Abstract
Objective
To examine independent associations of serum 25-hydroxyvitamin D (25(OH)D), parathyroid hormone (PTH) and calcium with a range of cardiovascular risk factors in adolescents.
Design
Cross-sectional population-based study.
Setting
A nationally representative sample of the US adolescent population.
Participants
Healthy adolescents (aged 12–19) participating in the National Health and Nutrition Examination Survey (NHANES) between 2001 and 2006. Numbers varied between 740 and 5609 for given exposure and outcome associations.
Main outcome measures
Systolic blood pressure (SBP), diastolic blood pressure (DBP), lipids (triglycerides, low-density lipoprotein cholesterol (LDL-C) and high-density lipoprotein cholesterol (HDL-C)), fasting insulin and glucose, postload glucose and glycohaemoglobin (HbA1c).
Results
25(OH)D was inversely associated with SBP (−0.068 standard deviations (SD), 95% CI −0.118 to −0.018), and positively associated with HDL-C (0.101; 0.040 to 0.162) and HbA1c (0.073; 0.021 to 0.125) after adjustment for gender, age, ethnicity, socioeconomic status and waist circumference. In adjusted models, PTH was inversely associated with triglycerides (−0.115; −0.188 to −0.042) and LDL-C (−0.133; −0.207 to −0.060). In adjusted models, calcium was positively associated with fasting insulin (0.110; 0.060 to 0.160), postload glucose (0.116; 0.000 to 0.232), HbA1c (0.079; 0.035 to 0.123), triglycerides (0.182; 0.122 to 0.242), HDL-C (0.049; 0.010 to 0.088) and LDL-C (0.137; 0.080 to 0.195). The associations of each exposure with risk factors remained after mutual adjustment for each other.
Conclusion
Higher calcium levels might be a more important predictor of increased cardiovascular risk in adolescents than lower 25(OH)D levels or PTH levels, but the findings require replication in additional studies and examination in prospective studies.
INTRODUCTION
Recent epidemiological evidence has linked vitamin D deficiency with several non-skeletal diseases, including cardiovascular disease1,2 and risk factors3 in general adult populations. Some evidence from randomised controlled trials (RCTs) suggests that higher vitamin D is causally related to lower blood pressure,4,5 triglycerides and tumour necrosis factor a (an inflammatory marker)6 in adults but one large RCT (N=36 282) reported no effect of an intervention with combined vitamin D and calcium supplements on coronary events.7 By regulating calcium homoeostasis, serum 25-hydroxyvitamin D (25(OH)D—the measured circulating form of vitamin D), parathyroid hormone (PTH) and calcium are closely related to each other.8 It has been suggested that variation in PTH mediates the association of 25(OH)D with cardiovascular risk factors.9 Serum calcium levels have also been positively associated with cardiovascular disease and risk factors in adults.10
To date, few studies have examined associations of 25(OH)D with cardiovascular risk factors in children or adolescents.11,12 To our knowledge, no studies have examined PTH or calcium in relation to cardiovascular risk factors in children or adolescents, or a combination of the three components (25(OH)D, PTH and calcium). Vitamin D deficiency and insufficiency may be common in children, though the prevalence of deficiency in this age group is difficult to discern owing to debates about the appropriate threshold for defining this.8 Furthermore, cardiovascular risk factors in adolescence track into adulthood13 and variation in risk factors in childhood correlate with future cardiovascular disease incidence similarly to adult levels.14 Thus, identifying modifiable risk factors relating to cardiovascular risk in adolescence may improve prevention of later disease.
In this study, we conducted an analysis of adolescent (12–19 years of age) participants of the post-millennial National Health and Nutrition Examination Survey (NHANES) series with the following aims: (1) to examine associations of 25(OH)D, PTH and calcium with cardiovascular risk factors (blood pressure, fasting and 2 h post-load glucose, fasting insulin, glycohaemoglobin (HbA1c) and lipid measures); (2) to compare the magnitude of associations of 25(OH)D, PTH and calcium with cardiovascular risk factors; (3) to assess whether associations of 25(OH)D, PTH and calcium with cardiovascular risk factors are independent of each other or not.
METHODS
Participants and measures
NHANES is a freely available, complex multistage probability sample of the civilian non-institution-alised population of the USA. Data on adolescents (12–19 years of age) from three cross-sectional surveys covering 2001–2, 2003–4 and 2005–6 were combined for analysis. Full details of the survey and data for scientific use are available at http://www.cdc.gov/nchs/nhanes.htm. Details of measures used here are also included in the online supplementary material.
Statistics
The analyses were adjusted for sampling probability (via weights) and cluster effects using the svy procedures in Stata version 10. Where distributions appeared non-normal, we used natural log transformations (fasting insulin, fasting and postload glucose, triglycerides and PTH).
Univariable linear and logistic regression was used to test trends of participants’ characteristics across fifths of the 25(OH)D, PTH and calcium distributions. Non-linearity of associations (specifically curvilinear associations) between exposures and outcomes was tested by including a quadratic term in the regression models. In multivariable models the following potential confounders were included: age, gender, ethnicity, poverty income ratio (PIR), waist circumference (or body mass index (BMI)). Because of missing data we examined possible confounding by smoking and outdoor physical activity only in the subgroup with these data. In a final model we mutually adjusted for each of the three exposure variables. In all regression models, 25(OH)D, logged PTH, calcium and outcomes were used on their SD scale (z-scores) so that the magnitude of regression coefficients between models was directly comparable. Since we combined three surveys and the NHANES methodology suggests the possibility of drift in 25 (OH)D assaying performance across cycles of the survey, we also conducted all analyses separately in the 2-year cycles and compared results with the combined analysis.
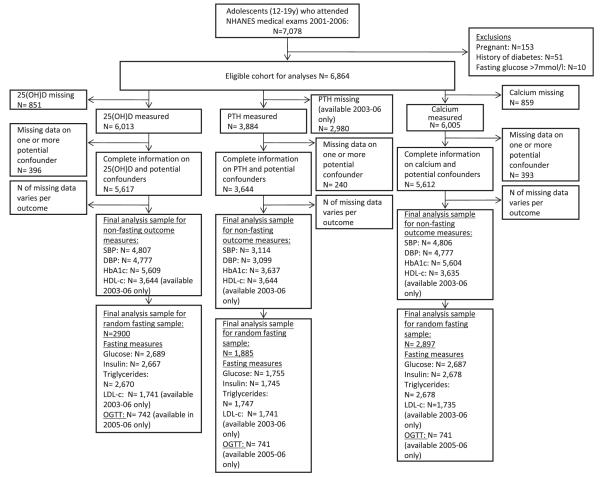
Our sample consisted of adolescents aged 12–19 who attended a medical examination (N=7078). Pregnant participants (N=153), those with a history of diabetes (N=51) and those with fasting glucose >7 mmol/l (N=10) were excluded. Numbers with available data for each association varied because some outcomes and exposures were available only at certain clinic times or in certain cycles of the study. Figure 1 shows the numbers available for each analysis. We used maximum numbers for all analyses, ensuring that group sizes in any series of multivariable analyses were equal. We also compared results with those from a single analytical sample (N=1460) with complete data on all of 25(OH)D, PTH, calcium, potential confounders and outcomes (excluding postload glucose, which was only available for a small subsample).
Figure 1. Eligible participants and those included in analyses of associations of 25(OH)D, PTH and calcium with CVD risk factors.
CVD, cardiovascular disease; DBP, diastolic blood pressure; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; NHANES, National Health and Nutrition Examination Survey; OGTT, oral glucose tolerance test; 25(OH)D, 25-hydroxyvitamin D; PTH, parathyroid hormone; SBP, systolic blood pressure.
RESULTS
The weighted mean 25(OH)D in our sample was 50.4 nmol/l (SE=0.29). Weighted mean corrected calcium was 2.35 mmol/l (SE=0.001) and the weighted geometric mean of PTH was 38.3 ng/ml (SE log PTH=0.007). 25(OH)D and log PTH were weakly inversely associated with each other (Pearson’s r=−0.27, p<0.001), as were log PTH and calcium (Pearson’s r =−0.18, p<0.001). 25(OH)D and calcium were not correlated (Pearson’s r=0.02, p=0.14). There was no evidence of non-linear associations between 25(OH)D, PTH and calcium (all p values ≥0.11). There was also no strong evidence that any of the associations we examined differed between male and female subjects (all p values for interaction ≥0.08), except for the associations between 25(OH)D and fasting insulin (p for interaction=0.03), low-density lipoprotein cholesterol (LDL-C) (p=0.01) and fasting triglycerides (p<0.001). For the associations noted above results are presented separately for male and female subjects as well as combined. There was no strong evidence that associations differed by ethnicity (all p values for interactions ≥0.13), with one exception—associations between 25(OH)D and fasting insulin.
Supplemental table 1 shows the characteristics of participants by fifths of the 25(OH)D distribution. Male gender, lighter pigmentation of the skin, the proportion above the poverty threshold, having engaged in recent outdoor activity, having used vitamin D supplements recently and high-density lipoprotein cholesterol (HDL-C) all increased linearly across fifths of 25(OH)D. BMI, waist circumference, systolic blood pressure (SBP), fasting insulin, postload glucose, HbA1c and PTH all decreased linearly across fifths of 25(OH)D. There was weak evidence of triglycerides increasing across fifths of the 25(OH)D distribution.
Supplemental table 2 shows participants’ characteristics across fifths of the PTH distribution. Age, the proportion above the poverty threshold, current smoking, triglycerides, LDL-C, 25(OH)D and calcium decreased across the PTH distribution. BMI, waist circumference and fasting insulin increased across fifths of the PTH distribution. A weak inverse trend was observed between vitamin D supplementation and PTH.
Supplemental table 3 shows participants’ characteristics by fifth of serum calcium. Black ethnicity, BMI, waist circumference, insulin, postload glucose, HbA1c, triglycerides, and LDL-C increased linearly across fifths of the calcium distribution. Age, Mexican ethnicity, poverty and PTH decreased across the calcium distribution. There was no evidence of non-linearity in any exposure–outcome association (all quadratic p values >0.10).
Table 1 shows multivariable associations between z-scored 25(OH)D and cardiovascular risk factors. In age- and gender-adjusted models (model 1), there were inverse associations between 25(OH)D and SBP, fasting insulin and HbA1c and positive associations with HDL-C and triglycerides. There was evidence for an inverse association between 25(OH)D and LDL-C in male subjects only. There was no strong evidence of associations with other outcomes. With further adjustments for ethnicity and PIR (model 2), the unexpected positive association with triglycerides levels attenuated to the null. The direction of the point estimate for HbA1c changed with adjustment for ethnicity and PIR, suggesting an (unexpected) increase in HbA1c with higher 25(OH)D. Other associations were largely unchanged. The main factor influencing the change in direction of association with HbA1c was adjustment for ethnicity. After adjustment for waist circumference (model 3), the inverse association of 25(OH)D with SBP remained. The association of 25(OH)D with fasting insulin attenuated to the null in female subjects, but remained in male subjects. With further adjustment for waist circumference (model 3) the only associations in the expected direction (low 25(OH)D being related to increased levels of adverse risk factors) were with HDL-C (SD per SD change 25(OH)D 0.101; 95% CI 0.040 to 0.162) and SBP (−0.068; 95% CI −0.118 to −0.018). On their original scales these associations show that for each greater nmol/l of 25(OH)D, HDL-C increases on average by 0.002 (95% CI 0.001 to 0.002) mmol/l and SBP decreases on average by −0.032 (95% CI −0.055 to −0.008) mm Hg.
Table 1. Multivariable associations of 25-hydroxyvitamin D with cardiovascular risk factors in US adolescents 2001–6.
| Model 1 |
Model 2 |
Model 3 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk factors | SD | N | Coefficient | 95% CI | p Value | Coefficient | 95% CI | p Value | Coefficient | 95% CI | p Value |
| SBP (mm Hg) | 10.3 | 4807 | −0.131 | −0.175 to −0.087 | <0.001 | −0.137 | −0.185 to −0.088 | <0.001 | −0.068 | −0.118 to −0.018 | 0.009 |
| DBP (mm Hg) | 10.7 | 4777 | −0.016 | −0.063 to 0.031 | 0.49 | −0.043 | −0.100 to 0.014 | 0.14 | −0.046 | −0.105 to 0.014 | 0.13 |
| Fasting glucose (mmol/l) | 0.44 | 2689 | −0.017 | −0.088 to 0.053 | 0.62 | −0.065 | −0.147 to 0.017 | 0.12 | −0.033 | −0.114 to 0.048 | 0.42 |
| Fasting insulin (mU/l)* | 0.72 | 2667 | −0.161 | −0.214 to −0.109 | <0.001 | −0.167 | −0.221 to −0.112 | <0.001 | −0.033 | −0.079 to 0.013 | 0.16 |
| Male subjects only | 0.75 | 1392 | −0.216 | −0.288 to −0.144 | <0.001 | −0.279 | −0.367 to −0.191 | <0.001 | −0.089 | −0.163 to −0.015 | 0.02 |
| Female subjects only | 0.67 | 1275 | −0.111 | −0.176 to −0.047 | 0.001 | −0.061 | −0.130 to 0.009 | 0.08 | 0.022 | −0.037 to 0.081 | 0.45 |
| Postload glucose* | 0.24 | 742 | −0.069 | −0.201 to 0.063 | 0.28 | −0.104 | −0.252 to 0.045 | 0.16 | −0.052 | −0.179 to 0.075 | 0.40 |
| Glycohaemoglobin (%) | 0.34 | 5609 | −0.057 | −0.102 to −0.011 | 0.02 | 0.049 | −0.002 to 0.100 | 0.06 | 0.073 | 0.021 to 0.125 | 0.007 |
| Fasting triglycerides (mmol/l)* | 0.48 | 2670 | 0.073 | 0.010 to 0.136 | 0.02 | −0.021 | −0.090 to 0.048 | 0.54 | 0.059 | −0.014 to 0.132 | 0.11 |
| Male subjects only | 0.50 | 1392 | −0.027 | −0.100 to 0.047 | 0.47 | −0.148 | −0.236 to −0.060 | 0.001 | −0.033 | −0.120 to 0.055 | 0.46 |
| Female subjects only | 0.46 | 1278 | 0.164 | 0.077 to 0.250 | <0.001 | 0.096 | 0.000 to 0.192 | 0.05 | 0.144 | 0.049 to 0.239 | 0.004 |
| HDL-C (mmol/l)† | 0.33 | 3644 | 0.069 | 0.015 to 0.123 | 0.01 | 0.172 | 0.117 to 0.226 | <0.001 | 0.101 | 0.040 to 0.162 | 0.002 |
| LDL-C (mmol/l)† | 0.69 | 1741 | −0.020 | −0.092 to 0.052 | 0.57 | −0.022 | −0.101 to 0.058 | 0.58 | 0.023 | −0.062 to 0.108 | 0.58 |
| Male subjects only | 0.72 | 934 | −0.105 | −0.207 to −0.004 | 0.04 | −0.116 | −0.231 to −0.001 | 0.05 | −0.057 | −0.187 to 0.073 | 0.38 |
| Female subjects only | 0.65 | 807 | 0.053 | −0.063 to 0.168 | 0.36 | 0.064 | −0.065 to 0.194 | 0.32 | 0.096 | −0.035 to 0.227 | 0.15 |
z-Score of log transformed values; SD of log-transformed variable represented.
Measured from 2003 to 2006 only.
Model 1—age and gender adjusted; model 2—as model 1 plus ethnicity, PIR; model 3—as model 2 plus waist circumference.
DBP, diastolic blood pressure; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; PIR, poverty index ratio; SBP, systolic blood pressure.
The positive association with HbA1c (0.073) remained in model 3. When analyses were stratified by ethnicity, the positive association between 25(OH)D and HbA1c was present in all ethnic groups except ‘Other’. As noted above there was evidence that the association of 25(OH)D with fasting insulin varied by ethnicity (see supplemental table 4), with the association being null in white and black participants, but inverse in Mexican Americans and ethnic groups defined as ‘Other’.
Table 2 shows multivariable associations between PTH and cardiovascular risk factors. In age and gender adjusted analyses (model 1), PTH was inversely associated with LDL-C and triglycerides. PTH was also positively associated with systolic blood pressure and fasting insulin. There was no evidence for associations with other cardiovascular risk factors. The associations of PTH with fasting insulin attenuated after adjustment for ethnicity and PIR (model 2), but the inverse associations with LDL-C and triglycerides remained after these adjustments and also after further controlling for waist circumference (model 3).
Table 2. Multivariable associations of parathyroid hormone with cardiovascular risk factors in US adolescents 2003–6.
| Model 1 |
Model 2 |
Model 3 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk factors | SD | N | Coefficient | 95% CI | p Value | Coefficient | 95% CI | p Value | Coefficient | 95% CI | p Value |
| SBP (mm Hg) | 10.3 | 3114 | 0.045 | −0.005 to 0.095 | 0.08 | 0.039 | −0.017 to 0.095 | 0.17 | 0.022 | −0.038 to 0.081 | 0.46 |
| DBP (mm Hg) | 10.8 | 3099 | 0.017 | −0.037 to 0.071 | 0.52 | 0.023 | −0.031 to 0.078 | 0.39 | 0.022 | −0.032 to 0.076 | 0.40 |
| Fasting glucose (mmol/l) | 0.44 | 1755 | 0.023 | −0.047 to 0.094 | 0.51 | 0.034 | −0.035 to 0.103 | 0.32 | 0.020 | −0.049 to 0.088 | 0.57 |
| Fasting insulin (mU/l)* | 0.75 | 1745 | 0.069 | −0.007 to 0.145 | 0.07 | 0.054 | −0.023 to 0.131 | 0.17 | 0.004 | −0.045 to 0.052 | 0.88 |
| Postload glucose* | 0.24 | 741 | 0.014 | −0.123 to 0.151 | 0.83 | 0.023 | −0.111 to 0.156 | 0.72 | 0.000 | −0.119 to 0.118 | 0.99 |
| Glycohaemoglobin (%) | 0.33 | 3637 | 0.033 | −0.024 to 0.089 | 0.25 | −0.008 | −0.065 to 0.049 | 0.77 | −0.014 | −0.071 to 0.044 | 0.63 |
| Fasting triglycerides (mmol/l)* | 0.47 | 1747 | −0.124 | −0.207 to −0.041 | 0.005 | −0.085 | −0.168 to −0.003 | 0.04 | −0.115 | −0.188 to −0.042 | 0.003 |
| HDL-C (mmol/l) | 0.33 | 3644 | −0.002 | −0.044 to 0.040 | 0.92 | −0.026 | −0.069 to 0.017 | 0.23 | −0.005 | −0.043 to 0.033 | 0.79 |
| LDL-C (mmol/l) | 0.68 | 1741 | −0.111 | −0.185 to −0.036 | 0.005 | −0.120 | −0.195 to −0.046 | 0.003 | −0.133 | −0.207 to −0.060 | 0.001 |
z-Score of log transformed values.
Model 1—age and gender adjusted; model 2—as model 1 plus ethnicity, PIR; model 3—as model 2 plus waist circumference.
DBP, diastolic blood pressure; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; PIR, poverty index ratio; SBP, systolic blood pressure.
Table 3 shows multivariable associations between calcium and cardiovascular risk factors. Calcium was positively associated with insulin, postload glucose, HbA1c, triglycerides and LDL-C in age- and gender-adjusted analyses (model 1). There was no strong evidence of associations with SBP or diastolic blood pressure (DBP) or HDL-C. After the inclusion of ethnicity and PIR in the models, the associations of calcium with insulin, postload glucose, HbA1c, triglycerides and LDL-C remained. After the addition of adjustment for waist circumference, calcium was positively associated with all cardiovascular risk factors except SBP and DBP, and fasting glucose. The order of magnitude of associations of higher calcium with adverse risk factors observed in the final model was: fasting triglycerides (0.182), LDL-C (0.137), postload glucose (0.116), fasting insulin (0.110) and HbA1c% (0.079). On their original scales these associations show that for each greater mmol/l of calcium, on average, fasting triglycerides increase by 3.19 (95% CI 2.18 to 4.69) mmol/l, postload glucose increases by 1.53 (95% CI 1.02 to 2.29) mmol/l, fasting insulin increases by 2.88 (95% CI 1.78 to 4.66) pmol/l and HbA1c increases by 0.31% (95% CI 0.13% to 0.48%).
Table 3. Multivariable associations of corrected calcium with cardiovascular risk factors in US adolescents 2001–6.
| Model 1 |
Model 2 |
Model 3 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk factors | SD | N | Coefficient | 95% CI | p Value | Coefficient | 95% CI | p Value | Coefficient | 95% CI | p Value |
| SBP (mm Hg) | 10.33 | 4806 | 0.023 | −0.013 to 0.059 | 0.21 | 0.008 | −0.029 to 0.044 | 0.68 | −0.017 | −0.051 to 0.017 | 0.32 |
| DBP (mm Hg) | 10.74 | 4777 | −0.025 | −0.066 to 0.017 | 0.24 | −0.028 | −0.070 to 0.013 | 0.18 | −0.028 | −0.069 to 0.013 | 0.18 |
| Fasting glucose (mmol/l) | 0.44 | 2687 | −0.004 | −0.056 to 0.047 | 0.86 | 0.020 | −0.034 to 0.074 | 0.46 | 0.004 | −0.050 to 0.058 | 0.88 |
| Fasting insulin (mU/l)* | 0.72 | 2678 | 0.175 | 0.123 to 0.227 | <0.001 | 0.175 | 0.124 to 0.225 | <0.001 | 0.110 | 0.060 to 0.160 | <0.001 |
| Postload glucose* | 0.24 | 741 | 0.159 | 0.028 to 0.290 | 0.02 | 0.111 | −0.004 to 0.227 | 0.06 | 0.116 | 0.000 to 0.232 | 0.05 |
| Glycohaemoglobin (%) | 0.34 | 5604 | 0.106 | 0.062 to 0.151 | <0.001 | 0.087 | 0.042 to 0.131 | 0.01 | 0.079 | 0.035 to 0.123 | 0.04 |
| Fasting triglycerides (mmol/l)* | 0.48 | 2678 | 0.169 | 0.106 to 0.232 | <0.001 | 0.217 | 0.150 to 0.284 | <0.001 | 0.182 | 0.122 to 0.242 | <0.001 |
| HDL-C (mmol/l)† | 0.33 | 3635 | 0.023 | −0.019 to 0.065 | 0.26 | 0.006 | −0.037 to 0.048 | 0.78 | 0.049 | 0.010 to 0.088 | 0.02 |
| LDL-C (mmol/l)† | 0.68 | 1737 | 0.150 | 0.091 to 0.209 | <0.001 | 0.151 | 0.092 to 0.211 | <0.001 | 0.137 | 0.080 to 0.195 | <0.001 |
z-Score of log transformed values.
Measured from 2003 to 2006 only.
Model 1—age and gender adjusted; model 2—as model 1 plus ethnicity, PIR; model 3—as model 2 plus waist circumference.
DBP, diastolic blood pressure; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; PIR, poverty index ratio; SBP, systolic blood pressure.
Table 4 displays multivariable associations of 25(OH)D, PTH and calcium with cardiovascular risk factors in a subsample of participants with complete data on 25(OH)D, PTH and calcium and potential confounders. The first model included all potential confounders, and the second model also included mutual adjustment for 25(OH)D, PTH and calcium. In general, the associations of each exposure of interest with risk factors observed in fully adjusted models remained after adjustment for the two additional exposures.
Table 4. Multivariable and mutually adjusted models for 25-hydroxyvitamin D (25(OH)D), parathyroid hormone (PTH) and corrected calcium with cardiovascular risk factors in US adolescents 2003–6.
| Vitamin D |
PTH |
Calcium |
|||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Confounder-Controlled |
Mutually adjusted model |
Confounder-Controlled |
Mutually adjusted model |
Confounder-Controlled |
Mutually adjusted model |
||||||||
| Risk factors | N | Coeff. | 95% CI | Coeff. | 95% CI | Coeff. | 95% CI | Coeff. | 95% CI | Coeff. | 95% CI | Coeff. | 95% CI |
| SBP (mm Hg) | 3106 | −0.061 | −0.123 to 0.001 | −0.059 | −0.118 to 0.001 | 0.021 | −0.038 to 0.081 | 0.011 | −0.047 to 0.069 | −0.012 | −0.056 to 0.032 | 0.002 | −0.044 to 0.048 |
| DBP (mm Hg) | 3091 | −0.085 | −0.150 to −0.019 | −0.080 | −0.145 to −0.015 | 0.022 | −0.032 to 0.076 | 0.003 | −0.056 to 0.062 | −0.036 | −0.086 to 0.013 | −0.021 | −0.075 to 0.032 |
| Fasting glucose (mmol/l) | 1749 | −0.006 | −0.121 to 0.108 | −0.003 | −0.119 to 0.114 | 0.020 | −0.049 to 0.089 | 0.022 | −0.046 to 0.090 | 0.010 | −0.058 to 0.079 | 0.015 | −0.054 to 0.085 |
| Fasting insulin (mU/l)* | 1742 | −0.016 | −0.075 to 0.043 | −0.027 | −0.083 to 0.028 | 0.003 | −0.045 to 0.052 | 0.018 | −0.031 to 0.067 | 0.126 | 0.068 to 0.185 | 0.134 | 0.076 to 0.192 |
| Postload glucose* | 740 | −0.052 | −0.178 to 0.075 | −0.066 | −0.199 to 0.066 | 0.000 | −0.118 to 0.119 | 0.011 | −0.107 to 0.129 | 0.130 | 0.005 to 0.255 | 0.141 | 0.010 to 0.271 |
| Glycohaemoglobin (%) | 3625 | 0.095 | 0.027 to 0.163 | 0.085 | 0.020 to 0.151 | −0.014 | −0.072 to 0.044 | 0.023 | −0.034 to 0.080 | 0.113 | 0.052 to 0.173 | 0.106 | 0.048 to 0.165 |
| Fasting triglycerides (mmol/l)* | 1741 | 0.065 | −0.032 to 0.162 | 0.020 | −0.072 to 0.111 | −0.115 | −0.188 to −0.042 | −0.077 | −0.145 to −0.009 | 0.229 | 0.151 to 0.307 | 0.210 | 0.122 to 0.298 |
| HDL-C (mmol/l) | 3632 | 0.101 | 0.040 to 0.162 | 0.100 | 0.043 to 0.158 | −0.004 | −0.042 to 0.034 | 0.022 | −0.011 to 0.055 | 0.049 | 0.010 to 0.088 | 0.040 | 0.001 to 0.079 |
| LDL-C (mmol/l) | 1735 | 0.018 | −0.066 to 0.103 | −0.033 | −0.113 to 0.047 | −0.134 | −0.207 to −0.061 | −0.110 | −0.179 to −0.040 | 0.221 | 0.144 to 0.297 | 0.202 | 0.130 to 0.273 |
z-Score of log transformed values.
Confounder-Controlled model adjusted for age, gender, ethnicity, PIR and waist circumference.
Mutually adjusted models:
Vitamin D as confounder-Controlled, plus PTH and calcium.
PTH as confounder-Controlled, plus 25(OH)D and calcium.
Calcium as confounder-Controlled, plus 25(OH)D and PTH.
DBP, diastolic blood pressure; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; PIR, poverty index ratio; SBP, systolic blood pressure.
Exchanging BMI for waist circumference produced identical results as did excluding participants who recently smoked. Adjusting for physical activity in a subgroup analysis of participants with physical activity measures also produced similar results. Excluding nine participants who reported using drugs that might influence levels or metabolism of the exposures examined here did not alter results. When we conducted all regression models in each 2-year survey cycles individually, similar associations were observed to those presented from the cycles combined. Analyses using a single analytic sample (N=1460), with complete data on all exposures, outcomes and covariables, resulted in no substantial differences from the results presented for maximal sample sizes (supplemental tables 5–7).
DISCUSSION
We examined associations of 25(OH)D (vitamin D), PTH and calcium with cardiovascular disease risk factors in adolescents. Our expectation, based on studies in adults, was that lower 25(OH)D, higher PTH and higher calcium would be associated with increased adverse levels of cardiovascular risk factors. We found that higher calcium was most consistently associated with adverse cardiovascular risk factors; being associated with higher fasting insulin, HbA1c, postload glucose, LDL-C and triglyceride levels. Low serum 25(OH)D was associated with higher SBP and lower HDL-C. We also found some unexpected associations in the opposite direction to that expected from findings in adults. Notably, higher PTH levels were only associated with beneficial risk factors, lower LDL-C and triglycerides, and higher calcium was associated with higher HDL-C.
A major strength of this study is the comparison of associations of all three of 25(OH)D, PTH and calcium with a wide range of cardiovascular risk factors in adolescents. The participants were a healthy unselected sample. No other studies have examined the associations here prospectively in adolescents. The main limitation is that our study was cross-sectional. Although there is the possibility of assay drift between surveys, we repeated analyses in each 2-year cycle separately and observed similar results to those of our main models. It was not possible to adjust for seasonality in the NHANES dataset and seasonality is correlated with vitamin D levels owing to varying ultraviolet B exposure. Seasonality is unlikely to be causally related to cardiovascular risk factors and is unlikely to explain the associations seen, but omitting adjustment for it may result in underestimation of vitamin D associations.15 No repeat measurements of 25(OH)D, PTH or calcium were available and therefore we cannot account for regression dilution bias, which may bias results towards the null. However, season-specific measurements of 25(OH)D have been shown to correlate over time,16 and single measures of 25 (OH)D, PTH and calcium have previously been shown to be associated with bone outcomes with the direction and magnitude expected from their known biological actions.17 Caution should be taken in interpreting results in relation to populations other than US adolescents.
Our finding that vitamin D is inversely associated with SBP and positively associated with HDL-C is in accordance with previous studies in adults,3,18 and previous analyses of NHANES children and adolescents.11,12 Experimental evidence suggests that vitamin D negatively regulates the renin–angiotensin system to lower blood pressure,19 though a possible relation to cholesterol metabolism is currently unclear. We also found a positive association of 25(OH)D with HbA1c in confounder-adjusted models (HbA1c was absent from previous analyses of NHANES adolescents/children). The positive association emerged only with adjustment for ethnicity, suggesting that ethnicity may mask a positive association of 25(OH)D with HbA1c; however, we found no evidence of an interaction between 25(OH)D and ethnicity in relation to HbA1c. This finding was unexpected, given the inverse association of vitamin D with type 2 diabetes in adults,20 and replication in further studies is warranted. In male subjects, we found evidence of an inverse association between 25(OH)D and fasting insulin. Some experimental evidence has also linked higher vitamin D status with improved glycaemic control.21 Nonetheless, the lack of any association of 25(OH)D with fasting or postload glucose (or fasting insulin in female subjects) here suggests that any influence of vitamin D on insulin-glucose metabolism may not emerge fully until adulthood.
In general, no strong evidence was found for associations between PTH and cardiovascular risk factors, but unexpected inverse associations were observed for LDL-C and triglycerides. As with the positive association of 25(OH)D with HbA1c these associations might be due to chance and require replication to rule this out. Overall, despite positive associations of PTH with cardiovascular disease in prospective studies of adults,22 our results suggest that variation in PTH in healthy adolescents is not related to cardiovascular risk.
In fully adjusted models, calcium was positively associated with a wide range of cardiovascular risk factors (all lipid measures, fasting insulin, postload glucose and HbA1c) and most of these associations were stronger than associations of 25(OH)D with risk factors. Thus, our results suggest that variation in calcium levels may be a more robust marker of cardiovascular risk in adolescents than vitamin D or PTH. Our findings are consistent with prospective evidence of positive associations of serum calcium with cardiovascular disease and risk factors in adult populations,10 and experimental evidence suggesting that extracellular calcium may underlie the regulation of insulin sensitivity.23
Profiles of 25(OH)D, PTH and calcium may vary between adolescence and adulthood: PTH levels increase during puberty,24 which produces a concomitant increase in the conversion of 25(OH)D to 1,25-dihydroxyvitamin D (the biologically active form of vitamin D).25,26 It is suggested that this exacerbates a decrease in serum 25(OH)D,8,26 though this has not been observed universally.27 Thus, associations of 25(OH)D, PTH and calcium with cardiovascular disease risk factors as well as with other outcomes may differ by age and this might explain why we did not find universally similar associations to those seen in adult studies. Data on pubertal stage is not available in NHANES and we could not account for this in our analyses. Possibly, the equivalent associations in studies of adults are explained by residual confounding by lifestyle characteristics, drugs or comorbidities that are less likely to confound associations in an adolescent age group.
We identified statistical evidence of gender×25(OH)D interactions with three outcomes and an ethnicity×25(OH)D interaction with one outcome. These four interactions represent 7% of the 54 interactions that we have examined and may be chance findings. They should be treated with caution unless robustly replicated in other studies.
Further prospective studies in adolescents are required. The use of genetic variants that are robustly associated with variation in 25(OH)D, PTH or calcium levels could be used in an instrumental variable analysis (Mendelian randomisation studies) to examine causal effects of these exposures on cardiovascular risk factors in adolescents28 The recent findings of such variants in genome-wide analyses of vitamin D in adults suggests this possibility,29 though it would be useful to demonstrate that these variants relate to levels in adolescents and to identify robust genetic variants associated with PTH and calcium. If prospective studies and Mendelian randomisation studies do confirm associations of lower vitamin D and higher calcium with adverse cardiovascular risk factors then funding for RCTs would be required to confirm causation, and to test the effectiveness of supplements to optimise serum levels of these to reduce cardiovascular risk.
Supplementary Material
Acknowledgements
We thank the participants and staff who make the NHANES data available.
Funding: UK Medical Research Council (MRC). 20 Park Crescent, London, W1B 1AL, UK. Other Funders: Wellcome Trust.
Work on this study is funded by an UK MRC Grant (G0701603). The UK Medical Research Council (MRC) (G0600705) and the University of Bristol provide core funding for the UK MRC Centre of Causal Analyses in Translational Epidemiology, where all authors work. AF is funded by a UK MRC research fellowship. DW is funded by a Wellcome Trust 4-year PhD studentship in molecular, genetic and lifecourse epidemiology [WT083431MA].
Footnotes
Competing interests: None.
Ethics approval: This study was conducted with the approval of the Part of NHANES protocol.
Provenance and peer review: Not commissioned; externally peer reviewed.
REFERENCES
- 1.Kilkkinen A, Knekt P, Aro A, et al. Vitamin D status and the risk of cardiovascular disease death. Am J Epidemiol. 2009;170:1032–9. doi: 10.1093/aje/kwp227. [DOI] [PubMed] [Google Scholar]
- 2.Wang TJ, Pencina MJ, Booth SL, et al. Vitamin D deficiency and risk of cardiovascular disease. Circulation. 2008;117:503–11. doi: 10.1161/CIRCULATIONAHA.107.706127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Martins D, Wolf M, Pan D, et al. Prevalence of cardiovascular risk factors and the serum levels of 25-hydroxyvitamin D in the United States: data from the Third National Health and Nutrition Examination Survey. Arch Intern Med. 2007;167:1159–65. doi: 10.1001/archinte.167.11.1159. [DOI] [PubMed] [Google Scholar]
- 4.Krause R, Buhring M, Hopfenmuller W, et al. Ultraviolet B and blood pressure. Lancet. 1998;352:709–10. doi: 10.1016/S0140-6736(05)60827-6. [DOI] [PubMed] [Google Scholar]
- 5.Pfeifer M, Begerow B, Minne HW, et al. Effects of a short-term vitamin D3 and calcium supplementation on blood pressure and parathyroid hormone levels in elderly women. J Clin Endocrinol Metab. 2001;86:1633–7. doi: 10.1210/jcem.86.4.7393. [DOI] [PubMed] [Google Scholar]
- 6.Zittermann A, Frisch S, Berthold HK, et al. Vitamin D supplementation enhances the beneficial effects of weight loss on cardiovascular disease risk markers. Am J Clin Nutr. 2009;89:1321–7. doi: 10.3945/ajcn.2008.27004. [DOI] [PubMed] [Google Scholar]
- 7.Hsia J, Heiss G, Ren H, et al. Calcium/vitamin D supplementation and cardiovascular events. Circulation. 2007;115:846–54. doi: 10.1161/CIRCULATIONAHA.106.673491. [DOI] [PubMed] [Google Scholar]
- 8.Holick MF. Vitamin D deficiency. N Engl J Med. 2007;357:266–81. doi: 10.1056/NEJMra070553. [DOI] [PubMed] [Google Scholar]
- 9.Snijder MB, van Dam RM, Visser M, et al. Adiposity in relation to vitamin D status and parathyroid hormone levels: a population-based study in older men and women. J Clin Endocrinol Metab. 2005;90:4119–23. doi: 10.1210/jc.2005-0216. [DOI] [PubMed] [Google Scholar]
- 10.Lind L, Skarfors E, Berglund L, et al. Serum calcium: a new, independent, prospective risk factor for myocardial infarction in middle-aged men followed for 18 years. J Clin Epidemiol. 1997;50:967–73. doi: 10.1016/s0895-4356(97)00104-2. [DOI] [PubMed] [Google Scholar]
- 11.Reis JP, von Muhlen D, Miller ER, III, et al. Vitamin D status and cardiometabolic risk factors in the United States adolescent population. Pediatrics. 2009;124:e371–9. doi: 10.1542/peds.2009-0213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kumar J, Muntner P, Kaskel FJ, et al. Prevalence and associations of 25-hydroxyvitamin D deficiency in US children: NHANES 2001–2004. Pediatrics. 2009;124:e362–70. doi: 10.1542/peds.2009-0051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Webber LS, Srinivasan SR, Wattigney WA, et al. Tracking of serum lipids and lipoproteins from childhood to adulthood: The Bogalusa Heart Study. Am J Epidemiol. 1991;133:884–99. doi: 10.1093/oxfordjournals.aje.a115968. [DOI] [PubMed] [Google Scholar]
- 14.McCarron P, Smith GD, Okasha M, et al. Blood pressure in young adulthood and mortality from cardiovascular disease. Lancet. 2000;355:1430–1. doi: 10.1016/S0140-6736(00)02146-2. [DOI] [PubMed] [Google Scholar]
- 15.Wang Y, Jacobs EJ, McCullough ML, et al. Comparing methods for accounting for seasonal variability in a biomarker when only a single sample is available: insights from simulations based on serum 25-hydroxyvitamin D. Am J Epidemiol. 2009;170:88–94. doi: 10.1093/aje/kwp086. [DOI] [PubMed] [Google Scholar]
- 16.Hofmann JN, Yu K, Horst RL, et al. Long-term variation in serum 25-hydroxyvitamin D concentration among participants in the prostate, lung, colorectal, and ovarian cancer screening trial. Cancer Epidemiol Biomarkers Prev. 2010;19:927–31. doi: 10.1158/1055-9965.EPI-09-1121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Brot C, Jørgensen N, Madsen OR, et al. Relationships between bone mineral density, serum vitamin D metabolites and calcium:phosphorus intake in healthy perimenopausal women. J Intern Med. 1999;245:509–16. doi: 10.1046/j.1365-2796.1999.00474.x. [DOI] [PubMed] [Google Scholar]
- 18.Forman JP, Giovannucci E, Holmes MD, et al. Plasma 25-hydroxyvitamin D levels and risk of incident hypertension. Hypertension. 2007;49:1063–9. doi: 10.1161/HYPERTENSIONAHA.107.087288. [DOI] [PubMed] [Google Scholar]
- 19.Li YC, Kong J, Wei M, et al. 1,25-Dihydroxyvitamin D3 is a negative endocrine regulator of the renin-angiotensin system. J Clin Invest. 2002;110:229–38. doi: 10.1172/JCI15219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mattila C, Knekt P, Mannisto S, et al. Serum 25-hydroxyvitamin D concentration and subsequent risk of type 2 diabetes. Diabetes Care. 2007;30:2569–70. doi: 10.2337/dc07-0292. [DOI] [PubMed] [Google Scholar]
- 21.Norman AW, Frankel JB, Heldt AM, et al. Vitamin-D deficiency inhibits pancreatic-secretion of insulin. Science. 1980;209:823–5. doi: 10.1126/science.6250216. [DOI] [PubMed] [Google Scholar]
- 22.Hagstrom E, Hellman P, Larsson TE, et al. Plasma parathyroid hormone and the risk of cardiovascular mortality in the community. Circulation. 2009;119:2765–71. doi: 10.1161/CIRCULATIONAHA.108.808733. [DOI] [PubMed] [Google Scholar]
- 23.Levy J. Abnormal cell calcium homeostasis in type 2 diabetes mellitus: a new look on old disease. Endocrine. 1999;10:1–6. doi: 10.1385/ENDO:10:1:1. [DOI] [PubMed] [Google Scholar]
- 24.Prentice A, Goldberg GR, Schoenmakers I. Vitamin D across the lifecycle: physiology and biomarkers. Am J Clin Nutr. 2008;88:500S–6S. doi: 10.1093/ajcn/88.2.500S. [DOI] [PubMed] [Google Scholar]
- 25.Tylavsky FA, Ryder KA, Lyytikainen A, et al. Vitamin D, parathyroid hormone, and bone mass in adolescents. J Nutr. 2005;135:2735S–8S. doi: 10.1093/jn/135.11.2735S. [DOI] [PubMed] [Google Scholar]
- 26.Aksnes L, Aarskog D. Plasma concentrations of vitamin D metabolites in puberty: effect of sexual maturation and implications for growth. J Clin Endocrinol Metab. 1982;55:94–101. doi: 10.1210/jcem-55-1-94. [DOI] [PubMed] [Google Scholar]
- 27.Krabbe S, Christiansen C, Hummer L. Serum vitamin D metabolites are not related to growth rate, bone mineral content, or serum alkaline phosphatase in male puberty. Calcif Tissue Int. 1986;38:127–9. doi: 10.1007/BF02556871. [DOI] [PubMed] [Google Scholar]
- 28.Lawlor DA, Harbord RM, Sterne JA, et al. Mendelian randomization: using genes as instruments for making causal inferences in epidemiology. Stat Med. 2008;27:1133–63. doi: 10.1002/sim.3034. [DOI] [PubMed] [Google Scholar]
- 29.Wang TJ, Zhang F, Richards JB, et al. Common genetic determinants of vitamin D insufficiency: a genome-wide association study. Lancet. 2010;376:180–8. doi: 10.1016/S0140-6736(10)60588-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.