Abstract
Advances in brachytherapy treatment planning systems have allowed the opportunity for brachytherapy to be planned intraoperatively as well as preoperatively. The relative advantages and disadvantages of each approach have been the subject of extensive debate, and some contend that the intraoperative approach is vital to the delivery of optimal therapy. The purpose of this study was to determine whether high-quality permanent prostate implants can be consistently achieved using a preoperative planning approach that allows for, but does not necessitate, intraoperative optimization. To achieve this purpose, we reviewed the records of 100 men with intermediate-risk prostate cancer who had been prospectively treated with brachytherapy monotherapy between 2006 and 2009 at our institution. All patients were treated with iodine-125 stranded seeds; the planned target dose was 145 Gy. Only 8 patients required adjustments to the plan on the basis of intraoperative findings. Consistency and quality were assessed by calculating the correlation coefficient between the planned and implanted amounts of radioactivity and by examining the mean values of the dosimetric parameters obtained on preoperative and 30 days postoperative treatment planning. The amount of radioactivity implanted was essentially identical to that planned (mean planned radioactivity, 41.27 U vs. mean delivered radioactivity, 41.36 U; R2 = 0.99). The mean planned and day 30 prostate V100 values were 99.9% and 98.6%, respectively. The mean planned and day 30 prostate D90 values were 186.3 Gy and 185.1 Gy, respectively. Consistent, high-quality prostate brachytherapy treatment plans can be achieved using a preoperative planning approach, mostly without the need for intraoperative optimization. Good quality assurance measures during simulation, treatment planning, implantation, and postimplant evaluation are paramount for achieving a high level of quality and consistency.
Keywords: prostate cancer, brachytherapy, iodine-125
Introduction
Transperineal interstitial permanent prostate brachytherapy is a routinely used treatment modality for men with localized prostate cancer.1 Advances in brachytherapy treatment planning systems have allowed the brachytherapist to model the radiation dose to target volumes and organs at risk more rapidly than ever before. This evolution has created a dichotomy in brachytherapy treatment planning: the opportunity for brachytherapy to be planned intraoperatively as well as preoperatively.
Typically, there are two general accepted methods to prostate brachytherapy treatment planning.2-5 In the first method, intraoperative or real time treatment planning, all procedures are carried out in the operating room on the day of the implant. These procedures include, setting up the patient, determination of the prostate volume, treatment planning and implantation of the patient with pre-ordered radioactive sources. In the second method namely pre-planning, the prostate implant process is broken up into two stages. In the first stage, the patient undergoes an ultrasound simulation sometimes also referred to as volume study. The simulation consists of, setting up the patient in an identical position as the actual implant, determination of the prostate volume and other prostate dimensions, evaluation for pubic bone interference and overall evaluation to decide whether the patient is a candidate for prostate brachytherapy. Upon completion of their simulation, the patient goes home and returns for their implant at a later scheduled date, typically within two to four weeks. During this period, a treatment plan is generated and seeds are ordered based on the treatment plan needle loading pattern. Quality assurance is performed on the seeds, which are stranded, placed into needles, and, sterilized. In the second stage of the pre-planning method, patients are setup in the operating room in an identical position as that of their simulation, and implanted based on the treatment plan.
The relative advantages and disadvantages of each approach have been the subject of extensive debate.2-5 Proponents of the pre-planning technique cite that the technique is more cost effective because treatment planning and quality assurance is performed outside the operating room. Having a plan prior to the implant procedure saves valuable operating room time and allows for the use of pre-loaded needles which entail little or no seed waste. Commonly cited advantages of intraoperative treatment planning over preoperative planning include improved accuracy of prostate volume studies, elimination of the need for a preplan volume study and the ability to adjust to unanticipated operative findings. Proponents of the intraoperative approach suggest that these advantages result in improved consistency and quality, as assessed by postimplant dosimetry.6 At our institution, we have adopted a hybrid approach in which the treatments are planned on the basis of preoperative imaging but modern brachytherapy treatment planning software is on hand in the operating room in case variation from the predefined treatment plan is necessary (intraoperative optimization). The goal of preoperative planning is to ensure an optimal treatment plan, while intraoperative optimization functions as a quality control step when needed. The purpose of this study was to evaluate our quality assurance process in patients treated using our approach by comparing the total radioactivity planned with the total radioactivity implanted. Furthermore, we sought to determine whether high-quality implantation results, as determined by postimplant dosimetry parameters, could be consistently achieved at our center using our planning approach.
Materials and Methods
We reviewed the medical records of 100 consecutive patients with intermediate-risk prostate cancer who had been prospectively treated at The University of Texas MD Anderson Cancer Center on an institutional review board-approved protocol. Patients received treatment from 2006 through 2009. All patients received permanent prostate brachytherapy as monotherapy with iodine-125 (I-125) seeds (Oncura, Plymouth Meeting, PA). Stranded seeds with a radioactivity of 0.497 U/seed were used in all cases to obtain a prescribed dose of 145 Gy to the target volume.
All patients underwent evaluation for prostate brachytherapy, including a detailed history and physical examination, laboratory testing, and pelvic computed tomography (CT) or magnetic resonance imaging (MRI). CT scans were obtained to evaluate the patients for potential pubic bone interference with needle insertion. Eligibility criteria for participation in the protocol included the following: clinical tumor stage T1 or T2; no evidence of gross extra-capsular extension, regional node involvement, or metastatic disease; and either a maximum Gleason score ≤ 7 and prostate-specific antigen (PSA) score < 10 ng/ml or a Gleason score ≤ 6 and a PSA score between 10 ng/ml and 15 ng/ml. In addition, patients could not have received hormone therapy.
All patients underwent scanning for treatment simulation in the dorsal-lithotomy position with a urinary catheter in place approximately 3 to 4 weeks prior to the implant procedure. Simulation scanning consisted of transrectal ultrasonography to determine the prostate volume and CT to determine pubic arch interference. Ultrasound images of the prostate were captured at 5-mm intervals and transferred to the VariSeed treatment planning system (Varian Medical Systems, Milpitas, CA); the prostate, seminal vesicles, rectum, urethra, and bladder were contoured on these images. A planning target volume with a margin of 3 mm around the prostate was generated, except posteriorly, where there was no margin. A treatment plan was then generated for each patient using the following planning parameter guidelines: prostate volume receiving at least the prescription dose (V100) > 95%, prostate volume receiving at least 150% of the prescription dose (V150) < 60%, prostate volume receiving at least 200% of the prescription dose (V200) < 20% and prescription dose that covers 90% of the prostate volume (D90) >100%; urethra volume receiving at least 200% of the prescription dose (U200) = 0%; and rectum volume receiving at least 100% of the prescription dose (R100) < 1 cm3. The amount of radioactivity, number and position of seeds, and loading pattern were optimized to meet these parameters. Two additional sterilized needles (1 with 2 stranded seeds and the other with 3) were purchased for each patient in addition to the planned stranded seeds. The extra I-125 stranded seeds were reserved for use at the treating physician's discretion in case intraoperative optimization planning indicated they were needed.
The physician who performed the simulation reproduced the patient position in the operating room. Radioactive sources were inserted into the prostate under ultrasound guidance according to the preoperative treatment plan. A coronal fluoroscopic image was obtained when the planned sources had been placed but before the implant procedure was concluded; these images were compared with those used in treatment planning to confirm the optimal source distribution. On the rare occasions when the planned implant seed positions or dose distribution did not match with the delivered seed positions or dose distributions, the implant was optimized in the operating room and additional stranded seeds were implanted to ensure complete target coverage while also ensuring both V150 < 60% and R100 < 1 cm3. This intraoperative optimization was performed using intraoperative ultrasound images and the VariSeed software to fully assess the necessity and consequences of any changes to the original plan. The number of patients that intraoperative optimization adjustments were made was documented.
After the procedure, all patients underwent immediate CT to evaluate prostate coverage and determine the postimplant day 0 (day of implant) dosimetric values. All patients underwent repeat CT on day 30 to evaluate the postimplant D30 (1 month after implant) dosimetry so that dosimetric data could be re-evaluated. Postoperative dose distributions and dosimetric values were determined on the basis of the day 30 CT scans. The postimplant structures were delineated on the day 30 CT images, with the prostate volume referenced to that on the preoperative planning sonogram. The target volume's position was determined relative to the base plane and the prostate-rectum interface, as determined at the time of strand placement.7
The planned dosimetry parameters were compared with the postimplant CT-based measurements taken on day 30. The amount of radioactivity planned was compared with the radioactivity actually implanted in terms of both amount of radioactivity and number of seeds. Correlations between the amount of activity planned and amount of activity implanted were generated by calculating R2 values. R2 is the coefficient of determination which is a statistical measure of how well the regression line approximates the real data points. R2 provides the goodness of fit of the data with an R2 value of 1.0 indicating that the regression line perfectly fits the data.
Results
The amount of radioactivity implanted was essentially identical to the amount of radioactivity predicted by the preoperative plan (Fig. 1). The average numbers of seeds planned and implanted were 83.04 (mean activity, 41.27 U) and 83.22 (mean activity, 41.36 U), respectively. Intraoperative adjustments were made for only 8 of the 100 patients (8%) whose records were reviewed. The mean planned prostate D90 value was 186.3 Gy (range, 160-200 Gy; standard deviation [SD], 6.2 Gy) and the mean day 30 prostate D90 value was 185.1 Gy (range, 140-220 Gy; SD, 14.4 Gy). The planned and postimplant prostate V100 values were similar (Fig. 2): The mean planned prostate V100 value was 99.9% (range, 99-100%; SD, 0%) and the mean day 30 prostate V100 value was 98.6% (range, 90-100%; SD, 1.5%). The intended rectal dose constraint (R100) was also routinely achieved, without significant deviation from the planned value (Fig. 3): The mean planned R100 value was 0.40 cm3 (range, 0-0.9 cm3; SD, 0.23 cm3), and the mean day 30 R100 value was 0.31 cm3 (range, 0-2.3 cm3; SD, 0.42 cm3).
Fig. 1.

Amount of radioactive planned versus amount of radioactivity implanted and results of the correlation analysis.
Fig. 2.
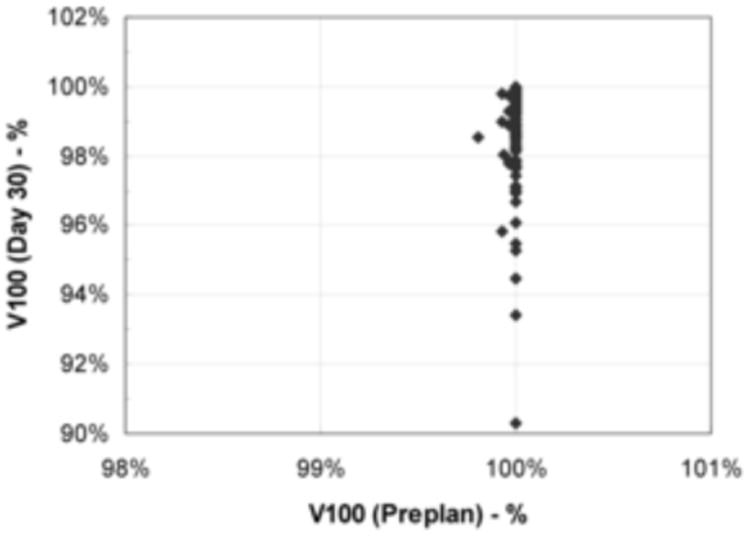
Planned prostate V100 values versus day 30 prostate V100 values.
Fig. 3.

Planned rectum R100 values versus day 30 rectum R100 values.
Discussion
In our study, we found a strong correlation between the planned and implanted amount of radioactivity, indicating that the preoperative plan was routinely executed in the operating room as intended. In the dichotomous world of prostate brachytherapy dosimetry, some have suggested that intraoperative (real-time) planning improves postimplant dosimetry. Zelefsky et al. reported apparent improvements in postimplant dose-volume histogram parameters after intraoperative planning compared with the parameters observed using a preoperative planning technique.6 Indeed, high-quality implants can be achieved using an intraoperative planning approach. However, our data demonstrate that an intraoperative approach is not required to achieve consistent, high-quality results. It is our opinion that the treatment planning philosophy of the brachytherapist (whether pre-planning or intraoperative planning) is not a major factor as compared to the process of quality assurance. The quality assurance process is essentially identical for either school of thought regardless of when planning is done and depends almost entirely upon the attention paid to appropriate patient selection, simulation, treatment planning, source delivery, and postimplant dosimetry.
A primary goal of the prostate brachytherapist, regardless of the chosen methodology, is to consistently achieve high-quality implantation results, as determined by postimplant dosimetry. Using accepted dosimetry standards of implant quality,8,9 we were able to demonstrate consistently high-quality results using a preoperative planning technique that allowed for, but mostly did not require, intraoperative adjustments. Furthermore, our study is unique in that we were able to demonstrate a strong correlation between the planned dosimetric features of the treatment using transrectal ultrasound images and the actual dosimetric features determined on postoperative CT images in our cohort.
Currently, the best gauge of permanent prostate brachytherapy implantation quality is postimplant dosimetry. Postimplant prostate V100 and D90 values predict biochemical tumor control following prostate brachytherapy, while the doses to surrounding nontarget structures have been correlated with the risk of adverse effects.10-15 We found a mean day 30 V100 value of almost 99%, a mean D90 value of just over 185 Gy, and a mean R100 value of 0.31 cm3, which demonstrates that high-quality implantation results can be achieved using a preoperative planning approach. Other investigators have also reported satisfactory postoperative dose-volume histogram parameters using a preoperative planning approach.16-18 Both Merrick et al. and Grimm et al. reported a median V100 > 90% and a D90 range of 145-160 Gy using CT-based dosimetry on postimplant days 0 and 1. Our study complements those data and confirms that the early positive results can persist over time by reporting that predefined dosimetric quality metrics were still achieved 30 days after the implant process. The mean D90 value calculated using CT images increased by an average of 23 Gy from day 0 to day 30, providing a quantitative assessment of edema resolution over this time interval.
In our study, the correlation between the amount of activity planned and the amount of activity delivered had an R2 value of 0.99, suggesting that the preoperative plan was routinely executed without variance. Our data are consistent with those of others who have shown a strong correlation between preoperative ultrasound-based and postimplant CT-based dosimetric values.18 Our study improves upon previously reported series, in that we were able to analyze a retrospectively collected dataset that included comprehensive preimplant and postimplant dosimetric information for each patient. In addition, our study not only provides data on target volume coverage but also includes an organ-at-risk assessment.
One potential limitation of our study is the inherent subjectivity of the target organ and organ-at-risk delineations on postimplant CT images. In an attempt to minimize the variance, we contoured the prostate volume relative to the initial plan. The planes of the implanted seeds at the base of the prostate and at the posterior prostate were used as points of reference, considering that these positions were conclusively verified at the time of implantation. The treating physician, who was primarily involved in all stages of the process, defined all contours. Although this approach does not exclude the possibility of error, we believe it optimizes consistency, despite the limitations of postimplant organ delineation. Implementation of efficient MRI-based treatment planning is likely to improve treatment accuracy in the future.19,20
Thus, consistent, high-quality implantation can be achieved using a preoperative planning approach. Despite its availability, intraoperative optimization was rarely necessary in this series. Regardless of the temporal relationship between the treatment planning and the actual implant procedure, good quality assurance measures – patient evaluation, simulation, treatment planning, implantation, and postimplant evaluation – at every step of the process are instrumental in achieving a high level of consistency.
Acknowledgments
The University of Texas MD Anderson Cancer Center is supported by the National Institutes of Health through grant CA16672.
Footnotes
Presented at the 52nd Annual Meeting of the American Association of Physicists in Medicine, July 18-22, 2010, Philadelphia, PA.
Conflict of Interest Statement: The authors have no conflicts of interest to disclose.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Potters L, Klein EA, Kattan MW, et al. Monotherapy for stage T1-T2 prostate cancer: radical prostatectomy, external beam radiotherapy, or permanent seed implantation. Radiother Oncol. 2004;71:29–33. doi: 10.1016/j.radonc.2003.12.011. [DOI] [PubMed] [Google Scholar]
- 2.Stone NN, Hong S, Lo YC, et al. Comparison of intraoperative dosimetric implant representation with postimplant dosimetry in patients receiving prostate brachytherapy. Brachytherapy. 2003;2:17–25. doi: 10.1016/S1538-4721(03)00005-9. [DOI] [PubMed] [Google Scholar]
- 3.Ishiyama H, Nakamura R, Satoh T, et al. Differences between intraoperative ultrasound-based dosimetry and postoperative computed tomography-based dosimetry for permanent interstitial prostate brachytherapy. Brachytherapy. 2010;9:219–23. doi: 10.1016/j.brachy.2009.09.007. [DOI] [PubMed] [Google Scholar]
- 4.Zelefsky MJ, Zaider M. Low-dose-rate brachytherapy for prostate cancer: preplanning vs. intraoperative planning-intraoperative planning is best. Brachytherapy. 2006;5:143–4. doi: 10.1016/j.brachy.2006.04.001. [DOI] [PubMed] [Google Scholar]
- 5.Blasko JC. Low-dose-rate brachytherapy for prostate cancer: preplanning vs. intraoperative planning-preplanning is best. Brachytherapy. 2006;5:139–42. doi: 10.1016/j.brachy.2006.04.004. [DOI] [PubMed] [Google Scholar]
- 6.Zelefsky MJ, Yamada Y, Marion C, et al. Improved conformality and decreased toxicity with intraoperative computer-optimized transperineal ultrasound-guided prostate brachytherapy. Int J Radiat Oncol Biol Phys. 2003;55:956–63. doi: 10.1016/s0360-3016(02)04142-1. [DOI] [PubMed] [Google Scholar]
- 7.Vassiliev ON, Kudchadker RJ, Swanson DA, et al. Displacement of periurethral stranded seeds and its dosimetric consequences in prostate brachytherapy. Brachytherapy. 2011;10:401–408. doi: 10.1016/j.brachy.2011.01.002. [DOI] [PubMed] [Google Scholar]
- 8.Rosenthal SA, Bittner NH, Beyer DC, et al. American Society for Radiation Oncology (ASTRO) and American College of Radiology (ACR) Practice Guideline for the Transperineal Permanent Brachytherapy of Prostate Cancer. Int J Radiat Oncol Biol Phys. 2011;79:335–41. doi: 10.1016/j.ijrobp.2010.08.045. [DOI] [PubMed] [Google Scholar]
- 9.Nag S, Beyer D, Friedland J, et al. American Brachytherapy Society (ABS) recommendations for transperineal permanent brachytherapy of prostate cancer. Int J Radiat Oncol Biol Phys. 1999;44:789–99. doi: 10.1016/s0360-3016(99)00069-3. [DOI] [PubMed] [Google Scholar]
- 10.Urbanic JJ, Lee WR. Update on brachytherapy in localized prostate cancer: the importance of dosimetry. Curr Opin Urol. 2006;16:157–61. doi: 10.1097/01.mou.0000193391.77469.39. [DOI] [PubMed] [Google Scholar]
- 11.Stock RG, Stone NN, Tabert A, et al. A dose-response study for I-125 prostate implants. Int J Radiat Oncol Biol Phys. 1998;41:101–8. doi: 10.1016/s0360-3016(98)00006-6. [DOI] [PubMed] [Google Scholar]
- 12.Kollmeier MA, Stock RG, Stone N. Biochemical outcomes after prostate brachytherapy with 5-year minimal follow-up: importance of patient selection and implant quality. Int J Radiat Oncol Biol Phys. 2003;57:645–53. doi: 10.1016/s0360-3016(03)00627-8. [DOI] [PubMed] [Google Scholar]
- 13.Papagikos MA, Deguzman AF, Rossi PJ, et al. Dosimetric quantifiers for low-dose-rate prostate brachytherapy: is V(100) superior to D(90)? Brachytherapy. 2005;4:252–8. doi: 10.1016/j.brachy.2005.09.001. [DOI] [PubMed] [Google Scholar]
- 14.Zelefsky MJ, Kuban DA, Levy LB, et al. Multi-institutional analysis of long-term outcome for stages T1-T2 prostate cancer treated with permanent seed implantation. Int J Radiat Oncol Biol Phys. 2007;67:327–33. doi: 10.1016/j.ijrobp.2006.08.056. [DOI] [PubMed] [Google Scholar]
- 15.Sherertz T, Wallner K, Merrick G, et al. Factors predictive of rectal bleeding after 103Pd and supplemental beam radiation for prostate cancer. Brachytherapy. 2004;3:130–5. doi: 10.1016/j.brachy.2004.05.005. [DOI] [PubMed] [Google Scholar]
- 16.Grimm PD, Blasko JC, Sylvester JE, et al. Technical improvement in permanent seed implantation: a two-stage brachytherapy system. Description and comparison with current technique. Brachytherapy. 2004;3:34–40. doi: 10.1016/j.brachy.2003.07.001. [DOI] [PubMed] [Google Scholar]
- 17.Merrick GS, Butler WM, Dorsey AT, et al. Effect of prostate size and isotope selection on dosimetric quality following permanent seed implantation. Tech Urol. 2001;7:233–40. [PubMed] [Google Scholar]
- 18.Al-Qaisieh B, Witteveen T, Carey B, et al. Correlation between pre- and postimplant dosimetry for iodine-125 seed implants for localized prostate cancer. Int J Radiat Oncol Biol Phys. 2009;75:626–30. doi: 10.1016/j.ijrobp.2009.04.002. [DOI] [PubMed] [Google Scholar]
- 19.Frank SJ, Tailor RC, Kudchadker RJ, et al. Anisotropy characterization of I-125 seed with attached encapsulated cobalt chloride complex contrast agent markers for MRI-based prostate brachytherapy. Med Dosim. 2011;36:200–5. doi: 10.1016/j.meddos.2010.03.004. [DOI] [PubMed] [Google Scholar]
- 20.Frank SJ, Stafford RJ, Bankson JA, et al. A novel MRI marker for prostate brachytherapy. Int J Radiat Oncol Biol Phys. 2008;71:5–8. doi: 10.1016/j.ijrobp.2008.01.028. [DOI] [PubMed] [Google Scholar]


