Abstract
Background
The gene for New Delhi metallo-β-lactamase 1 (NDM-1) has been reported to be transmitted via plasmids which are easily transferable and capable of wide distribution. We report the isolation of two NDM-1 producing strains and possible in vivo transfer of bla NDM-1 in a patient.
Methods
Clinical samples were collected for bacterial culture and antibiotic susceptibility testing from a patient during a 34-day hospitalization. The presence of bla NDM-1 was detected by PCR and sequencing. Plasmids of interest were sequenced. Medical records were reviewed for evidence of association between the administration of antibiotics and the acquisition of the NDM-1 resistance.
Results
A NDM-1 positive Raoultella planticola was isolated from blood on the ninth day of hospitalization without administration of any carbapenem antibiotics and a NDM-1 positive Escherichia coli was isolated from feces on the 29th day of hospitalization and eight days after imipenem administration. The bla NDM-1 was carried by a 280 kb plasmid pRpNDM1-1 in R. planticola and a 58 kb plasmid pEcNDM1-4 in E. coli. The two plasmids shared a 4812 bp NDM-1-ISCR1 element which was found to be excisable from the plasmid as a free form and transferrable in vitro to a NDM-1 negative plasmid from E. coli.
Conclusion
bla NDM-1 was embedded in an ISCR1 complex class 1 integron as a novel 4812 bp NDM-1-ISCR1 element. The element was found to be able to self excise to become a free form, which may provide a new vehicle for NDM-1 dissemination. This mechanism could greatly accelerate the spread of NDM-1 mediated broad spectrum β-lactam resistance.
Introduction
Carbapenems often represent last-resource drugs for Gram negative bacterial infections. The most common mechanism of carbapenem resistance in Gram negative bacteria is the production of carbapenemases, including metallo-β-lactamases (MBLs). The New Delhi metallo-beta-lactamase (NDM-1), a recently identified novel type of MBL can hydrolyze virtually all β-lactams, adding further burden to an already high level of antibiotic resistance. The emergence and global spread of NDM-1 mediated broad spectrum β-lactam resistance has become a major concern worldwide [1], [2].
NDM-1 was first identified in a carbapenem-resistant Klebsiella pneumoniae strain isolated from urine and an Escherichia coli strain isolated from feces of the same patient that was resistant to almost all antibiotics tested except colistin and tigecycline in 2009 in Sweden [3]. NDM-1 positive K. pneuomoniae and E. coli were soon detected in many other countries, including Australia, Canada, Germany, China, France, Japan, the Nordic countries, United States and many others [4]–[8]. In addition, reported NDM-1 positive bacterial species extended to Acinetobacter spp, Citrobacter freundii, Enterobacter cloacae, Morganella morganii, Providencia spp, and Pseudomonas aeruginosa [9]–[11]. The world has been alarmed by the wide dissemination of NDM-1 isolates, for which new and effective antibiotics are currently unavailable. The gene bla NDM-1 encoding NDM-1 has been mostly reported to be on plasmids, which could rapidly disseminate and spread between different bacterial species by cell-to-cell transfer of the plasmids. However the plasmids carrying bla NDM-1 are different in sizes, ranging from 40 kb to 400 kb [9], suggesting that direct transfer by plasmid is only one means of NDM-1 dissemination. Multiple mobile genetic elements are also associated with bla NDM-1 dissemination [4], [11], [12].
The insertion sequence common region 1 (ISCR1) is a well recognized system of gene capture. Its role in antibiotic resistance dissemination was first noted by the evidence that a wide variety of antimicrobial determinants are found in the immediate vicinity of ISCR1 [13], [14]. ISCR1 is capable of mobilizing its upstream sequences by a process of rolling circle transposition [15]. Association of the ISCR1 element with bla NDM-1 has been observed in plasmids pNDM-HK(HQ451074) [16], pMR0211(JN687470) [17] and in K. pneumoniae strain 05-506 [3] and P. aeruginosa [18]. However the role of the ISCR1 element in the transfer of the NDM-1 within and between bacterial species is unclear.
Here, we report that we identified two strains of different species (Raoultella planticola and E. coli) carrying NDM-1 isolated 20 days apart from the same patient and showed that bla NDM-1 was located on two unrelated plasmids. The bla NDM-1 was embedded in an ISCR1 complex class 1 integron, which is most likely responsible for bla NDM-1 transfer between R. planticola and E. coli in the patient in vivo.
Materials and Methods
Ethics statement
The feces, urine, sputum and blood samples were obtained with the written informed consent from the patient. This study was reviewed and approved by the ethics committee of the National Institute for Communicable Disease Control and Prevention, China CDC, according to the medical research regulations of the Ministry of Health, China. This research was conducted within China.
The Patient
The patient was a 51-year-old farmer from a small village of Gansu Province, who has never visited any cities in China or other countries that have reported NDM-1. On October 1, 2010, the patient was admitted to Lanzhou medical school hospital because of severe multiple left rib fractures as a result of traffic accident. Surgical operation was performed three hours after the accident. A second operation was performed five days later on the 6th of October. The patient was hospitalized for 34 days and discharged on November 6, 2010. The blood, urine, sputum and feces were sampled for isolation of bacteria and antibiotic susceptibility testing as standard patient care except that some samples also were collected for the investigation of NDM-1.
Bacterial isolation, antibiotics resistance testing and molecular typing
Species identification was performed using the Microscan WalkAway 40 S1 identification system (Dade Behring, Deerfield, IL, USA). The 16s rDNA and rpoB gene sequences were also analyzed to confirm the species identity. Susceptibility of the isolates to antibiotic was determined by the E-test methodology (BioMerieux SA, La Balme-les-Grottes, France). Modified Hodge test was performed to screen for carbapenemases. The results were interpreted according to the 2011 Clinical and Laboratory Standards Institute recommendation [19]. The relationship of NDM-1 positive and negative E. coli strains isolated from the same fecal samples was analyzed by PFGE (Pulsed Field Gel Electrophoresis)-Xba I digestion method, following the protocol previously described [20].
PCR primers and detection of bla NDM-1
PCR primers for detecting bla NDM-1 and for confirming the genetic environment of the bla NDM-1 are listed in Table S1 in File S1. Plasmids were analyzed and sized by the PFGE-S1 nuclease method [21]. Separation of large fragments from restriction enzyme digestion was done with PFGE by using of a CHEF-DR III apparatus (Bio-Rad, Hercules, CA) for 7 h at 6 V/cm and 14°C with initial and final pulse times of 5 s and 20 s, respectively. Southern hybridization was performed with a 560 bp segment of the bla NDM-1 gene as a probe using the ECL direct nucleic acid labeling and detection system (GE Healthcare, Buckinghamshire, UK).
Conjugation and transformation of bla NDM-1 plasmids
Conjugation experiments were carried out by the solid surface methods with E. coli J53 Azr as the recipient [4]. Transconjugants were selected on Luria-Bertani agar plates containing sodium azide (100 mg/L) and ampicillin (100 mg/L), confirmed by PCR amplification of the bla NDM-1 for NDM-1 positive donors and bla OXA-30 gene for NDM-1 negative donors and sequencing of the PCR products. The plasmid DNA of NDM-1 positive strain E. coli EcNDM1was used for transformation and E. coli JM109 was used as the recipient.
Whole genome sequencing and sequencing of plasmids
The whole genome of the isolate R. planticola was sequenced using a combined strategy of Roche 454 Genome Sequencer FLX System (Roche/454 Life Sciences, Branford, CT, USA) and Illumina sequencing technology (Illumina, San Diego, CA, USA). The Burrows-Wheeler Alignment (BWA) tool was used to map all the Illumina reads to the scaffolds generated by 454 Newbler. The inter- and intra-scaffold gaps were filled by local assembly of Roche and Illumina reads. The gaps between contigs were closed by targeted PCR amplification and sequencing of the PCR products with BigDye terminator chemistry on an ABI 3730 capillary sequencer.
Plasmid DNA extracted from E. coli EcNDM1 was transformed into E. coli JM109, and a carbapenems resistant transformant carrying plasmid pEcNDM1-4 named JM109 (pEcNDM1-4) was recovered. Plasmid pEcNDM1-4 extracted from the E. coli JM109 (pEcNDM1-4) was used to construct a Tn5 transposon library according to the manufacturer's recommendations (Epicenter, Madison, WI) [22]. Each transposon clone was sequenced using the BigDye terminator chemistry on an ABI 3730 capillary sequencer, first with primer FP-V and primer RP-V to obtain unique sequences flanking the transposon and then by PCR walking sequencing.
The pEcNDM1-4 equivalent plasmid in NDM-1 negative strain E. coli EcNDMneg, named pEcNDMneg-4, was sequenced by targeted sequencing. Twenty pairs of overlapping primers were designed according to the sequences of plasmid pEcNDM1-4 (Table S1 in File S1) and used for the amplification of pEcNDMneg-4 as overlapping PCR products which were sequenced.
Nucleotide sequences accession numbers
Sequences of the NDM-1 carrying plasmids were submitted to the GenBank database with accession numbers JX515588, JX469383 and JX515587 for pRpNDM1-1, pEcNDM1-4 and pEcNDMneg-4 respectively.
Results
Isolation of NDM-1 positive R. planticola and E. coli
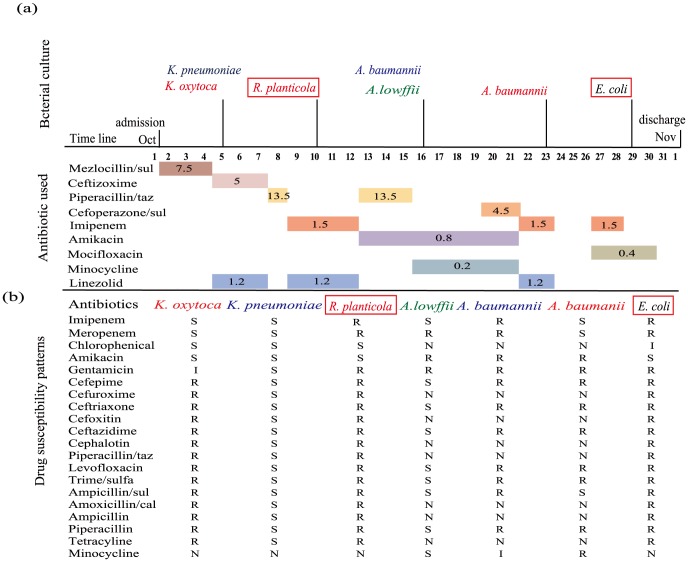
Bacterial culture was attempted on four occasions to guide clinical care of the patient during hospitalization. From the blood culture of October 9th, 2010, a NDM-1 positive R. planticola strain, RpNDM1, was isolated. On October 29th, 2010, a NDM-1 positive E. coli strain, EcNDM1, was isolated from a fecal sample of the same patient. Additionally from the same fecal sample a multidrug resistant NDM-1 negative E. coli strain, EcNDMneg, was also isolated. We did not detect any NDM-1 positive bacteria from any other patients in the same ward. Medical records of the patient were reviewed for any evidence of association between administration of antibiotics and dissemination of bla NDM-1. Before the isolation of the NDM-1 positive R. planticola, only mezlocillin/sulbactum, ceftizoxime and linezolid were used and no carbapenems were used. However the NDM-1 positive E. coli was isolated after administration of imipenem, piperacillin/tazobactum, amikacin, minocycline and linezolid (Figure 1).
Figure 1. Time line of antibiotics treatment, microbiological investigation and isolation of NDM-1 positive R. planticola and E. coli.
(a): Time line of events during the 34-day hospitalization of the patient. Bacterial species isolated at a particular time point are shown above the time line while antibiotics used and their durations are marked below the time line. The sources of strains are colour coded: red, from blood; blue, from sputum; green: from urine; black, from feces. Red boxed: NDM-1 positive bacteria. The number in bracket after a given antibiotic is dosage of the antibiotic used (g/day). (b): Antibiotics resistance profiles of bacteria isolated. Interpretation of results: S, susceptible; I, intermediate; R, resistant; N, not done.
Characterization of strains isolated from the patient
Both RpNDM1 and EcNDM1 were resistant to carbapenems, penicillins and cephalosporins and sensitive to amikacin, colistin sulphate and tigecycline. Additionally, EcNDM1 was also resistant to aztreonam, gentamicin and ciprofloxacin (Table 1). The two E. coli strains EcNDM1 and EcNDMneg had identical resistance patterns with the sole exception that the former was also resistant to carbapenems. PCR sequencing of the bla NDM-1 from both RpNDM1 and EcNDM1 revealed 100% identity with the published bla NDM-1 sequence [3]. PFGE-Xba I analysis of EcNDM1 and EcNDMneg showed identical PFGE patterns (Figure 2b), suggesting that the two strains had the same origin.
Table 1. Antimicrobial susceptibility patterns of R. planticola KpNDM1, E. coli EcNDMneg, E. coli EcNDM1-4 and their transformants.
| Antibiotics | Minimum inhibitory concentration (mg/L) | |||||
| R. planticola | E. coli | E. coli | E. coli | E. coli | E. coli | |
| RpNDM1 | EcNDM1 | EcNDMneg | EcNDMneg (4812) | JM109 (pEcNDM1-4) | JM109 | |
| TZP | >256 | >256 | 16 | 32 | >256 | 0.75 |
| CTX | >256 | >256 | >256 | >256 | >256 | 0.25 |
| ATM | 0.094 | 32 | 32 | 32 | 0.064 | 0.064 |
| GEN | 4 | 16 | 16 | 16 | 0.125 | 0.094 |
| AMK | 3 | 6 | 6 | 6 | 1.5 | 0.5 |
| CIP | 0.75 | >32 | >32 | >32 | 0.5 | 0.032 |
| IMP | >32 | >32 | 0.19 | 16 | 16 | 0.19 |
| MEM | >32 | >32 | 0.23 | 16 | 16 | 0.016 |
| CST | 0.5 | 0.75 | 0.5 | 0.5 | 0.125 | 0.125 |
| TGC | 0.75 | 0.38 | 0.38 | 0.38 | 0.38 | 0.25 |
| MHT* | positive | positive | negative | positive | positive | negative |
TZP: piperacillin/tazobactam; CTX: cefotaxime; ATM: aztreonam; GEN: gentamicin; AMK: amikacin; CIP: ciprofloxacin; IPM: imipenem; MEM: meropenem; CST: colistin; TGC: tigecycline.
MHT*: Modified hodge test was performed and interpreted according to 2011 CLSI recommendation.
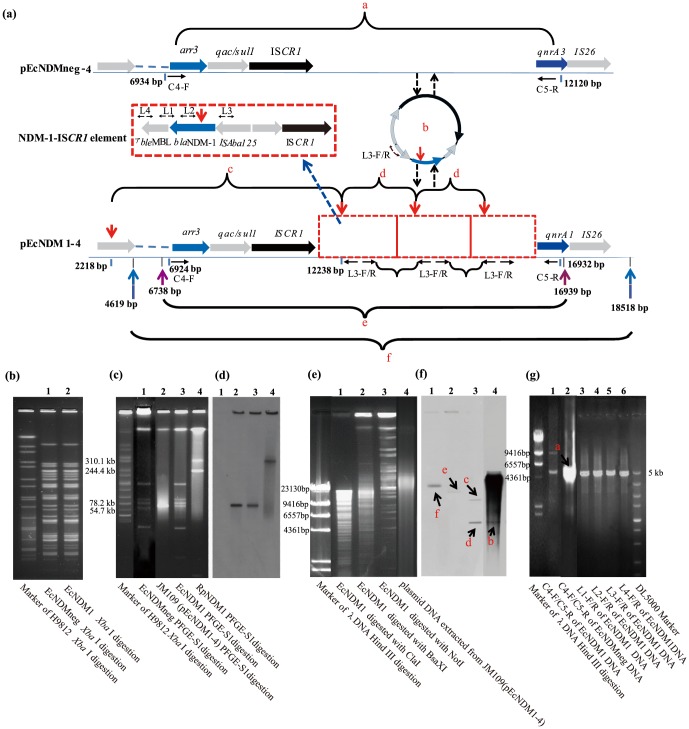
Figure 2. The characterization and mobilization model of the 4812-1-ISCR 1 element.
(a): Genetic context of plasmids pEcNDM1-4 and pEcNDMneg-4 and mobilization of the 4812 bp NDM-1-ISCR1 element. Red boxes denote 4812 bp NDM-1-ISCR1 element; Horizontal arrow denote the direction of primers; Red downward pointing arrows denote NotI restriction site; Blue upward pointing arrows denote ClaI restriction site; Purple upward pointing arrows BsaX1 restriction site; Lowercase red letters denote corresponding fragments in (f) and (g). (b): PFGE of Xba I digested patterns of E. coli strains EcNDM1 and EcNDMneg. (c): PFGE of S1 digested plasmid profiles from E. coli EcNDM1, R. planticola RpNDM1 and transformant E. coli JM109 (pEcNDM1-4). (d): Southern blot hybridization of (c) with a bla NDM-1 probe. (e): PFGE of plasmid pEcNDM1-4 with or without restriction enzyme digestion stained with ethidium bromide. (f): Southern blot hybridization of (e) with a bla NDM-1 probe. Lowercase red letters denote fragments expected as depicted in (a). (g): PCR amplification for confirmation of the structure of the NDM-1-ISCR1 element with primer pairs as shown in (a).
The bla NDM-1 location and plasmid analysis
PFGE of S1 digested plasmid DNA and Southern hybridization were performed to examine plasmid profiles and the bla NDM-1 location of E. coli EcNDM1, EcNDMneg and R. planticola RpNDM1. RpNDM1 had two large plasmids, designated as pRpNDM1-1 and pRpNDM1-2 of approximately 280 kb and 240 kb respectively and bla NDM-1 was found to be on the larger plasmid pRpNDM1-1 by Southern hybridization (Figure 2c and 2d). EcNDM1 had five plasmids ranging from 25 kb to 180 kb, which were named as pEcNDM1-1 to pEcNDM1-5 in descending order of their sizes. The bla NDM-1 was found to be on the 60 kb plasmid pEcNDM1-4 by Southern hybridization (Figure 2d). E. coli strain EcNDMneg shared an identical plasmid profile with EcNDM1. The plasmid pEcNDMneg-4 equivalent to plasmid pEcNDM1-4 in EcNDMneg was found to have no hybridization signal with the bla NDM-1 probe (Figure 2c and 2d).
Conjugation was performed with EcNDM1, EcNDMneg and RpNDM respectively as donor and E. coli J53 Azr as recipient. Both pEcNDM1-4 and pEcNDMneg-4 were found to be a conjugative plasmid with a relatively high transfer frequency at approximately 1×10−3 per donor cell. However pRpNDM1-1 failed to transfer into E. coli J53 Azr by conjugation.
Sequence analysis of plasmids pRpNDM1-1, pEcNDM1-4 and pEcNDMneg-4
Plasmid pRpNDM1-1 was sequenced by whole genome sequencing of RpNDM1 using a combination of Roche 454 sequencing with 283 867 paired-end reads generated for a 18.48 -fold coverage and Illumina sequencing with 15 713 532 75-bases paired end reads for an approximately 285.7-fold coverage. Most of the Roche and Illumina reads were assembled into 252 contigs and 23 scaffolds. Gaps were closed and genome structure confirmed by targeted PCR and Sanger sequencing. The chromosome and the two plasmids of RpNDM1 were completely assembled. The bla NDM-1 carrying plasmid pRpNDM1-1 was 277 682 bp in size and belongs to the IncH group by sequence homology analysis [23], [24]. The plasmid encodes 261 coding sequences (CDSs), of which 136 encode proteins with homology to proteins of known functions and the remaining 125 (47.9%) encode hypothetical proteins. The bla NDM-1 was found within a 39 594 bp antibiotic resistance region, starting from a Tn3-like 38 bp invert repeat (IR) upstream of merR (encoding putative transcriptional regulator) at bp 176 784 to the end of the interrupted 38 bp IR of Tn5036 at bp 216 377 and was flanked by a 5 bp direct repeat, characteristic of Tn3-family transposons. Besides bla NDM-1, six antibiotic resistance genes, aacA4 (resistance to aminoglycoside), catB8 (resistance to chloramphenicol), sul1 (resistance to sulphonamides), aadA2 (resistance to aminoglycoside), bla CTX-M-9 (resistance to lactam antibiotics), arr3 (resistance to rifampin) and a truncated dfrA27 (resistance to trimethoprim) with a 113 bp deletion in the middle of the gene were also found in this region (Figure 3a).
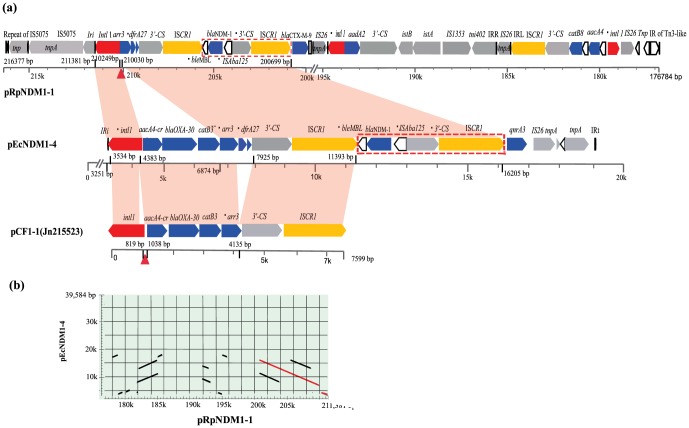
Figure 3. Schematic representation of the DNA sequences surrounding the bla NDM-1 in plasmid pEcNDM1-4, pRpNDM1-1 and homologous sequences in plasmid pCF1-1 to illustrate the relationship of the three plasmids.
(a): Illustration of the adjacent genes of NDM-1-ISCR1 of pRpNDM1-1, pEcNDM1-4 and homologous sequences in plasmid pCF1-1. Arrows represent ORFs and their direction of transcription; Filled triangles show the same 218 bp insertion in pCF1-1 and in pRpNDM1-1, compared with pEcNDM1-4; Open rectangle represent hypothetical proteins between bla CTX-M-9 and IS26 in plasmid pRpNDM1-1; Pink shades across the plasmids indicates more than 99% identity between sequences. (b): MUMer-based genomic display between pEcNDM1-4 and pRpNDM1-1. The corresponding homologous sequences represented in (a) were highlighted in red lines.
pRpNDM1-1 was found to share 86% of its sequence by length with Klebsiella oxytoca strain E718 plasmid pKOX-R1 (GenBank accession number CP003684), showing three segments of co-linearity, although genetic arrangement of the segments was slightly different (Figure S2 in File S1). In comparison to pKOX-R1, pRpNDM1-1 carries an additional 32 395 bp antibiotic resistance region, starting from downstream of IS26 at position bp 177 631 to upstream of class 1 integron 5′CS at position bp 21 0027, carrying 7 antibiotic resistance genes including bla NDM-1. Interestingly, Klebsiella oxytoca strain E718, a NDM-1 carrying strain, isolated from Taiwan harbored another plasmid, pKOX_NDM1 in addition to pKOX-R1. The bla NDM-1 was found to be located on pKOX_NDM1, which had little similarity with pRpNDM1-1 except for a 100% identical segment of 1294 bp carrying 5′-ISAba125-bla NDM-1-ble MBL-3′, which indicates that the NDM-1 acquisition by plasmid pRpNDM1-1 has a complex history (Figure S2 in File S1).
The plasmid pEcNDM1-4 was sequenced using the EZ-Tn5 transposition system. The genetic structure and the sequence of the plasmid were further confirmed by PCR and sequencing of the PCR products using 19 pairs of overlapping primers. The plasmid was 58 228 bp and was found to share a backbone of approximately 51 kb with plasmid pKp96 [25] at 99% DNA sequence identity including the complete array of genes for replication, plasmid transfer, partition and stabilization (Figure S1 in File S1). The backbone of pEcNDM1-4 was interspersed by four insertions. Insertion-1 (bp 4384 to bp 7924) was comprised of bla OXA-30 (resistance to lactam antibiotics), catB3 (resistance to chlorophenical), a truncated arr3 and a truncated dfrA27. The truncated dfrA27 is unusual as it contained a 113 bp deletion in the middle of the gene and is identical to that found in plasmid pRpNDM1-1. Insertion-2 (bp 11 394 to bp 16 257) was comprised of a truncated ble MBL(resistance to bleomycin), bla NDM-1, a truncated ISAba125, a truncated qacE1/sul1 and an intact ISCR1. Insertion-3 (bp 17 091 to bp 17 909) was comprised of a mobile element IS26. Insertion-4 (bp 35 885 to bp 36 170) was comprised of a partial fipA encoding a fertility inhibition protein (Figure S1 in File S1).
The plasmid pEcNDMneg-4 was sequenced by targeted PCR sequencing using the 19 pairs of primers that were used to confirm the genetic structure of pEcNDM1-4 (Table S1 in File S1). Except for 2 pairs of primers, 17 pairs of the primers successfully amplified products and the sequences obtained from sequencing the PCR products were identical to that of pEcNDM1-4. We found that pEcNDMneg-4 was completely co-linear and identical to pEcNDM1-4 except for the region where the 2 pairs of primers failed. The gap was closed using new primers and the plasmid pEcNDMneg-4 was 53 416 bp and was 100% identical to plasmid pEcNDM1-4 except for a 4812 bp deletion which contained a truncated ble MBL, bla NDM-1, a truncated ISAba125, the class 1 integron 3′CS (qacE1/sul1) and an ISCR1 element. This 4812 bp NDM-1-ISCR1 element is discussed in detail below.
Comparison of the two bla NDM-1 carrying plasmids, pRpNDM1-1 and pEcNDM1-4
The two bla NDM-1 carrying plasmids, pRpNDM1-1 and pEcNDM1-4, were unrelated in plasmid backbone and genetic arrangement, except for a 9332 bp-segment between bp 6874 and bp 16 205 in pEcNDM1-4 and between bp 210 030 and bp 200 699 in pRpNDM1-1 which was 100% identical (Figure 3b). The 9332 bp-segment consisted of a truncated arr3, an unusually truncated dfrA27, 3′-CS, ISCR1, a truncated ble MBL, bla NDM-1, a truncated ISAba125 and a second copy of 3′-CS and an ISCR1 with a truncted qacE1. This segment was located in a complex class 1 integron in both pRpNDM1-1 and pEcNDM1-4. In comparison to the complex class 1 integron in pRpNDM1-1, the complex class 1 integron in pEcNDM1-4 contained a 218 bp deletion in the 5′ CS and immediately downstream of the 5′ CS an additional gene cassette array aacA4-bla OXA-30-catB3 which is prevalent in Enterobacteriaceae plasmids, such as plasmid pCF1-1 (Figure 3a).
The discovery and characterization of the 4812 bp NDM-1-ISCR1 element
When the plasmid DNA extracted from JM109 (pEcNDM1-4) was probed with the bla NDM-1 probe, a 5 kb band (Figure 2f, labelled b) hybridized with the bla NDM-1 probe at low intensity although this band cannot be seen on the agarose gel (Figure 2e), in addition to the heavy smear band that corresponds to the bla NDM-1 carrying 58 kb plasmid. The 5 kb band was unexpected, suggesting that a free form of a 5 kb DNA molecule carrying bla NDM-1 exists in the cell.
Using primer pair C4-F and C5-R designed to amplify the sequence between arr3 and qnrA3 (Figure 2a), two fragments of approximately 5 kb and 10 kb were amplified from pEcNDM1-4 (Figure 2g, Lane 1) and one fragment of approximately 5 kb was amplified from pEcNDMneg-4 (Figure 2g, Lane 2). The latter was much more intense, although similar amounts of templates were used for the 2 PCR amplifications. Cloning and sequencing of the 5 kb and 10 kb agarose gel purified fragments from the pEcNDM1-4 PCR amplification revealed that the 10 kb fragment corresponds to the region between arr3 to qnrA3 (see Figure 2a) and the 5 kb fragment corresponds to the same region but is devoid of the 4812 bp NDM-1-ISCR1 element. We also sequenced the 5 kb PCR product from pEcNDMneg-4. The sequences of the two 5 kb fragments were 100% identical. These results suggest that the 4812 bp NDM-1-ISCR1 element can be excised from the plasmid. The 5 kb band observed in the Southern hybridization of undigested plasmid DNA extract described above must be the 4812 bp NDM-1-ISCR1 element, providing further evidence of the existence of a free form of the 4812 bp NDM-1-ISCR1 element.
Four pairs of primers, L1F/R to L4F/R, facing diverging directions were designed to confirm the structure of the 4812 bp NDM-1-ISCR1 element. An approximately 5 kb fragment was amplified from EcNDM1 using each of the four pairs of primers (Figure 2g). Sequencing of these PCR products showed that they all corresponded to the 4812 bp NDM-1-ISCR1 element, albeit the sequences started from different positions as the primers anneal at different sites. These PCR experiments confirmed the presence of the 4812 bp NDM-1-ISCR1 element. However the PCR results cannot determine whether the NDM-1-ISCR1 element is in a circular form or in tandem multiple copies since the diverging primer pairs can amplify a product in both scenarios.
We then used restriction mapping to determine whether there is more than one copy of the NDM-1-ISCR1 element on the plasmid. There is a NotI site at bp 12 238 located in the 4812 bp NDM-1-ISCR1 element and a NotI site upstream at bp 2218 (Figure 2a). When the plasmid pEcNDM1-4 was digested by NotI and probed with the bla NDM-1 probe, a 10 kb hybridization band was observed as expected (Figure 2e and 2f). A 5 kb hybridization band was also observed and was approximately double of the intensity of the 10 kb hybridization band, suggesting that there were double copies of the 5 kb band relative to the 10 kb band. We then used 2 more restriction enzymes, ClaI and BsaXI, to further determine whether there is a single copy or multiple copies of the NDM-1-ISCR1 element. The ClaI (restriction sites flanking the bla NDM-1 gene at bp 4619 and bp 18 518) and BsaXI (restriction sites flanking the bla NDM-1 gene at bp 6738 and bp 16 939) digestions were expected to generate 13 kb and 10 kb fragments respectively. However when plasmid pEcNDM1-4 was digested by ClaI and BsaXI and probed with the bla NDM-1 probe, a 23 kb band and a 20 kb band were hybridized with the bla NDM-1 probe respectively, which is 10 kb larger than expected (Figure 2f). The extra 10 kb indicates that there are 2 more copies of the NDM-1-ISCR1 element present in tandem. However this result is inconsistent with the C4-F/C5-R PCR results which only detected a 5 kb and a 10 kb band corresponding to 0 or 1 copy of the NDM-1-ISCR1 element. The discrepancy can be explained by that the C4-F/C5-R PCR failed to amplify the 20 kb amplicon that contains 3 copies of the NDM-1-ISCR1 element because of the length limit of the PCR amplification. This also explains the large difference in band intensity observed between pEcNDM1-4 and pEcNDMneg-4 in the C4-F/C5-R PCR described above. There must be a very small proportion of the pEcNDM1-4 carrying 0 or 1 copy of the NDM-1-ISCR1 element with the majority carrying 3 copies. Therefore the pEcNDM1-4 size is expected to vary from 53 416 kb (0 copy of the NDM-1-ISCR1 element) to 67 852 kb (3 copies of the NDM-1-ISCR1 element), which may also partly explain the smear observed in Figure 2e and Figure 2f of the total plasmid extract. Note that the assembled pEcNDM1-4 sequence has only one copy of the NDM-1-ISCR1 element with a size of 58 228 bp, due to the limitation of sequence assembly that cannot resolve the repeats.
Considering all of the results above, the 4812 bp NDM-1-ISCR1 element exists in two forms in EcNDM1 as illustrated in Figure 2a. One was located on the plasmid predominantly in three tandem copies, while the other was an extra-plasmid and extra-chromosomal free form. The latter must have been released from the plasmid in small quantities during replication, which may have resulted in some plasmids containing aberrant copies of the 4812 bp NDM-1-ISCR1 element.
The 4812 bp NDM-1-ISCR1 element was transferable by transformation to E. coli EcNDMneg
The transferability of the 4812 bp NDM1-ISCR1 element was examined by using E. coli strain EcNDMneg as a host which is sensitive to imipenem. The 4812 bp NDM-1-ISCR1 element was amplified by PCR using primer pair L5-F/R (Table S1 in File S1) and the PCR product was ligated by T4 DNA ligase to become circular. The PCR product was introduced into E. coli EcNDMneg by electroporation, and a carbapenem-resistant transformant named EcNDMneg (4812) was found. The susceptibility profile of EcNDMneg (4812) was identical to that of strain EcNDM1 (Table 1). Targeted PCR sequencing found that the 4812 bp NDM-1-ISCR1 element precisely inserted downstream of the existing ISCR1 in pEcNDM1-4 to recreate an identical gene arrangement to pEcNDM1-4.
Discussion
This case of isolating two NDM-1 positive strains of different species, R. planticola and E. coli isolated from the same patient provided an interesting scenario of emergence of NDM-1 within the same patient in vivo. The NDM-1 positive R. planticola strain was isolated when only mezlocillin/sulbactum, ceftizoxime and linezolid were used, suggesting that the emerging of NDM-1 was not due to administration of carbapenem antibiotics in this case. However, the NDM-1 positive E. coli was isolated after administration of imipenem which may have allowed the selection of an E. coli strain that possessed NDM-1. The 9332 bp fragment carrying an unusually truncated dfrA27 gene was shared by the two plasmids, suggesting an additional link between the two bla NDM-1 carrying plasmids pRpNDM-1 and pEcNDM1-4. The latter further harboured an additional gene cassette array, aacA4-cr-bla OXA-30-catB3, which is highly prevalent in Enterobacteriaceae, with a 218 bp deletion in the 5′-CS. This deletion would most likely have been generated when the gene cassette array was inserted in pEcNDM1-4. Therefore the likely process is that E. coli acquired bla NDM-1from R. planticola and additionally acquired the gene cassette array aacA4-bla OXA-30-catB3 from another Enterobacteriaceae plasmid, such as pCF1-1. The alternative is acquisition of bla NDM-1 by R. planticola from E. coli, which is also possible but less likely since acquisition of a 218 bp region to undo the deletion in pRpNDM1-1 is less probable. There is also a possibility that bla NDM-1was gained from another source by one or both species independently.
ISCR1 element was first observed in class 1 integrons In6 and In7 in 1993 [26]. Since then, ISCR1 elements were shown to have a close association with trimethoprim, quinolone, aminoglycoside resistance genes and several β-lactamase genes. Two copies of ISCR1 with a 3′CS upstream flanking different subtypes of qnr genes were also reported in China [27], France [17] and other countries [28]. bla NDM-1 has always been found to be carried on a structure consisting of a partial ISAba125 and a ble MBL gene with bla NDM-1 sandwiched between them [3]. ISCR1 elements adjacent to the conserved ISAba125-bla NDM-1- ble MBL structure have been reported [3], [16], [18]. However, this is the first time to report that bla NDM-1 was flanked by an ISCR1 element on both sides. The conserved 1596 bp ISAba125-bla NDM-1- ble MBL region was flanked by a copy of the integron 3′CS qacE1/sul and ISCR1 on both sides (Figure 3a) in the two NDM-1 carrying plasmids of different species R. planticola and E. coli.
ISCR1 is a known means of mobilizing its adjacent sequence via rolling circle transposition. Partridge and Hall [14] and Toleman et al. [15] demonstrated that rolling circle replication of the ISCR1 element can produce circular modules that include the ISCR1 and its upstream adjacent antibiotic resistance genes. These circular modules can then be rescued by recombination between homologous fragments. The 4812 bp NDM-1-ISCR1 element may be mobilized by rolling circle transposition. Our isolation of two nearly identical E. coli strains from the same fecal sample with only one being NDM-1 positive presented an interesting scenario for the acquisition of bla NDM-1 and provided a vehicle for experimental transfer of NDM-1 antibiotic resistance. Our in vitro transformation experiment using a synthesized circular 4812 bp NDM-1-ISCR1element showed that the element successfully transformed EcNDMneg containing plasmid pEcNDMneg-4 to a NDM-1 positive strain with the precise insertion of the element into the ISCR1 downstream in pEcNDMneg-4 to create an identical gene arrangement to pEcNDM1-4, demonstrating its mobility via ISCR1. However, in vitro transformation is artificial. There are likely other mechanisms of transfer.
The advantage for the bla NDM-1 gene embedding in a class 1 integron ISCR1 complex is that one of the most widely used mechanisms for the spread of antibiotics resistance across species is harnessed for NDM-1 dissemination. Therefore in addition to clonal expansion of a NDM-1 positive strain, NDM-1 can spread by multiple means including inter-strain spread within and between species through plasmids, transposons and the NDM-1-ISCR1 element. Given that strains carrying class 1 integrons or ISCR1 complex class1 integrons are common among bacteria [13], [15], [29], this is potentially a highly effective means for the spread of bla NDM-1.
In conclusion, bla NDM-1 was found to be embedded in an ISCR1 complex class 1 integron, which is most likely responsible for the bla NDM-1 transfer between R. planticola and E. coli in a patient in vivo. The novel 4812 bp NDM-1-ISCR1 element which can self excise to become a free form could provide a new vehicle for NDM-1 dissemination.
Supporting Information
Figure S1, Figure S2 and Table S1.
(DOC)
Acknowledgments
We thank Baoli Zhu (Institute of Microbiology, Chinese Academy of Science) for providing E. coli J53 Azr.
Funding Statement
This work was supported by the State Key Laboratory for Infectious Disease Prevention and Control of China [grant number 2012SKLID205] and the Priority Project on Infectious Disease Control and Prevention [grant number 2013ZX10004217] from the Ministry of Health and the Ministry of Science and Technology. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Shahid M (2011) Environmental dissemination of NDM-1: time to act sensibly. Lancet Infect Dis 11: 334–335. [DOI] [PubMed] [Google Scholar]
- 2. Walsh TR, Weeks J, Livermore DM, Toleman MA (2011) Dissemination of NDM-1 positive bacteria in the New Delhi environment and its implications for human health: an environmental point prevalence study. Lancet Infect Dis 11: 355–362. [DOI] [PubMed] [Google Scholar]
- 3. Yong D, Toleman MA, Giske CG, Cho HS, Sundman K, et al. (2009) Characterization of a new metallo-beta-lactamase gene, blaNDM-1, and a novel erythromycin esterase gene carried on a unique genetic structure in Klebsiella pneumoniae sequence type 14 from India. Antimicrob Agents Chemother 53: 5046–5054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Chen Y, Zhou Z, Jiang Y, Yu Y (2011) Emergence of NDM-1-producing Acinetobacter baumannii in China. J Antimicrob Chemother 66: 1255–1259. [DOI] [PubMed] [Google Scholar]
- 5. Bonomo RA (2011) New Delhi metallo-beta-lactamase and multidrug resistance: a global SOS? Clin Infect Dis 52: 485–487. [DOI] [PubMed] [Google Scholar]
- 6. Nordmann P, Poirel L, Carrer A, Toleman MA, Walsh TR (2011) How to detect NDM-1 producers. J Clin Microbiol 49: 718–721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Peirano G, Pillai DR, Pitondo-Silva A, Richardson D, Pitout JD (2011) The characteristics of NDM-producing Klebsiella pneumoniae from Canada. Diagn Microbiol Infect Dis 71: 106–109. [DOI] [PubMed] [Google Scholar]
- 8. Sole M, Pitart C, Roca I, Fabrega A, Salvador P, et al. (2011) First description of an Escherichia coli strain producing NDM-1 carbapenemase in Spain. Antimicrob Agents Chemother 55: 4402–4404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Kumarasamy KK, Toleman MA, Walsh TR, Bagaria J, Butt F, et al. (2010) Emergence of a new antibiotic resistance mechanism in India, Pakistan, and the UK: a molecular, biological, and epidemiological study. Lancet Infect Dis 10: 597–602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Jovcic B, Lepsanovic Z, Suljagic V, Rackov G, Begovic J, et al. (2011) Emergence of NDM-1 metallo-beta-lactamase in Pseudomonas aeruginosa clinical isolates from Serbia. Antimicrob Agents Chemother 55: 3929–3931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Hu H, Hu Y, Pan Y, Liang H, Wang H, et al. (2012) Novel plasmid and its variant harboring both a blaNDM-1 gene and type IV secretion system in clinical isolates of Acinetobacter lwoffii . Antimicrob Agents Chemother 56: 1698–1702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Sekizuka T, Matsui M, Yamane K, Takeuchi F, Ohnishi M, et al. (2011) Complete sequencing of the blaNDM-1-positive IncA/C plasmid from Escherichia coli ST38 isolate suggests a possible origin from plant pathogens. PLoS One 6: e25334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Toleman MA, Bennett PM, Walsh TR (2006) Common regions e.g. orf513 and antibiotic resistance: IS91-like elements evolving complex class 1 integrons. J Antimicrob Chemother 58: 1–6. [DOI] [PubMed] [Google Scholar]
- 14. Partridge SR, Hall RM (2003) In34, a complex In5 family class 1 integron containing orf513 and dfrA10. Antimicrob Agents Chemother 47: 342–349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Toleman MA, Bennett PM, Walsh TR (2006) ISCR elements: novel gene-capturing systems of the 21st century? Microbiol Mol Biol Rev 70: 296–316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Ho PL, Lo WU, Yeung MK, Lin CH, Chow KH, et al. (2011) Complete sequencing of pNDM-HK encoding NDM-1 carbapenemase from a multidrug-resistant Escherichia coli strain isolated in Hong Kong. PLoS One 6: e17989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Mammeri H, Van De Loo M, Poirel L, Martinez-Martinez L, Nordmann P (2005) Emergence of plasmid-mediated quinolone resistance in Escherichia coli in Europe. Antimicrob Agents Chemother 49: 71–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Janvier F, Jeannot K, Tesse S, Robert-Nicoud M, Delacour H, et al. (2013) Molecular characterization of blaNDM-1 in a sequence type 235 Pseudomonas aeruginosa isolate from France. Antimicrob Agents Chemother 57: 3408–3411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Institute CaLS (2011) Performance standards for antimicrobial susceptibility testing; twenty first informational supplement M100-S21. Clinical and Laboratory Standard Institute, Wayne, PA. [Google Scholar]
- 20. Beutin L, Kaulfuss S, Herold S, Oswald E, Schmidt H (2005) Genetic analysis of enteropathogenic and enterohemorrhagic Escherichia coli serogroup O103 strains by molecular typing of virulence and housekeeping genes and pulsed-field gel electrophoresis. J Clin Microbiol 43: 1552–1563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Ho PL, Shek RH, Chow KH, Duan RS, Mak GC, et al. (2005) Detection and characterization of extended-spectrum beta-lactamases among bloodstream isolates of Enterobacter spp. in Hong Kong, 2000–2002. J Antimicrob Chemother 55: 326–332. [DOI] [PubMed] [Google Scholar]
- 22. Vidal JE, Chen J, Li J, McClane BA (2009) Use of an EZ-Tn5-based random mutagenesis system to identify a novel toxin regulatory locus in Clostridium perfringens strain 13. PLoS One 4: e6232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Villa L, Poirel L, Nordmann P, Carta C, Carattoli A (2012) Complete sequencing of an IncH plasmid carrying the blaNDM-1, blaCTX-M-15 and qnrB1 genes. J Antimicrob Chemother 67: 1645–1650. [DOI] [PubMed] [Google Scholar]
- 24. Huang TW, Wang JT, Lauderdale TL, Liao TL, Lai JF, et al. (2013) Complete sequences of two plasmids in a blaNDM-1 positive Klebsiella oxytoca isolate from Taiwan. Antimicrob Agents Chemother 57: 4072–4076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Shen P, Jiang Y, Zhou Z, Zhang J, Yu Y, et al. (2008) Complete nucleotide sequence of pKP96, a 67 850 bp multiresistance plasmid encoding qnrA1, aac(6′)-Ib-cr and blaCTX-M-24 from Klebsiella pneumoniae . J Antimicrob Chemother 62: 1252–1256. [DOI] [PubMed] [Google Scholar]
- 26. Stokes HW, Tomaras C, Parsons Y, Hall RM (1993) The partial 3′-conserved segment duplications in the integrons In6 from pSa and In7 from pDGO100 have a common origin. Plasmid 30: 39–50. [DOI] [PubMed] [Google Scholar]
- 27. Ma J, Zeng Z, Chen Z, Xu X, Wang X, et al. (2009) High prevalence of plasmid-mediated quinolone resistance determinants qnr, aac(6′)-Ib-cr, and qepA among ceftiofur-resistant Enterobacteriaceae isolates from companion and food-producing animals. Antimicrob Agents Chemother 53: 519–524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Chen YT, Liao TL, Liu YM, Lauderdale TL, Yan JJ, et al. (2009) Mobilization of qnrB2 and ISCR1 in plasmids. Antimicrob Agents Chemother 53: 1235–1237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Rodriguez-Martinez JM, Poirel L, Canton R, Nordmann P (2006) Common region CR1 for expression of antibiotic resistance genes. Antimicrob Agents Chemother 50: 2544–2546. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1, Figure S2 and Table S1.
(DOC)