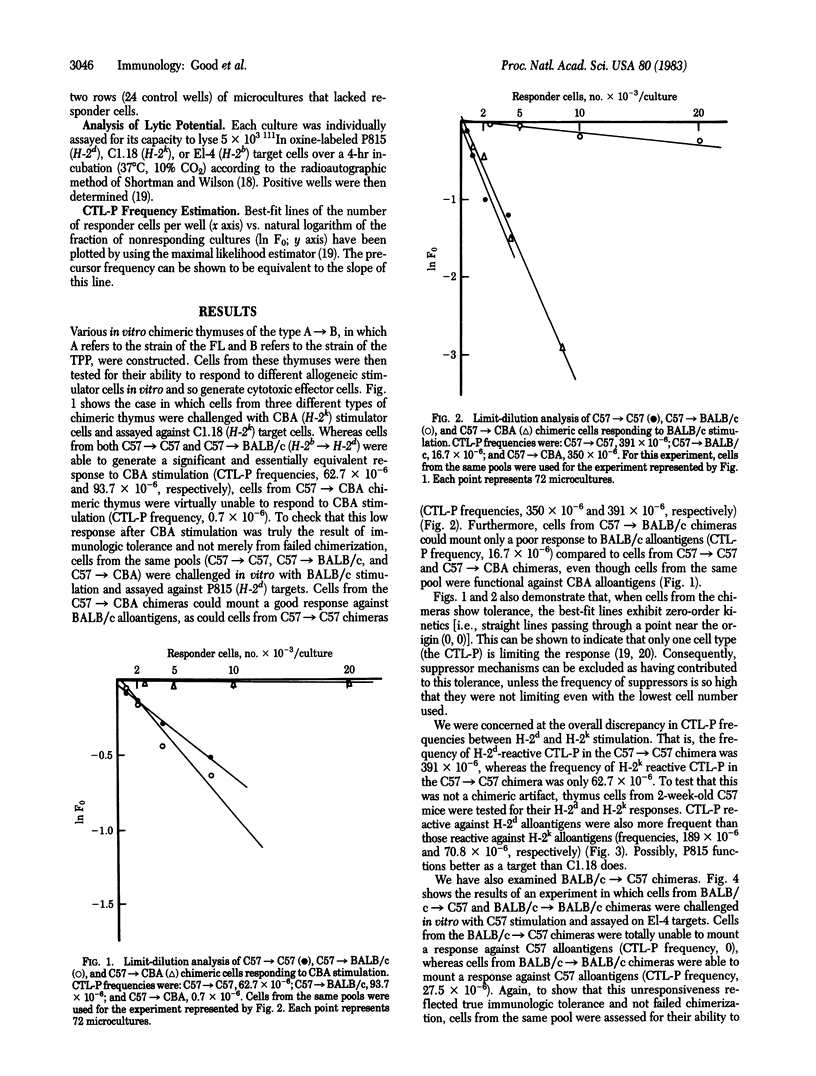
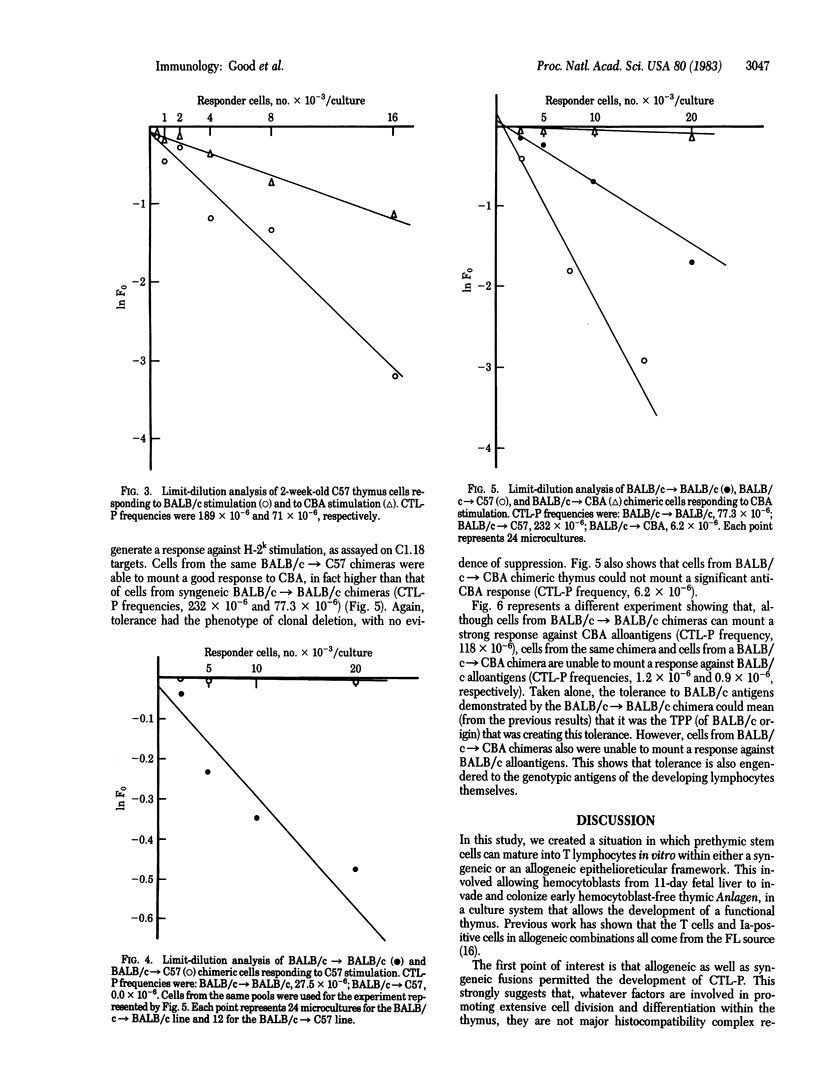
Abstract
Chimeric thymus, formed by fusing the prelymphoid third pharyngeal pouches of fetal mice with fetal liver, have been allowed to develop entirely in vitro. Syngeneic and allogeneic chimeras were prepared and both types of thymus were shown to contain substantial numbers of functional cytotoxic T lymphocyte precursors reactive against "third party" alloantigens. However, alloreactivity specific for H-2 antigens present on either the third pharyngeal pouch or the fetal liver was minimal. In three different allogeneic chimeric thymuses, the frequencies of cytotoxic T lymphocyte precursors reactive to H-2 antigens present on the third pharyngeal pouches were reduced to 1%, 4%, and 0% of control values, whereas, in the one allogeneic chimera tested for alloreactivity to H-2 antigens present on the fetal liver, the cytotoxic T lymphocyte precursor frequency was reduced to less than 1% of control values. The phenotype of the H-2 tolerance is shown to be one of functional clonal deletion of the cytotoxic T lymphocyte precursor.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BILLINGHAM R. E., BRENT L., MEDAWAR P. B. Actively acquired tolerance of foreign cells. Nature. 1953 Oct 3;172(4379):603–606. doi: 10.1038/172603a0. [DOI] [PubMed] [Google Scholar]
- Bunce J. V., Mason D. W. The toleration of rat thymocytes to xenogeneic erythrocytes: kinetics of induction and recovery. Eur J Immunol. 1981 Nov;11(11):889–896. doi: 10.1002/eji.1830111108. [DOI] [PubMed] [Google Scholar]
- Dorsch S., Roser B. Suppressor cells in transplantation tolerance. I. Analysis of the suppressor status of neonatally and adoptively tolerized rats. Transplantation. 1982 May;33(5):518–524. [PubMed] [Google Scholar]
- Dorsch S., Roser B. Suppressor cells in transplantation tolerance. II. Identification and probable mode of action of chimeric suppressor T cells. Transplantation. 1982 May;33(5):525–529. [PubMed] [Google Scholar]
- Dröge W. Theoretical considerations and probability models for the somatic development of the T-cell repertoire. Cell Immunol. 1981 Jan 1;57(1):251–264. doi: 10.1016/0008-8749(81)90137-4. [DOI] [PubMed] [Google Scholar]
- Fontaine-Perus J. C., Calman F. M., Kaplan C., Le Douarin N. M. Seeding of the 10-day mouse embryo thymic rudiment by lymphocyte precursors in vitro. J Immunol. 1981 Jun;126(6):2310–2316. [PubMed] [Google Scholar]
- Good M. F., Nossal G. J. Characteristics of tolerance induction among adult hapten-specific T lymphocyte precursors revealed by clonal analysis. J Immunol. 1983 Jan;130(1):78–85. [PubMed] [Google Scholar]
- Gorczynski R. M., Khomasurya B., MacRae S., Short L. Analysis of early events occurring during neonatal induction of tolerance to histoincompatible cells in mice. Cell Immunol. 1981 Jan 1;57(1):183–200. doi: 10.1016/0008-8749(81)90131-3. [DOI] [PubMed] [Google Scholar]
- Gorczynski R. M., MacRae S. Suppression of cytotoxic response to histoincompatible cells. II. Analysis of the role of two independent T suppressor pools in maintenance of neonatally induced allograft tolerance in mice. J Immunol. 1979 Mar;122(3):747–752. [PubMed] [Google Scholar]
- Hasek M., Chutná J. Complexity of the state of immunological tolerance. Immunol Rev. 1979;46:3–26. doi: 10.1111/j.1600-065x.1979.tb00282.x. [DOI] [PubMed] [Google Scholar]
- Hilgert I. The involvement of activated specific suppressor T cells in maintenance of transplantation tolerance. Immunol Rev. 1979;46:27–53. doi: 10.1111/j.1600-065x.1979.tb00283.x. [DOI] [PubMed] [Google Scholar]
- Le Douarin N. M., Jotereau F. V. Tracing of cells of the avian thymus through embryonic life in interspecific chimeras. J Exp Med. 1975 Jul 1;142(1):17–40. doi: 10.1084/jem.142.1.17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Longo D. L., Schwartz R. H. Gene complementation. Neither Ir-GLphi gene need be present in the proliferative T cell to generate an immune response to Poly(Glu55Lys36Phe9)n. J Exp Med. 1980 Jun 1;151(6):1452–1467. doi: 10.1084/jem.151.6.1452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Longo D. L., Schwartz R. H. T-cell specificity for H-2 and Ir gene phenotype correlates with the phenotype of thymic antigen-presenting cells. Nature. 1980 Sep 4;287(5777):44–46. doi: 10.1038/287044a0. [DOI] [PubMed] [Google Scholar]
- Mandel T. E., Kennedy M. M. The differentiation of murine thymocytes in vivo and in vitro. Immunology. 1978 Aug;35(2):317–331. [PMC free article] [PubMed] [Google Scholar]
- Matsunaga T., Simpson E., Meo T. Allogeneic tolerance in embryo aggregation mouse chimeras studied by mixed lymphocyte culture and cell-mediated lympholysis. Transplantation. 1980 Jul;30(1):34–39. doi: 10.1097/00007890-198007000-00007. [DOI] [PubMed] [Google Scholar]
- Miller S. D., Sy M. S., Claman H. N. The induction of hapten-specific T cell tolerance using hapten-modified lymphoid membranes. II. Relative roles of suppressor T cells and clone inhibition in the tolerant state. Eur J Immunol. 1977 Mar;7(3):165–170. doi: 10.1002/eji.1830070310. [DOI] [PubMed] [Google Scholar]
- Morrissey P. J., Kruisbeek A. M., Sharrow S. O., Singer A. Tolerance of thymic cytotoxic T lymphocytes to allogeneic H-2 determinants encountered prethymically: evidence for expression of anti-H-2 receptors prior to entry into the thymus. Proc Natl Acad Sci U S A. 1982 Mar;79(6):2003–2007. doi: 10.1073/pnas.79.6.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nash A. A., Phelan J., Gell P. G., Wildy P. Tolerance and immunity in mice infected with herpes simplex virus: studies on the mechanism of tolerance to delayed-type hypersensitivity. Immunology. 1981 Jun;43(2):363–369. [PMC free article] [PubMed] [Google Scholar]
- Nossal G. J., Pike B. L. Clonal anergy: persistence in tolerant mice of antigen-binding B lymphocytes incapable of responding to antigen or mitogen. Proc Natl Acad Sci U S A. 1980 Mar;77(3):1602–1606. doi: 10.1073/pnas.77.3.1602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nossal G. J., Pike B. L. Functional clonal deletion in immunological tolerance to major histocompatibility complex antigens. Proc Natl Acad Sci U S A. 1981 Jun;78(6):3844–3847. doi: 10.1073/pnas.78.6.3844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pyke K. W., Bartlett P. F., Mandel T. E. Lymphocyte depletion of murine fetal thymus and subsequent chimeric recolonization in vitro. Thymus. 1983 Mar;5(2):95–104. [PubMed] [Google Scholar]
- Roser B., Dorsch S. The cellular basis of transplantation tolerance in the rat. Immunol Rev. 1979;46:55–86. doi: 10.1111/j.1600-065x.1979.tb00284.x. [DOI] [PubMed] [Google Scholar]
- Shortman K., Wilson A. A new assay for cytotoxic lymphocytes, based on a radioautographic readout of 111 In release, suitable for rapid, semi-automated assessment of limit-dilution cultures. J Immunol Methods. 1981;43(2):135–152. doi: 10.1016/0022-1759(81)90017-x. [DOI] [PubMed] [Google Scholar]
- Wagner H., Hardt C., Stockinger H., Pfizenmaier K., Bartlett R., Röllinghoff M. Impact of thymus on the generation of immunocompetence and diversity of antigen-specific MHC-restricted cytotoxic T-lymphocyte precursors. Immunol Rev. 1981;58:95–129. doi: 10.1111/j.1600-065x.1981.tb00351.x. [DOI] [PubMed] [Google Scholar]
- Zinkernagel R. M., Callahan G. N., Althage A., Cooper S., Klein P. A., Klein J. On the thymus in the differentiation of "H-2 self-recognition" by T cells: evidence for dual recognition? J Exp Med. 1978 Mar 1;147(3):882–896. doi: 10.1084/jem.147.3.882. [DOI] [PMC free article] [PubMed] [Google Scholar]


