Abstract
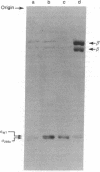
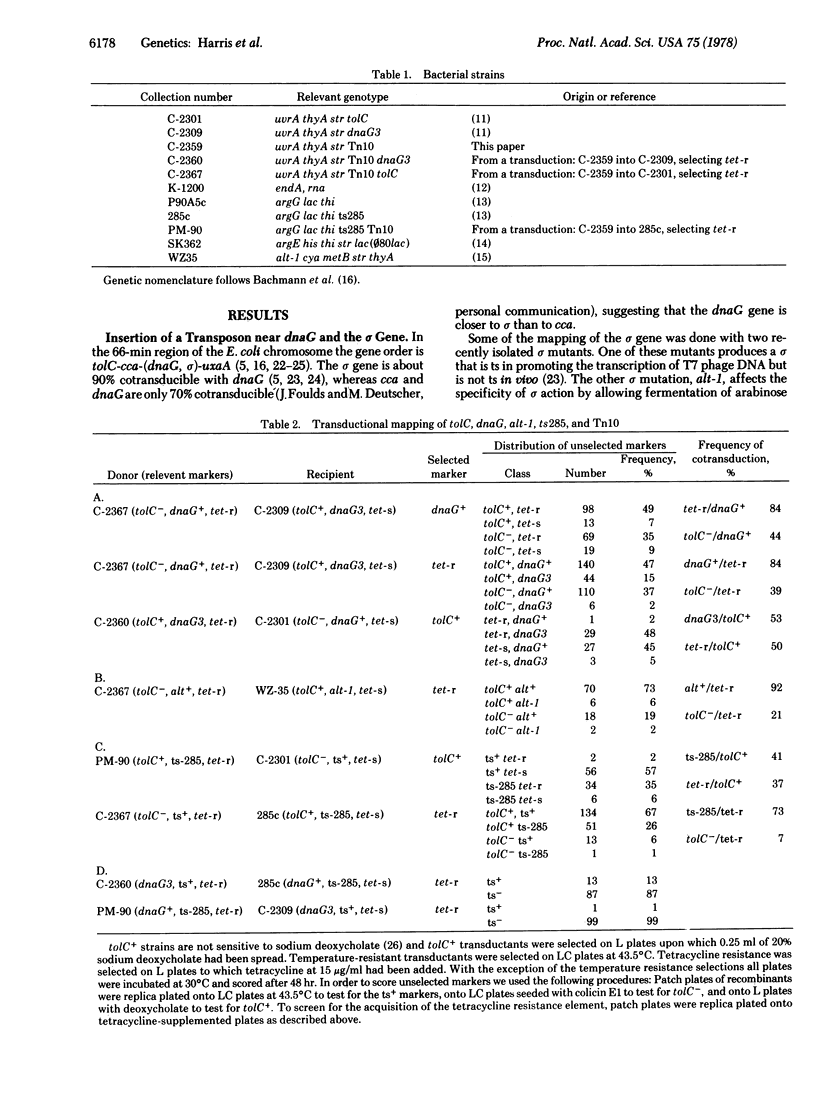
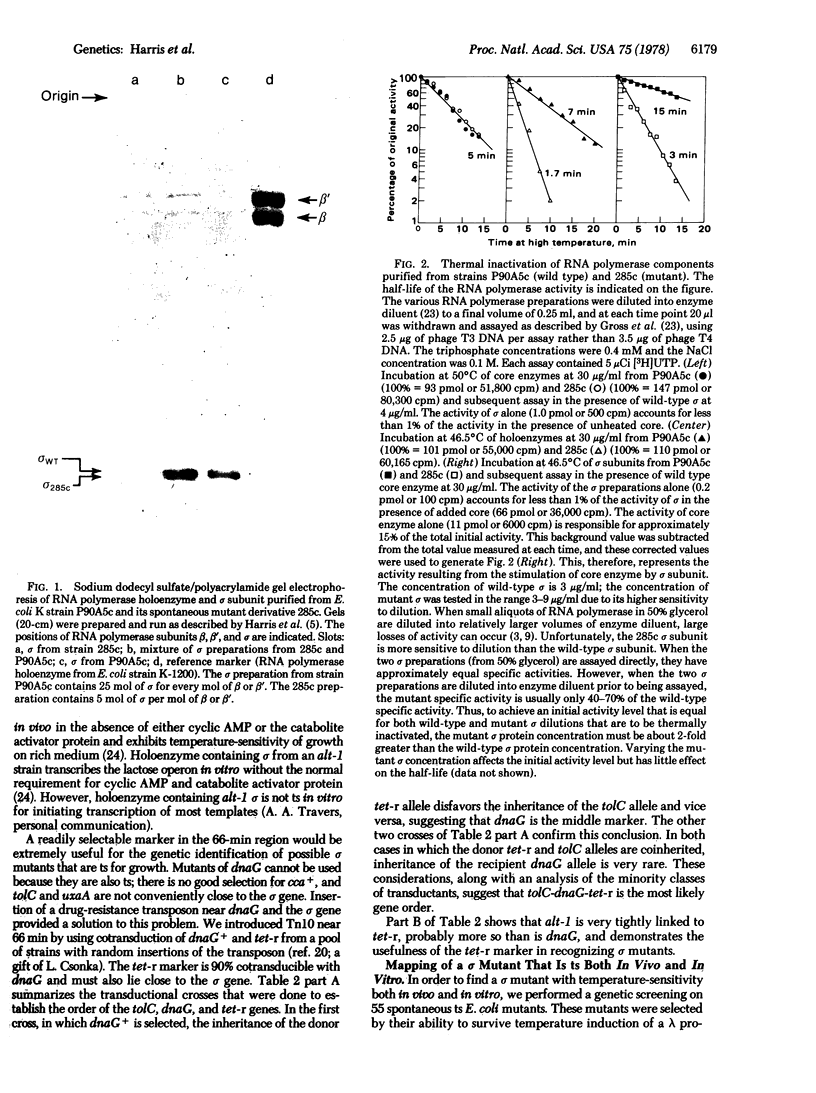
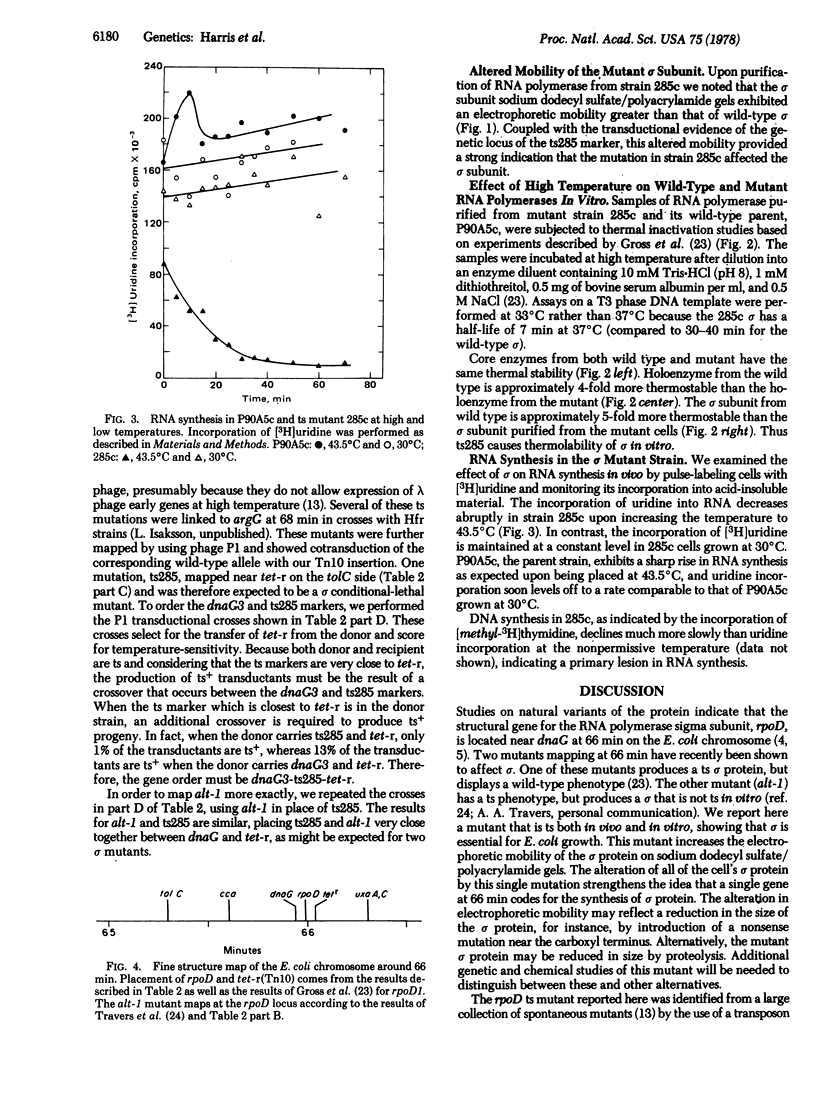
A gene affecting the sigma subunit of DNA-dependent RNA polymerase is tightly linked to dnaG at 66 min on the Escherichia coli chromosome. In order to create an easily selectable marker in this region, we inserted transposon-10, which carries a gene determining resistance to tetracycline (tet) near 66 min, and the order tolC-dnaG-sigma-tet was determined. We used frequency of contransduction with tet as a criterion to screen a collection of spontaneous temperature-sensitive Escherichia coli mutants that might affect the sigma subunit. One such mutant was found to map at the sigma locus. The sigma subunit isolated from this mutant is unstable at 46 degrees C in vitro and has an altered electrophoretic mobility. The temperature sensitivity of RNA synthesis in this mutant indicates that most transcription in E. coli is sigma dependent.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BERTANI G. Studies on lysogenesis. I. The mode of phage liberation by lysogenic Escherichia coli. J Bacteriol. 1951 Sep;62(3):293–300. doi: 10.1128/jb.62.3.293-300.1951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BERTANI G., WEIGLE J. J. Host controlled variation in bacterial viruses. J Bacteriol. 1953 Feb;65(2):113–121. doi: 10.1128/jb.65.2.113-121.1953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bachmann B. J., Low K. B., Taylor A. L. Recalibrated linkage map of Escherichia coli K-12. Bacteriol Rev. 1976 Mar;40(1):116–167. doi: 10.1128/br.40.1.116-167.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowden D. W., Twersky R. S., Calendar R. Escherichia coli deoxyribonucleic acid synthesis mutants: their effect upon bacteriophage P2 and satellite bacteriophage P4 deoxyribonucleic acid synthesis. J Bacteriol. 1975 Oct;124(1):167–175. doi: 10.1128/jb.124.1.167-175.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burgess R. R., Jendrisak J. J. A procedure for the rapid, large-scall purification of Escherichia coli DNA-dependent RNA polymerase involving Polymin P precipitation and DNA-cellulose chromatography. Biochemistry. 1975 Oct 21;14(21):4634–4638. doi: 10.1021/bi00692a011. [DOI] [PubMed] [Google Scholar]
- Burgess R. R., Travers A. A., Dunn J. J., Bautz E. K. Factor stimulating transcription by RNA polymerase. Nature. 1969 Jan 4;221(5175):43–46. doi: 10.1038/221043a0. [DOI] [PubMed] [Google Scholar]
- Calendar R., Lindahl G. Attachment of prophage P2: gene order at different host chromosomal sites. Virology. 1969 Dec;39(4):867–881. doi: 10.1016/0042-6822(69)90023-3. [DOI] [PubMed] [Google Scholar]
- Chamberlin M. J. The selectivity of transcription. Annu Rev Biochem. 1974;43(0):721–775. doi: 10.1146/annurev.bi.43.070174.003445. [DOI] [PubMed] [Google Scholar]
- Chen P. L., Carl P. L. Genetic map location of the Escherichia coli dnaG gene. J Bacteriol. 1975 Dec;124(3):1613–1614. doi: 10.1128/jb.124.3.1613-1614.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dürwald H., Hoffmann-Berling H. Endonuclease-I-deficient and ribonuclease I-deficient Escherichia coli mutants. J Mol Biol. 1968 Jul 14;34(2):331–346. doi: 10.1016/0022-2836(68)90257-x. [DOI] [PubMed] [Google Scholar]
- Foulds J., Hilderman R. H., Deutscher M. P. Mapping of the locus for Escherichia coli transfer ribonucleic acid nucleotidyltransferase. J Bacteriol. 1974 May;118(2):628–632. doi: 10.1128/jb.118.2.628-632.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez N., Wiggs J., Chamberlin M. J. A simple procedure for resolution of Escherichia coli RNA polymerase holoenzyme from core polymerase. Arch Biochem Biophys. 1977 Aug;182(2):404–408. doi: 10.1016/0003-9861(77)90521-5. [DOI] [PubMed] [Google Scholar]
- Gross C., Hoffman J., Ward C., Hager D., Burdick G., Berger H., Burgess R. Mutation affecting thermostability of sigma subunit of Escherichia coli RNA polymerase lies near the dnaG locus at about 66 min on the E. coli genetic map. Proc Natl Acad Sci U S A. 1978 Jan;75(1):427–431. doi: 10.1073/pnas.75.1.427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris J. D., Martinez I. I., Calendar R. A gene from Escherichia coli affecting the sigma subunit of RNA polymerase. Proc Natl Acad Sci U S A. 1977 May;74(5):1836–1840. doi: 10.1073/pnas.74.5.1836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinkle D. C., Chamberlin M. J. Studies of the binding of Escherichia coli RNA polymerase to DNA. II. The kinetics of the binding reaction. J Mol Biol. 1972 Sep 28;70(2):187–195. doi: 10.1016/0022-2836(72)90532-3. [DOI] [PubMed] [Google Scholar]
- Isaksson L. A., Sköld S. E., Skjöldebrand J., Takata R. A procedure for isolation of spontaneous mutants with temperature sensitive of RNA and/or protein. Mol Gen Genet. 1977 Nov 18;156(3):233–237. doi: 10.1007/BF00267177. [DOI] [PubMed] [Google Scholar]
- Kleckner N., Barker D. F., Ross D. G., Botstein D. Properties of the translocatable tetracycline-resistance element Tn10 in Escherichia coli and bacteriophage lambda. Genetics. 1978 Nov;90(3):427–461. doi: 10.1093/genetics/90.3.427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleckner N., Roth J., Botstein D. Genetic engineering in vivo using translocatable drug-resistance elements. New methods in bacterial genetics. J Mol Biol. 1977 Oct 15;116(1):125–159. doi: 10.1016/0022-2836(77)90123-1. [DOI] [PubMed] [Google Scholar]
- Lindahl L., Jaskunas S. R., Dennis P. P., Nomura M. Cluster of genes in Escherichia coli for ribosomal proteins, ribosomal RNA, and RNA polymerase subunits. Proc Natl Acad Sci U S A. 1975 Jul;72(7):2743–2747. doi: 10.1073/pnas.72.7.2743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindqvist B. H., Six E. W. Replication of bacteriophage P4 DNA in a nonlysogenic host. Virology. 1971 Jan;43(1):1–7. doi: 10.1016/0042-6822(71)90218-2. [DOI] [PubMed] [Google Scholar]
- Nakamura Y., Osawa T., Yura T. Chromosomal location of a structural gene for the RNA polymerase sigma factor in Escherichia coli. Proc Natl Acad Sci U S A. 1977 May;74(5):1831–1835. doi: 10.1073/pnas.74.5.1831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura Y. RNA polymerase mutant with altered sigma factor in Escherichia coli. Mol Gen Genet. 1978 Sep 20;165(1):1–6. doi: 10.1007/BF00270369. [DOI] [PubMed] [Google Scholar]
- Rabussay D., Geiduschek E. P. Phage T4-modified RNA polymerase transcribes T4 late genes in vitro. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5305–5309. doi: 10.1073/pnas.74.12.5305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silverstone A. E., Goman M., Scaife J. G. ALT: a new factor involved in the synthesis of RNA by Escherichia coli. Mol Gen Genet. 1972;118(3):223–234. doi: 10.1007/BF00333459. [DOI] [PubMed] [Google Scholar]
- Talkington C., Pero J. Promoter recognition by phage SP01-modified RNA polymerase. Proc Natl Acad Sci U S A. 1978 Mar;75(3):1185–1189. doi: 10.1073/pnas.75.3.1185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Travers A. A., Buckland R., Goman M., Le Grice S. S., Scaife J. G. A mutation affecting the sigma subunit of RNA polymerase changes transcriptional specificity. Nature. 1978 Jun 1;273(5661):354–358. doi: 10.1038/273354a0. [DOI] [PubMed] [Google Scholar]
- Wall J. D., Harriman P. D. Phage P1 mutants with altered transducing abilities for Escherichia coli. Virology. 1974 Jun;59(2):532–544. doi: 10.1016/0042-6822(74)90463-2. [DOI] [PubMed] [Google Scholar]
- Whitney E. N. The tolC locus in Escherichia coli K12. Genetics. 1971 Jan;67(1):39–53. doi: 10.1093/genetics/67.1.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolf B. The characteristics and genetic map location of a temperature sensitive DNA mutant of E. coli K12. Genetics. 1972 Dec;72(4):569–593. doi: 10.1093/genetics/72.4.569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zieg J., Kushner S. R. Analysis of genetic recombination between two partially deleted lactose operons of Escherichia coli K-12. J Bacteriol. 1977 Jul;131(1):123–132. doi: 10.1128/jb.131.1.123-132.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]