Abstract
Bladder cancer is often associated with recurrence and progression to invasive metastatic disease that have palliative therapeutic options. The use of traditional chemotherapeutic agents for bladder cancer management often suffer from toxicity and resistance concerns. This emphasizes the need for development of safer, natural, non-toxic compounds as chemotherapeutic/chemopreventive agents. Curcumin (diferuloylmethane) is a natural compound that has been known to possess anti-cancer properties in various cancers, including bladder cancer. However, the biological targets of curcumin are not well defined. Recently, it has been proposed that curcumin may mediate epigenetic modulation of expression of microRNAs (miRNAs). In this report, we define for the first time, that curcumin directly induces a tumor-suppressive miRNA, miR-203, in bladder cancer. miR-203 is frequently downregulated in bladder cancer due to DNA of its promoter. We studied the functional significance of miR-203 in bladder cancer cell lines and found that miR-203 has tumor suppressive properties. Also, we define Akt2 and Src as novel miR-203 targets in bladder cancer. Curcumin induces hypomethylation of the miR-203 promoter and subsequent upregulation of miR-203 expression. This leads to downregulation of miR-203 target genes Akt2 and Src that culminates in decreased proliferation and increased apoptosis of bladder cancer cells. This is the first report that shows a direct effect of curcumin on inducing epigenetic changes at a miRNA promoter with direct biological consequences. Our study suggests that curcumin may offer a therapeutic advantage in the clinical management of refractory bladder cancer over other standard treatment modalities.
Keywords: Curcumin, miR-203, bladder cancer, Src, Akt2
INTRODUCTION
Bladder cancer is a common malignancy affecting the genitourinary system that is associated with high mortality (1). The majority of tumors are urothelial cell carcinoma (UCC), and of these, 75% cases present with non-muscle invasive disease and ~20–30% present with muscle invasive disease. Though the non-muscle invasive disease has good prognosis, these cases are associated with high recurrence and progression to muscle invasive disease (2) which is often associated with metastases and poor survival (3). Although transurethral resection of bladder tumor is effective therapy, up to 45% of patients will have a recurrence within 1 year and there is risk of tumor progression to muscle invasive and/or metastatic cancer (4). This has led to the use of adjuvant therapy with intravesical agents which prevents recurrence but not tumor progression (4). Also, these chemotherapeutic regimens are often intolerable due to strong systemic toxicity and local irritation. Hence, there is an urgent need to develop better therapeutic interventions for this disease.
It is now being increasingly recognized that dietary factors possess chemopreventive and therapeutic potential in various malignancies, including bladder cancer. These factors have the advantage of being relatively non-toxic. Curcumin (diferuloylmethane), a natural compound present in turmeric, has been recognized as a promising anti-cancer drug and is being developed as a chemopreventive agent in various cancers (5–12). In vitro and in vivo preclinical studies have shown that curcumin has antioxidant, anti-inflammatory, antiproliferative, and proapoptotic activities. In bladder cancer, recent studies have shown that curcumin could prove an effective chemopreventive and chemotherapeutic agent (13). However, the molecular basis of the effects of curcumin are still not completely understood.
Curcumin targets diverse molecules associated with numerous biochemical and molecular cascades via direct molecular interactions and/or epigenetic modulation of gene expression (14). The molecular targets of curcumin include transcriptional factors, growth factors and their receptors, genes regulating cell proliferation and apoptosis (12, 15, 16). Also, it is being recognised that curcumin is an epigenetic agent (14) and epigenetic modulation by curcumin might play a major role in cancer treatment. Curcumin inhibits DNA methyltransferase I (DNMT1) and induces global genomic DNA hypomethylation (17), modulates histone acetyltransferases (HATs) and histone deacetylases (HDACs). Also, it has been proposed that epigenetic modulation of miRNA expression by curcumin may be an important mechanism underlying its biological effects (14, 18).
MicroRNAs (miRNAs) are small, noncoding RNAs that suppress gene expression posttranscriptionally via sequence-specific interactions with the 3’-untranslated regions (UTRs) of cognate mRNA targets (19) and control various key cellular processes such as proliferation, apoptosis, differentiation, and development (20). Alterations of miRNA expression have been described in various human cancers and can arise from either genetic or epigenetic changes (21). A significant proportion of miRNA genes (20–40%) are located close to CpG islands and it has been demonstrated that miRNAs are inactivated by epigenetic mechanisms (22). Thus, epigenetic drugs can cause their reexpression leading to downregulation of target oncogenes. Also, it has been proposed and supported by a few studies that dietary factors can modulate miRNA expression (18). However, this is an area that is largely underexplored and warrants further investigation.
In the present study, we explored the potential modulation of miRNA by curcumin in bladder cancer and identified that miR-203 expression is upregulated by curcumin. We also define a tumor suppressor role for miR-203 in bladder cancer in this study. It was observed that miR-203 is frequently downregulated in bladder cancer. We found that the miR-203 promoter is hypermethylated in bladder cancer cell lines and identified Src kinase and Akt2 as novel targets of miR-203 in bladder cancer. Restoration of miR-203 expression in bladder cancer cell lines inhibited proliferation, migration, invasion and induced cell cycle arrest and apoptosis. We also performed phenocopy experiments using siRNA to validate Src and Akt2 as miR-203 targets in bladder cancer. Importantly, curcumin attenuated the methylation of miR-203 promoter concomitant with an increase in expression of miR-203 target genes Akt2 and Src. Further, curcumin treatment of bladder cancer cell lines mimicked the effects of miR-203 reconstitution and miR-203 target gene knockdowns. Thus, we identify a novel curcumin regulated miR-203 regulatory loop that underlies human bladder cancer development.
MATERIALS AND METHODS
Cell culture
Non-malignant human uroepithelial cell line (SV-HUC-1) and bladder cancer cell lines (T24, J82 and TCCSUP) were obtained from the American Type Culture Collection. Cell line SV-HUC-1 was cultured in F12K medium, T24 in McCoy’s 5A medium, J82 and TCCSUP in Minimum essential medium (Eagle) (MEM), each supplemented with 10% fetal bovine serum (FBS) (Atlanta biologicals) and 1% penicillin/streptomycin (UCSF cell culture facility). For TCCSUP, media was also supplemented with non-essential amino acids and 1 mM sodium pyruvate. Cell lines were maintained in an incubator with a humidified atmosphere of 95% air and 5% CO2 at 37°C.
Treatment of cell lines with epigenetic drugs
Curcumin (Indofine Chemicals) was dissolved in dimethylsulfoxide (DMSO) and cells were treated with curcumin for 3 days. We initially tried different doses of curcumin (1–20 µM) and chose the optimal one (10 µM) for subsequent experiments. Higher doses (>10 µM) showed cytotoxic effects. Control cells were treated with the vehicle control (DMSO). Alternatively, cells were treated with 5-Aza (5 µM) for 3 days and TSA (100 ng) treatment was carried out for 24 hrs.
miRNA/siRNA transfections
Cells were plated in growth medium without antibiotics ~24hrs before transfections. Transient transfections of miRNA precursor (Ambion)/siRNA (Origene) was carried out by using Lipofectamine 2000 (Invitrogen) according to the manufacturers’s protocol. miR-203 precursor (PM10152) or negative control (miR-CON) (AM17110) (Ambion) was used for assays. All miRNA/siRNA transfections were for 72h.
Tissue samples
Formalin-fixed, paraffin-embedded (FFPE) bladder cancer samples were obtained from the Veterans Affairs Medical Center (San Francisco). Informed consent was obtained from all patients. All slides were reviewed and microdissected by a board certified pathologist for the identification of bladder cancer foci as well as adjacent normal epithelium.
RNA extraction and quantitative real-time PCR
Total RNA was extracted from microdissected FFPE tissues using an miRNeasy FFPE Kit (Qiagen) and an RNeasy mini kit (Qiagen) was used for RNA extraction from cultured cells. Mature miRNAs and other mRNAs were assayed using the TaqMan MicroRNA Assays and Gene Expression Assays, respectively, in accordance with the manufacturer's instructions (Applied Biosystems). Samples were normalized to RNU48 or GAPDH (Applied Biosystems), as indicated. The comparative Ct (threshold cycle) method was used to calculate the relative changes in gene expression using the 7500 Fast Real Time PCR System.
DNA Methylation Analysis
Genomic DNA was extracted from human bladder cell lines using a Genomic DNA extraction kit (Qiagen). Bisulfite modification of genomic DNA (1 µg) was done using the Epi-Tect Bisulfite kit (Qiagen) following the manufacturer’s directions. For methylation specific PCR (MSP), specific oligonucleotides were synthesized to amplify methylated or unmethylated miR-203 promoter regions (Table S2) (24). For bisulfite sequencing, bisulfite modified DNA was amplified using primers BS1 and BS2 (Table S2) to amplify ~500 bp region upstream of miR-203 TSS. The mapping of primers on the miR-203 promoter is shown schematically in Fig. 1C (upper panel). The amplification products were confirmed by electrophoresis on agarose gel, cloned into the pCR2.1-Topo vector using a TOPO TA Cloning Kit (Invitrogen). Ten to fifteen colonies were randomly chosen for culture, DNA was purified using PureLink Quick Plasmid Miniprep Kit (Invitrogen) and sequenced by an outside vendor (McLab).
Fig. 1. miR-203 expression is epigenetically downregulated in bladder cancer.
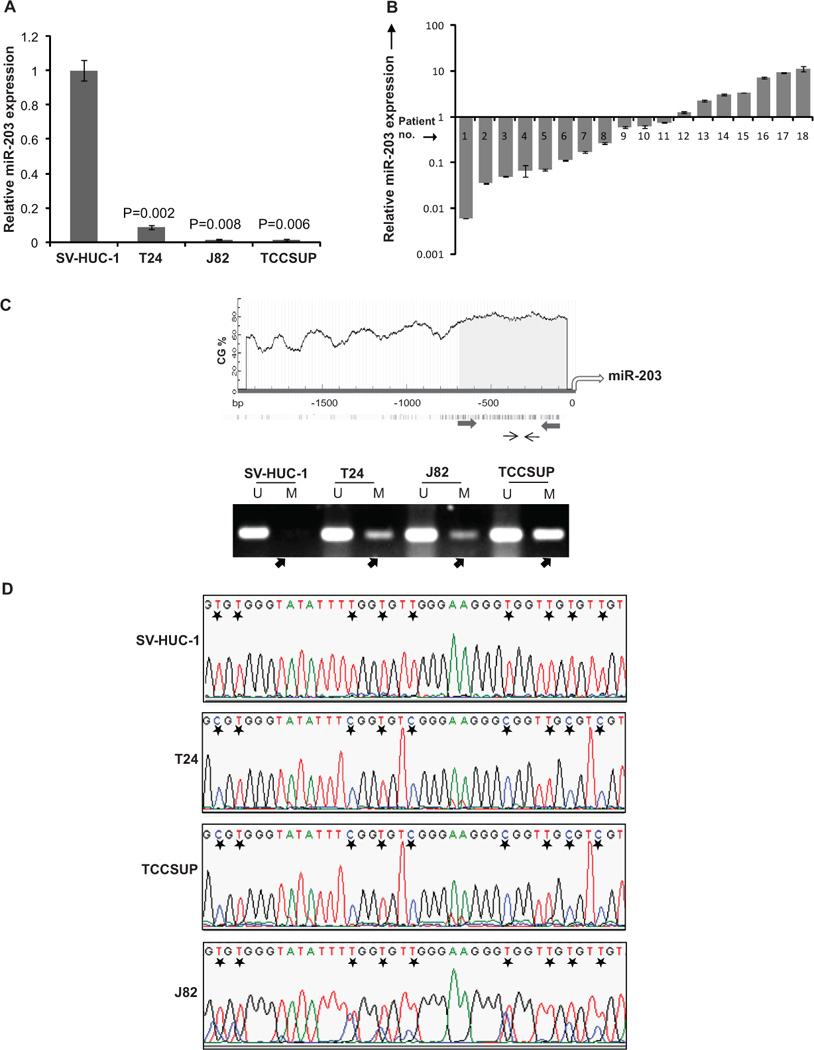
(A) Relative miR-203 expression levels in human bladder cell lines as assessed by RT-PCR. Data were normalized to the RNU48 signal and are represented as mean ± SEM. P-values are for each cell line relative to the SV-HUC1 cell line.
(B) Relative miR-203 expression levels in bladder cancer clinical specimens and patient-matched normal tissues as assessed by RT-PCR. Data were normalized to the RNU48 signal and are represented as mean ± SEM.
(C) Upper panel: Schematic representation of miR-203 5’-upstream region showing the CpG island flanking TSS. Percentage of C+G nucleotides (%CG) is shown and the density of CpG nucleotides are shown for a ~2 kb region upstream of miR-203. Thick convergent arrows depict the position of primers used for bisulfite sequencing and thin arrows indicate the position of MSP primers.
Lower panel: MS-PCR analysis of miR-203 5’ upstream region showing unmethylated (U) or methylated (M) DNA products. Methylation of miR-203 promoter is absent in normal SV-HUC-1 cell line but present in bladder cancer cell lines (indicated by arrows). The corresponding full length electrophoretic gel for this panel is represented in Fig. S1.
(D) Bisulfite DNA sequencing of the miR-203 promoter. A representative portion of the miR-203 promoter sequence is shown for SV-HUC-1 and bladder cancer cell lines (T24 J82, TCCSUP). Asterisks indicate individual CpG sites.
Cell viability, clonability, migratory, and invasion assays
Cell viability was determined at 24, 48 and 72 hours by using the CellTiter 96 AQueousOne Solution Cell Proliferation Assay Kit (Promega), according to the manufacturer's protocol. For colony formation assay, cells were transfected with miR-CON/miR-203 precursor or mock transfected and 24 hours post-transfection, cells were trypsinised, counted and seeded at low density (1000 cells/plate) in culture media and allowed to grow for one week. The cells were then stained with crystal violet and colonies were counted. Cytoselect Cell migration and invasion assay kit (Cell Biolabs, Inc.) was used for migration and invasion assays, according to the manufacturer's protocol as described in (23).
Flow Cytometry
The cells were harvested, washed with cold PBS, and resuspended in the nuclear stain DAPI for cell cycle analysis. Cells were stained with 7-AAD and Annexin-V-FITC using ANNEXIN V-FITC /7-AAD KIT (Beckman Coulter) for apoptosis analysis according to the manufacturer’s protocol. Stained cells were immediately analyzed by FACS (Cell Lab Quanta SC; Beckman Coulter).
Statistics
All quantified data represent an average of at least triplicate samples or as indicated. Error bars represent S.E.M. Statistical significance was determined by the Student’s t test, and two-tailed p < .05 were considered significant.
Supplemental data
The supplemental data includes supplemental experimental procedures and supplemental figures.
RESULTS
Curcumin is currently being developed as a natural epigenetic agent for prophylaxis and treatment of various cancers. To explore the potential epigenetic modulation of miRNA expression by curcumin in bladder cancer, we treated bladder cancer cell lines with 10uM curcumin or vehicle control (DMSO) for 72 hrs followed by miRNA microarray analysis (data not shown). This analysis identified miR-203, miR-26b and miR-1826 were significantly upregulated by curcumin treatment (Table S1). We focused on miR-203 as miR-203 is a very well studied miRNA that has been reported to be downregulated in various cancers (24, 25). However, the functional significance of this miRNA in bladder cancer is not fully known. Hence, we explored the functional role of miR-203 in bladder cancer in this study.
miR-203 expression is epigenetically downregulated in bladder cancer
To evaluate the role of miR-203 in bladder cancer, miR-203 expression was assayed in human bladder cell lines, which included normal immortalized uroepithelial cell line (SV-HUC-1) and bladder carcinoma cell lines (T24, J82 and TCCSUP). It was consistently observed that relative miR-203 expression is specifically attenuated in all bladder cancer cell lines (Fig. 1A). We extended our analysis to clinical malignancies by assessing miR-203 levels in microdissected bladder cancer tissues (n=18) and matched adjacent normal regions by real-time PCR (Fig. 1B). While the expression of miR-203 was unaltered in 1/18 cases (~5%) and higher in 6/18 cases (~33%), a major fraction of tissues (11/18, ~61%) showed lower miR-203 levels (<75%) relative to matched normal tissues (Fig. 1B). We further analysed whether miR-203 is silenced by epigenetic mechanisms in bladder cancer since the upstream genomic region flanking the transcriptional start site (TSS) of miR-203 has a CpG island. Methylation specific PCR (MSP) and bisulfite sequencing analysis of bisulfite modified DNA from bladder cell lines showed that miR-203 is unmethylated in SV-HUC-1 cells and considerably methylated in T24, J82 and TCCSUP cell lines (Fig. 1C, 1D). This pattern of DNA methylation was consistent with the miR-203 expression patterns observed in these cell lines suggesting that miR-203 locus is epigenetically downregulated in bladder cancer.
Curcumin and 5-Aza augment miR-203 expression in bladder cancer
We confirmed the microarray expression data showing upregulation of miR-203 by curcumin in bladder cancer cell lines. miR-203 expression was analysed in curcumin treated bladder cancer cell lines by miR qRT-PCR. Curcumin treatment increased miR-203 expression ~ 2-fold in all the cell lines (Fig. 2A and S2). Since promoter methylation can be reversed using specific epigenetic drugs like 5-Aza, we also treated T24 bladder cancer cell line with DNA demethylating agent, 5-Aza alone or in combination with HDAC inhibitor, Trichostatin A (TSA) in addition to curcumin. Treatment was followed by miR-203 expression analysis and miR-203 promoter methylation analysis. As expected, 5Aza-C augmented miR-203 expression and also significantly demethylated the miR-203 promoter (Fig. 2A, 2B). Curcumin in combination with 5-Aza led to further augmentation of miR-203 expression (Fig. 2A). Consistent with miR-203 expression data, methylation analysis showed that curcumin treatment leads to partial demethylation of the miR-203 promoter (Fig. 2B, 2C) suggesting that curcumin causes epigenetic modulation of miR-203 expression.
Fig. 2. Curcumin and 5-Aza augment miR-203 expression in T24 bladder cancer cell line.
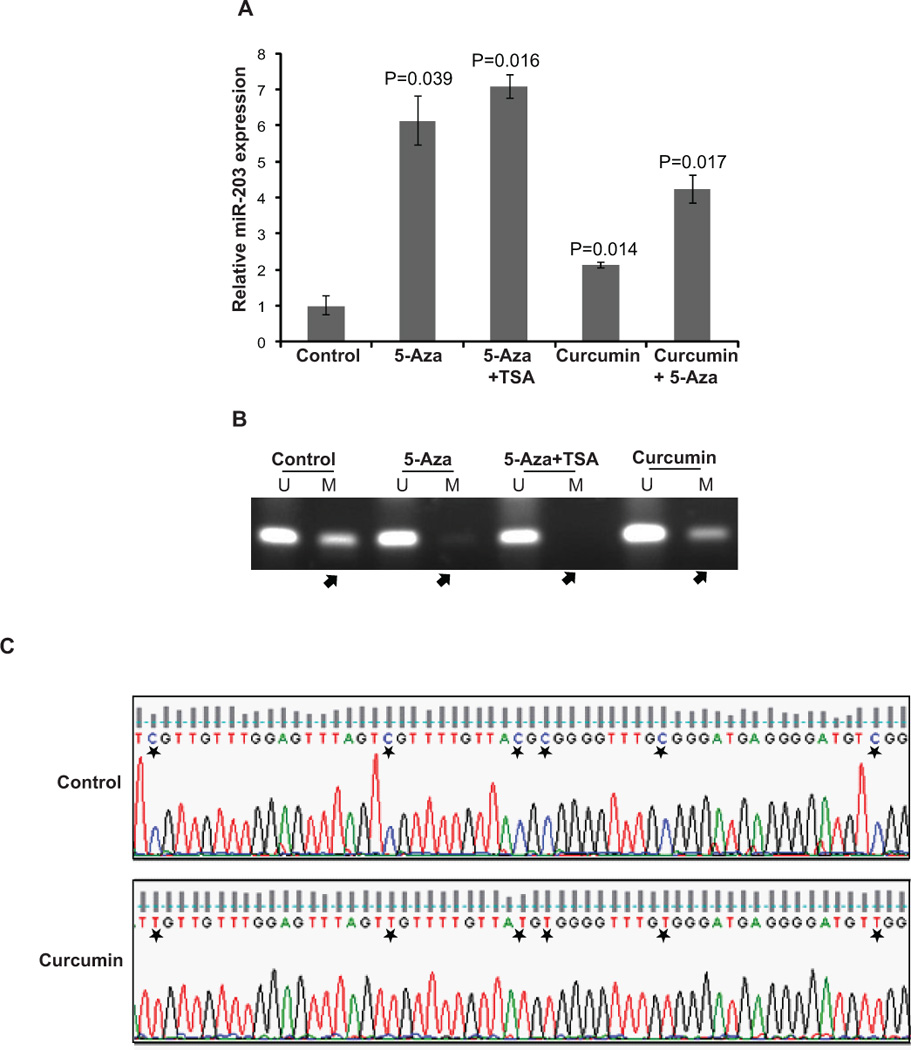
To examine the potential modulation of miR-203 by epigenetic drugs, T24 cells were treated with various epigenetic dugs either singly or in combination. Treatments included vehicle control (C) or 5-Aza (5 µM), 5-Aza (5 µM) +TSA (100ng), curcumin (10 µM), curcumin (10 µM) + 5-Aza (5 µM).
(A) Relative miR-203 expression was assessed after the various treatments by RT-PCR. Curcumin and 5-Aza alone or in combination significantly upregulated miR-203 expression.
(B) MS-PCR analysis of the human miR-203 upstream region following various treatments. Curcumin and 5-Aza treatment leads to partial demethylation of the miR-203 promoter (indicated by arrows). The corresponding full length electrophoretic gel for this section is shown in Fig. S3.
(C) Bisulfite DNA sequencing for miR-203 promoter following curcumin/control treatment. Curcumin treatment leads to demethylation of CpG sites in miR-203 promoter as indicated by asterisks.
Akt2 and Src are novel targets of miR-203 in bladder cancer
To identify potential targets of miR-203 in bladder cancer, we used two algorithms, miRANDA (26) and TargetScan (27), that predict the mRNA targets of a miRNA. Among the putative targets of miR-203, several proteins with oncogenic activity including c-Jun, Src, Yes1, Fos, MYB, ETS1, Survivin were represented. To validate miR-203 targets in bladder cancer, T24 and J82 cell lines were transfected with miR-203 precursor or control miRNA precursor (miR-CON) resulting in miR-203 overexpression as determined by RT-PCR analysis (Fig. 3A). We performed Western blot analysis for the putative targets using the transfectants. Interestingly, we found that reintroduction of miR-203 led to reduced protein levels of Akt2, Src kinase, c-jun and Survivin (Fig. 3B). The serine/threonine kinase Akt2/PKBβ is a member of the PI3K/Akt signaling pathway and Src is a non-receptor tyrosine kinase belonging to the Src family of kinases (SFK). Survivin is a small inhibitor of apoptosis (IAP) protein that is differentially expressed in cancer (28) and is also a miR-203 target in prostate cancer (23, 29). The 3’-UTR of Akt2, Src and Survivin mRNA have putative miR-203 binding sites (Fig. 3C). Luciferase reporter assays with control, Akt2, Src and Survivin 3’-UTR reporter constructs in miR-203/miR-CON expressing or mock transfected T24 cells (Fig. 3C) showed that miR-203 represses theses targets directly. We subsequently focused on miR-203 mediated regulation of the Src-Akt axis in bladder cancer and examined the downstream signaling events of the Src-Akt axis upon miR-203 overexpression in bladder cancer by Western blot analysis of key signaling molecules (Fig. 3D). Src is involved in multiple signaling pathways including PI3K/Akt, β-catenin/c-Myc/cyclin D1 that induce growth, survival and migration in various cancers (30). It was observed that p-Akt and p-β-catenin were reduced upon miR-203 reintroduction (Fig. 3D).
Fig. 3. Akt2 and Src are novel targets of miR-203 in bladder cancer.
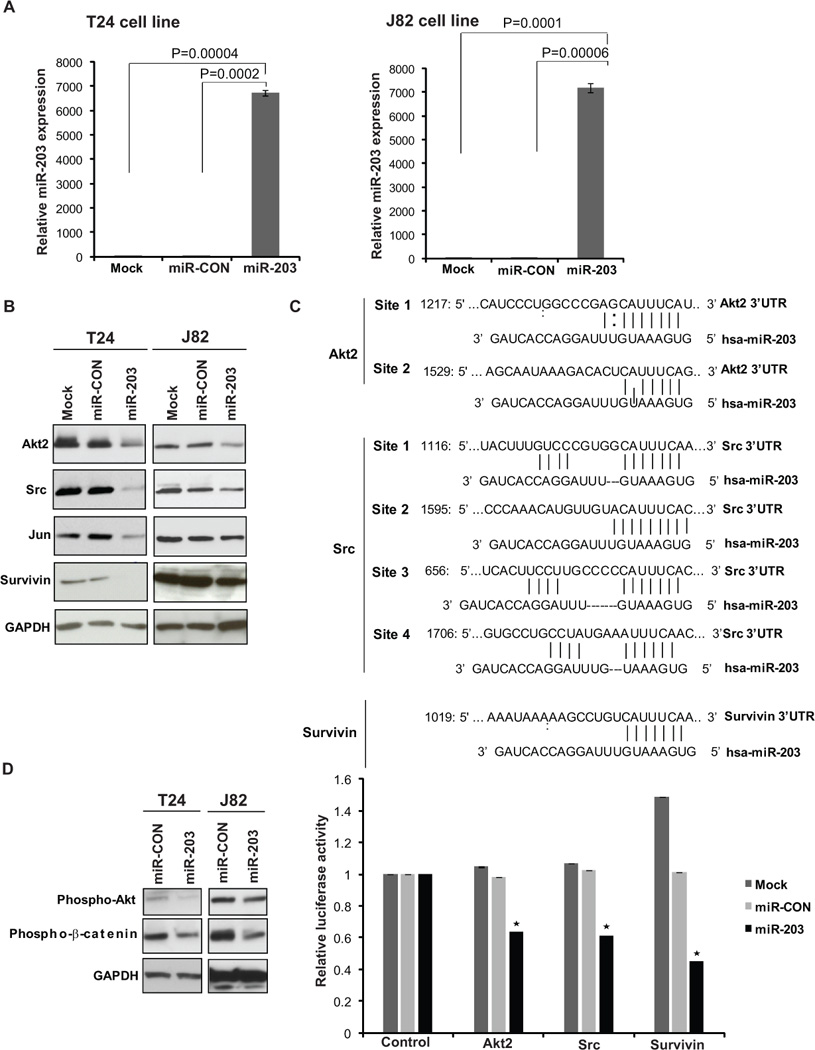
(A) Relative miR-203 expression in T24 cells (left panel) or J82 cells (right panel) transfected with a control miR/miR-203 or mock transfected cells as assessed by real time PCR.
(B) Western blot analysis for endogenous Akt2, Src, c-jun, survivin protein in T24/J82 cells transfected as indicated. GAPDH was used a loading control.
(C) Schematic representation of 3’-UTRs of Akt2, Src, Survivin showing putative miR-203 target site/sites and luciferase activity assays with the indicated 3’ UTR construct or control luciferase construct contranfected with mock/miR-CON/miR-203 in T24 cells. Firefly luciferase values were normalized to renilla luciferase activity and plotted as relative luciferase activity (* P< .05 as compared to miR-CON).
Restoration of miR-203 expression suppresses tumorigenicity in bladder cancer cells
Since both Akt2 and Src are known to be involved in cell growth, proliferation, survival and motility of cancer cells (31, 32), we sought to determine if miR-203 mediated downregulation of these kinases has effects on the these attributes of tumorigenicity in bladder cancer cells. Following transient transfections of miR-203/miR-CON precursors in T24 and J82 cell lines, various functional assays were performed (Fig. 4 and S4). A significant decrease in cell viability was observed over time in T24/J82 cells expressing miR-203 (Fig. 4A, S4A) as compared to cells expressing miR-CON. miR-203-transfected cells also had low colony formation ability, reduced invasiveness and migration as compared to control cells (Fig. 4B–C, S4B–C). FACS (fluorescence activated cell sorting) analysis showed that reexpression of miR-203 leads to a significant increase in the number of cells in the G0-G1 phase of the cell cycle compared to miR-CON suggesting that miR-203 causes a G0-G1 arrest in miR-203 transfected cells (Fig. 4D, S4D). Also, the apoptotic cell fractions (Early apoptotic + Apoptotic) were significantly increased upon miR-203 reexpression compared to control cells with a concomitant decrease in the viable cell population (Fig. 4E, S4E). These results indicate that suppression of Src/Akt by miR-203 inhibits bladder cancer cell proliferation, invasion, migration and induces apoptosis.
Fig. 4. Restoration of miR-203 expression suppresses tumorigenicity in vitro in T24 cell line.
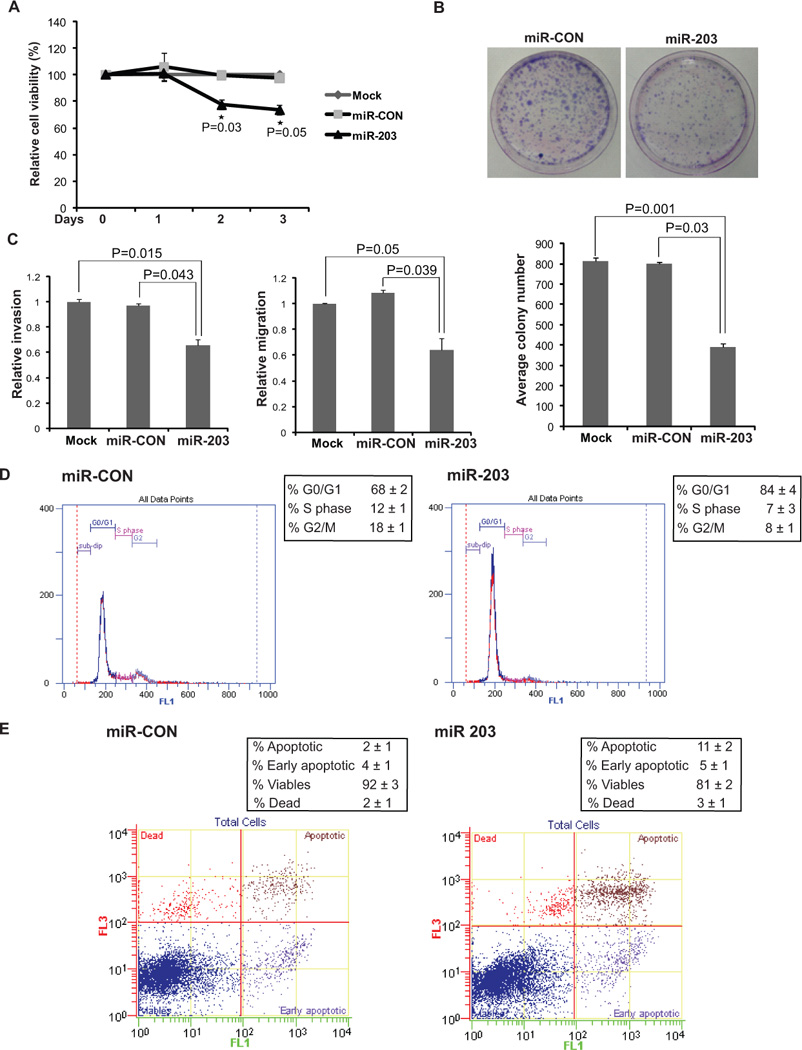
(A) Cellular viability assay showing that miR-203 reexpression significantly decreases the viability of T24 cells (* P< .05).
(B) Colony formation assay in T24 transfectants.
(C) Invasion assay (left panel) and migration assay (left panel) after indicated transfections.
(D) Cell cycle assay after miR-CON (left panel) or miR-203 (right panel) treatments showing induction of G0/G1 cell cycle arrest by miR-203 (P=0.050).
(E) Apoptosis assay showing induction of apoptosis by miR-203 overexpression (P= 0.026).
Akt2 and Src inhibition by siRNA mimics miR-203 reconstitution in T24 bladder cancer cells
Next we sought to determine if miR-203 mediates its anti-tumorigenic effects primarily through Src/Akt2 in bladder cancer. To address this, we performed phenocopy experiments where we treated T24 cells with Akt2 and Src siRNA followed by functional assays. We initially tested three sets of siRNA against both the genes to achieve efficient knockdown as assessed by RT-PCR and immunoblot analysis (Fig. 5A). si2 against both genes produced the most efficient knockdown and was used in subsequent experiments. These results show that siRNA inhibition of Akt2 and Src led to decreased cellular viability, migration and invasion of T24 cells (Fig. 5B–D). Apoptosis assay showed that apoptotic cell fractions (Early apoptotic + Apoptotic) were significantly increased upon Akt2 and Src knockdown compared to control siRNA-treated cells similar to that observed upon miR-203 reintroduction in T24 cells (Fig. 5E). These results suggest that inhibition of Akt2 and Src by miR-203 reintroduction is responsible for its anti-tumorigenic effects observed in bladder cancer.
Fig. 5. Akt2 and Src inhibition by siRNA mimics miR-203 reconstitution in T24 bladder cancer cells.
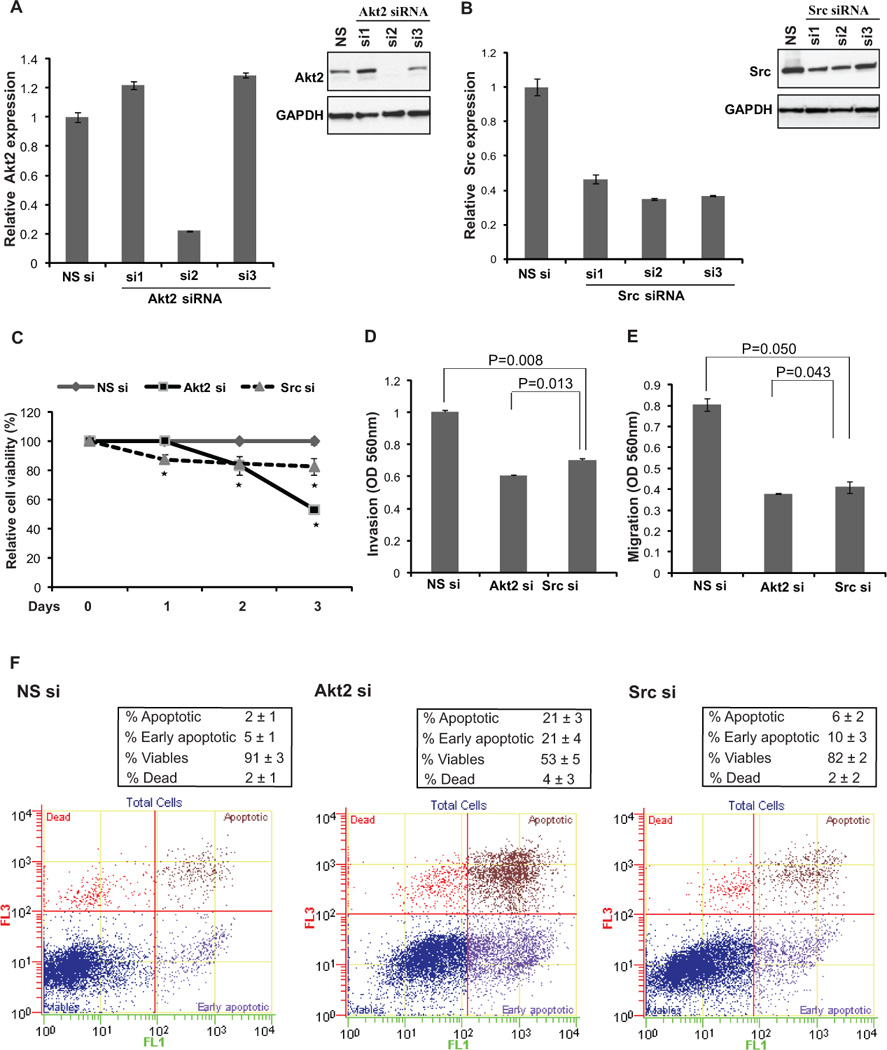
T24 cells were transfected with siRNA specific to Akt2 or Src and a control nonspecific (NS) siRNA at 50 nM for 72 h followed by various functional assays.
(A) Relative Akt2 and (B) Src mRNA expression and protein expression after siRNA transfections as assessed by real time PCR and immunoblotting respectively.
(C) Cellular viability assay after siRNA transfections indicating that Akt2 and Src knockdown significantly decreases the viability of T24 cells (* P< .05).
(D) Invasion assay and (E) Migration assay after indicated transfections.
(F) Apoptosis assay in T24 cells after NS siRNA (left panel) or Akt2 siRNA (middle panel) or Src siRNA (right panel) treatments.
Curcumin treatment mimics the effect of miR-203 reconstitution and Akt2/Src knockdown
In view of our present evidence that curcumin promotes hypomethylation of miR-203 promoter leading to augmentation of its expression, we asked if curcumin treatment mimics the effect of miR-203 reconstitution and also the effects of knockdown of miR-203 target genes. To examine this, we treated the T24 cell line with curcumin followed by cell viability, migration, invasion and apoptosis assays (Fig. 6). Curcumin treatment led to a significant decrease in cellular viability (Fig. 6A). Transwell assays showed that curcumin also impaired the migratory and invasive capabilities of bladder cancer cells (Fig. 6B). Flow cytometry analysis of Annexin V-FITC-7-AAD stained cells showed that compared to vehicle-treated control cells, curcumin caused a significant increase in apoptosis in T24 bladder cancer cells (Fig. 6C). These in vitro effects were similar to the effects seen upon miR-203 reexpression and Akt2/Src gene knockdown validating the curcumin-mediated regulation of the tumor suppressive functions of miR-203. To further consolidate these findings, we also determined the expression levels of miR-203 target genes Akt2/Src after curcumin treatment (Fig. 6D). It was observed that curcumin downregulated the expression of these two genes as assessed by Western blot analysis. These results indicate that the effects of curcumin were due to induction of miR-203 expression.
Fig. 6. Curcumin treatment mimics the effect of miR-203 reconstitution and Akt2/Src knockdown in T24 cells.
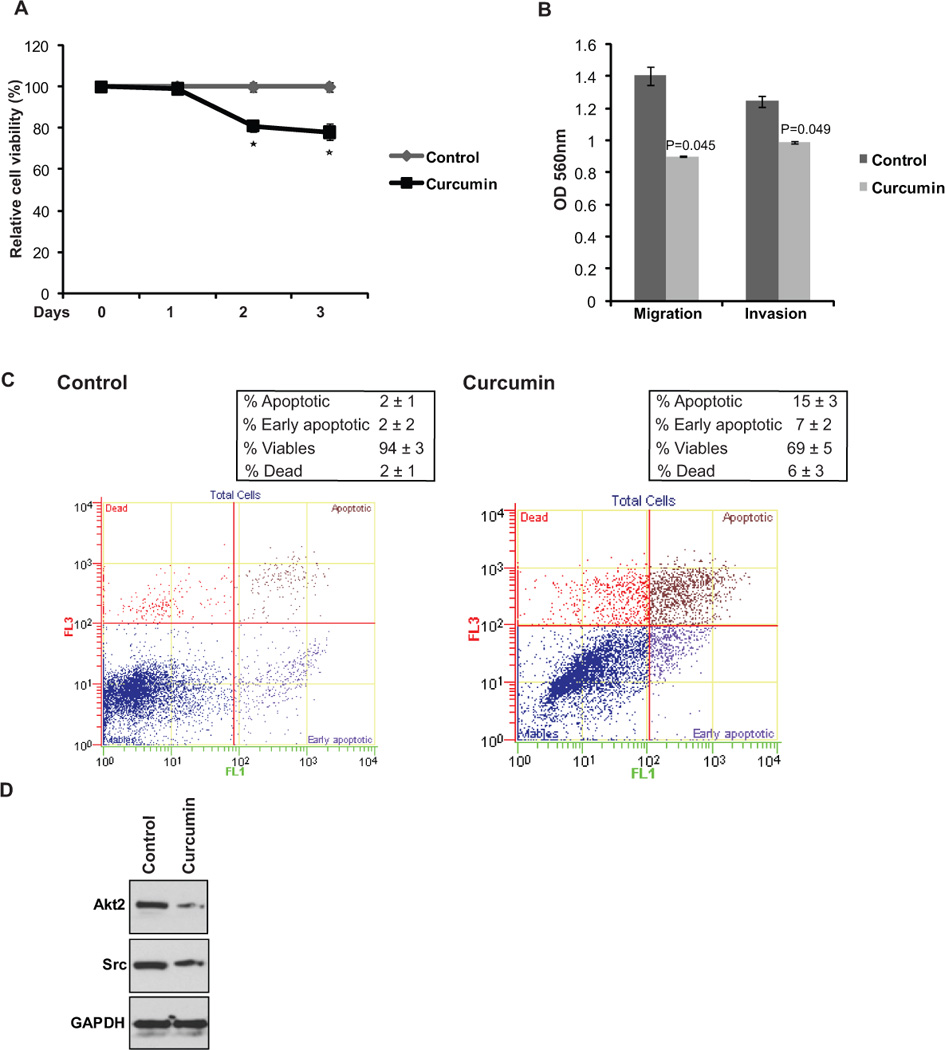
T24 cells were treated with vehicle control (DMSO) or curcumin (10 µM) followed by various functional assays.
(A) Cell viability assay after treatments (* P< .05).
(B) Invasion assay and migration assay upon conrol/curcumin treatment.
(C) Apoptosis assay in T24 cells after control (left panel) or curcumin (right panel) treatment.
(D) Western blot analysis in T24 cells treated as indicated. GAPDH was used a loading control.
DISCUSSION
In this report, we define for the first time, that curcumin directly induces a tumor-suppressive miRNA, miR-203, in bladder cancer cells. Our study suggests that curcumin leads to hypomethylation of the miR-203 promoter and subsequent upregulation of miR-203. miR-203 upregulation in turn, leads to downrgulation of miR-203 target genes Akt2 and Src that culminates in decreased proliferation and increased apoptosis of bladder cancer cells. Thus, we have identified a miRNA mediated regulatory loop that potentially underlies the prophylactic effects of curcumin in bladder cancer.
Over the last two decades, accumulating evidence has indicated that curcumin is a potent anti-cancer agent that has chemopreventive and chemotherapeutic potential. However, its precise mode of action is not completely understood, hindering its incorporation into the mainstream of clinical treatment regimens. It has been proposed that regulation of miRNAs by curcumin may underlie its chemopreventive and chemotherapeutic effects (33). A recent study showed that curcumin alters miRNA expression in human pancreatic cells, upregulating miR-22 and downregulating miR-199a* (34). Another study showed upregulation of miR- 15a and miR-16 expression in curcumin-treated MCF-7 breast cancer cells (35). However, there are very few reports on curcumin mediated regulation of miRNA expression and this area is largely unexplored. Our present study was focused on identifying curcumin regulated miRNAs in bladder cancer. Towards this, we performed miRNA microarray after curcumin treatment of bladder cancer cell lines and identified miR-203 as a curcumin-regulated miRNA.
MiRNAs represent potential disease biomarkers and novel therapeutic targets. In bladder cancer, several large-scale profiling studies describe altered expression of miRNAs (36) that varies with tumor type and molecular pathways (37). However, to extrapolate these findings to translational applications, single-miRNA focused studies in bladder cancer are needed to explore the molecular interactions of important miRNAs with their cognate targets. Here we investigated the functional significance of miR-203 in bladder cancer and identified novel targets for this microRNA. miR-203 was found to be downregulated in bladder cancer cell lines and in a significant proportion (61%) of clinical tissues. However, miR-203 expression was higher in 6/18 tissue samples that we analysed indicating that additional studies with more clinical samples are warranted to assess its prognostic significance.
We further showed that the miR-203 promoter is hypermethylated in bladder cancer cell lines. miR-203 has been reported to be epigenetically silenced in hematopoetic malignancies, hepatocellular carcinomas and oral cancers (24, 25, 38). However, this is the first report that shows epigenetic inactivation of this important miRNA in bladder cancer. Interestingly, we found that curcumin treatment leads to partial demethylation of the miR-203 promoter (Fig. 2B, 2C) suggesting that curcumin causes epigenetic modulation of miR-203 expression. These effects of curcumin on miR-203 expression and methylation were similar to those of the DNA demethylating agent, 5-aza. The toxicity associated with the use of epigenetic drugs like 5-Aza warrants investigations for safer, natural, non-toxic epigenetic agents. Curcumin is one such epigenetic agent (14). It has been reported that curcumin inhibits DNMT1 and induces global genomic DNA hypomethylation (17). Our results lend support to the hypothesis that epigenetic modulation of miRNA expression by curcumin may be an important mechanism underlying its biological effects (14, 18).
We also show that miR-203 has tumor suppressive effects in bladder cancer cells as miR-203 reconstitution resulted in decreased cellular viability, G0-G1 arrest and induction of apoptosis. Infact, the anti-proliferative function of this miRNA was indicated several years ago (29). An obstacle to understanding miR-203 function in cellular proliferation and apoptosis is the lack of experimentally validated targets. p63 is a well known target for miR-203 (39, 40). Other targets are still being defined (24, (23). We identified Akt2, Src, c-jun, survivin as miR-203 targets in bladder cancer. miR-203 mediated regulation of survivin is a highly significant finding as this nodal protein orchestrates extensive, tumor specific signaling networks and is an attractive drug target (41). However, in our present study, we focused on Akt2 and Src kinase as mediators of the role of miR-203 in bladder cancer cell proliferation and survival. The serine/threonine kinase Akt2/PKBβ is a core member of the phophatidylinositol 3-kinase (PI3K)/Akt signaling pathway that play key roles in fundamental cellular processes including proliferation, survival, migration and differentiation. Akt is activated by phospholipid binding and activation loop phosphorylation at Thr308 by PDK1 (4) and by phosphorylation within the carboxy terminus at Ser473. Akt functions as a cardinal nodal point for transducing extracellular (growth factors including insulin, IGF-1, EGF) and intracellular (such as mutated/activated receptor tyrosine kinases, PTEN, Ras and Src) signals. Akt2 is frequently amplified and overexpressed in human cancers (42, 43). In our present study, siRNA mediated inhibition of Akt2 led to decreased viability, migration and invasion of T24 bladder cancer cells along with induction of apoptosis.
Src, a prototypical member of SFK, also regulates key cellular processes including proliferation, survival, adhesion and motility (31, 32). Src can channel phosphorylation signals through PI3K/Akt, pathways and Ras/Raf/extracellular signal-regulated kinase 1/2 (Erk1/2). Elevated Src kinase activity has been reported in a wide range of human tumors with Src activity increasing with the disease progression (44, 45). In bladder cancer, Src activity and expression has been reported to be elevated in low grade bladder lesions (46) though there are conflicting reports on the role of Src in progression and metastasis of bladder cancer (47, 48). In this study, we inhibited Src expression in T24 cells and found increased apoptosis concomitant with decreased proliferation pointing to growth promoting activities of Src. Also, the effects of miR-203 overexpression on Src downregulation was coupled with downregulation of key signaling molecules of the Src-Akt axis including p-Akt and p-β-catenin. Overall, our results suggest that depletion of Akt2 and Src tends to phenocopy the effects of miR-203 reconstitution in bladder cancer. This lends support to our hypothesis that miR-203 primarily mediates its effects on cellular proliferation and survival through these targets in bladder cancer. Both Akt and Src represent potential targets for cancer therapy and there is currently much interest in developing agents to target these proteins (42, 49). Most importantly, in this study, curcumin treatment led to decreased expression of these oncogenic miR-203 targets concomitant with reduced proliferation and increased apoptosis of bladder cancer cells. Interestingly, the effects of curcumin were similar to the effects of Akt2/Src knockdown.
In conclusion, our study suggests that curcumin has the potential to be developed as an epigenetic agent for bladder cancer as it upregulates tumor suppressor miR-203 in bladder cancer by promoting DNA hypomethylation of the miR-203 promoter that in turn, leads to downregulation of its target oncogenes Akt2/Src and induction of apoptosis, inhibition of cellular proliferation, migration and invasion. To our knowledge this is the first report that defines the epigenetic effects brought about by curcumin on a miRNA promoter and its direct biological consequences. In view of our present results and the relative non-toxicity associated with the use of curcumin, we suggest that curcumin may offer an important therapeutic advantage in the clinical management of refractory bladder cancer over other standard treatments.
Supplementary Material
ACKNOWLEDGMENTS
We thank Dr. Roger Erickson for his support and assistance with the preparation of the manuscript. This study was supported by NIH grants R01CA130860, R01CA138642, VA REAP award, and Merit Review grants (PI: R.D.).
REFERENCES
- 1.Jemal A, Siegel R, Ward E, Hao Y, Xu J, Murray T, et al. Cancer statistics. CA Cancer J Clin. 2008;58:71–96. doi: 10.3322/CA.2007.0010. [DOI] [PubMed] [Google Scholar]
- 2.Dinney CP, McConkey DJ, Millikan RE, Wu X, Bar-Eli M, Adam L, et al. Focus on bladder cancer. Cancer Cell. 2004;6:111–116. doi: 10.1016/j.ccr.2004.08.002. [DOI] [PubMed] [Google Scholar]
- 3.Stein JP, Lieskovsky G, Cote R, Groshen S, Feng AC, Boyd S, et al. Radical cystectomy in the treatment of invasive bladder cancer: long-term results in 1,054 patients. J Clin Oncol. 2001;19:666–675. doi: 10.1200/JCO.2001.19.3.666. [DOI] [PubMed] [Google Scholar]
- 4.Williams SK, Hoenig DM, Ghavamian R, Soloway M. Intravesical therapy for bladder cancer. Expert Opin Pharmacother. 2010;11:947–958. doi: 10.1517/14656561003657145. [DOI] [PubMed] [Google Scholar]
- 5.Aggarwal BB, Kumar A, Bharti AC. Anticancer potential of curcumin: preclinical and clinical studies. Anticancer Res. 2003;23:363–398. [PubMed] [Google Scholar]
- 6.Everett PC, Meyers JA, Makkinje A, Rabbi M, Lerner A. Preclinical assessment of curcumin as a potential therapy for B-CLL. Am J Hematol. 2007;82:23–30. doi: 10.1002/ajh.20757. [DOI] [PubMed] [Google Scholar]
- 7.Li L, Aggarwal BB, Shishodia S, Abbruzzese J, Kurzrock R. Nuclear factor-kappaB and IkappaB kinase are constitutively active in human pancreatic cells, and their down-regulation by curcumin (diferuloylmethane) is associated with the suppression of proliferation and the induction of apoptosis. Cancer. 2004;101:2351–2362. doi: 10.1002/cncr.20605. [DOI] [PubMed] [Google Scholar]
- 8.Li L, Ahmed B, Mehta K, Kurzrock R. Liposomal curcumin with and without oxaliplatin: effects on cell growth, apoptosis, and angiogenesis in colorectal cancer. Mol Cancer Ther. 2007;6:1276–1282. doi: 10.1158/1535-7163.MCT-06-0556. [DOI] [PubMed] [Google Scholar]
- 9.Li L, Braiteh FS, Kurzrock R. Liposome-encapsulated curcumin: in vitro and in vivo effects on proliferation, apoptosis, signaling, and angiogenesis. Cancer. 2005;104:1322–1331. doi: 10.1002/cncr.21300. [DOI] [PubMed] [Google Scholar]
- 10.Li M, Zhang Z, Hill DL, Wang H, Zhang R. Curcumin, a dietary component, has anticancer, chemosensitization, and radiosensitization effects by down-regulating the MDM2 oncogene through the PI3K/mTOR/ETS2 pathway. Cancer Res. 2007;67:1988–1996. doi: 10.1158/0008-5472.CAN-06-3066. [DOI] [PubMed] [Google Scholar]
- 11.Liang T, Chen MJ, Zhou KY, Tang XD, Wang XG. Induction of apoptosis in human nasopharyngeal carcinoma cell line CNE-2Z by curcumin. Ai Zheng. 2004;23:1651–1654. [PubMed] [Google Scholar]
- 12.Nakamura K, Yasunaga Y, Segawa T, Ko D, Moul JW, Srivastava S, et al. Curcumin down-regulates AR gene expression and activation in prostate cancer cell lines. Int J Oncol. 2002;21:825–830. [PubMed] [Google Scholar]
- 13.Tian B, Wang Z, Zhao Y, Wang D, Li Y, Ma L, et al. Effects of curcumin on bladder cancer cells and development of urothelial tumors in a rat bladder carcinogenesis model. Cancer Lett. 2008;264:299–308. doi: 10.1016/j.canlet.2008.01.041. [DOI] [PubMed] [Google Scholar]
- 14.Fu S, Kurzrock R. Development of curcumin as an epigenetic agent. Cancer. 2010;116:4670–4676. doi: 10.1002/cncr.25414. [DOI] [PubMed] [Google Scholar]
- 15.Sarkar FH, Li Y, Wang Z, Kong D. Cellular signaling perturbation by natural products. Cell Signal. 2009;21:1541–1547. doi: 10.1016/j.cellsig.2009.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mukhopadhyay A, Bueso-Ramos C, Chatterjee D, Pantazis P, Aggarwal BB. Curcumin downregulates cell survival mechanisms in human prostate cancer cell lines. Oncogene. 2001;20:7597–7609. doi: 10.1038/sj.onc.1204997. [DOI] [PubMed] [Google Scholar]
- 17.Liu Z, Xie Z, Jones W, Pavlovicz RE, Liu S, Yu J, et al. Curcumin is a potent DNA hypomethylation agent. Bioorg Med Chem Lett. 2009;19:706–709. doi: 10.1016/j.bmcl.2008.12.041. [DOI] [PubMed] [Google Scholar]
- 18.Saini S, Majid S, Dahiya R. Diet, microRNAs and prostate cancer. Pharm Res. 2010;27:1014–1026. doi: 10.1007/s11095-010-0086-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136:215–233. doi: 10.1016/j.cell.2009.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Schickel R, Boyerinas B, Park SM, Peter ME. MicroRNAs: key players in the immune system, differentiation, tumorigenesis and cell death. Oncogene. 2008;27:5959–5974. doi: 10.1038/onc.2008.274. [DOI] [PubMed] [Google Scholar]
- 21.Ventura A, Jacks T. MicroRNAs and cancer: short RNAs go a long way. Cell. 2009;136:586–591. doi: 10.1016/j.cell.2009.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Calin GA, Sevignani C, Dumitru CD, Hyslop T, Noch E, Yendamuri S, et al. Human microRNA genes are frequently located at fragile sites and genomic regions involved in cancers. Proc Natl Acad Sci U S A. 2004;101:2999–3004. doi: 10.1073/pnas.0307323101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Saini S, Majid S, Yamamura S, Tabatabai ZL, Suh SO, Shahryari V, et al. Regulatory role of miR-203 in prostate cancer progression and metastasis. Clin Cancer Res. 2010;PMID:21159887. doi: 10.1158/1078-0432.CCR-10-2619. [DOI] [PubMed] [Google Scholar]
- 24.Furuta M, Kozaki KI, Tanaka S, Arii S, Imoto I, Inazawa J. miR-124 and miR-203 are epigenetically silenced tumor-suppressive microRNAs in hepatocellular carcinoma. Carcinogenesis. 2010;31:766–776. doi: 10.1093/carcin/bgp250. Epub 2009. [DOI] [PubMed] [Google Scholar]
- 25.Bueno MJ, Perez de Castro I, Gomez de Cedron M, Santos J, Calin GA, Cigudosa JC, et al. Genetic and epigenetic silencing of microRNA-203 enhances ABL1 and BCR-ABL1 oncogene expression. Cancer Cell. 2008;13:496–506. doi: 10.1016/j.ccr.2008.04.018. [DOI] [PubMed] [Google Scholar]
- 26.Betel D, Wilson M, Gabow A, Marks DS, Sander C. The microRNA.org resource: targets and expression. Nucleic Acids Res. 2008;36:D149–D153. doi: 10.1093/nar/gkm995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Grimson A, Farh KK, Johnston WK, Garrett-Engele P, Lim LP, Bartel DP. MicroRNA targeting specificity in mammals: determinants beyond seed pairing. Mol Cell. 2007;27:91–105. doi: 10.1016/j.molcel.2007.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Altieri DC. The case for survivin as a regulator of microtubule dynamics and cell-death decisions. Curr Opin Cell Biol. 2006;18:609–615. doi: 10.1016/j.ceb.2006.08.015. [DOI] [PubMed] [Google Scholar]
- 29.Cheng AM, Byrom MW, Shelton J, Ford LP. Antisense inhibition of human miRNAs and indications for an involvement of miRNA in cell growth and apoptosis. Nucleic Acids Res. 2005;33:1290–1297. doi: 10.1093/nar/gki200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chang YM, Bai L, Liu S, Yang JC, Kung HJ, Evans CP. Src family kinase oncogenic potential and pathways in prostate cancer as revealed by AZD0530. Oncogene. 2008;27:6365–6375. doi: 10.1038/onc.2008.250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Martin GS. The hunting of the Src. Nat Rev Mol Cell Biol. 2001;2:467–475. doi: 10.1038/35073094. [DOI] [PubMed] [Google Scholar]
- 32.Thomas SM, Brugge JS. Cellular functions regulated by Src family kinases. Annu Rev Cell Dev Biol. 1997;13:513–609. doi: 10.1146/annurev.cellbio.13.1.513. [DOI] [PubMed] [Google Scholar]
- 33.Li Y, Kong D, Wang Z, Sarkar FH. Regulation of microRNAs by natural agents: an emerging field in chemoprevention and chemotherapy research. Pharm Res. 2010;27:1027–1041. doi: 10.1007/s11095-010-0105-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sun M, Estrov Z, Ji Y, Coombes KR, Harris DH, Kurzrock R. Curcumin (diferuloylmethane) alters the expression profiles of microRNAs in human pancreatic cancer cells. Mol Cancer Ther. 2008;7:464–473. doi: 10.1158/1535-7163.MCT-07-2272. [DOI] [PubMed] [Google Scholar]
- 35.Yang J, Cao Y, Sun J, Zhang Y. Curcumin reduces the expression of Bcl-2 by upregulating miR-15a and miR*16 in MCF-7 cells. Med Oncol. 2010;27:1114–1118. doi: 10.1007/s12032-009-9344-3. [DOI] [PubMed] [Google Scholar]
- 36.Catto JW, Alcaraz A, Bjartell AS, De Vere White R, Evans CP, Fussel S, et al. MicroRNA in Prostate, Bladder, and Kidney Cancer: A Systematic Review. Eur Urol. 2011;59:671–681. doi: 10.1016/j.eururo.2011.01.044. [DOI] [PubMed] [Google Scholar]
- 37.Catto JW, Miah S, Owen HC, Bryant H, Myers K, Dudziec E, et al. Distinct microRNA alterations characterize high- and low-grade bladder cancer. Cancer Res. 2009;69:8472–8481. doi: 10.1158/0008-5472.CAN-09-0744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kozaki K, Imoto I, Mogi S, Omura K, Inazawa J. Exploration of tumor-suppressive microRNAs silenced by DNA hypermethylation in oral cancer. Cancer Res. 2008;68:2094–2105. doi: 10.1158/0008-5472.CAN-07-5194. [DOI] [PubMed] [Google Scholar]
- 39.Yi R, Poy MN, Stoffel M, Fuchs E. A skin microRNA promotes differentiation by repressing 'stemness'. Nature. 2008;452:225–229. doi: 10.1038/nature06642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lena AM, Shalom-Feuerstein R, Rivetti di Val Cervo P, Aberdam D, Knight RA, Melino G, et al. miR-203 represses 'stemness' by repressing DeltaNp63. Cell Death Differ. 2008;15:1187–1195. doi: 10.1038/cdd.2008.69. [DOI] [PubMed] [Google Scholar]
- 41.Altieri DC. Survivin, cancer networks and pathway-directed drug discovery. Nat Rev Cancer. 2008;8:61–70. doi: 10.1038/nrc2293. [DOI] [PubMed] [Google Scholar]
- 42.Cheng JQ, Lindsley CW, Cheng GZ, Yang H, Nicosia SV. The Akt/PKB pathway: molecular target for cancer drug discovery. Oncogene. 2005;24:7482–7492. doi: 10.1038/sj.onc.1209088. [DOI] [PubMed] [Google Scholar]
- 43.Kim D, Dan HC, Park S, Yang L, Liu Q, Kaneko S, et al. AKT/PKB signaling mechanisms in cancer and chemoresistance. Front Biosci. 2005;10:975–987. doi: 10.2741/1592. [DOI] [PubMed] [Google Scholar]
- 44.Frame MC. Src in cancer: deregulation and consequences for cell behaviour. Biochim Biophys Acta. 2002;1602:114–130. doi: 10.1016/s0304-419x(02)00040-9. [DOI] [PubMed] [Google Scholar]
- 45.Summy JM, Gallick GE. Src family kinases in tumor progression and metastasis. Cancer Metastasis Rev. 2003;22:337–358. doi: 10.1023/a:1023772912750. [DOI] [PubMed] [Google Scholar]
- 46.Fanning P, Bulovas K, Saini KS, Libertino JA, Joyce AD, Summerhayes IC. Elevated expression of pp60c-src in low grade human bladder carcinoma. Cancer Res. 1992;52:1457–1462. [PubMed] [Google Scholar]
- 47.Boyer B, Bourgeois Y, Poupon MF. Src kinase contributes to the metastatic spread of carcinoma cells. Oncogene. 2002;21:2347–2356. doi: 10.1038/sj.onc.1205298. [DOI] [PubMed] [Google Scholar]
- 48.Said N, Theodorescu D. Pathways of metastasis suppression in bladder cancer. Cancer Metastasis Rev. 2009;28:327–333. doi: 10.1007/s10555-009-9197-4. [DOI] [PubMed] [Google Scholar]
- 49.Green TP, Fennell M, Whittaker R, Curwen J, Jacobs V, Allen J, et al. Preclinical anticancer activity of the potent, oral Src inhibitor AZD0530. Mol Oncol. 2009;3:248–261. doi: 10.1016/j.molonc.2009.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


