Abstract
Background
Primary human hepatocytes are a useful in vitro model system to examine hepatic biochemical pathways, liver disorders and/or pharmacotherapies. This system can also be used for transport studies to investigate uptake and excretion of bile acids. Proper modeling of hepatic function requires careful attention to media components, and culture substrates and conditions.
Objectives
To examine the effects of different culture media and conditions on bile acid transport in cultured human hepatocytes.
Methods and Results
Hepatocytes cultured in Williams' medium E showed an increase in both uptake and excretion of taurocholate compared to cells cultured in Dulbecco's Modified Eagle Medium (DMEM). Supplementation of DMEM with glutathione or ascorbic acid did not compensate for the lower transport. The difference can be explained by lower mRNA expression of the transporter proteins sodium taurocholate cotransporting polypeptide (NTCP) and bile salt export pump (BSEP; ABCB11) when cultured in DMEM. Hepatocytes cultured in DMEM also display fewer and smaller bile canaliculi. Following extended time in culture supplementation of Williams' medium E with dexamethasone increased the expression of NTCP and BSEP.
Conclusion
Williams' medium E is superior to DMEM for transport studies in primary human hepatocytes. Supplementation with dexamethasone increase mRNA levels of NTCP and BSEP.
Keywords: primary human hepatocytes, bile acid transport, NTCP, BSEP, dexamethasone
Abbreviations: DMEM, Dulbecco's Modified Eagle Medium; NTCP, sodium taurocholate cotransporting polypeptide; BSEP, bile salt export pump; OATP, organic anion-transporting polypeptide; MRP2, multidrug resistance protein 2; WE, Williams' medium E; HBSS, Hank's Balanced Salt Solution; CgamF, cholylglycylamido-fluorescein; GSH, glutathione; AA, ascorbic acid
The study of in vitro transport of bile acids in hepatocytes provides a useful method for analyzing drug disposition, biliary clearance and different conditions causing cholestasis.
Under normal conditions bile acids are taken up by hepatocytes from portal circulation by high affinity sodium-dependent, sodium taurocholate cotransporting polypeptide (NTCP) and to a lesser extent by the sodium-independent organic anion-transporting polypeptides (OATPs) in the sinusoidal membrane.1,2 Within the hepatocyte, returning bile acids are mixed with newly synthesized bile acids and are secreted into bile. Conjugated bile acids are secreted from the hepatocyte canalicular membrane into bile by the bile salt export pump (BSEP; ABCB11) which is an adenosine triphosphate-binding cassette transporter. A small amount is also secreted by multidrug resistance protein 2 (MRP2).1,2
In cholestasis, bile flow from the liver to the intestine is impaired and as a consequence toxic bile acids and other metabolites are retained within the hepatocyte. In children, cholestasis with early onset accounts for a large proportion of the cases with severe liver disease with high mortality and morbidity. Cholestasis in infancy has many different causes and the etiology is unknown in 20–25% of affected patients.3 The disease can be caused by mutations in transporter proteins. For example, mutations in the ABCB11 gene, encoding for BSEP, are associated with progressive familial intrahepatic cholestasis type 2.4 Acquired cholestasis may be due to infections, drug-induced hepatocellular injury or total parenteral nutrition.5,6 To study the possible subtle difference between normal and diseased livers one needs to first investigate how bile acids are transported in liver cells from normal tissue. Thus, in order to use hepatocytes as a model system for cholestasis culture conditions that allow for bile acid transport in cultured cells have to be attained. Previous reports have investigated effects of different conditions for bile acid transport in rat and mouse hepatocytes7–9 but the knowledge on how different culture conditions affect bile acid transport in human hepatocytes is limited.
Two commonly used culture media for hepatocytes are Dulbecco's Modified Eagle Medium (DMEM) and Williams' medium E (WE), in this report we examined how bile acid transport is affected by culturing hepatocytes in these media. While there are many small differences between the components of DMEM and WE, two substantial differences are the presence of ascorbic acid and glutathione in WE. We therefore supplemented DMEM with these compounds to investigate if bile acid transport was affected. Dexamethasone is a synthetic glucocorticoid frequently included as a supportive or protective factor in hepatocyte culture media and even in flushing and transport solutions used to cold-preserve livers for transplantation. Since dexamethasone is such a common additive to culture media, its effects on transport activity in cultured hepatocytes were investigated.
The aim of the present study was to investigate the influence of different culture media, media supplements and time in culture for activity and expression of bile acid uptake and efflux transporters NTCP and BSEP in cultured healthy human hepatocytes.
Materials and methods
Isolation of Primary Human Hepatocytes
Normal human liver tissue was obtained from patients (n = 11, Table 1) undergoing surgical liver resection due to cancer or from donor livers that could not be used for transplantation. Approval to use parts of resected human liver specimens for research was given by the Ethics Committee at Karolinska Institutet and from the Institutional Review Board at University of Pittsburgh.
Table 1.
Demographics.
| Liver number | Age | Gender | Diagnosis |
|---|---|---|---|
| HH1261 | 18 | M | Neuroendocrine tumor |
| HH1270 | 77 | F | Deceased donor |
| HH1274 | 55 | F | Deceased donor |
| HH1281 | 16 | M | Deceased donor |
| HH1571 | 62 | F | Liver resection due to metastatic colon carcinoma |
| HH1591 | 12 | F | Deceased donor |
| HF79 | 39 | M | Liver resection due to suspected tumor in Chron's disease |
| HF82 | 49 | F | Liver resection due to metastatic pancreatic carcinoma |
| HF83 | 69 | F | Deceased donor |
| HF133 | 76 | M | Colon carcinoma |
| HF134 | 50 | M | Deceased donor |
| VF1 | 50 | M | Deceased donor |
F, female; M, male.
Hepatocytes were isolated by a three-step perfusion technique, utilizing EGTA and collagenase, as previously described by Strom et al10 and updated by Gramignoli et al11 1.5 million cells were plated onto 6 well culture dishes precoated with collagen I (3.3 mg/ml), after 24 h cells were over laid with matrigel (0.233 mg/ml) to further polarize the cells. Hepatocytes cultured in this sandwich configuration develop functional canalicular spaces between cells which allow transport studies. In all experiments, hepatocytes were cultured in cell culture media supplemented with amphoteracin, gentamicin and insulin (120 nM). In the first experiments cells were cultured in WE, WE and DMEM in equal parts, DMEM or in DMEM supplemented with glutathione (0.16 μM) or ascorbic acid (11 μM). In all subsequent experiments cells were cultured in WE without or with the addition of dexamethasone. The medium was changed 1 h after plating and then daily until harvesting. Following 17, 72 or 96 h in culture bile acid transport was measured and cells were harvested in Trizol for quantification of mRNA.
Bile Salt Transport Assay
Bile salt transport was measured by using 3H-Taurocholate as described previously12 with some minor changes, see Figure 1 for a cartoon illustrating the method. First, cell culture media was collected and cells washed with Hank's Balanced Salt Solution (HBSS) + (HBSS supplemented with CaCl2 (anhyd.), MgCl2 × 6 H2O, MgSO4 × 7 H2O) and incubated at 37 °C. The buffer was aspirated and cells loaded by adding HBSS+ containing non-labeled taurocholate and 3H-Taurocholate and incubated at 37 °C for 20 min. The cells were washed with ice cold HBSS+. Half of the wells received HBSS+ and the other half HBSS− (HBSS supplemented with EGTA) and the cells were then incubated for another 20 min at 37 °C. Aliquots of media were obtained from each well and counts per minute analyzed with a beta counter. The remaining fluid was quickly aspirated and the cells were lysed in buffer containing NaOH/SDS. Lysates were used for counting radioactivity and protein determination.13
Figure 1.
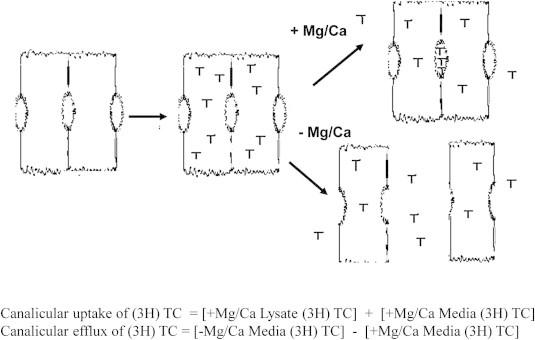
Bile salt transport assay.
Specific activity for uptake was defined by the sum of both effluxed and retained radioactivity in HBSS+ samples. Since desmosomes between the cells break in absence of Ca/Mg allowing the radioactivity to efflux, specific activity for canalicular transport was defined by the difference in efflux of 3H-Taurocholate in the absence and presence of Ca/Mg. To normalize different efflux capacities between different livers, efflux from each treatment was divided by total efflux for that liver.
Visualization of Bile Canaliculi
Bile canaliculi was visualized by treating cells cultured in DMEM or WE with cholylglycylamido-fluorescein (CGamF), a generous gift from Dr. Alan Hofmann, San Diego. CGamF is a fluorescein-labeled bile acid analog transported by NTCP and BSEP. Cells were washed as described for the transport assay and incubated with (CGamF) (2 μM) for 20 min, washed and photographed in the microscope. Cells were cultured without or with the addition of cyclosporine A (10 μM), an inhibitor of both NTCP and BSEP, which was used to identify transport-mediated efflux. Photographs were taken using a Nikon Eclipse TE2000-U, Roper Scientific camera (Photometrics Coolsnap ES), Metamorph software, Fryer.
Real-time polymerase chain reaction (PCR)
RNA was isolated using Trizol reagent (Invitrogen, Stockholm, Sweden), cDNA synthesis was performed using MultiScribe Reverse Transcriptase (Applied Biosystems, Stockholm, Sweden). The mRNA expression was quantified with Quantitative real-time PCR using Taqman probes from ABI, analysis was performed on an ABI Prism 7000 instrument (Applied Biosystems, Stockholm, Sweden). Cyclophilin was used as an endogenous control. To normalize different expression levels between different livers, mRNA expression from each treatment was divided by total expression level for that liver.
Statistics
Data are presented as means ± SEM. The significance of differences between treatments was tested by ANOVA followed by Tukey HSD (Figure 1) and by T-test (Figure 3) (IBM SPSS Statistics).
Figure 3.
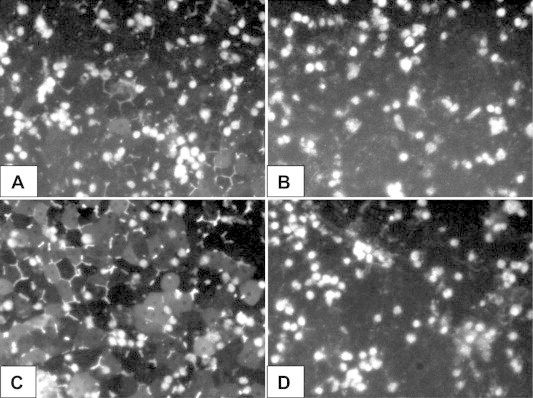
Visualization of bile canaliculi using cholylglycylamido-fluorescein (CGamF), n = 1 liver (HH1261). (A) Cells cultured in DMEM for 96 h. (B) Cells cultured in DMEM for 96 h and treated with cyclosporine. (C) Cells cultured in WE for 96 h. (D) Cells cultured in WE for 96 h and treated with cyclosporine.
Results
Cell Culture Media
In preliminary studies we noticed that the uptake and transport of bile acids appeared to be more efficient when hepatocytes were cultured in WE media compared to DMEM. As shown in Figure 2A taurocholate uptake was 3-fold higher in cells cultured in WE compared to cells cultured in DMEM (p ≤ 0.01). Supplementing DMEM with glutathione or ascorbic acid did not compensate for the lost uptake. Mixing WE and DMEM resulted in an intermediate uptake, Figure 2A. Efflux of taurocholate showed a pronounced difference between the different culture media. Bile acid efflux was 5-fold higher in cells cultured in WE compared to cells cultured in DMEM (p ≤ 0.001). The addition of glutathione or ascorbic acid to DMEM did not cause a significant increased efflux, Figure 2B.
Figure 2.
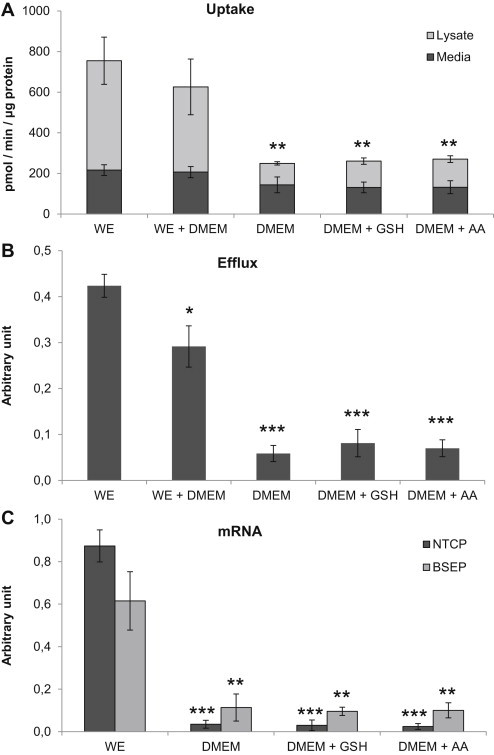
Bile acid transport in primary hepatocytes cultured for 96 h in Williams' medium E (WE), WE + DMEM, DMEM or DMEM supplemented with 0.16 μM glutathione (GSH) or 11 μM ascorbic acid (AA). (A) Uptake of taurocholate. (B) Efflux of taurocholate. (C) mRNA expression of NTCP and BSEP. Data represents means ± SEM, **p ≤ 0.01, ***p ≤ 0.01, n = 3 livers (HH1270, HH1274, HH1281).
mRNA expression analysis of NTCP and BSEP in the cells revealed that NTCP expression was 25 times higher in cells cultured in WE compared to cells cultured in DMEM (p ≤ 0.001), supplementing the DMEM did not compensate for the reduced levels, Figure 2C. BSEP mRNA was also higher in cells cultured in WE compared to cells cultured in DMEM, the difference was approximately 5 times (p ≤ 0.01).
To visualize the effect of culture media on bile canaliculi formation and function of the hepatocytes, cells were treated with CGamF. In cell cultures maintained in DMEM some bile canaliculi were detected, however, these were few and small, Figure 3A. When cells were cultured in WE numerous large canaliculi were seen, Figure 3C.
Dexamethasone and Time in Culture
We first determined how NTCP and BSEP mRNA expression was affected by the addition of dexamethasone (100 nM). Following 17 or 72 h in culture there was no difference in BSEP or NTCP mRNA expression in cells cultured without and with dexamethasone, Figure 4A and B. However, after 96 h in culture expression of both BSEP (p ≤ 0.05) and NTCP (p ≤ 0.01), was higher in cells supplemented with dexamethasone, Figure 4C.
Figure 4.
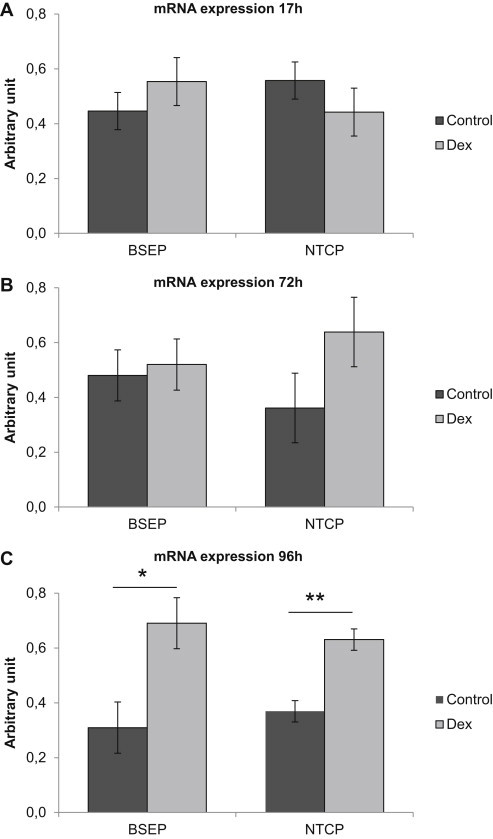
Effect of dexamethasone (100 nM) on mRNA expression of NTCP and BSEP. (A) mRNA expression in cells following 17 h in culture, n = 3 livers (HF133, HF134, VF1). (B) mRNA expression in cells following 72 h in culture, n = 3 livers (HF133, HF134, VF1). (C) mRNA expression in cells following 96 h in culture, n = 5 livers (HH1571, HH1591, HF79, HF82, HF83). Data represents means ± SEM, *p ≤ 0.05, **p ≤ 0.01.
We thereafter examined how bile acid transport is influenced by time in culture, without and with the addition of 100 nM dexamethasone. Uptake of taurocholate was not changed between 17 h and 72 h in culture, dexamethasone did not affect uptake, Figure 5A. There was a trend toward increased efflux of taurocholate following 72 h in culture compared to 17 h and after 72 h dexamethasone appeared to stimulate the efflux, Figure 5B.
Figure 5.
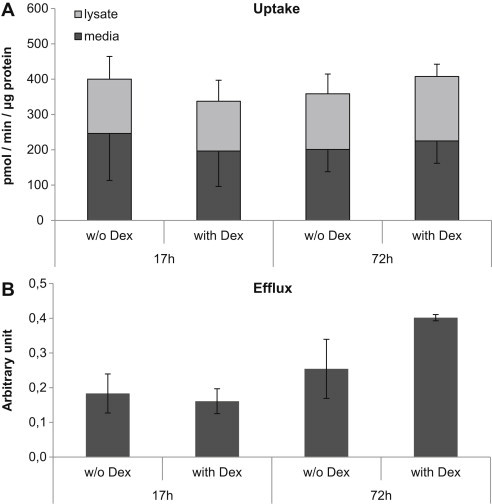
Effect of time in culture and dexamethasone (100 nM) on bile acid transport. (A) Uptake of taurocholate. (B) Efflux of taurocholate. Data represents means ± SEM, n = 3 livers (HF133, HF134, VF1).
Discussion
The results presented here show that the culture conditions can dramatically influence transport of bile acids. Taurocholate uptake and efflux are substantially decreased when human hepatocytes are cultured in DMEM compared to WE. Imaging of the transport using fluorescent bile acids revealed that cells cultured in DMEM also display fewer and smaller bile canaliculi. Supplementing DMEM with glutathione or ascorbic acid up to the same level as WE did not alter the transport. These results are to some extent in line with results from a previous study by Chandra et al using rat hepatocytes where formation of bile canaliculi was more extensive when cells were cultured in WE compared to DMEM. However, in that report no difference in taurocholate excretion was detected comparing DMEM and WE.9 Another study using rat hepatocytes also failed to detect any difference in excretion between cells cultured in DMEM and WE although they reported higher levels of BSEP protein in cells cultured in DMEM compared to WE.8 In our study mRNA expression of both NTCP and BSEP was much higher in cells cultured in WE compared to DMEM. These data indicates that cultured human hepatocytes behave differently than rat hepatocytes and that species differences exist and need to be considered in the design of transport experiments.
Dexamethasone is an inducer of several CYP450 enzymes including CYP3A. The addition of dexamethasone to incubation media is reported to improve attachment, survival, morphology and CYP expression of primary hepatocytes.14,15 Published reports with experiments with rat hepatocytes have not provided clarity with respect to the effects of dexamethasone on bile acid transport. This confusion could possibly be explained by the different culture conditions used by different groups. Warskulat et al show that the maintenance of Bsep expression in rat hepatocytes is dependent on the presence of dexamethasone (100 nM) in the medium.16 However, these results are in contrast to a study by Turncliff et al where dexamethasone did not alter Bsep expression but decreased the expression of Ntcp.17 Both these studies were performed using rat hepatocytes but one major difference is that Warskulat et al use a lower dexamethasone concentration (100 nM) compared to Turncliff et al who use 100 nM in their basal media (control cells) which they compare to cells treated with 100 μM dexamethasone. Luttringer et al investigated the effect of dexamethasone on hepatocyte transporters.18 According to their study, Ntcp mRNA expression is not affected by 100 nM nor by 1000 nM dexamethasone, however, the basal media for all cells contained hydrocortisone. Our results clearly demonstrate that concentrations that as low as 100 nM dexamethasone are sufficient to induce BSEP and NTCP expression as early as day 4 of culture in human hepatocytes. Thus, the lack of effect in the above-cited studies could be explained by the presence of dexamethasone and/or hydrocortisone in the basal media or temporal differences. Another possibility is that there is a difference in the regulation of these pathways between human and rat hepatocytes.
Hepatocytes may lose or gain function and gene expression upon isolation and culture. Therefore we investigated when the hepatocytes should be harvested for optimal bile acid transport. Several functions and enzymes are decreased or impaired upon hepatocyte isolation and prolonged incubation may allow hepatocytes time to regenerate canaliculi and to polarize.19,20 A study by Schuetz et al showed that there is a general increase in gene expression during the first 2 days of culture with regard to actin and tubulin.21 Following isolation, cytoskeletal proteins in hepatocytes need time to resynthesize and move to the appropriate location within the cell to restore functional activity for the excretion of bile acids. As we previously reported, bile acid synthesis is increased overtime in culture.22 There are however reports of decreased BSEP protein expression overtime in culture.23 Therefore we investigated if bile acid transport was lower after 72 h compared to 17 h after isolation. We could not detect any decrease in efflux comparing 17 h and 72 h in culture but instead there appears to be a trend toward increased efflux of bile acids with time in culture when cells are supplemented with dexamethasone.
To our knowledge this is the first report comparing bile acid transport in these different culture conditions in human hepatocytes. In conclusion, WE is superior to DMEM for transport studies in primary human hepatocytes and supplementation with dexamethasone increase mRNA levels of NTCP and BSEP.
Conflicts of interest
All authors have none to declare.
Acknowledgments
This work was supported by grants from Stiftelsen Samariten, H.K.H. Kronprinsessan Lovisas Förening för barnasjukvård/Stiftelsen Axel Tielmans minnesfond.
References
- 1.Wolkoff A.W., Cohen D.E. Bile acid regulation of hepatic physiology: I. Hepatocyte transport of bile acids. Am J Physiol Gastrointest Liver Physiol. 2003;284:G175–G179. doi: 10.1152/ajpgi.00409.2002. [DOI] [PubMed] [Google Scholar]
- 2.Kosters A., Karpen S.J. Bile acid transporters in health and disease. Xenobiotica. 2008;38:1043–1071. doi: 10.1080/00498250802040584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Fischler B., Papadogiannakis N., Nemeth A. Aetiological factors in neonatal cholestasis. Acta Paediatr. 2001;90:88–92. doi: 10.1080/080352501750064932. [DOI] [PubMed] [Google Scholar]
- 4.Strautnieks S.S., Bull L.N., Knisely A.S. A gene encoding a liver-specific ABC transporter is mutated in progressive familial intrahepatic cholestasis. Nat Genet. 1998;20:233–238. doi: 10.1038/3034. [DOI] [PubMed] [Google Scholar]
- 5.Trauner M., Fickert P., Stauber R.E. Inflammation-induced cholestasis. J Gastroenterol Hepatol. 1999;14:946–959. doi: 10.1046/j.1440-1746.1999.01982.x. [DOI] [PubMed] [Google Scholar]
- 6.Kelly D.A. Preventing parenteral nutrition liver disease. Early Hum Dev. 2010;86:683–687. doi: 10.1016/j.earlhumdev.2010.08.012. [DOI] [PubMed] [Google Scholar]
- 7.Swift B., Brouwer K.L. Influence of seeding density and extracellular matrix on bile acid transport and mrp4 expression in sandwich-cultured mouse hepatocytes. Mol Pharm. 2010;7:491–500. doi: 10.1021/mp900227a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Turncliff R.Z., Tian X., Brouwer K.L. Effect of culture conditions on the expression and function of Bsep, Mrp2, and Mdr1a/b in sandwich-cultured rat hepatocytes. Biochem Pharmacol. 2006;71:1520–1529. doi: 10.1016/j.bcp.2006.02.004. [DOI] [PubMed] [Google Scholar]
- 9.Chandra P., Lecluyse E.L., Brouwer K.L. Optimization of culture conditions for determining hepatobiliary disposition of taurocholate in sandwich-cultured rat hepatocytes. In Vitro Cell Dev Biol Anim. 2001;37:380–385. doi: 10.1007/BF02577575. [DOI] [PubMed] [Google Scholar]
- 10.Strom S.C., Pisarov L.A., Dorko K. Use of human hepatocytes to study P450 gene induction. Methods Enzymol. 1996;272:388–401. doi: 10.1016/s0076-6879(96)72044-x. [DOI] [PubMed] [Google Scholar]
- 11.Gramignoli R., Green M.L., Tahan V. Development and application of purified tissue dissociation enzyme mixtures for human hepatocyte isolation. Cell Transplant. 2011 doi: 10.3727/096368911X600939. Epub ahead of print. [DOI] [PubMed] [Google Scholar]
- 12.Kostrubsky V.E., Strom S.C., Hanson J. Evaluation of hepatotoxic potential of drugs by inhibition of bile-acid transport in cultured primary human hepatocytes and intact rats. Toxicol Sci. 2003;76:220–228. doi: 10.1093/toxsci/kfg217. [DOI] [PubMed] [Google Scholar]
- 13.Bradford M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- 14.Laishes B.A., Williams G.M. Conditions affecting primary cell cultures of functional adult rat hepatocytes. II. Dexamethasone enhanced longevity and maintenance of morphology. In Vitro. 1976;12:821–832. doi: 10.1007/BF02796367. [DOI] [PubMed] [Google Scholar]
- 15.Sidhu J.S., Omiecinski C.J. Modulation of xenobiotic-inducible cytochrome P450 gene expression by dexamethasone in primary rat hepatocytes. Pharmacogenetics. 1995;5:24–36. doi: 10.1097/00008571-199502000-00003. [DOI] [PubMed] [Google Scholar]
- 16.Warskulat U., Kubitz R., Wettstein M. Regulation of bile salt export pump mRNA levels by dexamethasone and osmolarity in cultured rat hepatocytes [In Process Citation] Biol Chem. 1999;380:1273–1279. doi: 10.1515/BC.1999.162. [DOI] [PubMed] [Google Scholar]
- 17.Turncliff R.Z., Meier P.J., Brouwer K.L. Effect of dexamethasone treatment on the expression and function of transport proteins in sandwich-cultured rat hepatocytes. Drug Metab Dispos. 2004;32:834–839. doi: 10.1124/dmd.32.8.834. [DOI] [PubMed] [Google Scholar]
- 18.Luttringer O., Theil F.P., Lave T. Influence of isolation procedure, extracellular matrix and dexamethasone on the regulation of membrane transporters gene expression in rat hepatocytes. Biochem Pharmacol. 2002;64:1637–1650. doi: 10.1016/s0006-2952(02)01382-5. [DOI] [PubMed] [Google Scholar]
- 19.LeCluyse E.L., Audus K.L., Hochman J.H. Formation of extensive canalicular networks by rat hepatocytes cultured in collagen-sandwich configuration. Am J Physiol. 1994;266:C1764–C1774. doi: 10.1152/ajpcell.1994.266.6.C1764. [DOI] [PubMed] [Google Scholar]
- 20.Tuschl G., Mueller S.O. Effects of cell culture conditions on primary rat hepatocytes-cell morphology and differential gene expression. Toxicology. 2006;218:205–215. doi: 10.1016/j.tox.2005.10.017. [DOI] [PubMed] [Google Scholar]
- 21.Schuetz E.G., Li D., Omiecinski C.J. Regulation of gene expression in adult rat hepatocytes cultured on a basement membrane matrix. J Cell Physiol. 1988;134:309–323. doi: 10.1002/jcp.1041340302. [DOI] [PubMed] [Google Scholar]
- 22.Ellis E., Goodwin B., Abrahamsson A. Bile acid synthesis in primary cultures of rat and human hepatocytes. Hepatology. 1998;27:615–620. doi: 10.1002/hep.510270241. [DOI] [PubMed] [Google Scholar]
- 23.Li N., Bi Y.A., Duignan D.B., Lai Y. Quantitative expression profile of hepatobiliary transporters in sandwich cultured rat and human hepatocytes. Mol Pharm. 2009;6:1180–1189. doi: 10.1021/mp900044x. [DOI] [PubMed] [Google Scholar]


