Abstract
Mutations of p53 cause not only loss of wild-type function but also gain of novel oncogenic functions (GOF). Accumulating evidence suggest that p53 hotspot mutations may confer different types and magnitudes of GOF. Here we add support to the heterogeneity of mutant p53 GOF by showing their unequal association with early tumor onset and spectrum of tumor types. We stratified Li-Fraumeni syndrome (LFS) patients according to carried p53 mutations using data from the updated p53 germline mutation database. When compared to loss-of-function nonsense mutations, the R282 GOF mutation associated with significantly earlier onset, while the G245 mutation displayed later onset. The R175, Y220, R248, R282 and nonsense mutations showed preferential distribution in certain cancer types, which varied in the age of onset. Multivariate COX regression model adjusting for cancer types and patient sex suggested that nonsense and G245 mutations had lower risk than R248 for early onset, suggesting unequal strengths of mutant GOF effects. Our results suggest that Li-Fraumeni syndrome can be subdivided into subtypes linking to unequal GOF effects of p53 mutations. These findings have potential implications in the prevention, early detection and targeted treatment of LFS tumors.
The TP53 gene is the most frequent target for mutation in human cancers, with over 50% of all observed tumors exhibiting mutation at this locus1. The tumor suppressive functions of wild-type p53 are often explained by its transcriptional functions in the nucleus. In response to oncogenic mutations or DNA damage, p53 triggers a transcriptional program of cell cycle arrest2, DNA repair3, senescence, apoptosis4, autophagy5, and metabolic remodeling6. The vast majority of p53 mutations in human cancer are missense mutations, sitting within the DNA-binding domain (amino acids 102–292) with hot spots at codons R175, Y220, G245, R248, R273 and R2827(illustrated in Figure 1a). Mutations are thought to inactivate p53 function by either disrupting its contact with DNA (R248W, R273H) or structurally destabilize the protein (R175H, Y220C, G245S, R248Q and R282W)8.
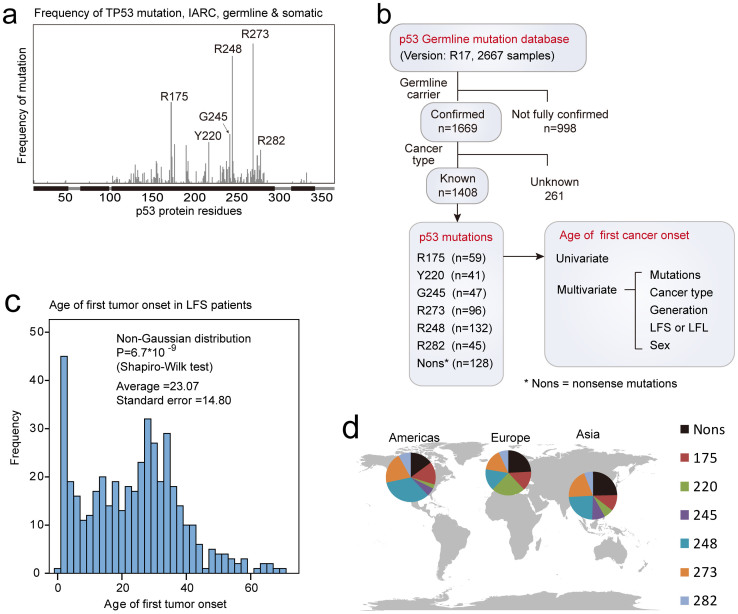
Figure 1. Hotspot mutations of p53 and design of the study.
(a) Positions of p53 hotspot gain-of-function (GOF) mutations. The x-axis indicates amino acid sequence of p53 protein, while the y-axis displays the frequency of p53 mutations in the IARC database. (b) Flowchart describing the design of the current study. The IARC p53 germline mutation dataset (release 17) were selected to retain only cases that were confirmed as germline carriers and diagnosed with cancers of known types. Further, cases were classified according to the mutations of p53 that they carried (number of cases indicated). The age of cancer diagnosis (onset) was compared between different groups using univariate Kaplan-Meier analysis. The effects of other factors (cancer type, generation, sex, and phenotype) were also analyzed by multivariate COX regression analysis. (c) The age of first cancer onset in LFS cases display non-Gaussian distribution. The x-axis indicates the age (years), and the y-axis shows the number of cases. (d) The distribution of TP53 germline mutations in different regions. Dataset were curated from published literatures, thus the plot represents source of data rather than natural occurrence of these mutations. The map was generated using a python script freely available from http://gsl-nagoya-u.net/appendix/software/worldmap/.
It is becoming increasingly evident that mutant p53 proteins not only lose their tumor-suppressor function but also acquire oncogenic gain of function (GOF)9,10. The most compelling support for GOF comes from mice engineered to harbour some of the hot spot tumor-associated p53 mutations. When compared to heterozygous or null (p53+/− or p53−/−) mice, animals with one mutant allele equivalent to human R175H or R273H showed more spontaneous carcinomas, sarcomas and lymphomas. In addition, unequal GOF effects have been displayed by different p53 hotspot mutations. Although R175H and R273H mutants can increase tumor metastasis, they do not shorten mouse survival time as compared to p53-null background11,12. Instead, the R248Q mutation was found with the abilities to accelerate tumor onset and shorten survival of mice13. These data suggest that mutant p53 may have different types and strengths of GOF effects, and thus should be considered as different proteins14.
Despite the findings in transgenic mouse models, limited evidence for mutant p53 GOF has been obtained from human cancers. Germline mutation of p53 is linked to an extremely rare autosomal dominant hereditary disorder named Li-Fraumeni syndrome (LFS), with a propensity to develop breast cancer, brain tumors, acute leukemia, soft tissue sarcomas, bone sarcomas, and adrenal cortical carcinoma15,16. However, it is still unclear if different p53 GOF mutations may confer equal susceptibility to cancers and display similar clinical features17,18. The onset of cancer is a result of many factors including the carcinogenic potential of certain activated oncogenes19, thus the GOF effects of p53 may affect the age of cancer onset. In this study, we compare the effects of p53 hotspot mutations (R175, Y220, G245, R248, R273, and R282) on the age of onset and the spectrum of tumors. Multivariate COX regression model was also employed to identify the factors that independently associate with earlier tumor onset. By these efforts we aim to seek evidence for unequal p53 GOF effects in human cancer onset, and present a proof-of-principle study on subtyping LFS according to the exact mutation form of p53.
Results
Germline mutations on R282 and G245 mark subgroups with distinct age of onset
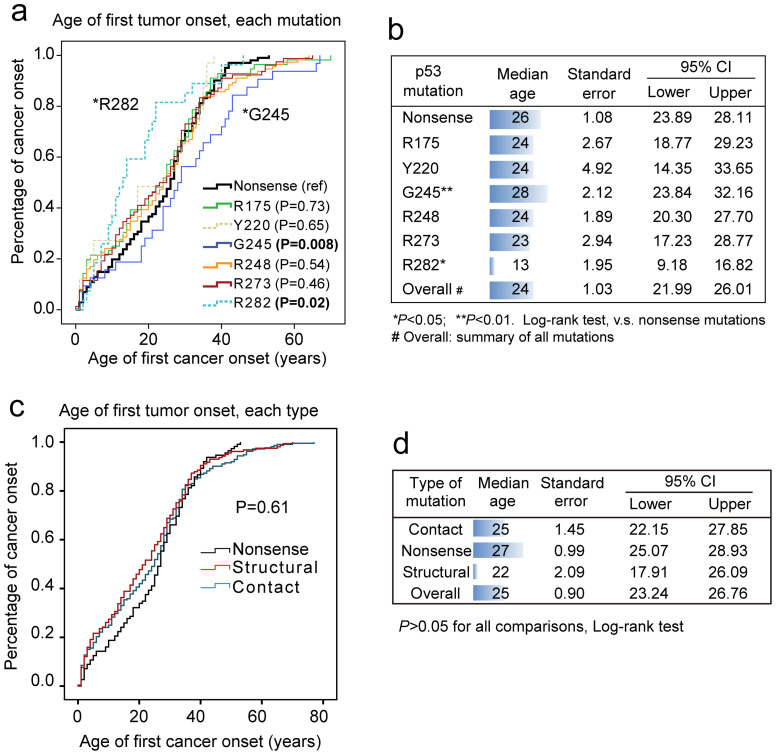
We firstly set stringent criteria to select the cases in the p53 germline mutation database (data processing flow chart shown in Figure 1b). The full dataset contained 2667 records, with 1669 cases fully confirmed as germline carriers of TP53 mutation. Further, data with known cancer types (1408 cases) were stratified according to p53 mutation (R175, Y220, G245, R273, R248, R282, or nonsense mutations). Only the age of first tumor onset for a given LFS case was included for further analysis. The age of first tumor onset in the selected LFS patients displayed non-Gaussian distribution (Figure 1c), and slightly different frequency of these mutations are found in Americas, Europe and Asia (Figure 1d). Univariate log-rank test revealed that mutation on R282 linked to significantly earlier onset than the nonsense (loss of function) mutations. Interestingly, the mutation on G245 associated with later onset when compared to nonsense mutations (Figure 2a&b). The other GOF mutations, including R175, Y220, R248 and R273 displayed no significant difference in this context. These results suggest that p53 GOF mutations are unequal in their effects on accelerating carcinogenesis, when considering any cancer onset as an event. When the structural and contact mutations were compared for the age of onset, the structural mutation displayed a trend for earlier onset. However, the difference was not statistically significant (Figure 2c&d).
Figure 2. Age of cancer onset (any type) compared between cases carrying different p53 hotspot mutations and loss-of-function (nonsense) mutations.
(a) Cases carrying mutations on the R282 position displayed significantly earlier onset (P = 0.02), while mutations on G245 residue showed later cancer onset (P = 0.008, both compared to nonsense mutations). No significant difference was found between other hotspot mutations. (b) The age of cancer onset (median with standard error and 95% confidence interval), are shown for each hotspot p53 mutation. (c) Univariate analysis showing the onset of first tumor in LFS patients carrying nonsense, structural or contact TP53 mutations. No significant difference was found between these groups. (d) The median ages of onset (with 95%CI) for different mutation types are indicated.
p53 GOF mutations associate with distinct tumor spectrum
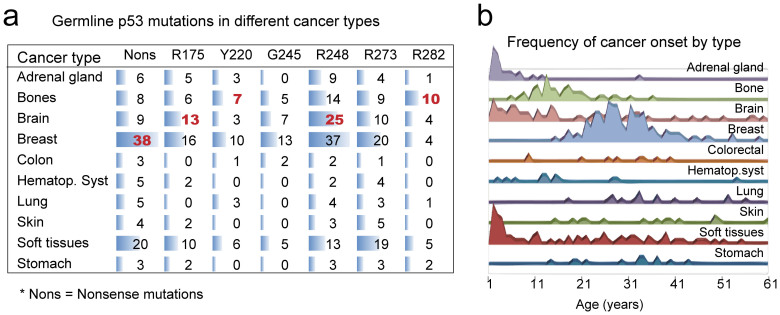
Further, we analyzed the tumor spectrum of different p53 germline mutations. To this end, the distribution of GOF mutations were compared between different cancer types using Chi-square test (Figure 3a&b). Interestingly, several mutations showed preferential distribution in certain cancer types. The R175 and R248 were enriched in brain tumors, while R282 and Y220 mutations were more frequently found in tumors of bones (P < 0.05). Moreover, the nonsense mutations were more prevalent in breast tumors (P < 0.05). Because Li-Fraumeni syndrome tumors in adrenal gland, brain, soft tissues and bone have much earlier onset than other types, the unbalanced distribution of p53 mutations in these tumor types (especially R282 in bone tumors) help to explain their unequal association with early onset.
Figure 3. Unequal distribution of p53 mutations in different cancer types affect the age of cancer onset.
(a) The distribution of each p53 mutation in the indicated tumor type. Horizontal bars in blue are used to indicate the quantities. Increased distributions with statistical significance are highlighted in red (Chi-square test, P < 0.05). Nonsense mutations are enriched in breast cancers, while R175 and R248 mutations are more frequent in brain tumors. The R282 and Y220 mutations are preferentially distributed in bone tumors. (b) The frequency of cancer onset as a function of time is plotted for each cancer type. Tumors in adrenal gland, bones, brain, hematopoietic system and soft tissue show earlier onset than other types. The dramatic enrichment of R282 mutation in bone tumors explains its association with early cancer onset.
Nonsense and G245 show weaker accelerating effect on cancer onset
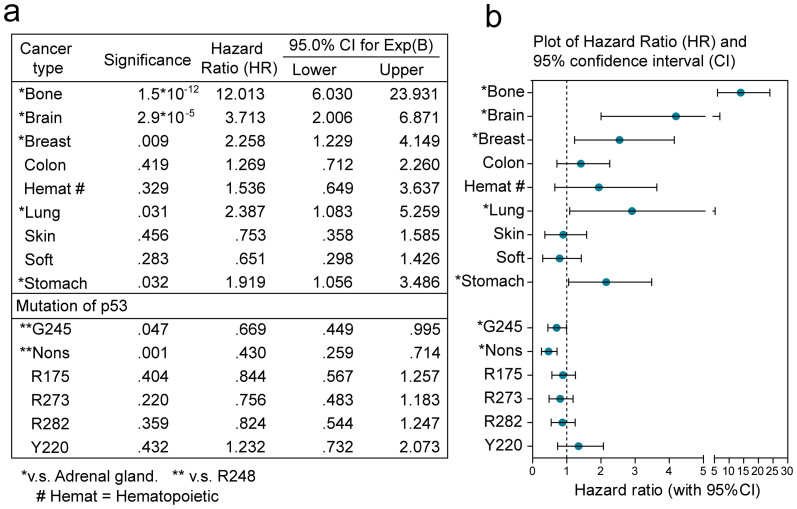
To evaluate the independent effect of p53 GOF on cancer onset, we used a multivariate COX regression model to adjust other factors including cancer types and sex of patient. The conditional forward regression model suggested that both cancer type and p53 mutation have significant effects on the age of cancer onset (Figure 4a&b). The patient sex was insignificant and thus not included in the model. The nonsense mutations were found with the weakest effect on accelerating cancer onset (P = 0.001, Hazard ratio = 0.430), followed by the G245 mutation (P = 0.047, Hazard ratio = 0.669). Other GOF mutations showed no significant difference with R248 mutation (reference condition in the model). It should be noted that the lack of significance may be due to the limited case numbers, and the results so far are at least sufficient to reject the assumption that p53 GOF mutations have equal effects on age of cancer onset.
Figure 4. Multivariate analysis on the effects of each p53 mutation on cancer onset.
(a) The potential effects of p53 mutation, cancer type, and patient sex on the age of cancer onset were analyzed by forward conditional COX regression model. The variables in the model include cancer types and mutations of p53. Tumors in several tissue types (including bone, brain, breast, lung and stomach) showed higher hazard ratios (HRs) than in adrenal gland, which was set as a reference condition. As for the germline allele status of TP53 gene, the nonsense and G245 mutations showed significantly later onset than the mutation on R248 (reference condition). Other hotspot mutations (including R175, R273, R282 and Y220) showed no significant difference in the age of cancer onset (P > 0.05). (b) Plot of hazard ratio and 95% confidence interval for each factor in the COX regression model.
Discussions
It is becoming increasingly evident that missense mutations may convert p53 from a tumor suppressor into an oncogene, and it is of both scientific and clinical interest to understand the GOF activities of mutant p53 ref. 20. While most studies focus on cellular and animal models to explore this link, our study evaluates the patterns and strengths of GOF effects on human cancer onset.
Using an integrated dataset of p53 germline mutations, we have systematically compared the effects of each p53 hotspot GOF mutation on cancer onset and spectrum. The results suggest that Li-Fraumeni syndrome (LFS) can be further subdivided to subtypes that are marked by different p53 GOF mutations. The LFS cases carrying R282 mutation showed even earlier tumor onset than those carrying nonsense mutations, but G245S mutation instead marked a subgroup of cases with an adverse trend (later onset than nonsense mutations). It is unexpected in the sense that missense mutations are generally assumed to display even higher oncogenic potential than nonsense mutations. Although it is unknown whether the exceptional effect of G245 mutation is due to partial loss of the wildtype function or gain of certain functions, these findings clearly demonstrated the heterogeneity within Li-Fraumeni syndrome.
The different effects of p53 germline mutations on tumor spectrum and age of onset suggest potential benefit for individualized healthcare practice according to which p53 mutation is carried by the family. For instance, patients bearing R175 and R248 mutations may be more frequently checked for brain tumors, while patients having R282 and Y220 mutations should be given special attention to potential osteosarcomas. In addition, the unequal biological activities of mutant p53 that are already evident in cellular and animal studies suggest that more targeted therapeutic strategies could be developed for the different subtypes of LFS patients.
Although this study is based on integrated dataset from many different studies, it still has rather limited number cases for each specific p53 mutation. To precisely determine the potentials of p53 GOF mutations on tumor spectrum and onset, future studies should include more LFS patients. In addition, the tumor grade, response to radiochemotherapies and overall/disease-free survival time should also be systematically compared between patients carrying different p53 germline mutations. Finally, more molecular biological studies are warranted to help understand the drastic effects of p53 mutations on tumor spectrum and onset. These efforts will benefit both the healthcare of LFS patients and improve our understanding on the molecular underpinning of cancer.
In conclusion, our results suggest that Li-Fraumeni syndrome can be further subdivided into subtypes according to which p53 mutation is carried. This study also provides further support to the unequal GOF effects of p53 mutations in human cancers.
Methods
P53 germline mutation and patient clinical data
We used data from the of the IARC TP53 germline mutation database (R17 release, November 2013) as described previously21. Only tumors in individuals that have been screened for TP53 mutation and identified as a germline mutation carrier were included. In addition, tumors without detailed information about the primary site (cancer type) were excluded for further study. Since the LFS cases may have multiple tumors, only the age of first tumor onset for a given LFS case was included for further analysis. Missense mutations on the same codon (e.g., R273H and R273C) were classified into one group (R273). The list of p53 hotspot mutations have described elsewhere22.
Univariate analysis on age of cancer onset
The univariate analysiswas carried out in R program using the “survival” package as described previously23. The median age of cancer onset, standard error and 95% confidence interval were respectively determined for each patient group. The differences between nonsense and each missense mutation were compared using the Log-rank test. Differences with P < 0.05 were considered statistically significant.
Multivariate analysis on age of cancer onset
The Cox regression model included p53 mutations (nonsense, R175, Y220, G245, R248, R273, R282), cancer type (adrenal cortical, bone, brain, breast, hematopoietic system, lung, skin, soft tissue, and stomach) and patient sex (male, female) as variants. All variables were defined as categorical, with R248 and adrenal cortical respectively set as reference conditions for p53 mutations and cancer types (hazard ratios were in relation to reference conditions). The conditional forward COX regression model was used to analyze the effects of different variables, and insignificant factors were excluded from the final model. The P-values for mutations were calculated after adjustment for the factors of cancer type and patient sex. The hazard ratios and 95% confidence intervals were also determined for each variable in the model. The effects of different p53 mutations were compared to R248 mutation, which had the most number of cases (n = 132).
Distribution of p53 mutations in different cancer types
The Chi-square test was used to compare the distribution of p53 mutations in different cancer types. For each mutation, the numbers of cases with or without the specific type of cancer were calculated and compared using Chi-square test. Taking the R248 mutation as an example, 25 of cases R248 mutation were diagnosed with brain tumors, while 87 cases had other tumors. These were compared to the sum of all other mutations with or without brain tumors (46 and 281, respectively) using Chi-square test. The P-value (0.0406) was below 0.05, and thus the difference was considered as statistically significant.
Author Contributions
J.X., J.Q., Y.H. collected and analyzed data; J.W., X.Z., H.C. and J.Y.F. collected data; J.X. designed the study and wrote the paper.
Acknowledgments
This project was supported by grants from the National Basic Research Program of China 973 Program (2010CB5293), the National High Technology Research and Development Program of China 863 Program (2012AA02A504), the Program for Innovative Research Team of Shanghai Municipal Education Commission, and National Natural Science Foundation of China (30971330, 31371420, 81320108024) to J.Y. Fang. The project was also partially supported by National Natural Science Foundation of China (81000861, 81322036, and 81272383), Shanghai “Oriental Scholars” project (2013XJ), Shanghai Science and Technology Commission “Pujiang Project” (13PJ1405900), and Shanghai Natural Science Foundation (12ZR1417900) to J. Xu. The sponsors of this study had no role in the collection of the data, the analysis and interpretation of the data, the decision to submit the manuscript for publication, or the writing of the manuscript.
References
- Lane D. P. & Crawford L. V. T antigen is bound to a host protein in SV40-transformed cells. Nature 278, 261–263 (1979). [DOI] [PubMed] [Google Scholar]
- Xu J. et al. RhoGAPs attenuate cell proliferation by direct interaction with p53 tetramerization domain. Cell Rep 3, 1526–1538 (2013). [DOI] [PubMed] [Google Scholar]
- Slee E. A. & Lu X. Requirement for phosphorylation of P53 at Ser312 in suppression of chemical carcinogenesis. Sci Rep 3, 3105 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo L. et al. Ionizing radiation induces a dramatic persistence of p53 protein accumulation and DNA damage signaling in mutant p53 zebrafish. Oncogene 32, 4009–4016 (2013). [DOI] [PubMed] [Google Scholar]
- Yang P.-M. et al. Zebularine inhibits tumorigenesis and stemness of colorectal cancer via p53-dependent endoplasmic reticulum stress. Sci. Rep. 3, 3219; 10.1038/srep03219 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maddocks O. D. et al. Serine starvation induces stress and p53-dependent metabolic remodelling in cancer cells. Nature 493, 542–546 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muller P. A. & Vousden K. H. p53 mutations in cancer. Nat Cell Biol 15, 2–8 (2013). [DOI] [PubMed] [Google Scholar]
- Joerger A. C. & Fersht A. R. The tumor suppressor p53: from structures to drug discovery. Cold Spring Harb Perspect Biol 2, a000919 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melino G. p63 is a suppressor of tumorigenesis and metastasis interacting with mutant p53. Cell Death Differ 18, 1487–1499 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muller P. A. et al. Mutant p53 enhances MET trafficking and signalling to drive cell scattering and invasion. Oncogene 32, 1252–1265 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lang G. A. et al. Gain of function of a p53 hot spot mutation in a mouse model of Li-Fraumeni syndrome. Cell 119, 861–872 (2004). [DOI] [PubMed] [Google Scholar]
- Olive K. P. et al. Mutant p53 gain of function in two mouse models of Li-Fraumeni syndrome. Cell 119, 847–860 (2004). [DOI] [PubMed] [Google Scholar]
- Hanel W. et al. Two hot spot mutant p53 mouse models display differential gain of function in tumorigenesis. Cell Death Differ 20, 898–909 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freed-Pastor W. A. & Prives C. Mutant p53: one name, many proteins. Genes Dev 26, 1268–1286 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serra V. et al. Clinical response to a lapatinib-based therapy for a Li-Fraumeni syndrome patient with a novel HER2V659E mutation. Cancer Discov 3, 1238–1244 (2013). [DOI] [PubMed] [Google Scholar]
- Wang P. Y. et al. Increased oxidative metabolism in the Li-Fraumeni syndrome. N Engl J Med 368, 1027–1032 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu J. et al. Gain of function of mutant p53 by coaggregation with multiple tumor suppressors. Nat Chem Biol 7, 285–295 (2011). [DOI] [PubMed] [Google Scholar]
- Xu J. et al. Colorectal cancer cells refractory to anti-VEGF treatment are vulnerable to glycolytic blockade due to persistent impairment of mitochondria. Mol Cancer Ther 12, 717–724 (2013). [DOI] [PubMed] [Google Scholar]
- Xu J. S. et al. Single nucleotide polymorphisms in the mitochondrial displacement loop and age-at onset of renal cell carcinoma. Sci. Rep. 3, 2408; 10.1038/srep02408 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang P., Du C. W., Kwan M., Liang S. X. & Zhang G. J. The impact of p53 in predicting clinical outcome of breast cancer patients with visceral metastasis. Sci. Rep. 3, 2246; 10.1038/srep02246 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ognjanovic S., Olivier M., Bergemann T. L. & Hainaut P. Sarcomas in TP53 germline mutation carriers: a review of the IARC TP53 database. Cancer 118, 1387–1396 (2012). [DOI] [PubMed] [Google Scholar]
- Vikhanskaya F., Lee M. K., Mazzoletti M., Broggini M. & Sabapathy K. Cancer-derived p53 mutants suppress p53-target gene expression--potential mechanism for gain of function of mutant p53. Nucleic Acids Res 35, 2093–2104 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braun R. et al. Discovery Analysis of TCGA Data Reveals Association between Germline Genotype and Survival in Ovarian Cancer Patients. PLoS One 8, e55037 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]