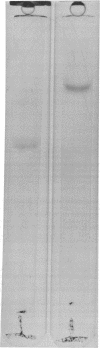
Abstract
Choleragen and its A protomer catalyzed the hydrolysis of NAD to ADP-ribose and nicotinamide. NADase activity was inhibited by gangliosides GM1 (galactosyl-N-acetylgalactosaminyl-[N-acetylneuraminyl]-galactosylglucosylceramide), GM2 (N-acetylgalactosaminyl-[N-acetylneuraminyl]-galactosylglucosylceramide), GM3 (N-acetylneuraminyl-galactosylglucosylceramide), and GD1a (N-acetylneuraminylgalactosyl-N-acetylgalactosaminyl-E1N-acetylneuraminyl]-galactosylglucosylceramide). These gangliosides also increased the intensity of the tryptophanyl fluorescence of the isolated A protomer (lambda max = 328 nm). GM1 but not GM2, GM3, and GD1a caused a "blue shift" in the fluorescence spectrum of the B protomer. These results are consistent with other evidence that the specificity of GM1 as the choleragen receptor resides in its carbohydrate moiety. The NADase activity of choleragen was similar to that of diphtheria toxin previously described [J. Kandel, R. J. Collier & D. W. Chung (1974) J. Biol. Chem. 249, 2088-2097]. As with diphtheria toxin, analogues of NAD were inhibitory, adenine being the most effective. Significant inhibition was also noted with adenosine, AMP, ADP-ribose, nicotinamide, nicotinamide mononucleotide, and NADP. NADP was hydrolyzed only slowly by choleragen. In the NADase reaction catalyzed by diphtheria toxin, water serves as an acceptor for the ADP-ribose moiety of NAD in lieu of the natural acceptor molecule, which is elongation factor II (Kandel et al., 1974). It seems probable that the natural protein acceptor for ADP-ribose in the reaction catalyzed by choleragen is adenylate cyclase or a protein component of a cyclase complex that regulates enzymatic activity.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bennett V., Craig S., Hollenberg M. D., O'Keefe E., Sahyoun N., Cuatrecasas P. Structure and function of cholera toxin and hormone receptors. J Supramol Struct. 1976;4(1):99–120. doi: 10.1002/jss.400040110. [DOI] [PubMed] [Google Scholar]
- Cuatrecasas P. Cholera toxin-fat cell interaction and the mechanism of activation of the lipolytic response. Biochemistry. 1973 Aug 28;12(18):3567–3577. doi: 10.1021/bi00742a033. [DOI] [PubMed] [Google Scholar]
- Cuatrecasas P. Gangliosides and membrane receptors for cholera toxin. Biochemistry. 1973 Aug 28;12(18):3558–3566. doi: 10.1021/bi00742a032. [DOI] [PubMed] [Google Scholar]
- Cuatrecasas P. Interaction of Vibrio cholerae enterotoxin with cell membranes. Biochemistry. 1973 Aug 28;12(18):3547–3558. doi: 10.1021/bi00742a031. [DOI] [PubMed] [Google Scholar]
- Finkelstein R. A., Boesman M., Neoh S. H., LaRue M. K., Delaney R. Dissociation and recombination of the subunits of the cholera enterotoxin (choleragen). J Immunol. 1974 Jul;113(1):145–150. [PubMed] [Google Scholar]
- Fishman P. H., McFarland V. W., Mora P. T., Brady R. O. Ganglioside biosynthesis in mouse cells: glycosyltransferase activities in normal and virally-transformed lines. Biochem Biophys Res Commun. 1972 Jul 11;48(1):48–57. doi: 10.1016/0006-291x(72)90342-7. [DOI] [PubMed] [Google Scholar]
- Fishman P. H., Moss J., Vaughan M. Uptake and metabolism of gangliosides in transformed mouse fibroblasts. Relationship of ganglioside structure to choleragen response. J Biol Chem. 1976 Aug 10;251(15):4490–4494. [PubMed] [Google Scholar]
- Gill D. M. Involvement of nicotinamide adenine dinucleotide in the action of cholera toxin in vitro. Proc Natl Acad Sci U S A. 1975 Jun;72(6):2064–2068. doi: 10.1073/pnas.72.6.2064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gill D. M., King C. A. The mechanism of action of cholera toxin in pigeon erythrocyte lysates. J Biol Chem. 1975 Aug 25;250(16):6424–6432. [PubMed] [Google Scholar]
- Heyningen S Van Cholera toxin: interaction of subunits with ganglioside GM1. Science. 1974 Feb 15;183(4125):656–657. doi: 10.1126/science.183.4125.656. [DOI] [PubMed] [Google Scholar]
- Holmgren J., Lönnroth I., Månsson J., Svennerholm L. Interaction of cholera toxin and membrane GM1 ganglioside of small intestine. Proc Natl Acad Sci U S A. 1975 Jul;72(7):2520–2524. doi: 10.1073/pnas.72.7.2520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holmgren J., Lönnroth I., Svennerholm L. Tissue receptor for cholera exotoxin: postulated structure from studies with GM1 ganglioside and related glycolipids. Infect Immun. 1973 Aug;8(2):208–214. doi: 10.1128/iai.8.2.208-214.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Honjo T., Nishizuka Y., Hayaishi O. Diphtheria toxin-dependent adenosine diphosphate ribosylation of aminoacyl transferase II and inhibition of protein synthesis. J Biol Chem. 1968 Jun 25;243(12):3553–3555. [PubMed] [Google Scholar]
- Kandel J., Collier R. J., Chung D. W. Interaction of fragment A from diphtheria toxin with nicotinamide adenine dinucleotide. J Biol Chem. 1974 Apr 10;249(7):2088–2097. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Moss J., Fishman P. H., Manganiello V. C., Vaughan M., Brady R. O. Functional incorporation of ganglioside into intact cells: induction of choleragen responsiveness. Proc Natl Acad Sci U S A. 1976 Apr;73(4):1034–1037. doi: 10.1073/pnas.73.4.1034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moss J., Manganiello V. C., Vaughan M. Hydrolysis of nicotinamide adenine dinucleotide by choleragen and its A protomer: possible role in the activation of adenylate cyclase. Proc Natl Acad Sci U S A. 1976 Dec;73(12):4424–4427. doi: 10.1073/pnas.73.12.4424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mullin B. R., Aloj S. M., Fishman P. H., Lee G., Kohn L. D., Brady R. O. Cholera toxin interactions with thyrotropin receptors on thyroid plasma membranes. Proc Natl Acad Sci U S A. 1976 May;73(5):1679–1683. doi: 10.1073/pnas.73.5.1679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Penick R. J., Meisler M. H., McCluer R. H. Thin-layer chromatographic studies of human brain gangliosides. Biochim Biophys Acta. 1966 Apr 4;116(2):279–287. doi: 10.1016/0005-2760(66)90010-5. [DOI] [PubMed] [Google Scholar]
- Sahyoun N., Cuatrecasas P. Mechanism of activation of adenylate cyclase by cholera toxin. Proc Natl Acad Sci U S A. 1975 Sep;72(9):3438–3442. doi: 10.1073/pnas.72.9.3438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Staerk J., Ronneberger J., Wiegandt H., Ziegler W. Interaction of ganglioside G Gtet1 and its derivatives with choleragen. Eur J Biochem. 1974 Oct 1;48(1):103–110. doi: 10.1111/j.1432-1033.1974.tb03747.x. [DOI] [PubMed] [Google Scholar]
- Van Heyningen S., King C. A. Short communications. Subunit A from cholera toxin is an activator of adenylate cyclase in pigeon erythrocytes. Biochem J. 1975 Jan;146(1):269–271. doi: 10.1042/bj1460269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Heyningen W. E., Carpenter C. C., Pierce N. F., Greenough W. B., 3rd Deactivation of cholera toxin by ganglioside. J Infect Dis. 1971 Oct;124(4):415–418. doi: 10.1093/infdis/124.4.415. [DOI] [PubMed] [Google Scholar]
- Yohe H. C., Rosenberg A. Interaction of triiodide anion with gangliosides in aqueous iodine. Chem Phys Lipids. 1972 Dec;9(4):279–294. doi: 10.1016/0009-3084(72)90015-1. [DOI] [PubMed] [Google Scholar]