Abstract
This study sought to evaluate the effects of a single session of exercise on the expression of Hsp70, of c-jun N-terminal kinase (JNK), and insulin receptor substrate 1 serine 612 (IRSser612) phosphorylation in the skeletal muscle of obese and obese insulin-resistant patients. Twenty-seven volunteers were divided into three experimental groups (eutrophic insulin-sensitive, obese insulin-sensitive, and obese insulin-resistant) according to their body mass index and the presence of insulin resistance. The volunteers performed 60 min of aerobic exercise on a cycle ergometer at 60 % of peak oxygen consumption. M. vastus lateralis samples were obtained before and after exercise. The protein expressions were evaluated by Western blot. Our findings show that compared with paired eutrophic controls, obese subjects have higher basal levels of p-JNK (100 ± 23 % vs. 227 ± 67 %, p = 0.03) and p-IRS-1ser612 (100 ± 23 % vs. 340 ± 67 %, p < 0.001) and reduced HSP70 (100 ± 16 % vs. 63 ± 12 %, p < 0.001). The presence of insulin resistance results in a further increase in p-JNK (460 ± 107 %, p < 0.001) and a decrease in Hsp70 (46 ± 5 %, p = 0.006), but p-IRS-1ser612 levels did not differ from obese subjects (312 ± 73 %, p > 0.05). Exercise reduced p-JNK in obese insulin-resistant subjects (328 ± 33 %, p = 0.001), but not in controls or obese subjects. Furthermore, exercise reduced p-IRS-1ser612 for both obese (122 ± 44 %) and obese insulin-resistant (185 ± 36 %) subjects. A main effect of exercise was observed in HSP70 (p = 0.007). We demonstrated that a single session of exercise promotes changes that characterize a reduction in cellular stress that may contribute to exercise-induced increase in insulin sensitivity.
Keywords: Exercise, Insulin resistance, Obesity, Heat shock protein, Cellular stress
Introduction
According to the World Health Organization, obesity (defined as body mass index (BMI) greater than 30 kg/m2) is a worldwide epidemic (WHO 2013). In 2008, 1.4 billion adults were overweight and more than half a billion were obese. The worldwide projections for 2015 are approximately 2.3 billion overweight and 700 million obese adults (WHO 2011). High-income countries are not the only ones affected by the obesity epidemic, because the condition is also prevalent in low- and middle-income countries (Finucane et al. 2011). In Brazil, for example, approximately 50 and 16 % of the adult population is overweight and obese, respectively (Brazilian Ministry of Health 2011).
Obesity is associated with an increased risk for the development of numerous metabolic complications, including insulin resistance (Shafrir 1996), glucose intolerance (Sinha et al. 2002) and type 2 diabetes (Colditz et al. 1990; Sasai et al. 2010). Long-term insulin resistance leads to an increase in the activity of pancreatic β cells to compensate for the low insulin sensitivity and increased glucose intolerance, which can lead to β cell dysfunction and, clinically, to type 2 diabetes. Specifically, skeletal muscle insulin resistance plays a causal role in the development of clinical insulin resistance and, consequently, type 2 diabetes (Hotamisligil 2010).
The involvement of stress kinases in the development of insulin resistance in skeletal muscle has been suggested. It has been proposed that the activation of mitogen-activated protein kinases, such as c-jun N-terminal kinase (JNK), can disrupt the insulin signaling pathway through the phosphorylation of serine residues in the insulin receptor substrate (IRS) (Aguirre et al. 2000; Lee et al. 2003). In obesity, the mechanisms involved in the increase of JNK phosphorylation seem to be related to the state of chronic low-grade inflammation associated with excess adipose tissue and greater expression of inflammatory cytokines, such as TNF-α, which are known to cause JNK phosphorylation (Davis 2000; Uysal et al. 1997). Chung et al. (2008) reported that JNK phosphorylation in the skeletal muscle of obese insulin-resistant individuals is higher than in paired healthy subjects. Indeed, the study of Hirosumi et al. (2002) emphasized the importance of JNK in the development of insulin resistance by showing that JNK knockout animals (JNK−/−) did not develop obesity-induced insulin resistance. Together, these studies clearly show the involvement of JNK phosphorylation in obesity and the development of insulin resistance.
Recently, some studies have shown an inhibitory activity of the heat shock protein 70 (HSP70) on JNK, and, consequently, insulin resistance. Chung et al. (2008) showed that heat shock therapy, transgenic muscle overexpression, and pharmacologic means to overexpress HSP70 in mice, either specifically in skeletal muscle or systemically, protected the animals against diet- or obesity-induced hyperglycemia, hyperinsulinemia, glucose intolerance, and insulin resistance. The authors provided evidence that HSP70 induction is highly associated with the prevention of JNK phosphorylation by identifying an essential role for HSP70 in blocking inflammation and preventing insulin resistance in the context of genetic obesity or high-fat feeding in animals.
Exercise has been widely recommended for the prevention and treatment of obesity and other related metabolic diseases. Although it has been well established that exercise is effective for the improvement of insulin sensitivity (Garetto et al. 1984; Richter et al. 1989), the mechanisms are not fully understood. It is possible that some of the benefits of exercise for obese insulin-resistant subjects could be mediated in part by alterations in JNK and IRS serine phosphorylation. In this context, Ropelle et al. (2006) demonstrated in rats that a single bout of exercise inhibits high-fat diet-induced insulin resistance in muscle with a parallel reduction in JNK activity; however, the HSP70 expression induction was not evaluated.
Because HSP70 can interfere in intracellular signaling (such as JNK phosphorylation) and act as an anti-inflammatory effector (Park et al. 2001), it is possible that this protein participates in the improvement of insulin sensitivity induced by exercise. Considering this evidence, this study sought to evaluate the effects of a single session of exercise on the expression of HSP70, activation of JNK and IRSser612 phosphorylation in the skeletal muscle of obese and obese insulin-resistant patients. We hypothesize that a single moderate intensity aerobic session of exercise will increase HSP70 and reduce activation of JNK and IRSser612 phosphorylation in the skeletal muscle of obese insulin-resistant patients.
Methods
Subjects
Twenty-seven non-smoking subjects (9 men and 18 women) recruited from the surrounding community were studied. All the subjects met the following inclusion criteria: age between 18 and 55 years, not being engaged in a regular exercise program, stable body weight (±2 kg) for at least 3 months prior to the study, no participation in a body weight reduction program for at least 6 months before the study, and no use of anti-inflammatory medications or other hypoglycemic agents known to affect metabolism. In addition, the subjects had not been diagnosed with type 2 diabetes mellitus (confirmed by the oral glucose tolerance test) or any acute or chronic diseases (self-reported). Before agreeing to participate in the study, the subjects gave their written informed consent, which was approved by the institutional review board of the local institution, and underwent medical screening.
Initially, each subject had the waist circumference measured (midway between the lower rib margin and the iliac crest) and the BMI calculated. The percentage of body fat was estimated using a tetrapolar bioimpedance apparatus (Biodynamics®, 450, USA). Total cholesterol, its fractions, and triglycerides were determined in serum after 12 h of fasting.
After the initial screening, the subjects were allocated to one of three groups according to the presence of obesity (BMI ≥ 30 kg/m2) and insulin resistance (based on the homeostatic model assessment 1 of insulin resistance (HOMA1-IR)) (Matthews et al. 1985). The cutoff adopted for the HOMA-IR index was based on the study of Geloneze et al. (2006). Each experimental group consisted of nine paired subjects (six women and three men): obese (OB) BMI ≥ 30 kg/m2 and HOMA1-IR < 2.71, obese insulin-resistant (OBR) BMI ≥ 30 kg/m2 and HOMA1-IR ≥ 2.71, and the control group (CTRL) BMI ≥ 18.5 and ≤ 24.9 kg/m2 and HOMA1-IR < 2.71.
Maximum exercise test
The subjects had a maximum exercise test conducted on an electronically braked cycle ergometer (Corival, Medgraphics, USA) using a ramp protocol (Myers and Bellin 2000). The power was increased every 60 s at an individual rate, which was based on the subject's exercise history questionnaire (Veterans Specific Activity Questionnaire), to induce fatigue within 8 to 12 min. Electrocardiography was conducted by a medical cardiologist, and no symptoms of coronary heart disease were observed during the exercise stress test. Expired air was analyzed breath by breath during the test using a portable metabolic cart (K4b2, Cosmed, Italy), calibrated as recommended by the manufacturer. Heart rate (HR) was recorded every minute using a HR transmitter strap (S810i series TM, Polar, USA). The subjects had to achieve at least three of the criteria proposed by Poole et al. (2008) for the determination of the peak oxygen consumption (VO2peak).
Muscle biopsy
Two muscle biopsies were performed in the m. vastus lateralis of the dominant leg using the Bergström technique (Bergström and Hultman 1966) under local anesthesia (2 % lidocaine with epinephrine). Samples were harvested by suction and frozen in liquid nitrogen until analysis. The first biopsy was performed in the morning at least 7 days prior to the exercise session, between 2 to 3 h after the standardized breakfast (241 kcal of total energy intake, 59 % carbohydrate, 18 % protein, and 23 % fat). The second biopsy was performed 30 min after the end of the exercise session (described below).
Aerobic exercise session
Before the single exercise session, the subjects were asked to refrain from vigorous exercise, caffeine, and alcoholic beverages for at least 24 h. The subjects ingested a standardized breakfast, described above, 2 h before the start of exercise. Subjects were considered hydrated (urine specific gravity (USG) lower than 1.030 (mean of 1.016 ± 0.008, ranging from 1.002 to 1.030). Then, the subjects rested for 30 min in the supine position.
The exercise session consisted of a 5-min warm-up cycling at 20 % of the individual peak power achieved in the maximum exercise test. The workload was then increased to 50 % of the peak power for 60 min (three sets of 20 min separated by 5 min intervals of passive rest). This intensity was chosen to require a metabolic rate of 60 % of the subject's peak oxygen consumption (VO2peak), which was confirmed during each trial by measuring oxygen consumption in the first 10 min of the second set employing open-circuit spirometry, as described above. The subjects exercised for another 3 min to cool down. Water was provided ad libitum during the passive rest intervals, and subjects were hydrated after the exercise period (mean USG of 1.011 ± 0.006, ranging from 1.000 to 1.022). HR was recorded every 5 min. While performing the exercise session, mean values of room temperature (p = 0.17) and relative air humidity (p = 0.4) were similar for the three groups (OBR 23.8 ± 0.64 °C and 70 ± 6 %, OB 23.4 ± 0.9 °C and 73 ± 9 %, and CTRL 22.8 ± 0.9 °C and 76 ± 9 %) respectively.
Determination of protein expression and phosphorylation in skeletal muscle
Muscle samples (approximately 50 mg) were lysed by maceration in ice in 100 μL of lysis buffer (1 % Triton X100; 100 mM Tris pH 8.0, 20 % glycerol, 0.2 mM EDTA) containing protease inhibitor cocktail (Complete Protease Inhibitor Cocktail, Roche, Germany) and phosphatase inhibitor cocktail (Phosphatase Inhibitor Cocktail PhosStop, Roche, Germany). The lysates were centrifuged at 2700×g for 20 min at 4 °C, and the supernatant containing the cytosolic protein was collected. Protein concentration in the extracts was determined by the Bradford method (Bradford 1976) using bovine serum albumin (BSA; 1 mg/ml) as standard. Approximately 50–150 μg of total protein was fractionated on polyacrylamide gel (8–12 %) containing sodium dodecyl sulfate (20 %) (SDS-PAGE). The samples were distributed in the gel where CTRL, OB, and OBR were matched by gender and age. The electrophoresis was performed at 90 V for about 18 h at a temperature between 4 and 8 °C.
Proteins were transferred to previously activated (10 s in 100 % methanol) polyvinylidene fluoride membranes (Millipore, EUA), wetted in transfer buffer (25 mMTris, 192 mM glycine, 20 % methanol). Transference was performed at constant amperage of 350 mA for 2 h at 4 °C. Transference efficiency was verified by Ponceau S staining (0.5 %) in every membrane. The membranes were then blocked with 5 % BSA in TBS-Tween buffer (150 mM NaCl, 10 mM Tris, pH 7.6, and 0.1 % Tween-20) for 1 h at room temperature under gentle agitation. The membranes were incubated with primary antibody (1:1,000, 5 % BSA in TBS-Tween buffer) (Hsp70 (Cell Signaling, #4872) SAPK/JNK (Cell Signaling, #9252), phospho-SAPK/JNK (Thr183/Tyr185, Cell Signaling, # 9251), IRS-1 (59G8, Cell Signaling, #2390), phospho-IRS-1(Ser612, Cell Signaling, #3203) (C15H5), GAPDH (14C10) (Cell Signaling, # 2118)) at 4 °C, under gentle agitation for approximately 18 h. The membranes were washed in TBS-Tween, three times for 5 min, and incubated for 120 min at room temperature with peroxidase-conjugated secondary antibody (1:2,000, Cell Signaling). After washing three times for 5 min with TBS-Tween, the membranes were incubated in enhanced chemiluminescence solution for 15 min, protected from light. The membranes were then exposed to X-ray film (Hyperfilm, Amersham, USA) that was developed using developer and fixer (RP X-Omat, Kodak, USA) according to the manufacturer's recommendations. The film was scanned, and the bands were analyzed using Scion Image software (Scion Corporation based on NIH Image; National Institutes of Health, Scion Corporation, Frederick, MD, USA).
Statistical analysis
The software Statistica (v8.0, StatSoft, Inc.) was used for statistical analysis. Sample size was estimated based on the study of O'Leary et al. (2006), using the fasting plasma insulin concentration as the reference variable. The Shapiro–Wilk test was used to assess the normality of the data. One-way analysis of variance (one-way ANOVA) was performed to compare the characterization data among groups. We used a two-way repeated measure ANOVA to evaluate the effect of exercise on the parameters studied in the different groups, followed by Tukey's post hoc test when a significant F value was observed. We used the Pearson test to evaluate the correlation between muscle Hsp70 expression and JNK phosphorylation, and JNK phosphorylation and plasma insulin concentration. A significance level of p ≤ 0.05 was used. Data are presented as means ± SEM.
Results
Subject characteristics
Subject characteristics are presented in Table 1. Age (p = 0.91), height (p = 0.72), maximal power output (p = 0.60), and VO2peak (p = 0.46) were not different among groups. As planned, the OBR and OB had a higher body weight (p < 0.001), BMI (p < 0.001), body fat percentage (p < 0.001), and waist circumference (p < 0.001) compared to CTRL, but they were not different when compared to each other (p > 0.05). There were no differences among the three groups with respect to plasma glucose (p = 0.14), total serum cholesterol (p = 0.13), and serum HDL-C (p = 0.32), and LDL-C (p = 0.44). Higher serum concentrations of triglycerides (p = 0.03), VLDL-C (p = 0.02), and insulin (p < 0.001) were observed in OBR, as well as a higher HOMA1-IR (p < 0.001), compared to the other groups (Table 1).
Table 1.
Clinical and metabolic characteristics and exercise testing of subjects
| Variables | CTRL (n = 8) | OB (n = 9) | OBR (n = 9) | P value |
|---|---|---|---|---|
| Gender (male/female) | 3/6 | 3/6 | 3/6 | – |
| Age (years) | 37 ± 8 | 38 ± 9 | 40 ± 8 | 0.91 |
| Body weight (kg) | 62.5 ± 13.2 | 88.3 ± 13.6a | 96.3 ± 16.6a | <0.001 |
| Height (cm) | 166.0 ± 12.0 | 162.0 ± 12.0 | 166.0 ± 12.0 | 0.72 |
| BMI (kg/m2) | 22.5 ± 2.4 | 33.6 ± 2.9a | 34.8 ± 2.7a | <0.001 |
| Body fat (%) | 23.3 ± 3.9 | 34.2 ± 7.3a | 35.5 ± 5.1a | <0.001 |
| Waist circumference (cm) | 73.0 ± 8.0 | 95 ± 6.0a | 104.1 ± 10.9a | <0.001 |
| VO2peak (mL min−1 kg−1) | 27.5 ± 5.3 | 24.3 ± 5.0 | 23.1 ± 4.0 | 0.46 |
| Peak power (watts) | 133 ± 66 | 151 ± 75 | 151 ± 56 | 0.60 |
| Fasting insulin (μU/mL) | 5.0 ± 1.7 | 9.0 ± 2.0 | 20.2 ± 5.4b | <0.001 |
| Fasting glucose (mg/dL) | 80.0 ± 5.0 | 83.0 ± 8.0 | 83.0 ± 10.0 | 0.14 |
| HOMA1-IR ((mmol)(μU)/L2) | 1.0 ± 0.4 | 1.8 ± 0.5 | 4.1 ± 1.0b | <0.001 |
| Total cholesterol (mg/dL) | 176.0 ± 32.0 | 189.0 ± 32.0 | 217.0 ± 26.0 | 0.13 |
| LDL-C (mg/dL) | 110.0 ± 29.0 | 123.0 ± 34.0 | 135.0 ± 18.0 | 0.44 |
| HDL-C (mg/dL) | 51.0 ± 10.0 | 45.0 ± 8.0 | 46.0 ± 9.0 | 0.32 |
| VLDL-C (mg/dL) | 16.0 ± 6.0 | 20.9 ± 9.8 | 35.2 ± 16.0b | 0.02 |
| Triglycerides (mg/dL) | 79.0 ± 30.0 | 104.0 ± 49.0 | 176.0 ± 76.0b | 0.03 |
CTRL eutrophic insulin-sensitive, OB obese insulin-sensitive, OBR obese insulin-resistant, BMI body mass index, HOMA1-IR homeostatic model assessment 1 of insulin resistance, LDL-C low-density lipoprotein cholesterol, HDL-C high-density lipoprotein cholesterol, VLDL-C very low density lipoprotein cholesterol
aDifference between OB and OBR compared to CTRL
bDifference between OBR and CTRL and OB
JNK1/2 phosphorylation in skeletal muscle
JNK1/2 phosphorylation was higher in OB (p = 0.03) and OBR (p < 0.001) than in the CTRL group. OBR presented higher JNK1/2 phosphorylation than OB (p < 0.001). After the exercise session, a reduction in JNK1/2 phosphorylation was observed in OBR (p = 0.001). The exercise had no effect on JNK1/2 phosphorylation in CTRL (p = 0.93) and OB (p = 0.95) (Fig. 1a and b).
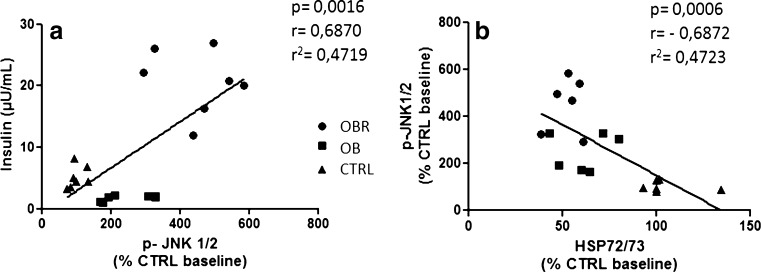
Fig. 1.
Expression and phosphorylation of skeletal muscle proteins before (baseline, white bars) and after (after exercise, black bars) a 1-h aerobic exercise session in eutrophic insulin-sensitive controls (CTRL), obese insulin-sensitive (OB), and obese insulin-resistant (OBR). Representative Western blot images of p-JNK and total JNK (a), p-IRS1ser612 and total IRS1 (c), and Hsp72/73 and GAPDH (e). Densitometric analysis of phosphorylated JNK to total JNK, n = 8 (b), IRS1, n = 5 (d), and Hsp72/73 to GAPDH, n = 8 (f). Data are reported as mean ± SEM. *p < 0.05 baseline vs after exercise; #p < 0.05 vs CTRL at the same time point; $p < 0.05 vs OB at the same time point
IRS-1 phosphorylation on serine 612 in skeletal muscle
Phosphorylation of IRS-1ser612 in both obese groups (OB and OBR) was higher than that of the CTRL (p < 0.001). There was no difference between OB and OBR in the p-IRS-1ser612 (p = 0.96). There was a decrease in p-IRS-1ser612 in the OB (p < 0.001) and OBR (p = 0.002) groups after the exercise session. There was no effect of the exercise session in the CTRL group (p = 0.99, Fig. 1c and d).
HSP70 (Hsp72/73) expression in skeletal muscle
The HSP70 expression at baseline and after the exercise session is presented in Fig. 1e and f. The OBR and OB groups had lower HSP70 expression than the CTRL group (p <0.001) at baseline. The OB had higher HSP70 expression than OBR (p = 0.006). There was a main effect of exercise on HSP70 expression after the exercise session (p = 0.007), but there was no interaction among the groups (p = 0.90). The GAPDH values did not change with exercise (p = 0.99), among or within the groups (p = 0.76) (CTRL 11,033±1,526 versus 10,836 ± 1,982 pixels, OB 11,401 ± 2,809 versus 10,411 ± 658 pixels, and OBR 10,875 ± 2,034 versus 10,431 ± 1,251 pixels, baseline and after exercise, respectively).
Correlation analyses
A positive correlation between basal JNK1/2 phosphorylation and fasting insulin concentration (r = 0.68, r2 = 0.47, p = 0.0006) was observed. We have also observed a negative correlation between JNK1/2 activation and Hsp70 expression (r = −0.68, r2 = 0.47, p = 0.0016) (Fig. 2a and b).
Fig. 2.
Correlation between the activation of JNK in the skeletal muscle and the fasting plasma insulin concentration (a) and between the activation of JNK and Hsp70 expression in the skeletal muscle (b)
Discussion
This study investigated the effects of a single session of aerobic exercise on molecular components that are directly involved in the development of obesity-induced insulin resistance. To our knowledge, this is the first study to investigate the response of a single session of exercise on the expression of HSP70, JNK, and IRS-1 serine residue phosphorylation in the skeletal muscle of OBR. Our findings show that at rest, OB individuals already have higher levels of p-JNK and p-IRS-1ser612 and reduced HSP70 expression in the skeletal muscle than paired CTRL. The presence of insulin resistance further increases JNK phosphorylation and decreases HSP70 expression, as shown in the OBR group. A single session of exercise reduced skeletal muscle JNK and p-IRS-1ser612 phosphorylation levels in OBR. A main effect of exercise on HSP70 expression was observed. Taken together, we showed that a single session of aerobic exercise is able to modulate intramuscular expression of molecules related to the insulin signaling pathway that are altered in OBR.
In the present study, we observed that activation of JNK in the skeletal muscle at rest was higher in OB than CTRL, with a further increase in OBR. A significant positive correlation between plasma insulin concentration and JNK phosphorylation in the skeletal muscle was also observed, a result that supports the participation of this stress kinase in the development of insulin resistance. Increased JNK phosphorylation in the skeletal muscle of obese insulin-resistant individuals has been reported in humans and animals. Hirosumi et al. (2002) reported that genetically and diet-induced obesity in animals is followed by a greater JNK1 activity in the skeletal muscle, adipose, and hepatic tissues. Moreover, Chung et al. (2008) also observed greater activation of JNK in the skeletal muscle of obese insulin-resistant subjects compared with healthy, eutrophic individuals. Therefore, our results confirm the ones found in the literature and expand them showing for the first time that human obesity per se increases JNK phosphorylation in the skeletal muscle, suggesting that obese subjects without any clinical signs of insulin resistance might already have altered skeletal muscle activation of stress kinases involved in the development of insulin resistance.
We showed for the first time that an aerobic exercise session reduced JNK phosphorylation in OBR, and this result is in accordance with previous observations in animals (Ropelle et al. 2006), which found that a single session of swimming exercise prevented high-fat diet-induced insulin resistance in parallel with reduced activation of JNK. Similarly, Schenk and Horowitz (2007) reported a session of exercise protected against insulin resistance induced by lipid infusion in healthy human subjects. The authors reported reduced activity of stress kinases evidenced by lower JNK and IKK/NFκB activation in the skeletal muscle. Therefore, our findings demonstrate that a single session of exercise is able to reduce a stress kinase casually involved in the development of insulin resistance.
A key component in the development of insulin resistance is the phosphorylation of serine residues of IRS-1 known to inhibit the insulin signaling pathway (Bandyopadhyay et al. 2005; Yi et al. 2007; De Fea and Roth, 1997; Geiger and Gupte 2011). In the present study, we observed that p-IRS-1ser612 in the skeletal muscle was higher in both obese groups, but the phosphorylation status was not modulated by the presence of insulin resistance. We speculate that OB might already have some impairment of intracellular insulin signaling, although insulin sensitivity, indirectly evaluated by the HOMA1-IR index, is still preserved. In addition, there are several other serine residues in the IRS-1 that may be potentially involved in the development of insulin resistance (Yi et al. 2007). After a single session of exercise, p-IRS-1ser612 decreased in both groups of obese subjects. Therefore, a single session of exercise might increase skeletal muscle insulin sensitivity and glucose uptake through a decrease in p-IRS-1ser612 and, thus, greater PI3-K-IRS-1 association (De Fea and Roth 1997).
Recent reports have identified an essential role for HSP70 in preventing insulin resistance in the context of genetic obesity or high-fat feeding (Chung et al. 2008). In the present study, HSP70 expression in the skeletal muscle of OB was lower than that of the CTRL and was even lower in OBR. Chung et al. (2008) had previously demonstrated that obese insulin-resistant subjects have lower muscle Hsp72 expression than eutrophic subjects. Additionally, Kurucz et al. (2002) reported a decreased expression of the Hsp72 mRNA in the skeletal muscle from patients with type 2 diabetes and that the Hsp72 mRNA correlated with glucose tolerance. However, to our knowledge, the present study is the first to demonstrate that OB already have lower skeletal muscle HSP70 than CTRL and that insulin resistance further reduces HSP70 expression in obese subjects. This finding suggests that a reduction in skeletal muscle HSP70 expression occurs before the insulin resistance in obesity and that insulin resistance can accentuate this reduction. In addition, an inverse relationship between HSP70 expression and JNK phosphorylation in the skeletal muscle was observed. It has been suggested that the reduction in HSP70 expression in insulin-resistant tissues may result from inhibition of the heat shock factor 1 (HSF1) (Geiger and Gupte 2011). Stress kinase hyperactivation, capable of serine phosphorylating HSF1, can repress the heat shock response in insulin-resistant tissues, thereby reducing HSP70 expression. Such kinases include glycogen synthase kinase 3, extracellular signal-regulated kinase and JNK (Kline and Morimoto 1997). Although the results of the present study do not provide direct support for this mechanism, we hypothesize that increased JNK phosphorylation might be involved in the reduction in HSP70 expression via inhibition of HSF1 in the obese subjects.
Previous studies have suggested that HSP70 induction interferes in JNK activity and results in an improvement in insulin sensitivity (Chung et al. 2008; Gupte et al. 2009). In the study by Chung et al. (2008), different means of increasing Hsp72 expression in the skeletal muscle protected mice from developing insulin resistance in response to a high-fat diet. In another study, Hsp72 induction, by means of heat exposure in rats receiving a high-fat diet, also improved glucose tolerance (Gupte et al. 2009). Using monocytes from obese subjects, Simar et al. (2012) observed that Hsp72 induction through heat exposure (2 h at 42 °C) was associated with decreased serine p-IRS-1ser312. In these studies, increased HSP70 expression was associated with a reduction in JNK and IKKβ activation. The Hsp70 overexpression might inhibit JNK phosphorylation by direct binding and abrogation of JNK phosphorylation by SAPK/ERK kinase 1 and MAP kinase kinase 7 (Park et al. 2001). HSP70 and JNK interaction might also impede JNK enzyme activity or inhibit JNK interaction with its substrate, c-Jun. Other potential mechanisms include HSP70 action on phosphatases that deactivate JNK such as MAP kinase phosphatase 1 (MKP1) and MKP3 or the dual leucine zipper-bearing kinase (Meriin et al. 1999; Daviau et al. 2006). This observation suggests a potential role of HSP70 in inhibiting signaling pathways related to insulin resistance induced by diet- or genetic-induced obesity.
Although the single session of exercise increased HSP70 expression, reduced JNK activity and p-IRS-1ser612 in the skeletal muscle, it cannot be assumed that these alterations promoted an increase in insulin sensitivity of tissues. However, we used an exercise protocol previously known to improve insulin sensitivity (Christ-Roberts et al. 2003). In addition, it is known that exercise can improve insulin-dependent and insulin-independent glucose uptake in the skeletal muscle, but other pathways, apart from those assessed in the present study, might also have a role in the improvement of insulin sensitivity.
In summary, we have demonstrated that a single session of exercise promotes changes that characterize reduction in stress (reduction on JNK activity, IRS-1 inhibition, and increase in HSP70 expression), which may contribute to physical exercise-induced increase in insulin sensitivity.
Acknowledgments
This work was supported by CAPES (PNPD-2455/2011), FAPEMIG (CDS APQ01621-10), and CNPq (477154/2011-5) grants. The authors wish to acknowledge Dr. Valmor Alberto Augusto Tricoli of the University of São Paulo for teaching us the muscle biopsy technique, and Dr. David Lee Nelson for reviewing the manuscript.
Conflict of interest
The authors have no conflict of interests.
References
- Aguirre V, Uchida T, Yenush L, Davis R, White MF. The c-Jun NH(2)-terminal kinase promotes insulin resistance during association with insulin receptor substrate-1 and phosphorylation of Ser(307) J Biol Chem. 2000;275:9047–9054. doi: 10.1074/jbc.275.12.9047. [DOI] [PubMed] [Google Scholar]
- Bandyopadhyay GK, Yu JG, Ofrecio J, Olefsky JM. Increased p85/55/50 expression and decreased phosphatidylinositol 3-kinase activity in insulin-resistant human skeletal muscle. Diabetes. 2005;54:2351–2359. doi: 10.2337/diabetes.54.8.2351. [DOI] [PubMed] [Google Scholar]
- Bergström J, Hultman E. Muscle glycogen synthesis after exercise: an enhancing factor localized to the muscle cells in man. Nature London. 1966;210:309–310. doi: 10.1038/210309a0. [DOI] [PubMed] [Google Scholar]
- Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Brazilian Ministry of Health. Vigitel Brazil 2010: surveillance of risk and protective factors for chronic diseases through telephone survey. Brasilia: Office of Strategic and Participative Management, Bureau of Health Surveillance, Ministry of Health, 2011. p. 37
- Christ-Roberts CY, Pratipanawatr T, Pratipanawatr W, Berria R, Belfort R, Mandarino LJ. Increased insulin receptor signaling and glycogen synthase activity contribute to the synergistic effect of exercise on insulin action. J Appl Physiol. 2003;95:2519–2529. doi: 10.1152/japplphysiol.00605.2003. [DOI] [PubMed] [Google Scholar]
- Chung J, Nguyen AK, Henstridge DC, Holmes AG, Chan MH, Mesa JL, Lancaster GI, Southgate RJ, Bruce CR, Duffy SJ, Horvath I, Mestril R, Watt MJ, Hooper PL, Kingwell BA, Vigh L, Hevener A, Febbraio MA. HSP72 protects against obesity-induced insulin resistance. PNAS. 2008;105:1739–1744. doi: 10.1073/pnas.0705799105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colditz GA, Willett WC, Stampfer MJ, Manson JE, Hennekens CH, Arky RA, Speizer FE. Weight as a risk factor for clinical diabetes in women. Am J Epidemiol. 1990;132:501–513. doi: 10.1093/oxfordjournals.aje.a115686. [DOI] [PubMed] [Google Scholar]
- Daviau A, Proulx R, Robitaille K, Di Fruscio M, Tanguay RM, Landry J, Patterson C, Durocher Y, Blouin R. Down-regulation of the mixed-lineage dual leucine zipper-bearing kinase by heat shock protein 70 and its co-chaperone CHIP. J Biol Chem. 2006;281:31467–31477. doi: 10.1074/jbc.M607612200. [DOI] [PubMed] [Google Scholar]
- Davis RJ. Signal transduction by the JNK group of MAP kinases. Cell. 2000;103:239–252. doi: 10.1016/S0092-8674(00)00116-1. [DOI] [PubMed] [Google Scholar]
- De Fea K, Roth RA. Protein kinase C modulation of insulin receptor substrate-1 tyrosine phosphorylation requires serine 612. Biochemistry. 1997;36:12939–12947. doi: 10.1021/bi971157f. [DOI] [PubMed] [Google Scholar]
- Finucane MM, Stevens GA, Cowan MJ, Danaei G, Lin JK, Paciorek CJ, Singh GM, Gutierrez HR, Lu Y, Bahalim AN, Farzadfar F, Riley LM, Ezzati M. National, regional, and global trends in body-mass index since 1980: systematic analysis of health examination surveys and epidemiological studies with 960 country-years and 9.1 million participants. Lancet. 2011;377:557–567. doi: 10.1016/S0140-6736(10)62037-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garetto LP, Richter EA, Goodman MN, Ruderman NB. Enhanced muscle glucose metabolism after exercise in the rat: the two phases. Am J Physiol Endocrinol Metab. 1984;246:E471–E475. doi: 10.1152/ajpendo.1984.246.6.E471. [DOI] [PubMed] [Google Scholar]
- Geiger PC, Gupte AA. Heat shock proteins are important mediators of skeletal muscle insulin sensitivity. Exerc Sport Sci Rev. 2011;39:34–42. doi: 10.1097/JES.0b013e318201f236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geloneze B, Repetto EM, Geloneze SR, Tambascia MA, Ermetice MN. The threshold value for insulin resistance (HOMAIR) in an admixtured population IR in the Brazilian Metabolic Syndrome Study. Diabetes Res Clin Pract. 2006;72:219–220. doi: 10.1016/j.diabres.2005.10.017. [DOI] [PubMed] [Google Scholar]
- Gupte AA, Bomhoff GL, Swerdlow RH, Geiger PC. Heat treatment improves glucose tolerance and prevents skeletal muscle insulin resistance in rats fed a high-fat diet. Diabetes. 2009;58:567–578. doi: 10.2337/db08-1070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirosumi J, Tuncman G, Chang L, Görgün CZ, Uysal KT, Maeda K, Karin M, Hotamisligil GS. A central role for JNK in obesity and insulin resistance. Nature. 2002;420:333–336. doi: 10.1038/nature01137. [DOI] [PubMed] [Google Scholar]
- Hotamisligil GS. Endoplasmic reticulum stress and the inflammatory basis of metabolic disease. Cell. 2010;140:900–917. doi: 10.1016/j.cell.2010.02.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kline MP, Morimoto RI. Repression of the heat shock factor 1 transcriptional activation domain is modulated by constitutive phosphorylation. Mol Cell Biol. 1997;17:2107–2115. doi: 10.1128/mcb.17.4.2107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurucz I, Morva A, Vaag A, Eriksson K, Huang X, Groop L, Koranyi L. Decreased expression of heat shock protein 72 in skeletal muscle of patients with type 2 diabetes correlates with insulin resistance. Diabetes. 2002;51:1102–1109. doi: 10.2337/diabetes.51.4.1102. [DOI] [PubMed] [Google Scholar]
- Lee YH, Giraud J, Davis RJ, White MF. c-Jun N-terminal kinase (JNK) mediates feedback inhibition of the insulin signaling cascade. J Biol Chem. 2003;278:2896–2902. doi: 10.1074/jbc.M208359200. [DOI] [PubMed] [Google Scholar]
- Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28:412–419. doi: 10.1007/BF00280883. [DOI] [PubMed] [Google Scholar]
- Meriin AB, Yaglom JA, Gabai VL, Zon L, Ganiatsas S, Mosser DD, Zon L, Sherman MY. Protein-damaging stresses activate c-Jun N-terminal kinase via inhibition of its dephosphorylation: a novel pathway controlled by HSP72. Mol Cell Biol. 1999;19:2547–2555. doi: 10.1128/mcb.19.4.2547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Myers J, Bellin D. Ramp exercise protocols for clinical and cardiopulmonary exercise testing. Sports Med. 2000;30:23–29. doi: 10.2165/00007256-200030010-00003. [DOI] [PubMed] [Google Scholar]
- O'Leary VB, Marchetti CM, Krishman RK, Stetzer BP, Gonzalez F, Kirwan JP. Exercise-induced reversal of insulin resistance in obese elderly is associated with reduced visceral fat. J Appl Physiol. 2006;100:1584–1589. doi: 10.1152/japplphysiol.01336.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park HS, Lee JS, Huh SH, Seo JS, Choi EJ. Hsp72 functions as a natural inhibitory protein of c-Jun N-terminal kinase. EMBO J. 2001;20:446–456. doi: 10.1093/emboj/20.3.446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poole DC, Wilkerson DP, Jones AM. Validity of criteria for establishing maximal O2 uptake during ramp exercise tests. Eur J Appl Physiol. 2008;102:403–410. doi: 10.1007/s00421-007-0596-3. [DOI] [PubMed] [Google Scholar]
- Richter EA, Mikines KJ, Galbo H, Kiens B. Effect of exercise on insulin action in human skeletal muscle. J Appl Physiol. 1989;66:876–888. doi: 10.1152/jappl.1989.66.2.876. [DOI] [PubMed] [Google Scholar]
- Ropelle ER, Pauli JR, Prada PO, de Souza CT, Picardi PK, Faria MC, Cintra DE, Fernandes MF, Flores MB, Velloso LA, Saad MJ, Carvalheira JB. Reversal of diet-induced insulin resistance with a single bout of exercise in the rat: the role of PTP1B and IRS-1 serine phosphorylation. J Physiol. 2006;577:997–1007. doi: 10.1113/jphysiol.2006.120006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sasai H, Sairenchi T, Iso H, Irie F, Otaka E, Tanaka K, Ota H, Muto T. Relationship between obesity and incident diabetes in middle-aged and older Japanese adults: the Ibaraki Prefectural Health Study. Mayo Clin Proc. 2010;85:36–40. doi: 10.4065/mcp.2009.0230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schenk S, Horowitz JF. Acute exercise increases triglyceride synthesis in skeletal muscle and prevents fatty acid-induced insulin resistance. J Clin Invest. 2007;117:1690–1698. doi: 10.1172/JCI30566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shafrir E. Development and consequences of insulin resistance: lessons from animals with hyperinsulinaemia. Diabetes Metab. 1996;22:122–131. [PubMed] [Google Scholar]
- Simar D, Jacques A, Caillaud C. Heat shock proteins induction reduces stress kinases activation, potentially improving insulin signalling in monocytes from obese subjects. Cell Stress Chaperones. 2012;17:615–621. doi: 10.1007/s12192-012-0336-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinha R, Fisch G, Teague BRN, Tamborlane WV, Banyas RNB, Allen KRN, Savoye MRD, Rieger VMD, Taksali SMPH, Barbetta GRD, Sherwin RSE, Caprio S. Prevalence of impaired glucose tolerance among children and adolescents with marked obesity. N Engl J Med. 2002;346:802–810. doi: 10.1056/NEJMoa012578. [DOI] [PubMed] [Google Scholar]
- Uysal KT, Wiesbrock SM, Marino MW, Hotamisligil GS. Protection from obesity-induced insulin resistance in mice lacking TNF-alpha function. Nature. 1997;389:610–614. doi: 10.1038/39335. [DOI] [PubMed] [Google Scholar]
- World Health Organization (2013) Global Health Observatory. Obesity. http://www.who.int/gho/ncd/risk_factors/obesity_text/en/index.html. Accessed 13 May 2013
- Yi Z, Langlais P, De Filippis EA, Luo M, Flynn CR, Schroeder S, Weintraub ST, Mapes R, Mandarino LJ. Global assessment of regulation of phosphorylation of insulin receptor substrate-1 by insulin in vivo in human muscle. Diabetes. 2007;56:1508–1516. doi: 10.2337/db06-1355. [DOI] [PubMed] [Google Scholar]