Abstract
In this issue of Blood, Benveniste et al report contrasting effects of canonical Notch signaling on human hematopoietic stem cells ex vivo and in vivo. Notch enhanced stem cell expansion in culture, but not in xenogeneic recipients.1
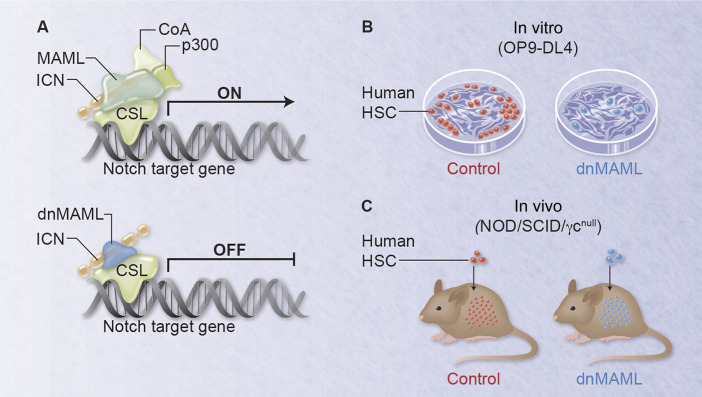
Canonical Notch signaling is required for in vitro but not in vivo expansion of human HSCs. (A) dominant-negative MAML (dnMAML) blocks transcriptional activation of Notch target genes downstream of all Notch receptors. ICN, intracellular Notch; CSL, CBF1/Su(H)/Lag-1, also called RBP-Jk; CoA, coactivator. (B) dnMAML inhibits maintenance and expansion of human HSCs in coculture with OP9 stromal cells expressing Delta-like4 Notch ligands (OP9-DL4). (C) dnMAML preserves in vivo expansion of human HSCs after transplantation into xenogeneic NOD/SCID/γcnull immunodeficient mice. Professional illustration by Alice Y. Chen.
For the past 60 years, studies in mice provided a sophisticated understanding of the hematopoietic hierarchy, a success story built on prospective purification, transplantation-based assays, and genetic tools. Although informative, these investigations leave open questions about conservation or divergence in mouse and human hematopoiesis. Recent developments using culture systems and immunodeficient xenogeneic hosts brought human hematopoiesis onto the center stage.
Notch signaling regulates multiple aspects of hematopoiesis in vertebrates. In mice, Notch was found to be essential during early T-lineage development. Stromal cell lines expressing Delta-like Notch ligands subsequently provided an unprecedented capacity to study mouse and human T-cell development. In parallel, ex vivo exposure of mouse or human hematopoietic progenitors to plate-bound or cell-bound Notch ligands was shown to enhance cytokine-mediated expansion, leading to preclinical testing and early clinical trials using Notch-expanded progenitors to boost hematopoietic recovery after cord blood transplantation.2,3 However, whether Notch regulates hematopoietic stem cell (HSC) expansion and function in vivo at physiological levels of signaling remains controversial. Using genetic strategies to block transcriptional activation downstream of all Notch receptors (Notch1-4), several studies showed preserved mouse HSC function in steady-state conditions and after transplantation into irradiated recipients.4,5 However, Notch2 inactivation delayed early hematopoietic reconstitution, although without effects at later time points.6 Endothelial-specific inactivation of Jagged1, encoding 1 of 5 mammalian Notch ligands, generated a microenvironment with decreased potential to maintain mouse HSCs.7 Beyond these divergent data in mice, the importance of Notch signaling in human HSCs had not been tested genetically in vivo, despite findings suggesting pathway activity in primitive hematopoietic progenitors within xenogeneic hosts.8
Benveniste et al directly compared the impact of canonical Notch signaling on highly purified human HSCs in vitro and in vivo, using identical strategies to inhibit Notch in both contexts.1 To enrich for cord blood HSCs, they combined surface markers and Rhodamine-123 exclusion. To block the effects of Notch in HSCs, they expressed a dominant-negative inhibitor of Mastermind-like (MAML) via lentiviral transduction. MAML proteins (MAML1-3) play an essential role in Notch-mediated transcriptional activation. After ligand binding and proteolytic receptor activation, intracellular Notch partners with CSL/RBP-Jk and MAML to mediate target gene activation (“canonical Notch signaling”). N-terminal MAML peptides bind Notch and CSL/RBP-Jk but fail to recruit transcriptional activators, thus exerting potent dominant-negative activity (see figure, panel A).4,9 Dominant-negative MAML (dnMAML) blocks the effects of MAML1-3 family members downstream of all Notch receptors, providing a unique genetic tool to capture the overall impact of canonical Notch signaling.9 Benveniste et al first built on their past work with OP9 stromal cells expressing Delta-like Notch ligands, now studying highly purified cocultured HSCs (see figure, panel B). Exposure to cytokines and Delta-like1/4 ligands led to Notch-driven progression along the T lineage, but also to expansion of cells maintaining a primitive HSC phenotype and in vivo reconstitution potential. The latter findings were reminiscent of work from Bernstein’s group using plate-bound Delta-like ligands.2,3 Importantly, dnMAML completely blocked both T-cell development and HSC maintenance/expansion, demonstrating that these in vitro effects were mediated by canonical Notch signaling. Next, Benveniste et al studied in vivo functions of Notch signaling by transferring dnMAML-expressing HSCs into irradiated NOD/SCID/γcnull neonates (see figure, panel C). This assay was performed in competition with tagged control HSCs, quantifying relative contribution to individual lineages and bone marrow HSCs 10 weeks after reconstitution. In contrast to in vitro findings, dnMAML expression had no impact on the capacity of human HSCs to expand and repopulate xenogeneic recipients, despite complete blockade of T-lineage development (a positive control for efficient Notch inhibition by dnMAML). These observations were reminiscent of findings in mice showing a similar dissociation between in vivo and in vitro effects.4 Finally, Benveniste et al advance our knowledge of human hematopoiesis by presenting a detailed characterization of cells similar to mouse lymphoid–primed multipotent progenitors.1
Thus, observations about canonical Notch signaling translate well from mouse to human HSCs, at least as assessed using HSC-specific pan-Notch inhibition in xenogeneic recipients.1,4 Benveniste et al relied on the same genetic strategy in vitro and in vivo, providing compelling external and internal controls for efficient Notch inhibition and highlighting the dissociation between in vitro and in vivo effects of the pathway. What might account for these divergent effects? The intensity of Notch signaling delivered to mouse or human HSCs could be lower in vivo than upon in vitro exposure to high-density plate-bound or cell-bound Notch ligands.2,3 Alternatively, Notch may exert functions on HSCs in vitro that are bypassed by other pathways in the richer in vivo HSC niche environment. As another possibility to account for divergent published observations about Notch and hematopoiesis, Notch may also exert noncanonical effects mediated independently of CSL/RBP-Jk and MAMLs that would not be inhibited by dnMAML expression or CSL/RBP-Jk loss, but still depend on Notch receptor cleavage by γ-secretase. For example, Delta-like1 ligands were suggested to rewire interleukin 6–mediated signal transducer and activator of transcription 3 signaling in CD34+ progenitors through noncanonical mechanisms that remain to be precisely delineated.10 These effects were identified in bulk CD34+ cells but not in highly purified HSCs, suggesting that they may operate mostly in progenitors downstream of HSCs. Finally, one should highlight that Benveniste et al focused on cell-autonomous effects of canonical Notch signaling in purified HSCs, thus not investigating: other functions of Notch signaling in downstream lineages, a role for Notch at early stages of reconstitution mediated by progenitors downstream of HSCs, possible functions of Notch in serial transplantation or in rare quiescent CD34– HSCs, or non–cell-autonomous effects that could play a role when Notch is not inhibited only in HSCs.6-8 Nevertheless, they show an experimental path forward by directly comparing in vitro and in vivo effects of canonical Notch signaling using a modern definition of human HSCs. These careful genetic and functional studies have fundamental significance in the study of human hematopoiesis and practical importance for the development of therapeutic strategies to modulate Notch signaling in patients.
Footnotes
Conflict-of-interest disclosure: The author declares no competing financial interests.
REFERENCES
- 1.Benveniste P, Serra P, Dervovic D, et al. Notch signals are required for in vitro but not in vivo maintenance of human hematopoietic stem cells and delay the appearance of multipotent progenitors. Blood. 2014;123(8):1167–1177. doi: 10.1182/blood-2013-07-505099. [DOI] [PubMed] [Google Scholar]
- 2.Varnum-Finney B, Brashem-Stein C, Bernstein ID. Combined effects of Notch signaling and cytokines induce a multiple log increase in precursors with lymphoid and myeloid reconstituting ability. Blood. 2003;101(5):1784–1789. doi: 10.1182/blood-2002-06-1862. [DOI] [PubMed] [Google Scholar]
- 3.Delaney C, Heimfeld S, Brashem-Stein C, Voorhies H, Manger RL, Bernstein ID. Notch-mediated expansion of human cord blood progenitor cells capable of rapid myeloid reconstitution. Nat Med. 2010;16(2):232–236. doi: 10.1038/nm.2080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Maillard I, Koch U, Dumortier A, et al. Canonical notch signaling is dispensable for the maintenance of adult hematopoietic stem cells. Cell Stem Cell. 2008;2(4):356–366. doi: 10.1016/j.stem.2008.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gao J, Graves S, Koch U, et al. Hedgehog signaling is dispensable for adult hematopoietic stem cell function. Cell Stem Cell. 2009;4(6):548–558. doi: 10.1016/j.stem.2009.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Varnum-Finney B, Halasz LM, Sun M, Gridley T, Radtke F, Bernstein ID. Notch2 governs the rate of generation of mouse long- and short-term repopulating stem cells. J Clin Invest. 2011;121(3):1207–1216. doi: 10.1172/JCI43868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Poulos MG, Guo P, Kofler NM, et al. Endothelial Jagged-1 is necessary for homeostatic and regenerative hematopoiesis. Cell Rep. 2013;4(5):1022–1034. doi: 10.1016/j.celrep.2013.07.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Anjos-Afonso F, Currie E, Palmer HG, Foster KE, Taussig DC, Bonnet D. CD34(-) cells at the apex of the human hematopoietic stem cell hierarchy have distinctive cellular and molecular signatures. Cell Stem Cell. 2013;13(2):161–174. doi: 10.1016/j.stem.2013.05.025. [DOI] [PubMed] [Google Scholar]
- 9.Maillard I, Weng AP, Carpenter AC, et al. Mastermind critically regulates Notch-mediated lymphoid cell fate decisions. Blood. 2004;104(6):1696–1702. doi: 10.1182/blood-2004-02-0514. [DOI] [PubMed] [Google Scholar]
- 10.Csaszar E, Wang W, Usenko T, et al. Blood stem cell fate regulation by Delta-1 mediated rewiring of IL-6 paracrine signaling [published online ahead of print November 15, 2013]. Blood. doi: 10.1182/blood-2013-08-520445. doi: [DOI] [PMC free article] [PubMed] [Google Scholar]