Dear Sir,
Due to the detrimental action of free radicals and reactive oxygen species (ROS), oxidative stress is involved in the pathogenesis of ageing and diseases such as inflammation, cancer, cardiovascular disorders and infections. Although this view remains still valid, during the last decade the understanding of oxidative stress has progressively changed and it has become clear that free radicals and reactive oxygen species also play a key role in cell biology and function. In fact, oxidation/reduction reactions are a primary mechanism for regulation of cell proliferation, death, and most notably, cell differentiation, which involves the function of several redox-sensitive molecular elements1.
The main intracellular antioxidant of mammalian cells, the tripeptide glutathione (GSH), plays a crucial role in such functions, together with other less abundant low molecular weight thiols (LMWT); in fact, besides participating in detoxicant reactions catalysed by glutathione transferase and glutathione peroxidase, the binding of GSH to proteins (protein S-glutathionylation) is a potent modulator of the molecular elements involved in cell proliferation, differentiation and death, both inside the cytoplasm (e.g. the transcription factor nuclear factor-κB), and on the cell surface (e.g. tumour necrosis factor-α receptor)1.
The cell surface enzyme gamma-glutamyltransferase (GGT) has a peculiar role in determining the biological action of GSH, as it catalyses the hydrolysis of the tripeptide GSH (gamma glutamylcysteinylglycine) to glutamate and the dipeptide cysteinyl-glycine (cys-gly). This latter is a potent reductant which promotes pro-oxidant reactions through the reduction of oxygen to superoxide anion radical and the generation of thiol radical. GGT is, therefore, able to transform an antioxidant (GSH) into a potent pro-oxidant (cys-gly), provided that sufficient oxygen is present in the environment2.
The isolation, characterisation, in vitro expansion and differentiation of human umbilical cord blood mesenchymal stem cells (hUCBMSC) are among the current aims of regenerative medicine, and achieving these goals is a pre-requisite for extensive use of this novel approach for the treatment of a number of human diseases; unfortunately, reliable procedures for efficient expansion and differentiation of hUCBMSC remain to be established, and oxidative stress caused by culture conditions might be a factor to take into account when devising adequate conditions for maintaining and differentiating stem cells in vitro3.
It is well known that standard cell culture conditions, which involve the exposure of cells to an atmosphere of air containing 5% CO2, are far from the physiologic situation. In fact, while the atmospheric oxygen tension is 160 mmHg, it reaches only 40 mmHg in blood, and decreases to 18 mmHg in foetal blood. Cell cultures are thus exposed to a potent oxidative stress even in basal conditions, and the gap between physiological and cell culture conditions is far wider if considering foetal tissues, which are subjected to very low oxygen concentrations in vivo4.
We decided to check whether decreasing oxygen tension in culture to very low levels, corresponding to those found in foetal tissues, is compatible with cell growth in vitro, whether it has appreciable effects on the status of intracellular and extracellular low-molecular weight thiols of cultured mesenchymal stem cells, and whether GGT acts as an oxygen-dependent trigger of oxidative stress in vitro.
Thirty-four human umbilical cord blood samples were harvested from term or preterm deliveries at the time of birth with the mothers’ consent. The mean volume of the cord blood units (CBU) was 113±12.3 mL without anticoagulant. Blood samples were processed within 24 hours of collection. The mononuclear cells were separated using Percoll (Amersham Biosciences, Uppsala, Sweden) and were suspended in culture medium (low-glucose Dulbecco’s modified Eagle’s medium; GIBCO) containing 20% foetal calf serum, 100 U/mL penicillin, 100 μg/mL streptomycin and 2 mM L-glutamine. Cells were then seeded at a density of 1×106 cells/cm2 in 25 cm2 culture flasks. The human mesenchymal stem cells were isolated using a method based on the adherence of fibroblast-like cells to the plastic substrate of the cell culture plate and on the concomitant lack of adherence of haematopoietic cells.
Cultures were maintained at 37 °C in an humidified atmosphere containing 5% CO2, with a change of culture medium and the removal of non-adherent cells every 3 days. When approximately 60–70% confluent, cells were detached with 0.05% trypsin and 0.02% EDTA and re-plated at a density of 104 cells/cm2 in 25 cm2 culture flasks. hUCBMSC colonies were isolated from 12 CBU (35.2%).
The hUCBMSC were analysed for epitope expression using a FACScan flow cytometer (Becton Dickinson, San Jose, California, USA). Cells were detached by trypsinisation, centrifuged, resuspended in phosphate-buffered salie (Euroclone, Milan, Italy) at a density of 0.5×106 cells/mL and stained according to the manufacturer’s instructions with the following monoclonal anti-human antibodies (BD): MHCI, MHCII, CD45, CD44, CD29, CD34, CD90, CD73 and CD14. At least 10,000 events were acquired for each sample. Data were analysed using the CELL QUEST software programme (Becton Dickinson, San Jose, California, USA).
Adherent hUCBMSC expressed the following cell-surface antigens: CD44, CD73, CD90, CD29. They did not express either haematopoietic lineage markers such as CD34 or monocyte-macrophage antigens such as CD14 and CD45. The cell population was positive for HLA class I but not for HLA class II.
For determinations concerning the effects of low oxygen tension on intracellular and extracellular LMWT, in four experiments, cell monolayers were split at the second passage and cultured in standard conditions or in a modified atmosphere containing O2 at a partial pressure of 6 mmHg, in both cases with a stable concentration of 5% CO2. Aliquots of cell culture media were collected 24 hours after seeding and proteins in the medium were precipitated by adding 10% trichloroacetic acid. Samples were then centrifuged at 15,000 g (10 min, 4 °C) and supernatants were collected and stored at 4 °C until thiol derivatisation. To determine intracellular LMWT, cell monolayers were washed twice with phosphate-buffered saline and cells were extracted in 10% trichloroacetic acid for 20 min at 20–25 °C. Acid extracts were then collected and stored at 4 °C until analysis, while cellular proteins were harvested in 0.1 M NaOH and stored at −20 °C until the protein content was determined (Bradford assay). The LMWT were determined as previously described5. Briefly, samples and thiol standards were incubated for 30 minutes at room temperature with tris-(2-carboxyethyl)-phosphine (TCEP; Molecular Probes, Eugene, Oregon, USA) in order to achieve disulfide reduction and then they were treated with thiol-reactive 7-fluorobenzo-2-oxa-1,3-diazole-4-sulfonate (SBD-F; Fluka; 60 minutes, 60 °C). Thiol concentrations were determined by a high-pressure liquid chromatography system equipped with a C-18 reverse-phase column (Resolve; Waters Corporation, Milford, Massachusetts, USA) and a fluorimetric detector (RF-551; Shimadzu Corporation, Kyoto, Japan - filter settings: 385 nm excitation, 515 nm emission). The mobile phase (5% methanol in 0.2 M KH2PO4, adjusted to pH 2.7 with H3PO4) was delivered at a flow rate of 1 mL/min.
Calibration was based on external standards using stock solutions (10 mM) of Cys, Cys-Gly and GSH, prepared in 0.12 M perchloric acid and stored at −20 °C; working solutions were prepared daily in 0.2 M Tris-HCl, pH 8.2, containing 20 mM EDTA and 1% (w/v) sodium dodecyl sulphate. A Shimadzu RF-551 fluorimetric detection system was used (filter settings: 385 nm excitation, 515 nm emission).
Protein content was determined by the Bradford method (Bio-Rad Laboratories, Hercules, California, USA). Data were analysed by Student’s t test for paired data.
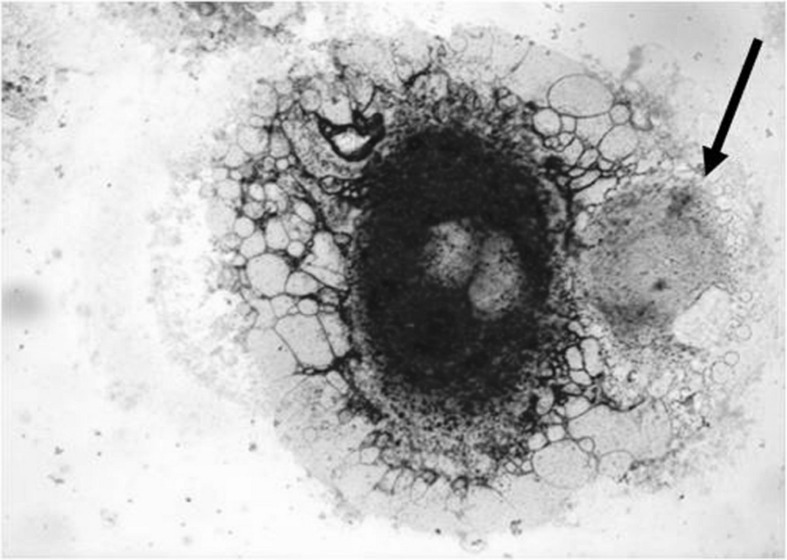
Cell monolayers were air-dried overnight at room temperature. After fixing (for 15 seconds) in phosphate-buffered acetone-formalin, flasks were washed three times in ice-cold double-distilled water to remove the fixative. GGT activity was revealed by using gamma-glutamyl-4-methoxy-2-naphthylamide as the substrate and Fast Garnet GBC as the chromogen, counterstaining nuclei with Mayer’s hemalum counterstain of nuclei (Figure 1).
Figure 1.
Cytochemical staining for GGT activity in umbilical cord blood cells.
Original magnification 100×. The red stain corresponds to GGT activity. The large, GGT-positive cell appears to contain a phagocytised GGT-negative cell (arrow).
The analysis of LMWT in low-oxygen conditions, when compared to standard conditions, showed a modest but significant increase of intracellular GSH concentration (98.2±1.7 vs 96.5±1.8 nmol mg/protein, P =0.029), a proportionally more marked increase in extracellular GSH (3.1±0.4 vs 1.4±0.3 nmol/mg, P <0.001) and intracellular cysteine (30.6±2.5 vs 19.7±2.2 nmol/mg protein, P =0.018). Intracellular cysteinylglycine, which was absent in standard culture conditions, was found only in low-oxygen conditions (2.3±0.25 nmol/mg protein). The presence of extracellular cysteinylglycine in culture media both in low-oxygen (2.4±0.2 nmol/mg protein) and in standard culture conditions (2.7±0.4 nmol/mg protein; P = ns) suggests that catalytically active GGT is expressed in umbilical cord blood cells. Accordingly, the cytochemical staining for GGT activity revealed the presence of the enzyme in correspondence of large cellular elements present in the cultures, thus suggesting that GGT might contribute to the redox reactions occurring at the cell surface. Indeed, the participation of GGT-derived extracellular cys-gly to pro-oxidant reactions and their dependence on the oxygen tension were confirmed by the appearance of intracellular cys-gly only in cells maintained in low-oxygen conditions. This result may suggest that although cys-gly is formed extracellularly in both conditions tested, only in the presence of low-oxygen tensions is it able to contribute to the intracellular LMWT pool. In contrast, in standard culture conditions the paradoxical pro-oxidant action of cys-gly can occur, thus possibly favouring the formation of extracellular mixed disulfide and reducing the recovery of LMWT (cys, cys-gly) required for intracellular GSH synthesis.
To our knowledge, this is the first study demonstrating the influence of oxygen tension on the balance of LMWT in cultured hUCBMSC. The results confirm the need for detailed exploration of the relationship between oxidative stress and proliferation/differentiation of human umbilical cord blood cells in vitro.
Footnotes
The Authors declare no conflicts of interest.
References
- 1.Takahashi M. Oxidative stress and redox regulation on in vitro development of mammalian embryos. J Reprod Dev. 2012;58:1–9. doi: 10.1262/jrd.11-138n. [DOI] [PubMed] [Google Scholar]
- 2.Pompella A, Visvikis A, Paolicchi A, et al. The changing faces of glutathione, a cellular protagonist. Biochem Pharmacol. 2003;66:1499–503. doi: 10.1016/s0006-2952(03)00504-5. [DOI] [PubMed] [Google Scholar]
- 3.Moussavi-Harami F, Duwayri Y, Martin JA, et al. Oxygen effects on senescence in chondrocytes and mesenchymal stem cells: consequences for tissue engineering. Iowa Orthop J. 2004;24:15–20. [PMC free article] [PubMed] [Google Scholar]
- 4.Thorp JA, Dildy GA, Yeomans ER, et al. Umbilical cord blood gas analysis at delivery. Am J Obstet Gynecol. 1996;175:517–22. doi: 10.1053/ob.1996.v175.a74401. [DOI] [PubMed] [Google Scholar]
- 5.Corti A, Paolicchi A, Franzini M, et al. The S-thiolating activity of membrane gamma-glutamyltransferase: formation of cysteinyl-glycine mixed disulfides with cellular proteins and in the cell microenvironment. Antioxid Redox Signal. 2005;7:911–8. doi: 10.1089/ars.2005.7.911. [DOI] [PubMed] [Google Scholar]