doi: 10.1016/j.semcdb.2011.08.003. [DOI] [PubMed] [Google Scholar]
- 18.Eijkelenboom A, Burgering BM. FoxOs: signalling integrators for homeostasis maintenance. Nat Rev Mol Cell Biol. 2013;14:83–97. doi: 10.1038/nrm3507. [DOI] [PubMed] [Google Scholar]
- 19.Dobson M, Ramakrishnan G, Ma S, Kaplun L, Balan V, Fridman R, Tzivion G. Bimodal regulation of FoxO3 by Akt and 14-3-3. Biochim Biophys Acta. 2011;1813:1453–1464. doi: 10.1016/j.bbamcr.2011.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhu QS, Rosenblatt K, Huang KL, Lahat G, Brobey R, Bolshakov S, Nguyen T, Ding Z, Belousov R, Bill K, et al. Vimentin is a novel Akt1 target mediating motility and invasion. Oncogene. 2011;30:457–470. doi: 10.1038/onc.2010.421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sivaramakrishnan S, Schneider JL, Sitikov A, Goldman RD, Ridge KM. Shear stress induced reorganization of the keratin intermediate filament network requires phosphorylation by protein kinase C ζ. Mol Biol Cell. 2009;20:2755–2765. doi: 10.1091/mbc.E08-10-1028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Phua DCY, Humbert PO, Hunziker W. Vimentin regulates scribble activity by protecting it from proteasomal degradation. Mol Biol Cell. 2009;20:2841–2855. doi: 10.1091/mbc.E08-02-0199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Vuoriluoto K, Haugen H, Kiviluoto S, Mpindi J-P, Nevo J, Gjerdrum C, Tiron C, Lorens JB, Ivaska J. Vimentin regulates EMT induction by slug and oncogenic H-Ras and migration by governing AXL expression in breast cancer. Oncogene. 2011;30:1436–1448. doi: 10.1038/onc.2010.509. [DOI] [PubMed] [Google Scholar]
- 24.Maxwell SA, Cherry EM, Bayless KJ. Akt, 14-3-3zeta, and vimentin mediate a drug-resistant invasive phenotype in diffuse large B-cell lymphoma. Leuk Lymphoma. 2011;52:849–864. doi: 10.3109/10428194.2010.551793. [DOI] [PubMed] [Google Scholar]
- 25.Wang RC, Wei Y, An Z, Zou Z, Xiao G, Bhagat G, White M, Reichelt J, Levine B. Akt-mediated regulation of autophagy and tumorigenesis through beclin 1 phosphorylation. Science. 2012;338:956–959. doi: 10.1126/science.1225967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Perlson E, Michaelevski I, Kowalsman N, Ben-Yaakov K, Shaked M, Seger R, Eisenstein M, Fainzilber M. Vimentin binding to phosphorylated Erk sterically hinders enzymatic dephosphorylation of the kinase. J Mol Biol. 2006;364:938–944. doi: 10.1016/j.jmb.2006.09.056. [DOI] [PubMed] [Google Scholar]
- 27.Yang JY, Zong CS, Xia W, Yamaguchi H, Ding Q, Xie X, Lang JY, Lai CC, Chang CJ, Huang WC, et al. Erk promotes tumorigenesis by inhibiting FoxO3a via MDM2-mediated degradation. Nat Cell Biol. 2008;10:138–148. doi: 10.1038/ncb1676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Xing H, Zhang S, Weinheimer C, Kovacs A, Muslin AJ. 14-3-3 proteins block apoptosis and differentially regulate MAPK cascades. EMBO J. 2000;19:349–358. doi: 10.1093/emboj/19.3.349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Thiery JP. Epithelial–mesenchymal transitions in tumour progression. Nat Rev Cancer. 2002;2:442–454. doi: 10.1038/nrc822. [DOI] [PubMed] [Google Scholar]
- 30.Valastyan S, Weinberg RA. Tumor metastasis: molecular insights and evolving paradigms. Cell. 2011;147:275–292. doi: 10.1016/j.cell.2011.09.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Schoumacher M, Goldman RD, Louvard D, Vignjevic DM. Actin, microtubules, and vimentin intermediate filaments cooperate for elongation of invadopodia. J Cell Biol. 2010;189:541–556. doi: 10.1083/jcb.200909113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Helfand BT, Mendez MG, Murthy SN, Shumaker DK, Grin B, Mahammad S, Aebi U, Wedig T, Wu YI, Hahn KM, et al. Vimentin organization modulates the formation of lamellipodia. Mol Biol Cell. 2011;22:1274–1289. doi: 10.1091/mbc.E10-08-0699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gilles C, Polette M, Zahm JM, Tournier JM, Volders L, Foidart JM, Birembaut P. Vimentin contributes to human mammary epithelial cell migration. J Cell Sci. 1999;112:4615–4625. doi: 10.1242/jcs.112.24.4615. [DOI] [PubMed] [Google Scholar]
- 34.Hu L, Lau SH, Tzang CH, Wen JM, Wang W, Xie D, Huang M, Wang Y, Wu MC, Huang JF, et al. Association of vimentin overexpression and hepatocellular carcinoma metastasis. Oncogene. 2004;23:298–302. doi: 10.1038/sj.onc.1206483. [DOI] [PubMed] [Google Scholar]
- 35.Rittling SR, Baserga R. Functional analysis and growth factor regulation of the human vimentin promoter. Mol Cell Biol. 1987;7:3908–3915. doi: 10.1128/mcb.7.11.3908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rittling SR, Coutinho L, Amram T, Kolbe M. AP-1/Jun binding sites mediate serum inducibility of the human vimentin promoter. Nucleic Acids Res. 1989;17:1619–1633. doi: 10.1093/nar/17.4.1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lilienbaum A, Paulin D. Activation of the human vimentin gene by the tax human T-cell leukemia virus. I. Mechanisms of regulation by the NF-kappa b transcription factor. J Biol Chem. 1993;268:2180–2188. [PubMed] [Google Scholar]
- 38.Rogel MR, Soni PN, Troken JR, Sitikov A, Trejo HE, Ridge KM.Vimentin is sufficient and required for wound repair and remodeling in alveolar epithelial cells FASEB J 2011253873–3883 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wu Y, Zhang X, Salmon M, Lin X, Zehner ZE. TGFbeta1 regulation of vimentin gene expression during differentiation of the C2C12 skeletal myogenic cell line requires Smads, AP-1 and SP1 family members. Biochim Biophys Acta. 2007;1773:427–439. doi: 10.1016/j.bbamcr.2006.11.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Krishnamachary B, Berg-Dixon S, Kelly B, Agani F, Feldser D, Ferreira G, Iyer N, LaRusch J, Pak B, Taghavi P, et al. Regulation of colon carcinoma cell invasion by hypoxia-inducible factor 1. Cancer Res. 2003;63:1138–1143. [PubMed] [Google Scholar]
- 41.Erler JT, Weaver VM. Three-dimensional context regulation of metastasis. Clin Exp Metastasis. 2009;26:35–49. doi: 10.1007/s10585-008-9209-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Adam SA, Gerace L. Cytosolic proteins that specifically bind nuclear location signals are receptors for nuclear import. Cell. 1991;66:837–847. doi: 10.1016/0092-8674(91)90431-w. [DOI] [PubMed] [Google Scholar]
- 43.Eriksson JE, He T, Trejo-Skalli AV, Harmala-Brasken AS, Hellman J, Chou YH, Goldman RD. Specific in vivo phosphorylation sites determine the assembly dynamics of vimentin intermediate filaments. J Cell Sci. 2004;117:919–932. doi: 10.1242/jcs.00906. [DOI] [PubMed] [Google Scholar]
- 44.Liu T, Guevara OE, Warburton RR, Hill NS, Gaestel M, Kayyali US. Regulation of vimentin intermediate filaments in endothelial cells by hypoxia. Am J Physiol Cell Physiol. 2010;299:C363–C373. doi: 10.1152/ajpcell.00057.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tang DD, Bai Y, Gunst SJ. Silencing of p21-activated kinase attenuates vimentin phosphorylation on Ser-56 and reorientation of the vimentin network during stimulation of smooth muscle cells by 5-hydroxytryptamine. Biochem J. 2005;388:773–783. doi: 10.1042/BJ20050065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ivaska J, Pallari HM, Nevo J, Eriksson JE. Novel functions of vimentin in cell adhesion, migration, and signaling. Exp Cell Res. 2007;313:2050–2062. doi: 10.1016/j.yexcr.2007.03.040. [DOI] [PubMed] [Google Scholar]
- 47.Bowden ET, Onikoyi E, Slack R, Myoui A, Yoneda T, Yamada KM, Mueller SC. Co-localization of cortactin and phosphotyrosine identifies active invadopodia in human breast cancer cells. Exp Cell Res. 2006;312:1240–1253. doi: 10.1016/j.yexcr.2005.12.012. [DOI] [PubMed] [Google Scholar]
- 48.Weaver AM. Invadopodia: specialized cell structures for cancer invasion. Clin Exp Metastasis. 2006;23:97–105. doi: 10.1007/s10585-006-9014-1. [DOI] [PubMed] [Google Scholar]
- 49.Paccione RJ, Miyazaki H, Patel V, Waseem A, Gutkind JS, Zehner ZE, Yeudall WA. Keratin down-regulation in vimentin-positive cancer cells is reversible by vimentin RNA interference, which inhibits growth and motility. Mol Cancer Ther. 2008;7:2894–2903. doi: 10.1158/1535-7163.MCT-08-0450. [DOI] [PubMed] [Google Scholar]
- 50.Le Clainche C, Carlier M. Regulation of actin assembly associated with protrusion and adhesion in cell migration. Physiol Rev. 2008;88:489–513. doi: 10.1152/physrev.00021.2007. [DOI] [PubMed] [Google Scholar]