Abstract
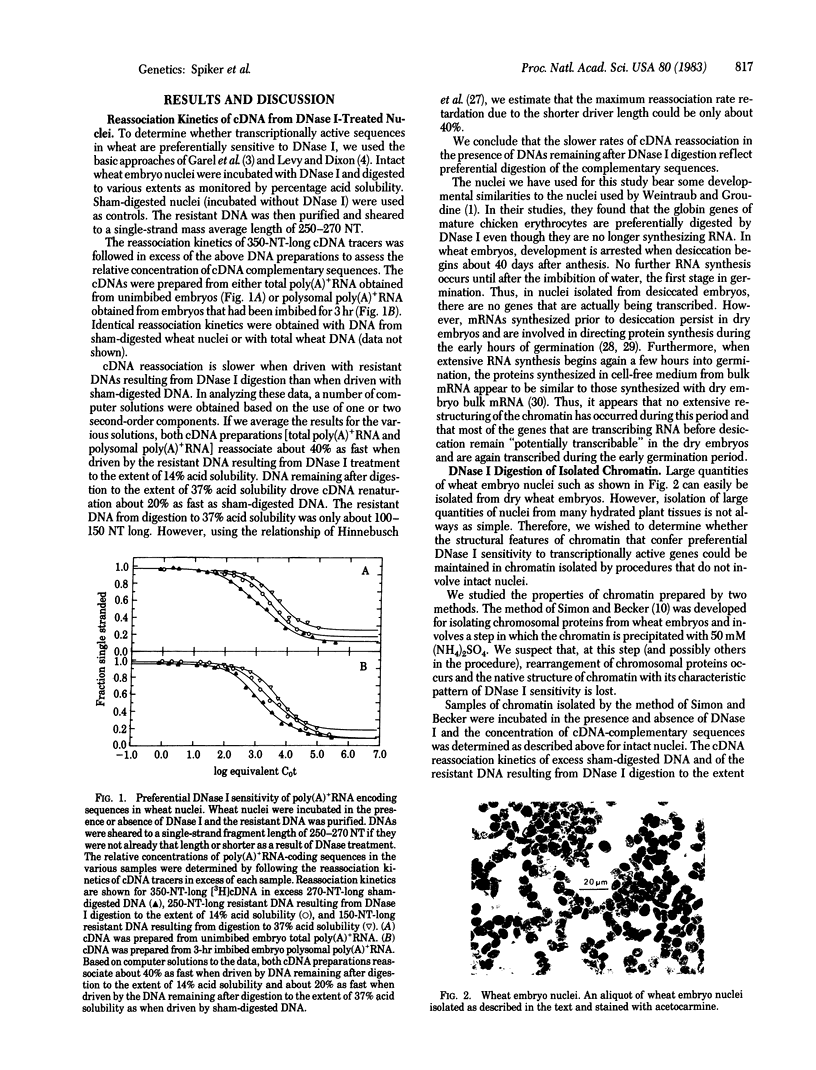
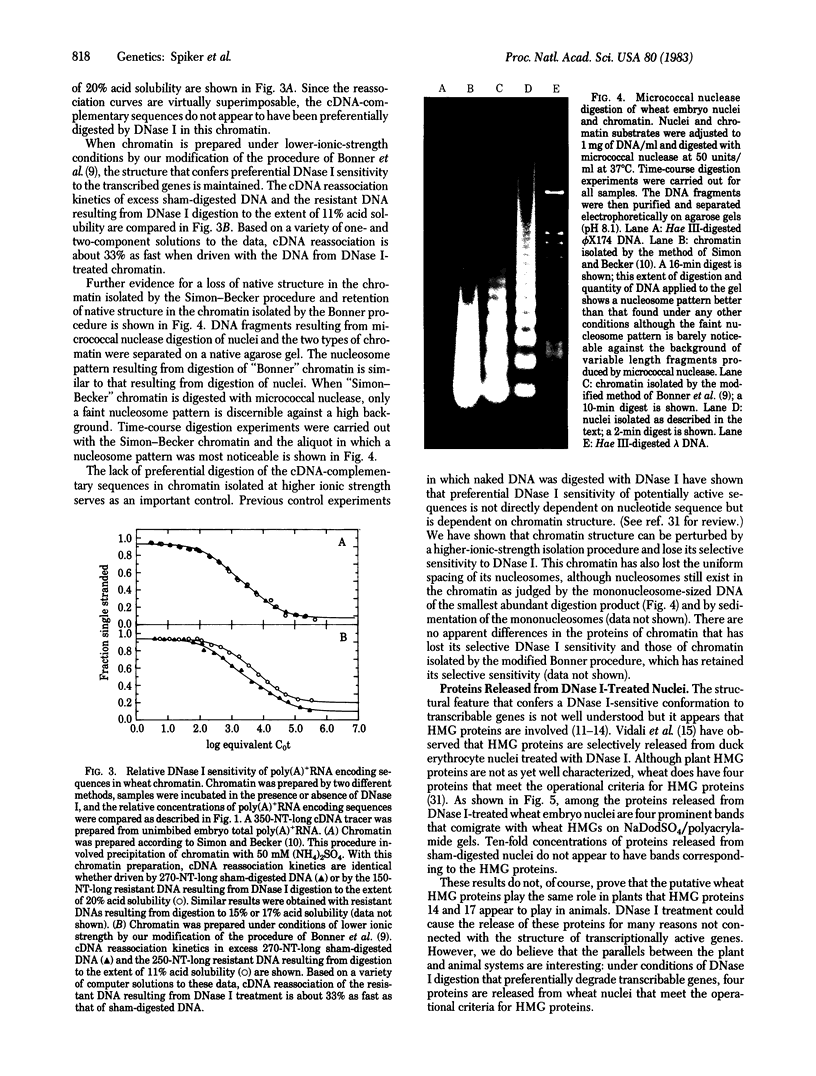
We have investigated the DNase I sensitivity of transcriptionally active DNA sequences in intact nuclei and isolated chromatin from embryos of wheat (Triticum aestivum L.). Nuclei or isolated chromatin was incubated with DNase I, and the extent of DNA digestion was monitored as percentage acid solubility. The resistant DNA and DNA from sham-digested controls were used to drive reassociation reactions with cDNA populations corresponding to either total poly(A)+RNA from unimbibed wheat embryos or polysomal poly(A)+RNA from embryos that had imbibed for 3 hr. Sequences complementary to either probe were depleted in DNase I-resistant DNA from nuclei and from chromatin isolated under low-ionic-strength conditions. This indicates that transcriptionally active sequences are preferentially DNase I sensitive in plants. In chromatin isolated at higher ionic strength, cDNA complementary sequences were not preferentially depleted by DNase I treatment. Therefore, the chromatin structure that confers preferential DNase I sensitivity to transcriptionally active genes appears to be lost when the higher-ionic-strength method of preparation is used. Treatment of wheat nuclei with DNase I causes the release of four prominent nonhistone chromosomal proteins that comigrate with wheat high mobility group proteins on NaDodSO4 gels.
Full text
PDF


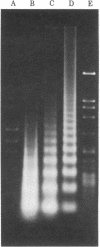
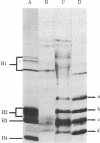
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Albanese I., Weintraub H. Electrophoretic separation of a class of nucleosomes enriched in HMG 14 and 17 and actively transcribed globin genes. Nucleic Acids Res. 1980 Jun 25;8(12):2787–2805. doi: 10.1093/nar/8.12.2787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bennett M. D. Nuclear DNA content and minimum generation time in herbaceous plants. Proc R Soc Lond B Biol Sci. 1972 Jun 6;181(1063):109–135. doi: 10.1098/rspb.1972.0042. [DOI] [PubMed] [Google Scholar]
- Britten R. J., Graham D. E., Neufeld B. R. Analysis of repeating DNA sequences by reassociation. Methods Enzymol. 1974;29:363–418. doi: 10.1016/0076-6879(74)29033-5. [DOI] [PubMed] [Google Scholar]
- Britten R. J., Kohne D. E. Repeated sequences in DNA. Hundreds of thousands of copies of DNA sequences have been incorporated into the genomes of higher organisms. Science. 1968 Aug 9;161(3841):529–540. doi: 10.1126/science.161.3841.529. [DOI] [PubMed] [Google Scholar]
- Brooker J. D., Tomaszewski M., Marcus A. Preformed Messenger RNAs and Early Wheat Embryo Germination. Plant Physiol. 1978 Feb;61(2):145–149. doi: 10.1104/pp.61.2.145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cuming A. C., Lane B. G. Wheat embryo ribonucleates XI. Conserved mRNA in dry wheat embryos and its relation to protein synthesis during early inhibition. Can J Biochem. 1978 Jun;56(6):365–369. doi: 10.1139/o78-058. [DOI] [PubMed] [Google Scholar]
- Garel A., Axel R. Selective digestion of transcriptionally active ovalbumin genes from oviduct nuclei. Proc Natl Acad Sci U S A. 1976 Nov;73(11):3966–3970. doi: 10.1073/pnas.73.11.3966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garel A., Zolan M., Axel R. Genes transcribed at diverse rates have a similar conformation in chromatin. Proc Natl Acad Sci U S A. 1977 Nov;74(11):4867–4871. doi: 10.1073/pnas.74.11.4867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giri C. P., Gorovsky M. A. DNase I sensitivity of ribosomal genes in isolated nucleosome core particles. Nucleic Acids Res. 1980 Jan 11;8(1):197–214. doi: 10.1093/nar/8.1.197-e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinnebusch A. G., Clark V. E., Klotz L. C. Length dependence in reassociation kinetics of radioactive tracer DNA. Biochemistry. 1978 Apr 18;17(8):1521–1529. doi: 10.1021/bi00601a026. [DOI] [PubMed] [Google Scholar]
- Klotz L. C., Zimm B. H. Size of DNA determined by viscoelastic measurements: results on bacteriophages, Bacillus subtilis and Escherichia coli. J Mol Biol. 1972 Dec 30;72(3):779–800. doi: 10.1016/0022-2836(72)90191-x. [DOI] [PubMed] [Google Scholar]
- Levy B., Dixon G. H. Renaturation kinetics of cDNA complementary to cytoplamic polyadenylated RNA from rainbow trout testis. Accessibility of transcribed genes to pancreatic DNase. Nucleic Acids Res. 1977 Apr;4(4):883–898. doi: 10.1093/nar/4.4.883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lohr D., Hereford L. Yeast chromatin is uniformly digested by DNase-I. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4285–4288. doi: 10.1073/pnas.76.9.4285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lohr D., Kovacic R. T., Van Holde K. E. Quantitative analysis of the digestion of yeast chromatin by staphylococcal nuclease. Biochemistry. 1977 Feb 8;16(3):463–471. doi: 10.1021/bi00622a020. [DOI] [PubMed] [Google Scholar]
- Mathis D., Oudet P., Chambon P. Structure of transcribing chromatin. Prog Nucleic Acid Res Mol Biol. 1980;24:1–55. doi: 10.1016/s0079-6603(08)60670-4. [DOI] [PubMed] [Google Scholar]
- Matsudaira P. T., Burgess D. R. SDS microslab linear gradient polyacrylamide gel electrophoresis. Anal Biochem. 1978 Jul 1;87(2):386–396. doi: 10.1016/0003-2697(78)90688-7. [DOI] [PubMed] [Google Scholar]
- McDonell M. W., Simon M. N., Studier F. W. Analysis of restriction fragments of T7 DNA and determination of molecular weights by electrophoresis in neutral and alkaline gels. J Mol Biol. 1977 Feb 15;110(1):119–146. doi: 10.1016/s0022-2836(77)80102-2. [DOI] [PubMed] [Google Scholar]
- Murray M. G., Cuellar R. E., Thompson W. F. DNA sequence organization in the pea genome. Biochemistry. 1978 Dec 26;17(26):5781–5790. doi: 10.1021/bi00619a027. [DOI] [PubMed] [Google Scholar]
- Pearson W. R., Davidson E. H., Britten R. J. A program for least squares analysis of reassociation and hybridization data. Nucleic Acids Res. 1977 Jun;4(6):1727–1737. doi: 10.1093/nar/4.6.1727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silflow C. D., Hammett J. R., Key J. L. Sequence complexity of polyadenylated ribonucleic acid from soybean suspension culture cells. Biochemistry. 1979 Jun 26;18(13):2725–2731. doi: 10.1021/bi00580a006. [DOI] [PubMed] [Google Scholar]
- Simon J. H., Becker W. M. A polyethylene glycol/dextran procedure for the isolation of chromatin proteins (histones and nonhistones) from wheat germ. Biochim Biophys Acta. 1976 Nov 12;454(1):154–171. doi: 10.1016/0005-2787(76)90362-2. [DOI] [PubMed] [Google Scholar]
- Sledziewski A., Young E. T. Chromatin conformational changes accompany transcriptional activation of a glucose-repressed gene in Saccharomyces cerevisiae. Proc Natl Acad Sci U S A. 1982 Jan;79(2):253–256. doi: 10.1073/pnas.79.2.253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spiker S., Mardian J. K., Isenberg I. Chomosomal HMG proteins occur in three eukaryotic kingdoms. Biochem Biophys Res Commun. 1978 May 15;82(1):129–135. doi: 10.1016/0006-291x(78)90586-7. [DOI] [PubMed] [Google Scholar]
- Thomas J. O., Kornberg R. D. An octamer of histones in chromatin and free in solution. Proc Natl Acad Sci U S A. 1975 Jul;72(7):2626–2630. doi: 10.1073/pnas.72.7.2626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson E. W., Lane B. G. Relation of protein synthesis in imbibing wheat embryos to the cell-free translational capacities of bulk mRNA from dry and imbibing embryos. J Biol Chem. 1980 Jun 25;255(12):5965–5970. [PubMed] [Google Scholar]
- Vidali G., Boffa L. C., Allfrey V. G. Selective release of chromosomal proteins during limited DNAase 1 digestion of avian erythrocyte chromatin. Cell. 1977 Oct;12(2):409–415. doi: 10.1016/0092-8674(77)90117-9. [DOI] [PubMed] [Google Scholar]
- Weintraub H., Groudine M. Chromosomal subunits in active genes have an altered conformation. Science. 1976 Sep 3;193(4256):848–856. doi: 10.1126/science.948749. [DOI] [PubMed] [Google Scholar]
- Weisbrod S., Groudine M., Weintraub H. Interaction of HMG 14 and 17 with actively transcribed genes. Cell. 1980 Jan;19(1):289–301. doi: 10.1016/0092-8674(80)90410-9. [DOI] [PubMed] [Google Scholar]
- Weisbrod S., Weintraub H. Isolation of a subclass of nuclear proteins responsible for conferring a DNase I-sensitive structure on globin chromatin. Proc Natl Acad Sci U S A. 1979 Feb;76(2):630–634. doi: 10.1073/pnas.76.2.630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weisbrod S., Weintraub H. Isolation of actively transcribed nucleosomes using immobilized HMG 14 and 17 and an analysis of alpha-globin chromatin. Cell. 1981 Feb;23(2):391–400. doi: 10.1016/0092-8674(81)90134-3. [DOI] [PubMed] [Google Scholar]