Abstract
OBJECTIVE:
The goal of this study was to determine changes in antibiotic-dispensing rates among children in 3 health plans located in New England [A], the Mountain West [B], and the Midwest [C] regions of the United States.
METHODS:
Pharmacy and outpatient claims from September 2000 to August 2010 were used to calculate rates of antibiotic dispensing per person-year for children aged 3 months to 18 years. Differences in rates by year, diagnosis, and health plan were tested by using Poisson regression. The data were analyzed to determine whether there was a change in the rate of decline over time.
RESULTS:
Antibiotic use in the 3- to <24-month age group varied at baseline according to health plan (A: 2.27, B: 1.40, C: 2.23 antibiotics per person-year; P < .001). The downward trend in antibiotic dispensing slowed, stabilized, or reversed during this 10-year period. In the 3- to <24-month age group, we observed 5.0%, 9.3%, and 7.2% annual declines early in the decade in the 3 plans, respectively. These dropped to 2.4%, 2.1%, and 0.5% annual declines by the end of the decade. Third-generation cephalosporin use for otitis media increased 1.6-, 15-, and 5.5-fold in plans A, B, and C in young children. Similar attenuation of decline in antibiotic use and increases in use of broad-spectrum agents were seen in other age groups.
CONCLUSIONS:
Antibiotic dispensing for children may have reached a new plateau. Along with identifying best practices in low-prescribing areas, decreasing broad-spectrum use for particular conditions should be a continuing focus of intervention efforts.
Keywords: antibiotics, otitis media, respiratory tract infections
What’s Known on This Subject:
Antibiotic use for children has decreased dramatically over the last 20 years. Programs encouraging judicious antibiotic use have focused both on decreasing overall antibiotic use and appropriate prescribing of broad-spectrum agents.
What This Study Adds:
Large declines in antibiotic rates were prominent in the early 2000s. This trend has attenuated, and use has leveled off in some age groups and locales; continued improvement in the use of broad-spectrum agents is possible.
Local and national efforts, such as the Centers for Disease Control and Prevention’s Get Smart Program, have tried to decrease inappropriate use of antibiotics, particularly for respiratory tract infections (RTIs),1–3 to minimize the development and spread of antimicrobial resistance.4,5 In addition, professional organizations, including the American Academy of Pediatrics, have long promoted judicious use of antibiotics by issuing treatment guidelines for common pediatric infections.6–18 Judicious use of antibiotics includes provider adherence to prescribing guidelines, not using antibiotics for probable viral infections, and using the narrowest spectrum agent that is active against the targeted pathogens.1,19 These efforts, coupled with the introduction of new vaccines and attention focused by the lay press on antibiotic misuse, have resulted in substantial decreases in antibiotic use for US children during the last 2 decades.20,21 Centers for Disease Control and Prevention investigators reported up to a 24% cumulative reduction in antibiotic use between 1993 and 2008 in children aged <14 years.22 For children aged <5 years, in whom antibiotic utilization is highest, an impressive 36% reduction in the use of antibiotics for RTIs occurred between 1995 and 2006.23
It is unknown whether the previous downward trend in antibiotic use will continue or if a leveling off of prescribing will soon be reached. Stabilization of antibiotic use rates may be appropriate if principles of judicious use are being followed or it may signal the need for new interventions. A recent study from a limited number of communities in Massachusetts indicated that stabilization of dispensing has occurred in children aged <6 years.24 Because geographic variation exists in antibiotic prescribing,21,25–27 we investigated trends in community antibiotic dispensing in children aged <18 years insured by 3 geographically distinct health plans to examine whether rates have leveled off at these sites. We also explored trends in antibiotic utilization for common infections among specific age groups and within drug categories to better understand prescribing and inform where additional interventions may be needed.
Methods
Setting
The study population included children 3 months to <18 years of age between September 1, 2000, and August 31, 2010, in 3 commercial health plans located in New England (A), the Mountain West (B), and the Midwest (C) regions of the United States. All health plans are members of the HMO Research Network.28 Plan A is a commercial health insurer that reimburses care provided by a large network of affiliated practices; plan B is an integrated health care delivery system with a multispecialty group practice; and plan C is an insurer with both an integrated health care delivery system and affiliated network practices. Children enrolled for a minimum of 7 consecutive days with concurrent pharmacy benefits were included. Each study year was defined as September 1 to August 31 to include an entire 12-month period with only 1 respiratory illness season.
This study was approved by the institutional review board of Harvard Pilgrim Health Care. Institutional review board approval was ceded to Harvard Pilgrim Health Care by the 2 other health plans.
Data Collection
A previously developed approach for analysis without transfer of personal health information was used to calculate rates of antibiotic dispensings per person-year of observation in each health plan.21,29,30 Within each study year, we calculated the number of days a subject spent in each of the age categories: 3 to <24 months and 2 to <4, 4 to <6, 6 to <12, and 12 to <18 years. A child could contribute data to multiple age groups as he or she aged and might contribute to 2 age groups in a single year.
Antibiotic data were captured similarly at all 3 sites.31 Dispensings of oral antibiotics were identified by using pharmacy claims data according to a previously developed list of National Drug Codes32 cross-indexed by using generic or brand name and grouped into antimicrobial subclasses. Subclass groupings included first-line penicillins (eg, amoxicillin), amoxicillin-clavulanate, erythromycin, second-generation macrolides (azithromycin, clarithromycin), and first-generation (eg, cephalexin), second-generation (eg, cefuroxime), and third-generation (eg, cefdinir, cefixime) cephalosporins. Other oral antibiotics were grouped as “other,” except for those used in adolescents, for whom we classed tetracyclines separately (Appendix 1). Antitubercular, antihelminthic, and parenteral preparations and topical antibiotics were excluded.
Encounter data were used to link each dispensing to the diagnosis assigned at the most recent outpatient visit (including urgent care or emergency department encounters) in the previous 3 days. If no visit occurred in this time frame, the dispensing was classified as “unlinked.” A previously described algorithm was used to identify a primary International Classification of Diseases, Ninth Revision, diagnosis for each visit, giving priority to any diagnosis of a bacterial infection and using a hierarchy of bacterial diagnoses21 (Appendix 2).
Data Analysis
Within each age group, annual population-based antibiotic-dispensing rates (number of antibiotic dispensings divided by the number of person-years aggregated across individuals) were determined according to health plan and year, and 95% confidence intervals (CIs) were calculated.21 Differences in total antibiotic rates between the first and last study years and among study sites were tested for statistical significance (P < .05) based on a Poisson regression model that accounted for person-years enrolled.
We sought to understand if the data were most consistent with a single linear trend throughout the study period or if the linear slope changed at some point (ie, a “2-slope” model). We tested each study year as a potential inflection point by fitting a model with separate linear slopes in the years preceding and the years after that point.24,33 Within each plan and age group, the year for which the model had the smallest Akaike information criterion was designated the most likely year for a change in slope. If this 2-slope model fit significantly better than the single-slope model (P < .05), we calculated the percent year-to-year decline preinflection and postinflection. CIs were examined for the rate of change in the postinflection period to see if data were consistent, statistically, with stabilization (defined as a 0% rate of change).
The percentage of visits for each diagnosis that was associated with an antibiotic dispensing was calculated, as was dispensing of each class of antibiotic for diagnoses of interest. All analyses were conducted by using SAS version 9.3 (SAS Institute, Inc, Cary, NC).
Results
During the 10-year study period, the total number of person-years observed across all 3 sites was 395 095 for children aged 3 to <24 months; 454 165 for children aged 2 to <4 years; 460 092 for children aged 4 to <6 years; 1 450 327 for children aged 6 to <12 years; and 1 570 713 for children aged 12 to <18 years.
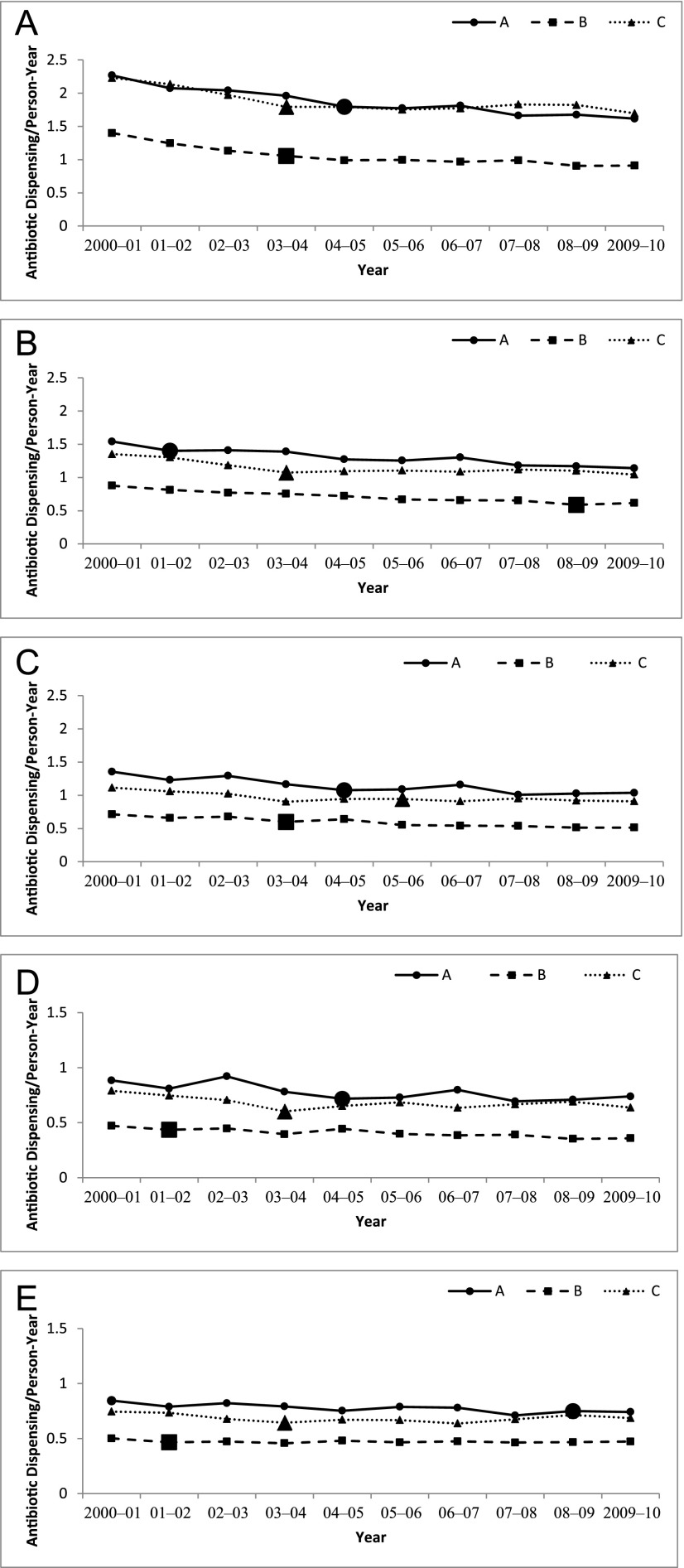
The overall annual antibiotic-dispensing rates in each age group and each plan were lower in 2009–2010 than 2000–2001 (P < .001 for 2009–2010 vs 2000–2001 at all sites and age groups) (Appendix 3). Children aged 3 to <24 months were the highest utilizers of antibiotics in all years. By 2009–2010, antibiotic rates were 1.62, 0.91, and 1.70 antibiotics per person-year in plans A, B, and C, respectively, down from starting rates of 2.27, 1.40, and 2.23 antibiotics per person-year in this age group. Minimal, although statistically significant, changes in dispensing occurred in children aged ≥6 years. Antibiotic dispensing varied considerably among the 3 health plans in each of the age groups (P < .001). Plan B consistently had lower visit rates and antibiotic-dispensing rates compared with plans A and C for each age group (Fig 1).
FIGURE 1.
Rates of antibiotic dispensing per person-year of enrollment for children aged as follows: A, 3 to 24 months; B, 2 to <4 years; C, 4 to <6 years; D, 6 to <12 years; and E, 12 to <18 years. Values are for each health plan (A–C) between 2000 and 2010. Note: axes differ for the last 2 age groups. Enhanced marker reflects year of greatest change in decline of antibiotic rate. Although 95% CIs were calculated, the results were too small to be visible on graphs.
Stabilization of Antibiotic Dispensing
Although rates decreased between 2000–2001 and 2009–2010 for all age groups and health plans, in all cases a model for a single downward slope was rejected in favor of a model with 2 slopes, indicating that the trend in antibiotic dispensing changed over the study period (P < .001 for all sites and age groups). Inflection points indicating the year of the change in slope are noted with larger markers in Fig 1. The particular year in which the change in slope occurred differed across age groups and sites. After each inflection point, the rate of decline slowed relative to the preinflection period or even reversed, meaning increasing rates were observed in the later period, although these increases were typically small (Table 1).
TABLE 1.
Two-Slope Regression Model
| Age Group | Plan | Preinflection % Decline/Year (95% CI) | Inflection Year | Postinflection % Decline/Year (95% CI) |
|---|---|---|---|---|
| 3 to <24 mo | ||||
| A | 5.0 (4.7 to 5.3) | 2004–2005 | 2.4 (2.1 to 2.6) | |
| B | 9.3 (8.6 to 10) | 2003–2004 | 2.1 (1.7 to 2.5) | |
| C | 7.2 (6.8 to 7.5) | 2003–2004 | 0.5 (0.3 to 0.7) | |
| 2 to <4 y | ||||
| A | 7.2 (5.9 to 8.5) | 2001–2002 | 2.8 (2.6 to 2.9) | |
| B | 4.4 (4.1 to 4.7) | 2008–2009 | –2.9 (–5.8 to –0.1) | |
| C | 7.1 (6.6 to 7.5) | 2003–2004 | 0.3 (0.1 to 0.6) | |
| 4 to <6 y | ||||
| A | 4.6 (4.3 to 5.0) | 2004–2005 | 1.7 (1.4 to 2.0) | |
| B | 4.3 (3.9 to 4.7) | 2006–2007 | 2.3 (1.3 to 3.2) | |
| C | 5.8 (5.3 to 6.4) | 2003–2004 | 0.3 (0.1 to 0.6) | |
| 6 to <12 y | ||||
| A | 4.2 (4.0 to 4.5) | 2004–2005 | 0.9 (0.7 to 1.1) | |
| B | 5.4 (3.5 to 7.3) | 2001–2002 | 2.7 (2.5 to 2.9) | |
| C | 7.1 (6.7 to 7.4) | 2003–2004 | –0.7 (–0.9 to –0.5) | |
| 12 to <18 y | ||||
| A | 1.7 (1.5 to 1.8) | 2007–2008 | –0.3 (–0.8 to 0.2)a | |
| B | 6.7 (5.0 to 8.4) | 2001–2002 | –0.1 (–0.2 to 0.1)a | |
| C | 4.9 (4.6 to 5.3) | 2003–2004 | –1.1 (–1.3 to –1.0) |
Decline in total antibiotic rate before inflection year compared with decline noted after inflection. Negative percentages indicate an increase in antibiotic prescribing rate during this time period.
For CIs that include 0, a decline of 0 is consistent with a stable rate of dispensing.
Children aged 3 to <24 months experienced the greatest overall decline in antibiotic use between 2000–2001 and 2009–2010. However, after the inflection year, the decline in dispensing was <3% per year for plans A and B and close to zero in plan C, indicating near stabilization of dispensing. Throughout the study period, dispensing for children aged 2 to <4 years was nearly one-third less than in the 3- to <24-month age group. In this age group, the rate of antibiotic dispensing continued to decline after the inflection point in plans A and C but at a slower rate. In plan B, however, after years of decreases, a 5% increase in dispensing was noted between 2008–2009 and 2009–2010 (0.59 to 0.62; P < .001). For the 4- to <6-year-olds and 6- to <12-year-olds, modest declines in dispensing continued in plans A and B, while the dispensing rate at plan C approached stabilization for 4- to 6-year-olds and increased for 6- to 12-year-olds. For adolescents (aged 12 to <18 years), antibiotic dispensing stabilized at plans A and B in 2007–2008 and 2001–2002, respectively, and increased at plan C after 2003–2004.
Trends in Dispensing According to Diagnosis
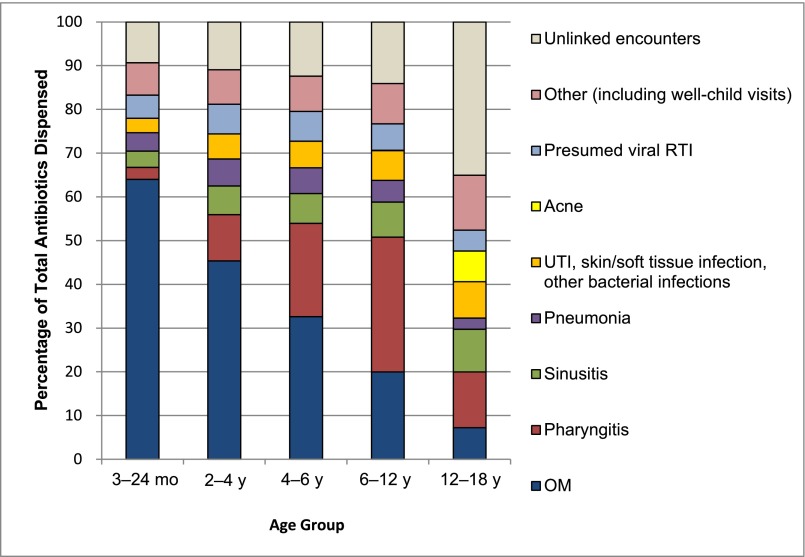
RTIs accounted for >75% of all antibiotics prescribed for children aged <12 years. Otitis media (OM) was the most common diagnosis for which antibiotics were prescribed for children aged <6 years, particularly in the 3- to <24-month age group (Fig 2). For children in the 3- to <24-month age group, the antibiotic-dispensing rate per person-year for OM declined at all sites between 2000–2001 and 2009–2010 (A: 1.40 to 1.05; B: 0.93 to 0.64; and C: 1.44 to 1.04 [P < .001]). This decrease in antibiotic prescribing for OM accounted for 54%, 59%, and 74% of the total drop in antibiotic use in plans A, B, and C, respectively. Whereas the OM diagnosis rate decreased in each plan (A: 2.23 to 1.72; B: 1.30 to 1.02; C: 2.50 to 1.71 [P < .001]), the percentage of OM cases treated once diagnosed showed only modest changes over the decade in 2 sites (A: 63% to 61%; C: 58% to 61%). A decrease in treatment fraction occurred at plan B (72% to 63%), which still accounts for a relatively small portion of the decrease in antibiotic use for OM on a population level.
FIGURE 2.
Distribution of diagnoses and antibiotic prescriptions in 2009–2010 among 3 health plans. UTI, urinary tract infection.
Among children aged 6 to <12 years, pharyngitis was the most common diagnosis associated with an antibiotic dispensing (Fig 2). The visit rate for adolescents (12 to <18 years) was lower than for younger children, but dispensing of antibiotics not linked to an encounter was common, accounting for 35% of dispensed antibiotics across sites by 2009–2010. Adolescents also had the highest fractions of illnesses coded as viral RTIs that were treated with an antibiotic, with large variation according to health plan (A: 24%; B: 8%; C: 26%).
Trends in Dispensing According to Antibiotic Class
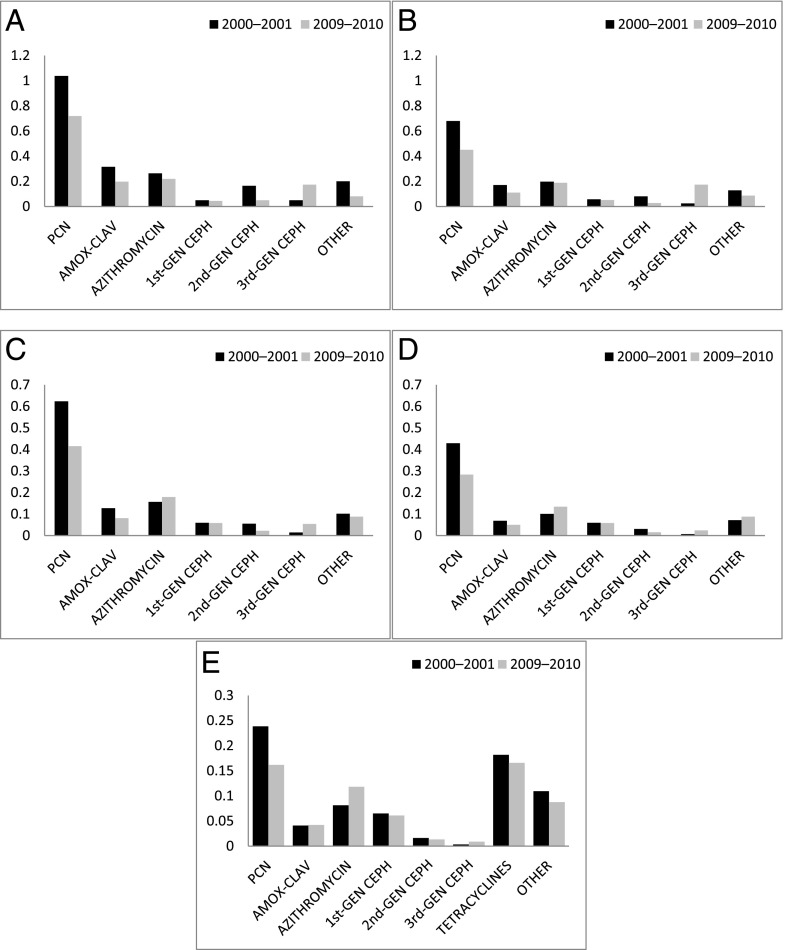
The majority of antibiotics dispensed for children aged <12 years included penicillins, cephalosporins, and second-generation macrolides (Fig 3). In contrast to younger children, among adolescents, first-line penicillin dispensings were followed closely by tetracycline dispensings, coincident with more visits for acne (Fig 2). Between 2000–2001 and 2009–2010, first-generation penicillins, amoxicillin-clavulanate, and second-generation cephalosporin use decreased in all age groups (Appendix 3 and 4).
FIGURE 3.
Distribution of antibiotic classes among health plans, 2000–2001 and 2009–2010, for children aged as follows: A, 3 to <24 months; B, 2 to <4 years; C, 4 to <6 years; D, 6 to <12 years; and E, 12 to <18 years. Changes in dispensing rate between 2000–2001 and 2009–2010 were statistically significant (P < .05) for all antibiotic classes. Note: y-axis scaled differently in lower panels. AMOX-CLAV, amoxicillin/clavulanate; CEPH, cephalosporins; GEN, generation; PCN, 1st - line penicillins.
Increases were noted in the use of broad-spectrum, third-generation cephalosporins (Appendix 4). OM was the leading diagnosis associated with third-generation cephalosporin use through age 12 years, followed by sinusitis and pharyngitis. In adolescents, broad-spectrum cephalosporins were prescribed frequently for sinusitis. (Table 2).
TABLE 2.
Distribution of Diagnoses Linked With Third-Generation Cephalosporin Dispensing at Each Health Plan in 2009–2010
| Age Group | Plan | OM (%) | Pneumonia (%) | Pharyngitis (%) | Sinusitis (%) | Other Bacterial Infections (%) | Presumed Viral RTI (%) | All Other (%)a |
|---|---|---|---|---|---|---|---|---|
| 3 to <24 mo | ||||||||
| A | 76 | 2 | 1 | 3 | 3 | 1 | 13 | |
| B | 81 | 3 | 1 | 4 | 2 | 0b | 9 | |
| C | 70 | 6 | 1 | 4 | 1 | 3 | 14 | |
| 2 to <4 y | ||||||||
| A | 60 | 4 | 5 | 7 | 4 | 3 | 16 | |
| B | 68 | 6 | 3 | 8 | 3 | 0b | 11 | |
| C | 57 | 7 | 5 | 8 | 3 | 4 | 16 | |
| 4 to <6 y | ||||||||
| A | 47 | 5 | 11 | 11 | 8 | 2 | 15 | |
| B | 61 | 5 | 4 | 12 | 6 | 0b | 12 | |
| C | 46 | 7 | 10 | 12 | 3 | 5 | 16 | |
| 6 to <12 y | ||||||||
| A | 33 | 4 | 16 | 15 | 8 | 2 | 20 | |
| B | 51 | 5 | 5 | 14 | 8 | 0b | 15 | |
| C | 31 | 7 | 15 | 16 | 5 | 4 | 21 | |
| 12 to <18 y | ||||||||
| A | 14 | 4 | 14 | 25 | 12 | 3 | 27 | |
| Bc | 27 | NAb | 6 | 20 | 9 | 0b | 29 | |
| C | 19 | 5 | 14 | 28 | 5 | 3 | 26 |
NA, not applicable.
Includes dispensings unlinked to diagnoses.
Less than 0.5%.
Due to an antibiotic rate <0.5% among multiple diagnoses in a combined category, total percentage may not add to 100%.
Macrolide dispensing at all plans in the 3- to <24-month age group and in plans A and B for those aged 2 to <4 years showed small decreases, but increases were noted in plan C, and in all plans for all other age groups. (Appendix 4). The majority of macrolides were prescribed for OM among those aged <6 years. In those aged 6 to <18 years, pharyngitis, viral RTIs, and pneumonia were the leading diagnoses treated with macrolides (Table 3).
TABLE 3.
Distribution of Diagnoses Linked With a Second-Generation Macrolide Dispensing at Each Health Plan in 2009–2010
| Age Group | Plan | OM (%) | Pneumonia (%) | Pharyngitis (%) | Sinusitis (%) | Other Bacterial Infections (%) | Presumed Viral RTI (%) | All Other (%)a |
|---|---|---|---|---|---|---|---|---|
| 3 to <24 mo | ||||||||
| A | 58 | 7 | 2 | 4 | NAb | 15 | 15 | |
| Bc | 49 | 12 | 3 | 2 | NAb | 8 | 20 | |
| C | 51 | 11 | 3 | 3 | NAb | 16 | 16 | |
| 2 to <4 y | ||||||||
| A | 38 | 13 | 7 | 7 | NAb | 20 | 15 | |
| B | 24 | 20 | 12 | 5 | NAb | 16 | 22 | |
| C | 34 | 15 | 9 | 5 | NAb | 21 | 16 | |
| 4 to <6 y | ||||||||
| A | 27 | 15 | 12 | 6 | NAb | 22 | 18 | |
| B | 16 | 23 | 20 | 6 | NAb | 14 | 20 | |
| C | 23 | 15 | 16 | 6 | NAb | 22 | 17 | |
| 6 to <12 y | ||||||||
| A | 16 | 15 | 20 | 8 | 1 | 20 | 20 | |
| B | 10 | 28 | 21 | 6 | NAb | 14 | 21 | |
| C | 16 | 14 | 21 | 9 | 1 | 21 | 19 | |
| 12 to <18 y | ||||||||
| A | 9 | 9 | 16 | 16 | 2 | 22 | 27 | |
| Bc | 7 | 27 | 8 | 11 | 2 | 13 | 31 | |
| C | 9 | 9 | 18 | 15 | 2 | 22 | 25 |
NA, not applicable.
Includes dispensings unlinked to diagnoses.
Less than 0.5%.
Due to an antibiotic rate <0.5% among multiple diagnoses in a combined category, total percentage may not add to 100%.
Discussion
The downward trend in antibiotic dispensing slowed substantially or reversed between 2000–2001 and 2009–2010 in these 3 plans and across all age groups. In the early part of this decade, rates declined relatively rapidly, particularly in the 3- to <24-month age group. Later in the decade, this decline slowed or even reversed, as noted in adolescents. This finding is similar to that of Greene et al24 in a smaller sample of children aged <6 years in Massachusetts. To our knowledge, our study results reflect the first clear evidence of stabilization of antibiotic-dispensing rates across geographically distinct regions of the United States.
Our findings provide compelling evidence that continued improvements in judicious antibiotic use are possible. We found that the decline in antibiotic use was driven more by a decline in the OM diagnosis rate than by changes in the management of OM once diagnosed. This finding may be due to guidelines and education aimed at narrowing the diagnostic criteria for acute OM, as well as use of the conjugated pneumococcal vaccine.18,34,35 Because OM continues to be the primary driver of antibiotics dispensed (particularly for those aged <6 years), any additional decline in antibiotic rates will likely be driven by changes in diagnosis and management of this common infection. In contrast, we found a relatively low proportion of antibiotics prescribed for presumed viral infections compared with previous studies,26,36,37 suggesting that targeting antibiotic treatment of episodes explicitly labeled as “viral” is unlikely to result in large decreases in antibiotic use.
Use of second-generation macrolides and third-generation cephalosporins continued despite attention to issues of overuse of these agents in the last decade.23,26,38–43 Macrolides are frequently prescribed for OM or associated with visits for diagnosed viral infections, particularly among adolescents. Broad-spectrum cephalosporins are most prescribed for OM, pharyngitis, and sinusitis. Clinical guidelines for management of these common childhood infections include clear statements that neither macrolides nor third-generation cephalosporins are appropriate initial agents14–17 and may contribute to increased costs of care.44 Although use of these drugs may be considered in cases of penicillin allergy or for clinical failure of a first-line agent, understanding the driving forces for the upward trend in use of these broad-spectrum agents is vital to designing interventions to reduce their unwarranted use.
We were surprised at the plan-level variation in antibiotic dispensing, with plan B having lower dispensing rates across all age groups. We do not know whether these findings represent differences in regional prescribing patterns, the influence of local health plan interventions, or the consequence of care-seeking patterns in these populations. Furthermore, even within a health plan, individual- and practice-level prescribing rates may vary. During this study period, a variety of antimicrobial stewardship efforts were implemented at the health plan, state, and national levels, including educational campaigns directed at both prescribers and patients.2,3,19,45 A recent paper by Hicks et al46 affirms the wide geographic variation in prescribing that align with data from our study. In addition, other concurrent antibiotic stewardship efforts and electronic medical record modifications specifically at plan B could have influenced the low prescribing rates noted.
Our study analysis (in 3 defined populations) complements data from studies using the National Ambulatory Medical Care Survey, which estimates rates from visit-level data. In contrast to these studies,22,25,26 our data represent all new and refilled ambulatory prescriptions during the study period for enrolled patients, not just those that occur at an office visit. As with all studies based on health insurance claims, miscoding by providers can occur. However, changes in coding practice would have to vary across years for our findings regarding stabilization of rates to be the result of miscoding. Our data also lack individual-level demographic data, such as race, gender, and ethnicity, which have been previously associated with modest differences in rates of antibiotic prescribing. Finally, these findings reflect data from 3 health plans and may not be representative of trends in other parts of the country, other health plan models, or other populations in these regions.
Optimal antibiotic prescribing balances the consequences of overtreatment of viral syndromes and undertreatment of bacterial infections, the diagnosis of which is not always certain.47,48 Use of antimicrobial stewardship programs, institution of clinical practice guidelines, and electronic medical record changes have been used to decrease unnecessary antibiotic prescribing, with varied success.49 Recent interventions have been reported to decrease inappropriate antibiotic use.45,50–52 Renewed efforts to decrease inappropriate broad-spectrum antibiotic use may be especially important given the current dispensing trends found in the current study. Although the optimal rate of antibiotic dispensing is unknown, providers in 1 health plan studied here consistently prescribed fewer antibiotics. Further reductions in total dispensing may be achievable by understanding the differences in approach between lower and higher prescribing plans. These data also suggest that after years of decline in rates based on general messages that promote judicious prescribing, further progress may require focus on particular age groups, diagnoses, or antibiotic classes. New interventions tailored to local prescribing patterns at the level of the health system, practice, or clinician may also be needed.
Conclusions
Our data suggest that the downward trend in antibiotic use for children, observed for more than a decade, is coming to an end. This finding seems to be true across all pediatric age groups. However, because the highest rates of antibiotic use continue to be in young children, decreasing broad-spectrum use for common conditions such as OM will need to be a continuing focus for intervention. Because prescribing rates vary considerably, further efforts to improve judicious antibiotic use are also likely to benefit from identifying best practices in low-prescribing areas and health care delivery systems.
Acknowledgments
The authors thank Adam Jackson, Frances Lanty, David Price (Kaiser Permanente-CO), and Peter Marshall (HealthPartners) for providing contextual background and perspectives regarding judicious antibiotic use at their respective institutions.
Glossary
- CI
confidence interval
- OM
otitis media
- RTI
respiratory tract infection
APPENDIX 1.
Oral Antibiotic Categories
| Antibiotic Category | Individual Antibiotics |
|---|---|
| First-line penicillins | Penicillin V potassium, ampicillin, amoxicillin, dicloxacillin, oxacillin |
| Second-line penicillins | Amoxicillin/clavulanate |
| First-generation macrolides | Erythromycin |
| Second-generation macrolides | Azithromycin, clarithromycin, telithromycin |
| First-generation cephalosporins | Cephalexin, cefadroxil |
| Second-generation cephalosporins | Cefuroxime, cefaclor, loracarbef, cefprozil |
| Third-generation cephalosporins | Cefdinir, cefixime, cefpodoxime, ceftibuten, cefditoren |
| Tetracyclines | Tetracycline, doxycycline, minocycline, oxytetracycline HCl, oxytetracycline HCl/ sulfamethoxazole /phenazopyridine |
| Other | Sulfamethoxazole-trimethoprim, clindamycin, ciprofloxacin, levofloxacin, moxifloxacin, norfloxacin, lomefloxacin, ofloxacin, vancomycin, metronidazole, nitrofurantoin, sulfisoxazole, trimethoprim, ery e-succ/sulfisoxazole, sulfisoxazole/phenazopyridine HCl |
HCl, hydrochloride.
APPENDIX 2.
Diagnosis Categories With Associated ICD-9 Codes
| Illness | Diagnoses | ICD-9 Codes | |
|---|---|---|---|
| RTIs | Presumed bacterial origin | Pneumonia | 033.0, 0.33.1, 0.33.8, 033.9, 041.81, 481–486, 487.0, 510–11, 513 |
| OM | 381, 382, 384.0–384.2 | ||
| Pharyngitis | 034, 041.0, 462, 463 | ||
| Sinusitis | 461, 473 | ||
| Presumed viral origin | Bronchitis | 466.0, 490–491 | |
| Influenza | 487–488 | ||
| Viral pneumonia/bronchiolitis | 079.6, 466, 480 | ||
| Viral URI/common cold | 079.3, 460, 464, 464.0–464.2, 464.4, 465, 786.2 | ||
| Non-RTIs | Bacterial diagnoses | Urinary tract infections | 590, 595, 599 |
| Skin and soft tissue infections | 035, 289.3, 376.01, 680-686, 771.4–771.5, 910–919 (0.1, 0.3, 0.5) | ||
| Acne | 706 | ||
| Other bacterial diseases, including meningitis, and sepsis | 002–004, 008–010, 036, 038, 041.09–041.12, 041.2–041.9, 079.88–079.98, 088.81, 098, 320, 360, 464.3 | ||
| Viral diagnoses | Other viral illnesses | 008.6–008.8, 047, 079, 790.8, 373.13, 383, 390, 391, 421, 475, 478.22, 478.24, 519.01, 523, 527.3, 528.3, 597, 614, 616, 730 | |
| Other | Other, including well-child care | All other codes |
ICD-9, International Classification of Diseases, Ninth Revision; URI, upper respiratory infection.
APPENDIX 3.
RTI Visit Rate Per Person-Year, Total Antibiotic-Dispensing Rate Per Person-Year, and Antibiotic Rate Per Person-Year for Penicillins and Amoxicillin-Clavulanate, 2000–2001 Compared With 2009–2010
| Age | Plan | RTI Visit Rate/Person-Year | Total Antibiotic Rate/Person-Year | Penicillin Rate/Person-Year | Amoxicillin-Clavulanate Rate/ Person-Year | ||||
|---|---|---|---|---|---|---|---|---|---|
| 2000–2001 | 2009–2010 | 2000–2001 | 2009–2010 | 2000–2001 | 2009–2010 | 2000–2001 | 2009–2010 | ||
| 3 to <24 mo | |||||||||
| A | 3.95 | 3.37 | 2.27 | 1.62 | 1.22 | 0.79 | 0.35 | 0.26 | |
| B | 2.49 | 2.17 | 1.40 | 0.91 | 0.80 | 0.51 | 0.20 | 0.11 | |
| C | 4.28 | 3.30 | 2.23 | 1.70 | 0.97 | 0.77 | 0.34 | 0.20 | |
| 2 to <4 y | |||||||||
| A | 2.39 | 2.09 | 1.54 | 1.14 | 0.82 | 0.54 | 0.20 | 0.15 | |
| B | 1.34 | 1.30 | 0.88 | 0.62 | 0.53 | 0.34 | 0.09 | 0.06 | |
| C | 2.30 | 1.82 | 1.35 | 1.05 | 0.60 | 0.44 | 0.18 | 0.11 | |
| 4 to <6 y | |||||||||
| A | 1.91 | 1.66 | 1.35 | 1.04 | 0.75 | 0.49 | 0.16 | 0.11 | |
| B | 0.96 | 0.97 | 0.71 | 0.51 | 0.46 | 0.30 | 0.05 | 0.04 | |
| C | 1.78 | 1.44 | 1.11 | 0.91 | 0.57 | 0.42 | 0.13 | 0.08 | |
| 6 to <12 y | |||||||||
| A | 1.19 | 1.08 | 0.88 | 0.74 | 0.50 | 0.34 | 0.08 | 0.07 | |
| B | 0.63 | 0.63 | 0.47 | 0.36 | 0.30 | 0.19 | 0.03 | 0.02 | |
| C | 1.25 | 0.99 | 0.79 | 0.64 | 0.42 | 0.28 | 0.08 | 0.05 | |
| 12 to <18 y | |||||||||
| A | 0.73 | 0.65 | 0.84 | 0.74 | 0.28 | 0.18 | 0.05 | 0.06 | |
| B | 0.45 | 0.48 | 0.50 | 0.47 | 0.18 | 0.13 | 0.02 | 0.02 | |
| C | 0.75 | 0.62 | 0.75 | 0.69 | 0.23 | 0.16 | 0.04 | 0.04 | |
All changes from 2000–2001 to 2009–2010 were statistically significant (P < .001).
APPENDIX 4.
Antibiotic-Dispensing Rate Per Person-Year for Second-Generation Macrolides and the Cephalosporins, 2000–2001 Compared With 2009–2010
| Age | Plan | Second-Generation Macrolide Rate/Person-Year | First-Generation Cephalosporin Rate/Person-Year | Second-Generation Cephalosporin Rate/Person-Year | Third-Generation Cephalosporin Rate/Person-Year | ||||
|---|---|---|---|---|---|---|---|---|---|
| 2000–2001 | 2009–2010 | 2000–2001 | 2009–2010 | 2000–2001 | 2009–2010 | 2000–2001 | 2009–2010 | ||
| 3 to <24 mo | |||||||||
| A | 0.27 | 0.23 | 0.05 | 0.04 | 0.11 | 0.06 | 0.08 | 0.13 | |
| B | 0.09 | 0.05 | 0.05 | 0.04 | 0.11 | NAa | 0.01 | 0.15 | |
| C | 0.34 | 0.30 | 0.05 | 0.05 | 0.25 | 0.07 | 0.04 | 0.22 | |
| 2 to <4 years | |||||||||
| A | 0.24 | 0.23 | 0.06 | 0.06 | 0.06 | 0.03 | 0.04 | 0.07 | |
| B | 0.06 | 0.06 | 0.05 | 0.05 | 0.04 | NAa | 0.01 | 0.08 | |
| C | 0.22 | 0.23 | 0.06 | 0.05 | 0.12 | 0.04 | 0.01 | 0.11 | |
| 4 to <6 y | |||||||||
| A | 0.20 | 0.23 | 0.06 | 0.07 | 0.05 | 0.03 | 0.03 | 0.05 | |
| B | 0.05 | 0.05 | 0.05 | 0.05 | 0.02 | NAa | NAa | 0.04 | |
| C | 0.16 | 0.21 | 0.06 | 0.05 | 0.08 | 0.03 | 0.01 | 0.07 | |
| 6 to <12 y | |||||||||
| A | 0.13 | 0.16 | 0.06 | 0.08 | 0.03 | 0.02 | 0.01 | 0.02 | |
| B | 0.03 | 0.05 | 0.04 | 0.04 | 0.01 | NAa | NAa | 0.01 | |
| C | 0.11 | 0.16 | 0.06 | 0.05 | 0.04 | 0.02 | NAa | 0.03 | |
| 12 to <18 y | |||||||||
| A | 0.11 | 0.14 | 0.07 | 0.08 | 0.02 | 0.01 | NAa | 0.01 | |
| B | 0.03 | 0.06 | 0.05 | 0.04 | 0.01 | 0.01 | NAa | NAa | |
| C | 0.08 | 0.13 | 0.06 | 0.06 | 0.02 | 0.02 | NAa | 0.01 | |
All changes from 2000–2001 to 2009–2010 were statistically significant (p <0.001). NA, not applicable.
Antibiotic rate <0.01 dispensing per person-year.
Footnotes
Dr Vaz conceptualized and designed the study, conducted the analyses and interpreted the data, and drafted the manuscript; Dr Kleinman conceptualized the study, supervised the analyses, and reviewed and revised the manuscript; Drs Raebel and Nordin contributed to data acquisition, assisted in interpretation of the results, and edited the manuscript for critical content; Mr Lakoma conducted the analyses, interpreted the data, and revised the manuscript; Ms Dutta-Linn interpreted the data and revised the manuscript; Dr Finkelstein conceptualized and designed the study, supervised all analyses, assisted in interpretation of the results, and edited the manuscript for critical content; and all authors approved the final manuscript as submitted.
FINANCIAL DISCLOSURE: The authors have indicated they have no financial relationships relevant to this article to disclose.
FUNDING: Dr Vaz was supported under T32 training grants 5T32HD066148-05 and 1T32HD075727-01. All phases of this study were supported by a National Institutes of Health grant (1K24HD060786). Funded by the National Institutes of Health (NIH).
POTENTIAL CONFLICT OF INTEREST: The authors have indicated they have no potential conflicts of interest to disclose.
COMPANION PAPER: A companion to this article can be found on page 533, and online at www.pediatrics.org/cgi/doi/10.1542/peds.2013-4016.
References
- 1.Centers for Disease Control and Prevention. Get Smart: pediatric treatment guidelines for URIs. Available at: www.cdc.gov/getsmart/campaign-materials/pediatric-treatment.html. Updated July1, 2013. Accessed July 8, 2013
- 2.Belongia EA, Knobloch MJ, Kieke BA, Davis JP, Janette C, Besser RE. Impact of statewide program to promote appropriate antimicrobial drug use. Emerg Infect Dis. 2005;11(6):912–920 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gonzales R, Corbett KK, Leeman-Castillo BA, et al. The “minimizing antibiotic resistance in Colorado” project: impact of patient education in improving antibiotic use in private office practices. Health Serv Res. 2005;40(1):101–116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Paschke AA, Zaoutis T, Conway PH, Xie D, Keren R. Previous antimicrobial exposure is associated with drug-resistant urinary tract infections in children. Pediatrics. 2010;125(4):664–672 [DOI] [PubMed] [Google Scholar]
- 5.Costelloe C, Metcalfe C, Lovering A, Mant D, Hay AD. Effect of antibiotic prescribing in primary care on antimicrobial resistance in individual patients: systematic review and meta-analysis. BMJ. 2010;340(18):c2096. [DOI] [PubMed] [Google Scholar]
- 6.Institute of Medicine. Antibiotic Resistance: Implications for Global Health and Novel Intervention Strategies. In: Choffnes E, Relman D, Mack A, eds. Washington, DC: National Academies Press; 2010. Available at: www.nap.edu/openbook.php?record_id=12925&page=1. Accessed July 15, 2013 [PubMed]
- 7.Institute of Medicine. Antimicrobial Resistance: Issues and Options In: Harrison PF, Lederberg J, eds. Washington, DC: National Academies Press; 1998. Available at: www.nap.edu/openbook.php?record_id=6121&page=1. Accessed July 15, 2013 [PubMed]
- 8.Schwartz B, Marcy SM, Phillips WR, Gerber MA, Dowell SF. Pharyngitis—principles of judicious use of antimicrobial agents. Pediatrics. 1998;101(suppl 1):171–174
- 9.Rosenstein N, Phillips WR, Gerber MA, Marcy SM, Schwartz B, Dowell SF. The common cold—principles of judicious use of antimicrobial agents. Pediatrics. 1998;101(suppl 1):181–184
- 10.O’Brien K, Dowell S, Schwartz B, Marcy S, Phillips W, Gerber M. Cough illness/bronchitis—principles of judicious use of antimicrobial agents. Pediatrics. 1998;101(suppl 1):178–181 [Google Scholar]
- 11.Schwartz B, Marcy SM, Phillips WR, Gerber MA, Dowell SF, Rosenstein N. Otitis media—principles of judicious use of antimicrobial agents. Pediatrics. 1998;101(suppl 1):165–171
- 12.O’Brien K, Dowell S, Schwartz B. Acute sinusitis—principles of judicious use of antimicrobial agents. Pediatrics. 1998;101(suppl 1):174–177
- 13.Dowell SF, Marcy SM, Phillips WR, Gerber MA, Schwartz B. Principles of judicious use of antimicrobial agents for pediatric upper respiratory tract infections. Pediatrics. 1998;101(suppl 1):163–165
- 14.Chow AW, Benninger MS, Brook I, et al. Infectious Diseases Society of America . IDSA clinical practice guideline for acute bacterial rhinosinusitis in children and adults. Clin Infect Dis. 2012;54(8):e72–e112 [DOI] [PubMed] [Google Scholar]
- 15.Lieberthal AS, Carroll AE, Chonmaitree T, et al. The diagnosis and management of acute otitis media. Pediatrics. 2013;131(3). Available at: www.pediatrics.org/cgi/content/full/131/3/e964 [DOI] [PubMed] [Google Scholar]
- 16.Shulman ST, Bisno AL, Clegg HW, et al. Infectious Diseases Society of America . Clinical practice guideline for the diagnosis and management of group A streptococcal pharyngitis: 2012 update by the Infectious Diseases Society of America. Clin Infect Dis. 2012;55(10):e86–e102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bradley JS, Byington CL, Shah SS, et al. Pediatric Infectious Diseases Society and the Infectious Diseases Society of America . The management of community-acquired pneumonia in infants and children older than 3 months of age: clinical practice guidelines by the Pediatric Infectious Diseases Society and the Infectious Diseases Society of America. Clin Infect Dis. 2011;53(7):e25–e76 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hersh AL, Jackson MA, Hicks LA; Committee on Infectious Diseases. Principles of judicious antibiotic prescribing for upper respiratory tract infections in pediatrics. Pediatrics. 2013;132(6):1146–1154 [DOI] [PubMed] [Google Scholar]
- 19.Centers for Disease Control and Prevention. A public health action plan to combat antimicrobial resistance. Available at: www.cdc.gov/drugresistance/actionplan/actionplan.html. Updated May 23, 2013. Accessed July 15, 2013
- 20.Chai G, Governale L, McMahon AW, Trinidad JP, Staffa J, Murphy D. Trends of outpatient prescription drug utilization in US children, 2002-2010. Pediatrics. 2012;130(1):23–31 [DOI] [PubMed] [Google Scholar]
- 21.Finkelstein JA, Stille C, Nordin J, et al. Reduction in antibiotic use among US children, 1996-2000. Pediatrics. 2003;112(3 pt 1):620–627 [DOI] [PubMed] [Google Scholar]
- 22.McCaig LF, Hicks L, Roberts R, Fairlie T, Centers for Disease Control and Prevention (CDC) . Office-related antibiotic prescribing for persons aged ≤ 14 years—United States, 1993-1994 to 2007-2008. MMWR Morb Mortal Wkly Rep. 2011;60(34):1153–1156 [PubMed] [Google Scholar]
- 23.Grijalva CG, Nuorti JP, Griffin MR. Antibiotic prescription rates for acute respiratory tract infections in US ambulatory settings. JAMA. 2009;302(7):758–766 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Greene SK, Kleinman KP, Lakoma MD, et al. Trends in antibiotic use in Massachusetts children, 2000-2009. Pediatrics. 2012;130(1):15–22 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Halasa NB, Griffin MR, Zhu Y, Edwards KM. Decreased number of antibiotic prescriptions in office-based settings from 1993 to 1999 in children less than five years of age. Pediatr Infect Dis J. 2002;21(11):1023–1028 [DOI] [PubMed] [Google Scholar]
- 26.Hersh AL, Shapiro DJ, Pavia AT, Shah SS. Antibiotic prescribing in ambulatory pediatrics in the United States. Pediatrics. 2011;128(6):1053–1061 [DOI] [PubMed] [Google Scholar]
- 27.Zhang Y, Steinman MA, Kaplan CM. Geographic variation in outpatient antibiotic prescribing among older adults. Arch Intern Med. 2012;172(19):1465–1471 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.HMORN. About the HMO Research Network. Available at: www.hmoresearchnetwork.org/about.htm. Accessed July 15, 2013
- 29.Brown JS, Holmes JH, Shah K, Hall K, Lazarus R, Platt R. Distributed health data networks: a practical and preferred approach to multi-institutional evaluations of comparative effectiveness, safety, and quality of care. Med Care. 2010;48(suppl 6):45–51 [DOI] [PubMed]
- 30.Maro JC, Platt R, Holmes JH, et al. Design of a national distributed health data network. Ann Intern Med. 2009;151(5):341–344 [DOI] [PubMed] [Google Scholar]
- 31.Andrade SE, Raebel MA, Boudreau D, et al. Automated data systems: health maintenance organizations/health plans. In: Strom BL, Kimmel SEHS, eds. Pharmacoepidemiology. 5th ed. West Sussex, UK: Wiley-Blackwell; 2012:163–188 [Google Scholar]
- 32.National Committee for Quality Assurance. HEDIS 2011 final NDC lists: antibiotic utilization Table ABX-A. Available at: www.ncqa.org/tabid/1274/Default.aspx. Accessed April 1, 2012
- 33.Yanagimoto T, Yamamoto E. Estimation of safe doses: critical review of the hockey stick regression method. Environ Health Perspect. 1979;32(October):193–199 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Taylor S, Marchisio P, Vergison A, Harriague J, Hausdorff WP, Haggard M. Impact of pneumococcal conjugate vaccination on otitis media: a systematic review. Clin Infect Dis. 2012;54(12):1765–1773 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.American Academy of Pediatrics Subcommittee on Management of Acute Otitis . Diagnosis and management of acute otitis media. Pediatrics. 2004;113(5):1451–1465 [DOI] [PubMed] [Google Scholar]
- 36.Nyquist AC, Gonzales R, Steiner JF, Sande MA. Antibiotic prescribing for children with colds, upper respiratory tract infections, and bronchitis. JAMA. 1998;279(11):875–877 [DOI] [PubMed] [Google Scholar]
- 37.Nash DR, Harman J, Wald ER, Kelleher KJ. Antibiotic prescribing by primary care physicians for children with upper respiratory tract infections. Arch Pediatr Adolesc Med. 2002;156(11):1114–1119 [DOI] [PubMed] [Google Scholar]
- 38.Linder JA, Bates DW, Lee GM, Finkelstein JA. Antibiotic treatment of children with sore throat. JAMA. 2005;294(18):2315–2322 [DOI] [PubMed] [Google Scholar]
- 39.Kronman MP, Hersh AL, Feng R, Huang YS, Lee GE, Shah SS. Ambulatory visit rates and antibiotic prescribing for children with pneumonia, 1994-2007. Pediatrics. 2011;127(3):411–418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Shapiro DJ, Gonzales R, Cabana MD, Hersh AL. National trends in visit rates and antibiotic prescribing for children with acute sinusitis. Pediatrics. 2011;127(1):28–34 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Steinman MA, Gonzales R, Linder JA, Landefeld CS. Changing use of antibiotics in community-based outpatient practice, 1991-1999. Ann Intern Med. 2003;138(7):525–533 [DOI] [PubMed] [Google Scholar]
- 42.Stille CJ, Andrade SE, Huang SS, et al. Increased use of second-generation macrolide antibiotics for children in nine health plans in the United States. Pediatrics. 2004;114(5):1206–1211 [DOI] [PubMed] [Google Scholar]
- 43.Vernacchio L, Vezina RM, Mitchell AA. Management of acute otitis media by primary care physicians: trends since the release of the 2004 American Academy of Pediatrics/American Academy of Family Physicians clinical practice guideline. Pediatrics. 2007;120(2):281–287 [DOI] [PubMed] [Google Scholar]
- 44.Piccirillo JF, Mager DE, Frisse ME, Brophy RH, Goggin A. Impact of first-line vs second-line antibiotics for the treatment of acute uncomplicated sinusitis. JAMA. 2001;286(15):1849–1856 [DOI] [PubMed] [Google Scholar]
- 45.Gonzales R, Corbett KK, Wong S, et al. “Get smart Colorado”: impact of a mass media campaign to improve community antibiotic use. Med Care. 2008;46(6):597–605 [DOI] [PubMed] [Google Scholar]
- 46.Hicks LA, Taylor TH, Jr, Hunkler RJ. US outpatient antibiotic prescribing, 2010. N Engl J Med. 2013;368(15):1461–1462 [DOI] [PubMed] [Google Scholar]
- 47.Weissman J, Besser RE. Promoting appropriate antibiotic use for pediatric patients: a social ecological framework. Semin Pediatr Infect Dis. 2004;15(1):41–51 [DOI] [PubMed] [Google Scholar]
- 48.Finkelstein JA, Stille CJ, Rifas-Shiman SL, Goldmann D. Watchful waiting for acute otitis media: are parents and physicians ready? Pediatrics. 2005;115(6):1466–1473 [DOI] [PubMed] [Google Scholar]
- 49.Arnold SR, Straus SE. Interventions to improve antibiotic prescribing practices in ambulatory care. Cochrane Database Syst Rev. 2005;(4):CD003539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Finkelstein JA, Davis RL, Dowell SF, et al. Reducing antibiotic use in children: a randomized trial in 12 practices. Pediatrics. 2001;108(1):1–7 [DOI] [PubMed] [Google Scholar]
- 51.Gerber JS, Prasad PA, Fiks AG, et al. Effect of an outpatient antimicrobial stewardship intervention on broad-spectrum antibiotic prescribing by primary care pediatricians: a randomized trial. JAMA. 2013;309(22):2345–2352 [DOI] [PubMed] [Google Scholar]
- 52.Forrest CB, Fiks AG, Bailey LC, et al. Improving adherence to otitis media guidelines with clinical decision support and physician feedback. Pediatrics. 2013;131(4). Available at: www.pediatrics.org/cgi/content/full/131/4/e1071 [DOI] [PubMed] [Google Scholar]