Abstract
Despite activating similar signaling cascades, the type I and type III interferons (IFNs) differ in their ability to antagonize virus replication. However, it is not clear whether these cytokines induce unique antiviral states, particularly in the liver, where the clinically important hepatitis B and C viruses cause persistent infection. Here, clustering and promoter analysis of microarray-based gene expression profiling was combined with mechanistic studies of signaling pathways to dynamically characterize the transcriptional responses induced by these cytokines in Huh7 hepatoma cells and primary human hepatocytes. Type I and III IFNs differed greatly in their level of interferon-stimulated gene (ISG) induction with a clearly detectable hierarchy (IFN-β > IFN-α > IFN-λ3 > IFN-λ1 > IFN-λ2). Notably, although the hierarchy identified varying numbers of differentially expressed genes when quantified using common statistical thresholds, further analysis of gene expression over multiple time points indicated that the individual IFNs do not in fact regulate unique sets of genes. The kinetic profiles of IFN-induced gene expression were also qualitatively similar with the important exception of IFN-α. While stimulation with either IFN-β or IFN-λs resulted in a similar long-lasting ISG induction, IFN-α signaling peaked early after stimulation then declined due to a negative feedback mechanism. The quantitative expression hierarchy and unique kinetics of IFN-α reveal potential specific roles for individual IFNs in the immune response, and elucidate the mechanism behind previously observed differences in IFN antiviral activity. While current clinical trials are focused on IFN-λ1 as a potential antiviral therapy, the finding that IFN-λ3 invariably possesses the highest activity among type III IFNs suggests that this cytokine may have superior clinical activity.
Keywords: interferon, innate immunity, cytokine signaling, liver
Introduction
The interferon (IFN) family of cytokines plays key roles in the defense against a variety of pathogens. IFNs are commonly classified into three distinct types based on amino acid sequence similarity and the receptor complex with which they interact. The type I IFNs (IFN-α, IFN-β, IFN-κ, IFN-ε, and IFN-ω) bind to the IFN-α receptor, type II IFN (IFN-γ) utilizes the IFN-γ receptor, and type III IFNs (IFN-λ1, IFN-λ2, and IFN-λ3) signal through the IFN-λ receptor. Although the type I and type III IFNs act through different receptors, their signaling cascades converge to phosphorylate STAT1 and STAT2, leading to the formation of the ISGF3 transcription complex and the activation of a variety of antiviral and antiproliferative pathways (1,2). Despite this convergence, the biological effectiveness of these two cytokine families appear to differ, both in terms of antiviral activity and in their ability to induce some interferon stimulated genes (ISGs) (1–6). In particular, a study by Marcello et al. showed that gene expression induced by IFN-α tended to peak at earlier time points than that of IFN-λ (7), suggesting that the relative activities may vary over time. However, it remains unclear whether type-I and type-III IFNs induce unique patterns of ISG expression, resulting in distinct antiviral states.
Within the type I IFN family, IFN-α and IFN-β are often considered similar in early signaling events and antiviral activity (8), although some differences have been noted. For example, IFN-β appears to be more potent in the activation of downstream cellular processes such as proliferation inhibition and apoptosis (9). A recent study examining gene expression in human amnion-derived WISH cells (a highly proliferative cervical cancer cell line) found that although IFN-α and IFN-β induced comparable levels of transcription at early time points, IFN-α-induced transcription declined sharply 8 hours post stimulation (10). The decrease was associated with the expression of IFN-stimulated gene USP18 (UBP43), which is proposed to interact with the IFN-α receptor and inhibit signaling through JAK1 (11). While this study examined only a small number of ISGs, it suggests that comparing the transcriptional signatures of IFNs after different lengths of exposure could provide useful answers to the question of whether the IFNs induce unique transcriptional signatures. It also remains unknown whether this IFN-α/β differential holds true in hepatocytes, since elevated levels of USP18 in the liver have been associated with non-responsiveness to IFN-α therapy in chronic HCV patients (12).
The differential response among the individual IFN-λs also remains poorly characterized. The initial characterization of the IFN-λ family reported IFN-λ1 to possess the strongest activity (1), a finding that was further corroborated by another group testing antiviral and antiproliferative activities (4). However, neither of these studies included IFN-λ3, since it was assumed that this cytokine would possess similar potency as IFN-λ2 based on the high similarity of their amino acid sequences (96%). Surprisingly, a recent study using a functional EMCV protection assay demonstrated that IFN-λ3 may in fact be the most potent member of the family (13). The basis for these functional differences is not clear as, until now, no study has compared the changes in global transcriptional profiles induced by all three IFN-λs over time.
In this paper, we combined highly time-resolved microarray-based gene expression profiling, clustering and promoter analysis, along with mechanistic studies of signaling pathways to characterize the dynamic response to type I and type III IFN stimulation in hepatocyte-derived hepatoma cells (Huh7) and primary human hepatocytes. We found that the type-I and type-III IFNs induced similar sets of ISGs, but the strength of this induction differed greatly between the individual cytokines. This hierarchy of expression led to wide variations in the numbers of genes that appeared to be differentially expressed according to statistical criteria and these quantitative differences may indeed produce distinct functional activities. In the case of IFN-α, the induction of an antiviral state appeared to be transient, with ISG expression starting to decline after 6 hours post-stimulation due to a negative feedback mechanism. In contrast, IFN-β and all three IFN-λs induced long-lasting gene expression. Our data also demonstrate that IFN-λ3 consistently possesses the highest activity among the type III IFNs over time, which has implications for the development of future antiviral and anticancer applications of these cytokines.
Methods
Cells and reagents
Human Huh7 hepatoma cells were cultured in Dulbecco's Modified Eagle's Medium (DMEM) with the addition of 10% heat-inactivated fetal bovine serum, 2 mM L-Glutamine, 100 U/ml penicillin, 100 μg/ml streptomycin, 1 mM sodium pyruvate, 1× MEM non-essential amino acids and 10 mM HEPES buffer (Invitrogen). Metabolism-qualified [cytochrome P450 (CYP1A2, 2B6, 2C8, 2C9, 2C19, 2D6, 3A4, UGT2B7) and UDP-glucuronosyltransferase] cryopreserved primary human hepatocytes (PHHs) from HIV, HBV, HCV-free donors were purchased from BD Biosciences. PHHs were cultured overnight in collagen-coated plates and unattached cells were removed before stimulation with IFN. Fresh PHHs were also obtained from the Cellular and Molecular Physiology Core facility of the Yale Liver Center. Recombinant human IFN-α2a and human IFN-β1a were purchased from PBL Interferon Source (Piscataway, NJ). Recombinant human IL-29 (IFN-λ1), IL-28A (IFN-λ2), and IL-28B (IFN-λ3) were purchased from R&D Systems (Minneapolis, MN). The pharmacological inhibitors SB203580, PD98059, SP600125, and LY294002 were obtained from Selleck Chemicals (Houston, TX). Rottlerin was purchased from Sigma-Aldrich (St. Louis, MO). Cycloheximide (CHX) was purchased from Santa Cruz Biotechnology (Santa Cruz, CA). Inhibitors were used at previously described effective concentrations (14–16).
Microarray analysis
Huh7 cells were seeded into 6-well plates at a density of 3 × 105 cells/well and cultured overnight before treatment with either 500 U/ml of IFN-α or IFN-β, or 10 ng/ml of IFN-λ1, IFN-λ2, or IFN-λ3. Total RNA was harvested and isolated pre-treatment and at 0.5, 1, 2, 4, 6, 12 and 24 hours post-treatment using RNeasy Mini Kit (Qiagen). All subsequent processing, hybridization to the Illumina HumanHT-12 microarray, and quality control analyses were carried out by the Yale Center for Genome Analysis using standard protocols. IFN incubation at each time point was performed in duplicate. RNA concentration was measured by Nanodrop ND-1000 Spectrophotometer and RNA quality was determined by the ratio of OD A260/A280 and A260/A230.
Data preparation and normalization
Microarray data underwent quantile normalization using the Beadarray package in Bioconductor (17). Basic quality control analyses revealed that one replicate for the IFN-λ3 treatment at 4 hours did not properly hybridize, possibly as a result of incomplete purification of the original samples. This sample was removed from the analyses. Gene expression data are available through GEO (http://www.ncbi.nlm.nih.gov/geo) using accession number GSE48400.
Differential Expression Analysis
Differentially expressed probesets were determined by comparing the duplicate samples from each time point with the four untreated samples. Significance was defined by two criteria: (1) a minimum absolute two-fold change in expression, and (2) a significant change in expression determined using the LIMMA package in BioConductor (18) with FDR (false discovery rate) cutoff of 0.05 by the method of Benjamini and Hochberg (19).
Clustering of significant probesets
Hierarchical clustering was performed in R using Ward's minimum variance method. The distance measure was defined as one minus the Pearson correlation of the log fold-changes over time. A set of 6 clusters was selected using manual inspection of the group structure.
See supplemental methods for additional details.
Results
Type I and III IFNs induce a stable hierarchy of gene expression
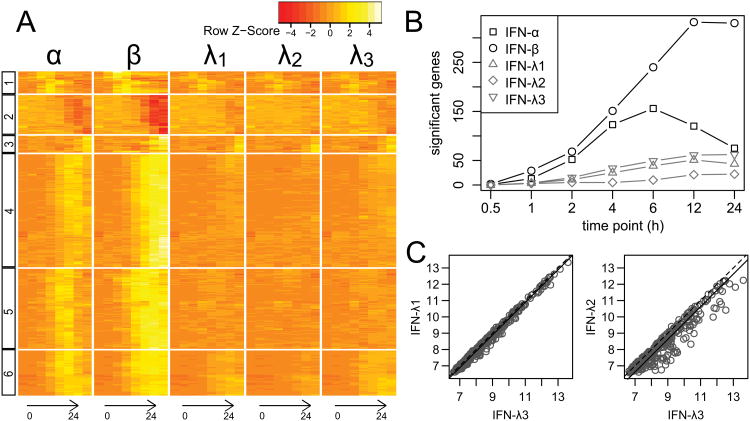
Although the majority of infectious microorganisms that reach the liver are rapidly eliminated (20), certain pathogens, including the hepatitis B and hepatitis C viruses, may successfully establish chronic infections. Both type I and type III IFNs are known to interfere with the replication of viruses that give rise to hepatic infections (21,22). To define the antiviral transcriptional states induced in hepatocytes by different IFNs, we performed a high-resolution time-course microarray experiment using a human hepatoma (Huh7) cell line exposed to the type I IFNs (IFN-α and IFN-β) and type III IFNs (IFN-λ1, IFN-λ2 and IFN-λ3). In order to ensure that expression levels were comparable, the amount of type I IFNs (500 U/ml) was chosen based on previous literature (23–25), where the effective concentration was defined by the cytokine's ability to inhibit EMCV replication. As the type III IFNs do not have a similar international unit system, we adopted a fixed amount for all the IFN-λs (10 ng/ml), which is similar to the concentration of type I IFN used on the basis of molarity, and is consistent with concentrations used in previous studies that examined HCV inhibition and ISG expression in Huh7 cells (7,22). Duplicate samples were collected at 0.5, 1, 2, 4, 6, 12, and 24 hours post treatment, along with four untreated samples, and microarray analysis was performed. After normalization, differential expression analysis of each treatment was performed by comparing each time point to the untreated samples, which revealed a total of 434 significantly differentially-expressed probesets (FDR<0.05 and |Fold change| ≥ 2), representing 368 unique genes (Figure 1a and Suppl. Table S1).
Figure 1. IFN treatment generates a stable hierarchy of gene expression.
Huh7 cells were treated with either IFN-α, IFN-β (500 U/ml), or IFN-λ1, IFN-λ2, IFN-λ3 (10 ng/ml), and gene expression was measured at 0, 0.5, 1, 2, 4, 6, 12, and 24 hours post stimulation. a) A total of 434 probesets (rows) were found to be significantly differentially-expressed for at least one time point by at least one IFN (columns). Hierarchical clustering identified six separate groups (numbered boxes). b) The number of genes significantly induced or suppressed by individual IFNs at each time point. c) Comparison of log2 expression values for genes induced after 12 hours of stimulation with IFN-λ1, IFN-λ2 or IFN-λ3. Equivalent expression is indicated by the 1-to-1 line (dotted line), while the solid line shows the line of best fit with slope set to 1.
In general, we found that the relative activity of each interferon was surprisingly consistent across genes, with IFN-β having the greatest effect on expression, followed by IFN-α, IFN-λ3, IFN-λ1, and IFN-λ2. This pattern was observed in both the overall number of significant genes (Figure 1b), as well as the expression levels of individual genes (Figure S1). Among the type III IFNs, we found that the activity of IFN-λ3 was slightly higher overall when compared to IFN-λ1, and both were approximately 30% higher on average compared to IFN-λ2 (Figure 1c). Interestingly, there were no significant genes found in the early time points where this pattern did not hold true (i.e., genes where, for example, IFN-λ1 was significant, but IFN-λ3 was not), and at 12 and 24 hours only a small number of genes did not adhere to this pattern.
Distinct time-dependent expression modules are associated with unique regulators
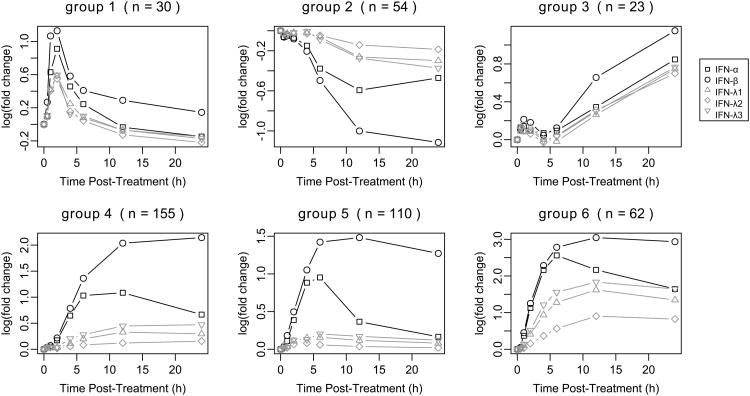
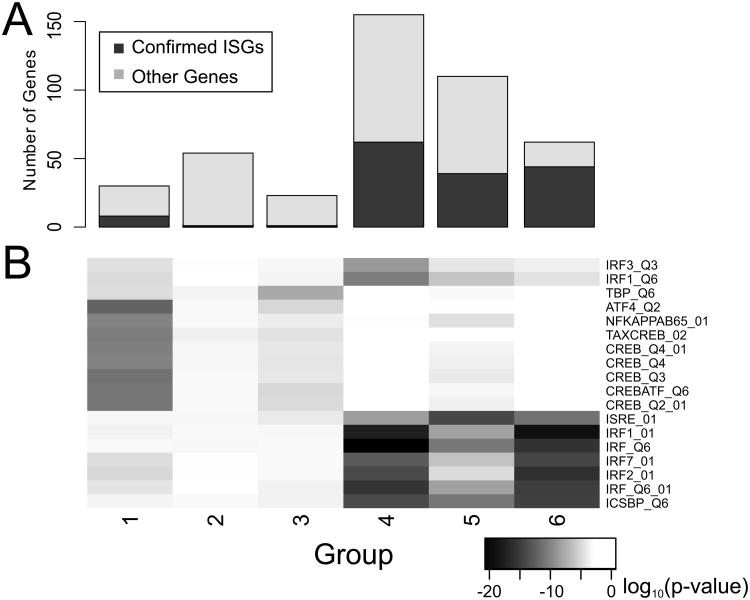
Although the relative expression levels of any given gene could be predicted by the stable hierarchy, significant deviations were observed in the time-dependent profiles of individual genes. In order to better understand this variability, the 434 significant probesets were clustered using a distance metric based on gene-gene correlations. Six groups were identified, each representing a distinct temporal pattern of gene expression (Figure 2 and Figure 1a; a list of the genes belonging to each group can be found in Suppl. Table 1). In general, each group appears to represent a common profile of regulation. For example, genes in group 1 (e.g. FOS, JUN, and SOCS3), were activated immediately upon exposure to IFN, many within 30 minutes, and returned to baseline levels after 4-6 hours. In contrast, genes in groups 4, 5, and 6, (e.g. MX1 and IRF9), generally showed a slower but more sustained response after IFN stimulation. Of note, many of the genes in these groups are generally accepted to be IFN-stimulated genes (ISGs), with group 6 containing the largest overall percentage of genes previously identified as IFN-sensitive (Figure 3a), based on a list compiled by Schoggins et al. (26).
Figure 2. IFN treatment induces six distinct temporal expression patterns.
Hierarchical clustering was carried out on the set of differentially expressed probesets using a distance matrix based on the pairwise correlation. A set of 6 groups was identified using manual analysis of the group structure (same as shown in Figure 1A). Each line indicates the average log2 fold change for genes in each group.
Figure 3. Temporal clusters are associated with common transcription factors.
a) The number of genes in each group is represented as a sum of genes previously described as confirmed ISGs (dark grey), and other genes or potential ISGs (light grey). b) Over-representation of genes containing the indicated transcription factor binding sites within each group was calculated using a hypergeometric test. The log10 (p-value) is shown for significantly enriched sites.
The number of differentially expressed genes generally mirrored the time-dependent expression patterns, with IFN-β producing both the greatest number of statistically significant genes (Figure 1b) and inducing the highest fold-changes (Figure 2). However, these patterns show that relying on differential-expression analysis alone presents an incomplete picture of the overall response. Even though the IFN-λ responses consistently produced the fewest number of differentially expressed genes when using common statistical thresholds, visual inspection of the cluster profiles in Figure 2 clearly shows that these cytokines are regulating genes within each of the 6 groups. The differences are simply due to the fact that the extent of up/down-regulation is quantitatively less. Thus, individual IFNs do not appear to target unique sets of genes.
The distinct temporal patterns exemplified by each cluster group could be the result of regulation by different transcription factors. To test this hypothesis, promoter analysis was carried out to identify transcription factor target genes (defined by putative binding sites in their promoter regions) that were over-represented within each of the clustered groups. We found that groups 4, 5, and 6 were all significantly enriched for genes containing Interferon Stimulatory Response Elements (ISREs) (Figure 3b), implying that these genes are direct targets of the ISGF3 complex, and therefore all typical IFN-stimulated genes. These groups were also enriched for many Interferon Regulatory Factor (IRF) binding sites, most likely because IRF binding sites closely resemble ISRE sites, and many sites can bind both ISGF3 and IRF (27). In contrast, group 1 was significantly enriched for cAMP-Responsive Element (CRE) binding sites, particularly the binding site for CREB/ATF1. Interestingly, neither factor was associated with group 2, which contained all of the down-regulated genes. Indeed, despite screening the transcription factor binding site definitions available in TRANSFAC, we could not identify any potential negative regulators. This suggests the existence of IFN-regulated factors that have yet to be identified.
The IFN-α response exhibits a unique kinetic profile suggesting a negative feedback
We observed a unique pattern of expression in IFN-α-treated cells, in which expression levels of the ISGs in groups 4-6, which were initially very similar to those in IFN-β treatment, peaked after 6 hours of stimulation and eventually decreased to the same magnitude as found in IFN-λ-treated cells. A similar pattern has previously been observed for a small number of IFN-stimulated genes (10). Here, we show that this difference in activity is consistent across all differentially expressed genes, including those that are downregulated. This pattern results in a large difference in the number of differentially regulated transcripts after 24 hours of stimulation (Figure 1b). Thus, depending on when gene expression is measured, it is possible to reach the conclusion that IFN-α and IFN-β induce equivalent transcriptional responses (<6 hours post-stimulation) or that IFN-α regulates a subset of the genes induced by IFN-β. Our results show that these cytokines target the same set of genes, but that only the IFN-α response is subject to a significant negative feedback.
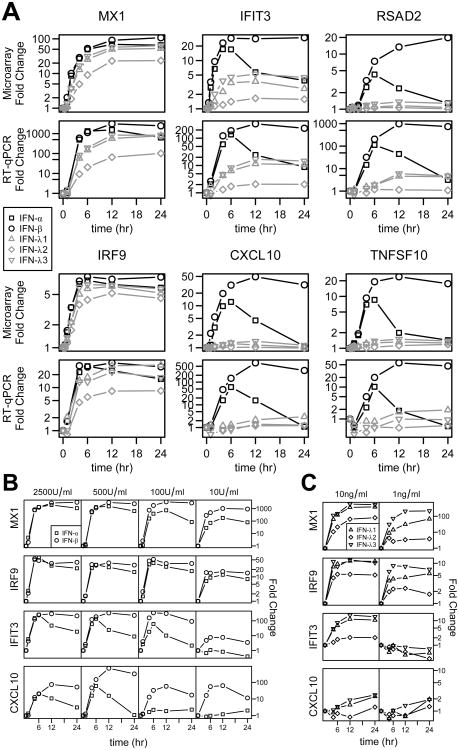
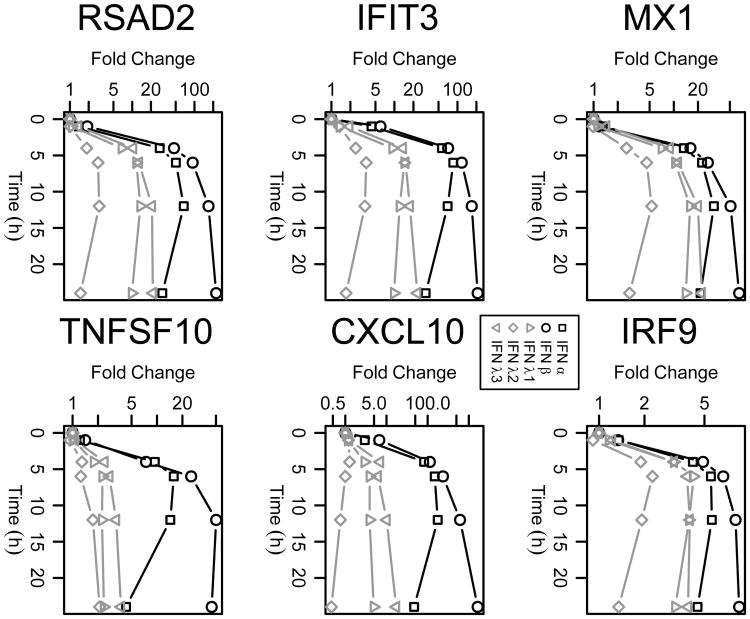
In order to confirm that the gene expression patterns were not confined to a particular concentration of the cytokines, we performed RT-qPCR to validate the expression of selected genes from expression groups 4-6 (MX1, IRF9, IFIT3, CXCL10, RSAD2, and TNFSF10) after treatment with the type I and type III IFNs (Figure 4a), and with varying dosages of the type I IFNs (10, 100, 500 and 2500 U/ml; Figure 4b) and the type III IFNs (1 and 10 ng/ml; Figure 4c). Consistent with our expectations, the expression patterns of the selected genes were comparable to those detected based on the microarray analysis, and the differences between the cytokines became even more pronounced when lower dosages were used. We next asked whether there are differences in the degradation or internalization rates of the cytokines in cell culture, which could potentially explain the discrepancy within the type I IFNs. To address this, IFN-α and IFN-β were added to media both with and without Huh7 cells present, and the samples were incubated for 24 hours. Conditioned media was then transferred to untreated Huh7 cells, and the ability of the cytokines to induce ISG expression was compared to that of freshly-added IFNs. We found that neither IFN-α nor IFN-β demonstrated a loss of activity after incubation for 24 hours in the presence or absence of Huh7 cells (Figure S2), indicating that both cytokines remain stable in cell culture and were not differentially affected by endocytosis over the 24-hour time course of the experiment. Thus we conclude that the patterns of expression found via microarray analysis are of biologically relevant origin, and do not stem from unaccounted-for technical variables.
Figure 4. RT-qPCR validation confirms unique pattern of activity for IFN-α-induced ISGs.
a) Huh7 cells were treated with IFN-α, IFN-β (500 U/ml), IFN-λ1, IFN-λ2, or IFN-λ3 (10 ng/ml), and gene expression of six ISGs from groups 4, 5, and 6 were quantified using RT-qPCR (bottom), and compared to fold-changes from the microarray data (top). Huh7 cells were treated with 10, 100, 500, or 2500 U/ml of IFN-α or IFN-β (b), or with 1 or 10 ng/ml of IFN-λ1, IFN-λ2, or IFN-λ3 (c) at fixed time points, and mRNA expression of MX1, IRF9, IFIT3 and CXCL10 were quantified by RT-qPCR.
Because cell lines may have inherent alterations in the IFN response compared to normal untransformed cells, primary human hepatocytes (PHHs) were used to confirm the different IFN responses. Cryopreserved PHHs were similarly treated with either 500 U/ml of IFN-α, IFN-β or 10 ng/ml of IFN-λ1, IFN-λ2, and IFN-λ3, and cells were collected at 1, 4, 6, 12, and 24 hours post stimulation. The expression of six representative ISGs was measured by RT-qPCR and both the overall hierarchy and the time-course pattern of expression closely resembled the patterns observed in Huh 7 cells (compare Figures 4 and 5). Over the 24-hour timeframe post stimulation, IFN-β exhibited the strongest activity among all five cytokines, and the ability of IFN-λ3 to induce ISG expression in PHHs was consistently more potent than the other two IFN-λs. The same set of experiments was also performed using fresh PHHs within 24 hours of isolation, with overall equivalent results (data not shown). Collectively, these findings suggest that the patterns observed using microarray analysis of Huh7 cells are consistent in primary cells across the set of genes differentially expressed.
Figure 5. IFN stimulation of primary human hepatocytes confirms hierarchy of IFN activity over time.
Primary human hepatocytes were seeded into collagen-coated 12-well plates before treatment with IFN-α, IFN-β (500 U/ml), IFN-λ1, IFN-λ2, or IFN-λ3 (10 ng/ml) for 0, 1, 4, 6, 12, or 24 hours. The expression of six ISGs from groups 4, 5, and 6 were quantified using RT-qPCR.
Expression hierarchy and temporal patterns are correlated with STAT1 phosphorylation
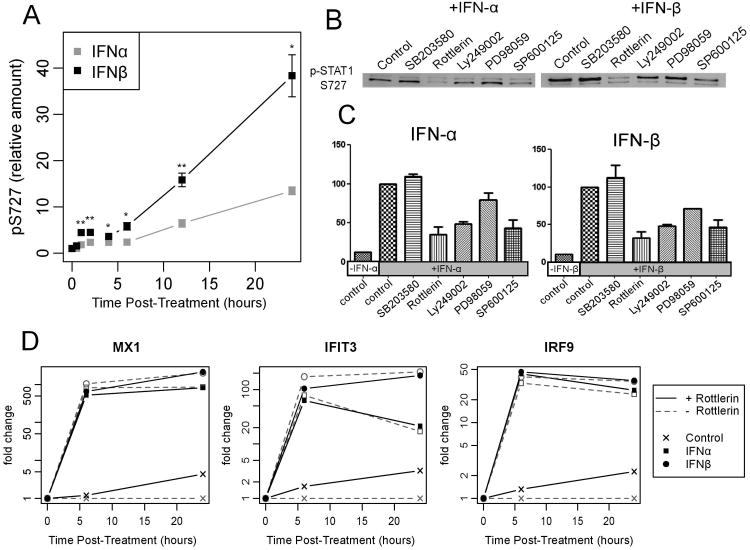
To identify the underlying signaling mechanism responsible for the observed gene expression hierarchy and temporal patterns, we investigated STAT1 phosphorylation states. In accordance with previous studies (10), high levels of STAT1 Y701 phosphorylation were apparent at early time points following treatment with either IFN-α or IFN-β (Figure S3a). However, in both cases, these phosphorylation levels decreased over time in a similar manner (Figure S3a). This contrasts with previous results in amnion-derived WISH cells (10), and suggest that a different mechanism may be responsible for the unique temporal expression profile following IFN-α treatment in hepatocytes. Therefore, we further investigated phosphorylation of the STAT1 serine residue (S727), which is also known to play a role in the maximal activation of ISG expression (28). Following stimulation, S727 phosphorylation on STAT1 was more potently induced by IFN-β at 12 and 24 hours post-treatment in comparison to S727 activation by IFN-α (Figure 6a and Figure S3a). This pattern raises the possibility that serine phosphorylation may play an important role the long-term gene induction observed following IFN-β treatment.
Figure 6. Type I IFNs induced ISG expression in a serine phosphorylation independent manner.
a) The levels of pSTAT1 S727 were measured at various time points after exposure of Huh7 cells to IFN-α (grey) and IFN-β (black). The mean (points) ± standard error (bars) was calculated from four independent experiments. b) Cells were treated with kinase inhibitors before stimulation with IFN-α and IFN-β for 6 hours. Cell lysates were used in Western Blotting and probed for serine phosphorylation. c) The relative amounts of pSTAT1 S727 in b) were quantified and normalized to an untreated control. d) Expression of selected ISGs was measured at various time points using RT-qPCR with (black) or without (grey) the presence of rottlerin following treatment with the type I IFNs.
In contrast to the type I IFNs, early strong STAT1 Y701 phosphorylation was not observed following treatment with any of the IFN-λs, possibly explaining the weaker overall gene activation by the type III IFNs (Figure S3b). The patterns of STAT1 S727 phosphorylation were similar for all three type-III IFNs, among which IFN-λ3 appeared to induce the highest level of phosphorylation, consistent with this cytokine's highest activity levels observed in the gene expression analysis. Overall, these results raise the possibility that STAT1 Y701 phosphorylation is responsible for the early, strong gene induction following type I IFN treatment relative to type III IFNs.
PKC- δ activity is required for STAT-1 S727 phosphorylation, but not ISG expression
In order to identify the kinase responsible for S727 phosphorylation, as well as to understand whether reduced S727 phosphorylation would lead to a change in the levels of signaling of either IFN-α or IFN-β, Huh7 cells were treated with a panel of specific inhibitors targeting different kinases prior to IFN stimulation. SB203580, rottlerin, Ly294002, PD98059, and SP600125 were applied to Huh7 cells to specifically inhibit the activities of p38 MAPK, PKC-δ, PI3K, MEK and JNK, respectively, and the cells were subsequently stimulated with either IFN-α or IFN-β for the next 6 hours. Overall, PKC-δ was found to be primarily responsible for S727 phosphorylation in the signaling pathways of both type I IFNs, with the kinase inhibitor rottlerin blocking a majority of the serine phosphorylation after 6 hours of stimulation (Figure 6b and 6c). We next asked whether the inhibition of PKC-δ with rottlerin would affect gene expression levels in IFN-treated cells. However, contrary to previous studies showing that serine phosphorylation is required for the maximal induction of ISGs (28), there was little change in the patterns of gene expression after either 6 or 24 hours of IFN stimulation in the presence of rottlerin (Figure 6d and Figure S4). Therefore, IFN-β elicits a greater magnitude of IFN-stimulated gene expression independent of its ability to induce higher levels of STAT S727 phosphorylation.
Discussion
In this study, we examined the temporal expression profiles of hepatic Huh7 cells and primary hepatocytes when treated with comparable concentrations of the type I IFNs, IFN-α and β, and the type III IFNs, IFN-λ1, λ2, and λ3. While all five cytokines induced the expression of a similar set of genes, the magnitude and pattern of induction varied greatly among them and a clearly detectable hierarchy was observed, with: IFN-β > IFN-α > IFN-λ3 > IFN-λ1 > IFN-λ2. This result contrasts previous studies, which have claimed that the type I and type III IFNs induce overlapping, but distinct sets of genes (7,29). Among the genes that were induced in our experiment, the hierarchy of expression remained consistent, and there were no genes specifically induced by IFN-λ. Conversely, although a number of genes only reached statistical significance after treatment with type I IFN, their expression in type III IFN-treated cells tended to follow a qualitatively similar temporal profile, demonstrating they were also induced, but to lower levels. Overall, our results suggest that the differences between the type I and type III IFNs are quantitative, rather than qualitative.
When the IFN-λ genes were first discovered (1), it was demonstrated that in the hepatic HepG2 cell line, IL-29 (IFN-λ1) was more active than IL-28A (IFN-λ2). In a follow-up study, it was confirmed that in terms of both antiviral and antiproliferative activities, IFN-λ1 was stronger than IFN-λ2 (4). Considering the high degree of sequence homology between IFN-λ2 and IFN-λ3, it was assumed that IFN-λ1 would be the most active member. Here, our data demonstrate that IFN-λ3 is the most potent and broad inducer of ISG expression, seen not only as a higher number of significantly induced genes at each time point (Figure 1b), but also as consistently higher expression levels over time. While increased ISG expression does not necessarily correlate with higher levels of functional antiviral effects, recent studies have suggested that IFN-λ3 does indeed have greater antiviral activity than both IFN-λ1 and IFN-λ2 (13). However, although consistent with previous studies, we cannot entirely rule out the possibility of varying proportions of active proteins among the IFN- λ preparations as a possible contributing factor to this result.
The difference in the relative strengths of the IFN-λ family members may be important for developing therapeutic strategies against chronic virus infections (such as HCV) and cancer. Currently, standard therapy for HCV involves a 48-week regimen of pegylated IFN-α and ribavirin, which can be both expensive and poorly tolerated, and is only effective in approximately 50% of patients. Recently, a Phase IIB clinical trial has been underway to test if pegylated recombinant IFN-λ1 can be used instead of IFN-α with same potency and much less side effects. Our results suggest that IFN-λ3 might be an even better substitute for IFN-α than IFN-λ1, supported by its potent activity in both Huh7 cells and primary hepatocytes. This idea is also substantiated by a number of recent GWAS studies (30–32) which have identified single nucleotide polymorphisms (SNPs) located in regions upstream of IL28B (the gene that encodes IFN-λ3) that are strongly correlated with the resolution of chronic HCV infection. With the advantages of restricted receptor expression when compared to IFN-α and stronger activity when compared to IFN-λ1, IFN-λ3 may prove itself to be a worthwhile subject to test for future HCV clinical studies.
Another intriguing aspect of our findings is the difference between the patterns of ISG expression in IFN-α and IFN-β-treated cells. It is generally accepted that IFN-α and IFN-β are very similar both in terms of early signaling events and antiviral activity (8). However, we show that with prolonged stimulation this correlation begins to break down, with expression in IFN-α-treated cells decreasing rapidly after 8 hours of stimulation. This pattern is consistent for the hundreds of ISGs examined by microarray, and broadly extends previous observations made in a limited number of genes (10). Importantly, we also observed a similar pattern in human hepatocytes suggesting that, in the liver, IFN-β induces a stronger and more persistent response compared to IFN-α.
In an effort to understand the factors that contribute to the unique pattern of IFN-α-induced ISG expression, we examined the phosphorylation kinetics of STAT1, the primary transcription factor associated with type-I IFN signaling. Although we did not observe the differences in STAT1 tyrosine phosphorylation which had been previously reported (10), IFN-β gave rise to significantly higher levels of STAT1 serine phosphorylation when compared to IFN-α, especially at later time points. Subsequently, after identifying PKC-δ as the predominant kinase involved in S727 phosphorylation, we found that the inhibition of PKC-δ activity by rottlerin led to decreased S727 phosphorylation but did not negatively affect the expression of the canonical ISGs, represented by MX1, IRF9, IFIT3 and CXCL10. Several previous studies have asserted a positive role for S727 phosphorylation on STAT1 in type I IFN-induced gene expression (28,33), although these papers utilized an ISRE-luciferase construct to represent the typical ISG induction by type I IFNs, potentially ignoring any inherent differences in the promoter regions of different ISGs. Here in our data, it seems that tyrosine phosphorylation is still the dominant factor in determining ISG induction. Despite this finding, however, it is still possible that a small subset of genes is affected by serine phosphorylation.
In summary, we find that treatment with the type I and type III IFNs creates a stable hierarchy of IFN-stimulated gene expression in Huh7 cells and primary hepatocytes, with IFN-β being the most active, followed by IFN-α, IFN-λ3, IFN-λ1, and IFN-λ2. This pattern was consistent from 30 minutes up to 24 hours after stimulation. The activity of IFN-α closely resembles that of IFN-β, but after 6 hours post stimulation the expression profiles of the stimulated ISGs diverge, apparently due, at least in part, to a negative feedback mechanism. In contrast to IFN- α, IFN-β or IFN-λ stimulation resulted in a similar long-lasting ISG expression increase. In conclusion, our results demonstrate that IFN signaling in human hepatocytes is represented by a stable hierarchy resulting from differing cytokine activity levels correlated with STAT1 phosphorylation states. This study deepens our understanding of the dynamics of IFN and hierarchy of cytokine activity levels, and provides new insights into the development of future antiviral therapy.
Supplementary Material
Acknowledgments
This project was partially funded by NIH NIDDK Grant: Silvio O. Conte Digestive Diseases Research Core Centers - P30 DK034989. The work of CRB was supported in part by NIH Grant T15 LM07056 from the National Library of Medicine. SD was supported in part by a fellowship from the China Scholarship Council.
References
- 1.Sheppard P, Kindsvogel W, Xu W, Henderson K, Schlutsmeyer S, Whitmore TE, et al. IL-28, IL-29 and their class II cytokine receptor IL-28R. Nat Immunol. 2003;4:63–68. doi: 10.1038/ni873. [DOI] [PubMed] [Google Scholar]
- 2.Kotenko SV, Gallagher G, Baurin VV, Lewis-Antes A, Shen M, Shah NK, et al. IFN-lambdas mediate antiviral protection through a distinct class II cytokine receptor complex. Nat Immunol. 2003;4:69–77. doi: 10.1038/ni875. [DOI] [PubMed] [Google Scholar]
- 3.Doyle SE, Schreckhise H, Khuu-Duong K, Henderson K, Rosler R, Storey H, et al. Interleukin-29 uses a type 1 interferon-like program to promote antiviral responses in human hepatocytes. Hepatology. 2006;44:896–906. doi: 10.1002/hep.21312. [DOI] [PubMed] [Google Scholar]
- 4.Meager A, Visvalingam K, Dilger P, Bryan D, Wadhwa M. Biological activity of interleukins-28 and -29: comparison with type I interferons. Cytokine. 2005;31:109–118. doi: 10.1016/j.cyto.2005.04.003. [DOI] [PubMed] [Google Scholar]
- 5.Ank N, West H, Bartholdy C, Eriksson K, Thomsen AR, Paludan SR. Lambda Interferon (IFN-λ), a Type III IFN, Is Induced by Viruses and IFNs and Displays Potent Antiviral Activity against Select Virus Infections In Vivo. J Virol. 2006;80:4501–4509. doi: 10.1128/JVI.80.9.4501-4509.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Pagliaccetti NE, Chu EN, Bolen CR, Kleinstein SH, Robek MD. Lambda and alpha interferons inhibit hepatitis B virus replication through a common molecular mechanism but with different in vivo activities. Virology. 2010;401:197–206. doi: 10.1016/j.virol.2010.02.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Marcello T, Grakoui A, Barba–Spaeth G, Machlin ES, Kotenko SV, Macdonald MR, et al. Interferons α and λ Inhibit Hepatitis C Virus Replication With Distinct Signal Transduction and Gene Regulation Kinetics. Gastroenterology. 2006;131:1887–1898. doi: 10.1053/j.gastro.2006.09.052. [DOI] [PubMed] [Google Scholar]
- 8.Marijanovic Z, Ragimbeau J, van der Heyden J, Uzé G, Pellegrini S. Comparable potency of IFNα2 and IFNβ on immediate JAK/STAT activation but differential down-regulation of IFNAR2. Biochemical Journal. 2007;407:141. doi: 10.1042/BJ20070605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Uzé G, Schreiber G, Piehler J, Pellegrini S. The receptor of the type I interferon family. Curr. Top. Microbiol. Immunol. 2007;316:71–95. doi: 10.1007/978-3-540-71329-6_5. [DOI] [PubMed] [Google Scholar]
- 10.Francois-Newton V, Livingstone M, Payelle-Brogard B, Uzé G, Pellegrini S. USP18 establishes the transcriptional and anti-proliferative interferon α/β differential. Biochem J. 2012;446:509–516. doi: 10.1042/BJ20120541. [DOI] [PubMed] [Google Scholar]
- 11.Malakhova OA, Kim KI, Luo JK, Zou W, Kumar KGS, Fuchs SY, et al. UBP43 is a novel regulator of interferon signaling independent of its ISG15 isopeptidase activity. EMBO J. 2006;25:2358–2367. doi: 10.1038/sj.emboj.7601149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chen L, Borozan I, Feld J, Sun J, Tannis LL, Coltescu C, et al. Hepatic Gene Expression Discriminates Responders and Nonresponders in Treatment of Chronic Hepatitis C Viral Infection. Gastroenterology. 2005;128:1437–1444. doi: 10.1053/j.gastro.2005.01.059. [DOI] [PubMed] [Google Scholar]
- 13.Dellgren C, Gad HH, Hamming OJ, Melchjorsen J, Hartmann R. Human interferon-[lambda]3 is a potent member of the type III interferon family. Genes Immun. 2008;10:125–131. doi: 10.1038/gene.2008.87. [DOI] [PubMed] [Google Scholar]
- 14.Kim K, Kim KH, Kim HY, Cho HK, Sakamoto N, Cheong J. Curcumin inhibits hepatitis C virus replication via suppressing the Akt-SREBP-1 pathway. FEBS Lett. 2010;584:707–712. doi: 10.1016/j.febslet.2009.12.019. [DOI] [PubMed] [Google Scholar]
- 15.Schulze-Bergkamen H, Fleischer B, Schuchmann M, Weber A, Weinmann A, Krammer PH, et al. Suppression of Mcl-1 via RNA interference sensitizes human hepatocellular carcinoma cells towards apoptosis induction. BMC Cancer. 2006;6:232. doi: 10.1186/1471-2407-6-232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hung JH, Lu YS, Wang YC, Ma YH, Wang DS, Kulp SK, et al. FTY720 induces apoptosis in hepatocellular carcinoma cells through activation of protein kinase C delta signaling. Cancer Res. 2008;68:1204–1212. doi: 10.1158/0008-5472.CAN-07-2621. [DOI] [PubMed] [Google Scholar]
- 17.Dunning MJ, Smith ML, Ritchie ME, Tavaré S. beadarray: R classes and methods for Illumina bead-based data. Bioinformatics. 2007;23:2183–2184. doi: 10.1093/bioinformatics/btm311. [DOI] [PubMed] [Google Scholar]
- 18.Smyth GK. Linear Models and Empirical Bayes Methods for Assessing Differential Expression in Microarray Experiments. [cited 2010 Oct 21];Statistical Applications in Genetics and Molecular Biology [Internet] 2004 3 doi: 10.2202/1544-6115.1027. Available from: http://www.bepress.com/sagmb/vol3/iss1/art3. [DOI] [PubMed] [Google Scholar]
- 19.Benjamini Y, Hochberg Y. Controlling the false discovery rate: a practical and powerful approach to multiple testing. Journal of the Royal Statistical Society. Series B Methodological. 1995;57:289–300. [Google Scholar]
- 20.Protzer U, Maini MK, Knolle PA. Living in the liver: hepatic infections. Nat Rev Immunol. 2012;12:201–213. doi: 10.1038/nri3169. [DOI] [PubMed] [Google Scholar]
- 21.Rehermann B, Nascimbeni M. Immunology of hepatitis B virus and hepatitis C virus infection. Nat Rev Immunol. 2005;5:215–229. doi: 10.1038/nri1573. [DOI] [PubMed] [Google Scholar]
- 22.Pagliaccetti NE, Eduardo R, Kleinstein SH, Mu XJ, Bandi P, Robek MD. Interleukin-29 Functions Cooperatively with Interferon to Induce Antiviral Gene Expression and Inhibit Hepatitis C Virus Replication. Journal of Biological Chemistry. 2008;283:30079–30089. doi: 10.1074/jbc.M804296200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chawla-Sarkar M, Leaman DW, Borden EC. Preferential induction of apoptosis by interferon (IFN)-beta compared with IFN-alpha2: correlation with TRAIL/Apo2L induction in melanoma cell lines. Clin Cancer Res. 2001;7:1821–1831. [PubMed] [Google Scholar]
- 24.Leaman DW, Chawla-Sarkar M, Jacobs B, Vyas K, Sun Y, Ozdemir A, et al. Novel growth and death related interferon-stimulated genes (ISGs) in melanoma: greater potency of IFN-beta compared with IFN-alpha2. J Interferon Cytokine Res. 2003;23:745–756. doi: 10.1089/107999003772084860. [DOI] [PubMed] [Google Scholar]
- 25.Shen H, Zhang M, Minuk GY, Gong Y. Different effects of rat interferon alpha, beta and gamma on rat hepatic stellate cell proliferation and activation. BMC Cell Biol. 2002;3:9. doi: 10.1186/1471-2121-3-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Schoggins JW, Wilson SJ, Panis M, Murphy MY, Jones CT, Bieniasz P, et al. A diverse range of gene products are effectors of the type I interferon antiviral response. Nature. 2011;472:481–485. doi: 10.1038/nature09907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Schmid S, Mordstein M, Kochs G, García-Sastre A, Tenoever BR. Transcription factor redundancy ensures induction of the antiviral state. J Biol Chem. 2010;285:42013–42022. doi: 10.1074/jbc.M110.165936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wen Z, Zhong Z, Darnell JE., Jr Maximal activation of transcription by Stat1 and Stat3 requires both tyrosine and serine phosphorylation. Cell. 1995;82:241–250. doi: 10.1016/0092-8674(95)90311-9. [DOI] [PubMed] [Google Scholar]
- 29.Kohli A, Zhang X, Yang J, Russell RS, Donnelly RP, Sheikh F, et al. Distinct and overlapping genomic profiles and antiviral effects of Interferon-λ and -α on HCV-infected and noninfected hepatoma cells. J Viral Hepat. 2012;19:843–853. doi: 10.1111/j.1365-2893.2012.01610.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ge D, Fellay J, Thompson AJ, Simon JS, Shianna KV, Urban TJ, et al. Genetic variation in IL28B predicts hepatitis C treatment-induced viral clearance. Nature. 2009;461:399–401. doi: 10.1038/nature08309. [DOI] [PubMed] [Google Scholar]
- 31.Suppiah V, Moldovan M, Ahlenstiel G, Berg T, Weltman M, Abate ML, et al. IL28B is associated with response to chronic hepatitis C interferon-[alpha] and ribavirin therapy. Nat Genet. 2009;41:1100–1104. doi: 10.1038/ng.447. [DOI] [PubMed] [Google Scholar]
- 32.Tanaka Y, Nishida N, Sugiyama M, Kurosaki M, Matsuura K, Sakamoto N, et al. Genome-wide association of IL28B with response to pegylated interferon-[alpha] and ribavirin therapy for chronic hepatitis C. Nat Genet. 2009;41:1105–1109. doi: 10.1038/ng.449. [DOI] [PubMed] [Google Scholar]
- 33.Uddin S, Sassano A, Deb DK, Verma A, Majchrzak B, Rahman A, et al. Protein kinase C-delta (PKC-delta) is activated by type I interferons and mediates phosphorylation of Stat1 on serine 727. J Biol Chem. 2002;277:14408–14416. doi: 10.1074/jbc.M109671200. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.