Abstract
The ability of cells to divide asymmetrically is essential for generating diverse cell types during development. The past 10 years have seen tremendous progress in our understanding of this important biological process. We have learned that localized phosphorylation events are responsible for the asymmetric segregation of cell fate determinants in mitosis, and that centrosomes and microtubules play important roles. The relevance of asymmetric cell division for stem cell biology has added a new dimension, and exciting connections between asymmetric cell division and tumorigenesis have begun to emerge.
Introduction
The development of multicellular organisms involves the specification of diverse cell types from a single fertilized egg. To generate this diversity, some cells can undergo an asymmetric cell division, during which they segregate protein or RNA determinants into one of the two daughter cells, thereby determining distinct cell fates.
The process of asymmetric cell division was originally described almost 100 years ago by Ed Conklin, who found that during division of early ascidian embryos an area of yellow cytoplasm always co-segregates with cells that will become muscle cells 1. It was not until 1994, however, that an asymmetrically segregating cell fate determinant from Drosophila melanogaster called Numb was functionally and molecularly characterized 2. During mitosis, Numb was found to localize into one edge of the cell, forming a crescent-shaped pattern, and to segregate into only one of the two daughter cells 2,3; in the absence of Numb normally different cells assume the same fate in D. melanogaster external sensory organs 4. These observations suggested that high levels of Numb in one of the two daughter cells cause the division to become asymmetric.
A similar asymmetric localization was found for Par proteins in C. elegans, which are also involved in other processes that require polarization 5-7. During the first division of the worm zygote, Par-3 8, Par-6 9 and PKC-3 10 accumulate at the anterior cell cortex and Par-1 11 and Par-2 12 accumulate posteriorly and segregate into one of the two daughter cells. In contrast to Numb in D. melanogaster, however, Par proteins are also required for many aspects of asymmetric cell division, including the establishment of different daughter cell sizes and the orientation and position of the mitotic spindle in C. elegans 6,7. In fact, it is the D. melanogaster homologues of the anterior Par proteins that direct the asymmetric localization of Numb into one of the two daughter cells 13-17.
A simple model of asymmetric cell division postulates that it is a three-step process in which the Par proteins set up a polarity axis in interphase 18. In mitosis, this axis is used both for spindle orientation and for the asymmetric localization of cell fate determinants. In telophase, the tight coordination of these two processes ensures that those determinants are inherited by only one of the two daughter cells
Since this model was proposed almost 10 years ago 18, new findings have emerged that have led to conceptual changes in this field of research. In this Review I highlight how recent discoveries have changed our view of how determinants are asymmetrically localized. I also summarize recent findings revealing a surprising role for centrosomes in maintaining the polarity axis over many divisions. Finally, I describe how the role of asymmetric cell division in mammalian development has been re-interpreted and how the connections between asymmetric cell division and tumorigenesis have opened unexpected and challenging avenues for this dynamic and rapidly moving field.
Asymmetric cell division: the basics
The mechanisms of asymmetric cell division have been derived from studies of invertebrates, and more specifically in D. melanogaster and C. elegans. Below, I describe the basic principles of this process in these organisms.
Asymmetric cell division in D. melanogaster
In Drosophila, most progress during the past ten years in understanding asymmetric cell division has been made in neuroblasts. Neuroblasts delaminate from the ventral neuroectoderm during embryogenesis. In embryos, they undergo up to 20 rounds of asymmetric cell division to generate the neurons of the larval nervous system and become quiescent at the end of embryogenesis. During larval stages of development, neuroblasts re-enter the cell cycle and continue to divide asymmetrically to generate the neurons of the adult fly brain 19. Several types of larval neuroblasts can be distinguished based on lineage and location (Fig. 1a), and unique markers exist to allow their identification (suppl. Fig 1). Most are type I neuroblasts, which divide into a large cell that remains a neuroblast and a smaller ganglion mother cell (GMC) that divides only once more into two terminally differentiating neurons. Type II neuroblasts are located in the dorso-posterior region of each central brain hemisphere and divide to give rise to a different cell lineage 20-22. Their smaller daughter cell becomes an intermediate neural precursor (INP) that continues to undergo self-renewing asymmetric divisions, generating one INP and one GMC. Furthermore, specialized kinds of type I neuroblasts exist in the mushroom bodies (MB) 19,23 and the optic lobes 24.
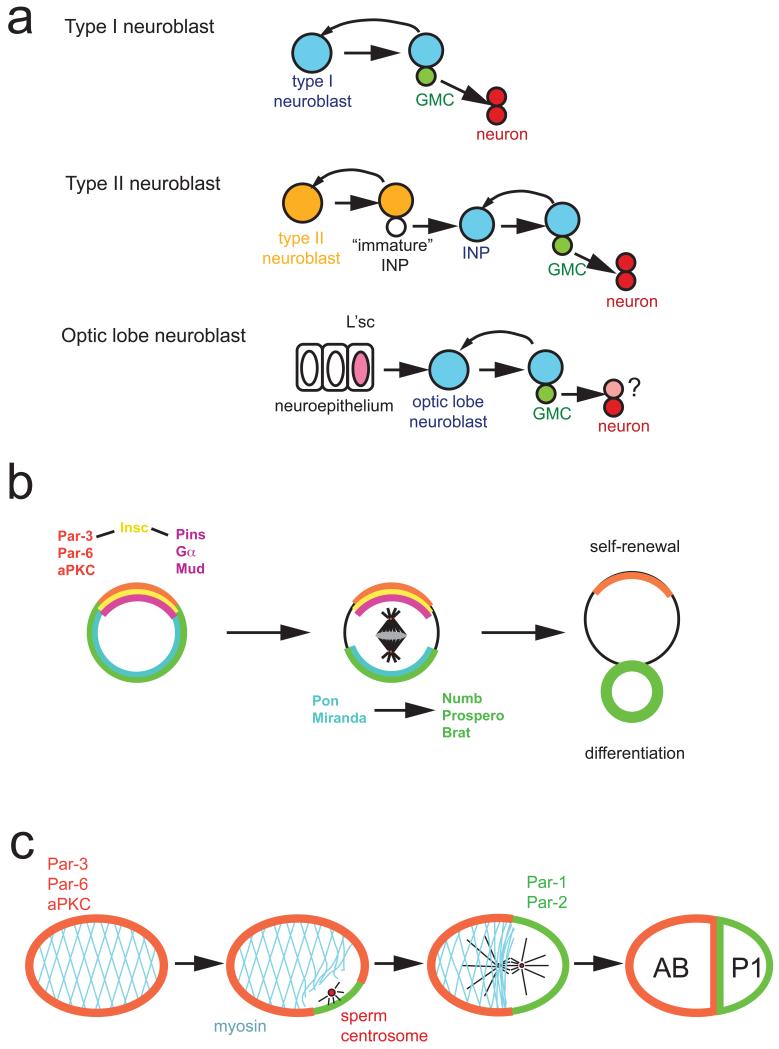
Figure 1. Models for asymmetric cell division.
a ∣ Drosophila type 1 neuroblasts (top, blue) divide asymmetrically into one neuroblast and a ganglion mother cell (GMC, green). The neuroblast self-renews while the GMC divides terminally into two neurons. Type II neuroblasts (middle, orange) divide into one self-renewing type II neuroblast and one intermediate neural precursor (INP). The INP starts expressing the neuroblast markers Asense and Deadpan to become a mature INP which divides asymmetrically into one GMC and one INP. In the optic lobe (bottom), neuroblasts arise from an epithelium. A wave of l(1)sc expression spreads through the epithelium and induces neuroblast formation. Optic lobe neuroblasts (blue) behave like type I neuroblasts although their precise lineage has not been determined. Differential expression of the markers Deadpan, Asense, Prospero and ElaV allows the unique identification of Individual cell types in type I, type II and optic lobe neuroblast lineages (see suppl. Fig. 1).
b ∣ In Drosophila neuroblasts, the apically localized Par-3, Par-6, aPKC complex is connected to the Pins, Gα, Mud complex by the adaptor protein Inscuteable (Insc). During mitosis, this apical complex directs both the orientation of the mitotic spindle and the asymmetric localization of the adaptor proteins Pon and Miranda and the cell fate determinants Numb, Prospero and Brat to the opposite, basal cell cortex. After mitosis, Numb, Prospero and Brat act together to prevent self-renewal and induce cell cycle exit and differentiation.
c ∣ In the C. elegans zygote, the anterior Par proteins Par-3, Par-6 and PKC-3 segregate into the anterior AB and the posterior Par-proteins Par-2 and Par-1 into the posterior P1 cell. Polarization starts after fertilization, when interactions between sperm centrosome and cortex allow Par-2 to accumulate. This initiates an anterior contraction of the cortical actin cytoskeleton, which allows anterior movement of Par-3 and Par-6.
The basic mechanism of asymmetric cell division is common to all D. melanogaster neuroblasts 25-28 (Fig. 1b). The endocytic protein 29 Numb and the translational inhibitor 30 Brat transiently accumulate at the basal plasma membrane in late prometaphase 3,31-33. Their asymmetric localization is facilitated by two adaptor proteins that localize asymmetrically at the same time as Numb and Brat. Brat localizes by binding to Miranda 31,33, and Numb localization is facilitated by (but not dependent on) the adaptor protein Pon 34,35. In type I neuroblasts and INPs, Miranda also transports the transcription factor Prospero into the GMC 36-40. Slightly after the basal determinants localize, the mitotic spindle is set up in an apical–-basal orientation so that the determinants are inherited by the basal daughter cell.
The asymmetric localization of basal determinants requires another set of proteins that accumulate at the apical cell cortex before mitosis. These include the PDZ domain-containing proteins Par-3 and Par-6 and the protein kinase aPKC 13-17 (which is the D. melanogaster homolog of C. elegans PKC-3). They also include the adaptor protein Inscuteable 41,42, which links Par-3–Par-6–aPKC to a second protein complex containing the heterotrimeric G-protein α-subunits Gαi 43 and the adaptor protein Pins 44,45,43. Pins binds to the microtubule-associated and dynein-binding protein Mud 46-48 and thereby provides a cortical attachment site for astral microtubules to ensure the apical-basal orientation of the mitotic spindle.
The initial apical localization of Par-3, Par-6 and aPKC is inherited from epithelial cells of the ventral neuroectoderm when neuroblasts delaminate 13,14,16,17. In these epithelial cells, Par-proteins localize apically and are required for establishing and maintaining apical basal polarity. In fact, Par-3, Par-6 and aPKC and their homologs in other organisms play a key role in almost all known cell polarity events including epithelial polarity, axon outgrowth, synapse formation and specification of the anterior-posterior body axis 6,7. How Par-proteins direct the asymmetric localization of cell fate determinants during asymmetric cell division and how their apical localization is maintained during subsequent neuroblast cell cycles has only become clear more recently and is reviewed below.
Asymmetric cell division in C. elegans
In C. elegans, the first cell division during development generates an anterior AB and a posterior P1 cell (for excellent reviews, see refs 49 and 50). Both the size and the fate of the two daughter cells are different, and the mechanisms that generate this asymmetry share exciting similarities with those that act in neuroblasts (Fig. 1c). Polarization of the zygote starts when the entire cortical actin cytoskeleton moves towards to the anterior pole 51. This movement is initiated by the sperm centrosome 52,53 and by the Rho-GEF CYK-4 54, which is contributed during fertilization and remains localized close to the posterior male pronucleus. As a result of anterior cortical movement, surface contractions that initially occur throughout the cell are progressively confined to the anterior half while the posterior side becomes smooth 50. Par-3, Par-6 and PKC-3 (the aPKC homolog in C. elegans) are uniformly cortical initially but concentrate anteriorly after fertilization. The anterior movement of cortical actomyosin is responsible for asymmetric localization of anterior Par proteins 51, although a second, myosin-independent mechanism has been described 55. Par-2 and Par-1 become enriched on the posterior, non-contracting cell cortex, and inhibitory interactions between the anterior and posterior Par proteins maintain their localization to opposite cortical domains. Par-2, for example, prevents the cortical localization of Par-3 56 and aPKC phosphorylates Par-2 56 this removes it from the plasma membrane. Thus, in contrast to D. melanogaster, Par-proteins are involved both in regulating asymmetric cell division but also in the symmetry breaking events that establish the anterior-posterior axis in the zygote.
The distinction between proteins establishing polarity and segregating determinants in C. elegans is not as clear as in D. melanogaster 57. Besides the Par-proteins, the asymmetric division of the zygote is influenced by the CCCH-Zn finger proteins Mex-1, Mex-5, Mex-6, Pos-1 and Pie-1, the RNA-binding proteins Mex-3 and Spn-4 and the homeodomain protein Pal-1 57. Pie-1 is inherited by the posterior P1 cell 58, where it blocks transcriptional elongation 59 and prevents the expression of genes that would promote somatic differentiation in the germline blastomeres 60. Mex-5 and Mex-3 segregate into the anterior AB daughter cell and inhibit the specification of muscle cell fate in its progenitors 61,62. The Par-proteins are essential for asymmetric segregation of Pie-1, Mex-5 and Mex-3. However, Mex-5 also acts redundantly with the highly related Mex-6 in regulating actin flow and anterior Par-protein accumulation 63. In fact, most of these proteins are also involved in the asymmetric segregation of other factors. With the notable exception of Pie-1 and Pal-1, they are therefore as polarity mediators rather than segregating determinants 57.
Asymmetric localization of determinants
The mechanisms that lead to the asymmetric localization of Numb, Prospero and Brat in D. melanogaster neuroblasts had remained a mystery for many years. Similarly, it was unclear, how cytoplasmic determinants are segregated into AB or P1 in C. elegans. Initial experiments using chemical inhibitors in D. melanogaster showed that the process does not require microtubules but depends on actin and myosin 36,64-66. This led to a model in which an actin and myosin-dependent process moves asymmetrically segregating cell fate determinants along the cell cortex to concentrate them on the basal side 18,67. Support for this model came from the demonstration that myosin VI is important for asymmetric cell division 68 and from the identification of the cytoskeletal protein Lgl as a major factor for the basal localization of Numb but not for the apical localization of Par proteins 69,70. Lgl binds to cytoplasmic non-muscle myosin 71,72, and this interaction is inhibited through phosphorylation by the apical protein aPKC 73,74. As a result, Lgl is inhibited on the apical cortex but active on the basal side, where it could potentially inhibit myosin. Consistent with this hypothesis, myosin II is concentrated apically in neuroblasts 75 and when it is inhibited by mutation or chemical inhibitors of Rho-kinase (ROCK), Numb and its interacting protein Miranda (see below) no longer concentrate on the basal side 75.
Although the cortical transport model is attractive, it has been challenged by several recent observations. The asymmetry in myosin localization is not observed in external sensory organs 76 and could not be confirmed in more recent reports, which actually describe myosin localization to the opposite, basal side of the neuroblast 77. Furthermore, the ROCK inhibitor used to demonstrate a requirement for myosin for Numb and Miranda localization can also inhibit aPKC 78. Finally, photobleaching (FRAP) experiments did not reveal unidirectional cortical transport of the Numb adaptor Pon 79. Instead, FRAP recovery rates showed that Pon and Numb rapidly exchange between cortex and cytoplasm and that local differences in cortical ‘on’ and ‘off’ rates, rather than cortical transport, are responsible for their asymmetric localization 80. Therefore, the cortical transport model has been replaced by more dynamic models in which differential mobility or cortical attachment of protein determinants to the apical and basal plasma membranes regulate their asymmetric localization. Below, I discuss how those models explain asymmetric determinant segregation in D. melanogaster and describe very similar models that explain the asymmetric localization of cytoplasmic proteins in C. elegans.
The prophase pathway: asymmetric phosphorylation
In D. melanogaster neuroblasts Numb is recruited to the plasma membrane through phospholipid interactions of positively charged amino acids in its N-terminus 65. Next to those residues are several phosphorylation sites for aPKC, mutation of which to Alanine abolishes the asymmetric localization of Numb in mitosis 81. These observations suggest that aPKC phosphorylation neutralizes positive charges and thereby inhibits the membrane association of Numb 76 (Fig. 2a).
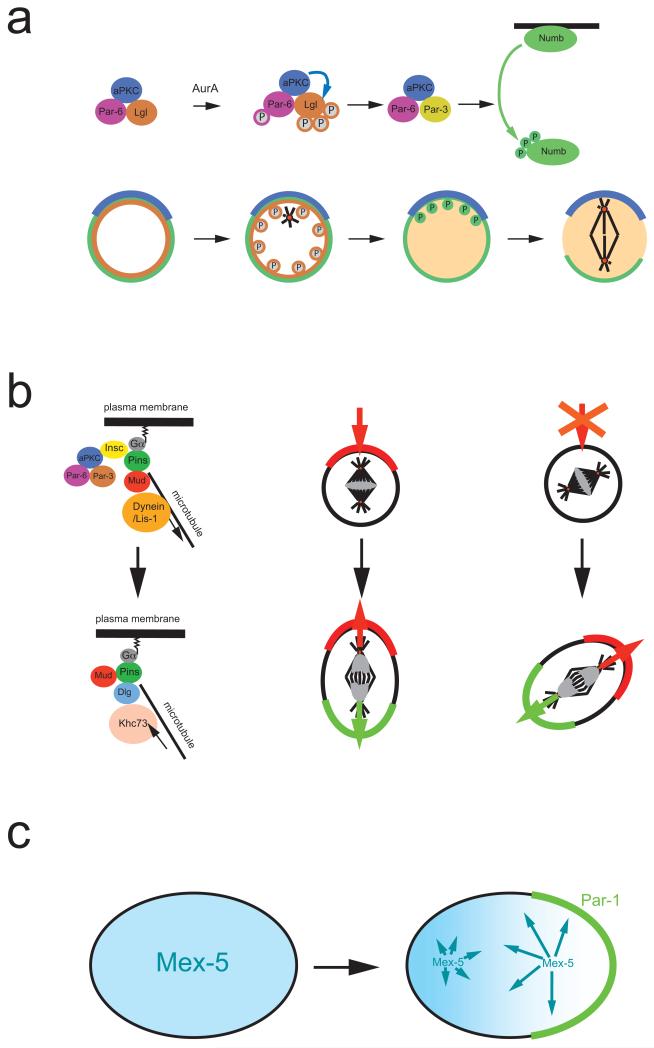
Figure 2. Asymmetric segregation of protein determinants.
a ∣ In Drosophila melanogaster neuroblasts, activation of the kinase Aurora A triggers a subunit exchange in the apically localized Par-3–aPKC complex. Aurora A phosphorylates Par-6, which in turn activates aPKC, leading to Lgl phosphorylation and exit from the complex. Lgl is exchanged for Par-3, which acts as an adaptor that allows aPKC to phosphorylate of Numb. Phosphorylated Numb is released into the cytoplasm. Since aPKC is restricted to the apical cortex, Numb is retained on the basal side and segregates into the basal daughter cell.
b ∣ Left side: In metaphase, Gα, Pins and Mud establish a cortical attachment site for astral microtubules to orient the mitotic spindle. In telophase, however, it is the mitotic spindle that influences cortical polarity via a pathway involving the kinesin Khc-73 and the protein Dlg. Right side: Normally, the telophase pathway is not essential. When components of the apical complex are missing, however, it will rescue the formation of opposing cortical domains in ana and telophase. The new polarity axis aligns with the mitotic spindle and no longer always with apical-basal polarity.
c ∣ In C. elegans, Mex-5 (blue) and Pie-1 (not shown) exist as fast and slow diffusing forms. The faster diffusing form of Mex-5 is more abundant posteriorly while more rapidly diffusable Pie-1 is anterior, resulting in the asymmetric distribution of the cytoplasmic proteins. For Mex-5, phosphorylation by posteriorly localized Par-1 may be responsible for the faster diffusion rate.
In interphase, aPKC is in a complex with Par-6 and Lgl, and this complex cannot phosphorylate Numb, presumably because the substrate-binding site is blocked. On entry into mitosis, the kinase Aurora A phosphorylates the aPKC binding partner Par-6 76, leading to activation of and consequent phosphorylation of Lgl. This reduces Lgl’s affinity for Par-6 and aPKC, thereby allowing Par-3 to enter the complex 82. Par-3 can bind to both Numb and aPKC and might act as an adaptor between kinase and substrate. This subunit exchange initiates the phosphorylation of Numb because aPKC only phosphorylates Numb when it is bound to Par-3 but not when bound to Lgl. Therefore, the function of Lgl is not to recruit determinants to the cortex, as previously thought, but to regulate the substrate specificity and maybe also activity of aPKC. In lgl mutants, for example, it is premature aPKC phosphorylation rather than myosin defects that prevent Numb localization, and the effects of nonphosphorylatable Lgl overexpression occur owing to aPKC inhibition 78 rather than active recruitment of asymmetric determinants to the cortex. These new findings have converted Numb localization from a complete mystery to one of the best understood events that occur in a mitotic cell.
In fact, aPKC-dependent phosphorylation is a general mechanism for asymmetric protein localization during mitosis, at least in D. melanogaster. aPKC can also phosphorylate the adaptor protein Miranda 76 and regulates its cortical localization similarly to how it controls Numb 78. Furthermore, the E3 ubiquitin ligase Neuralized, which segregates asymmetrically in sensory organ precursor cells, contains aPKC consensus sites in its N-terminal phosphoinositide-binding domain 83, suggesting that it might also be regulated by aPKC. This new model of phosphorylation dependent asymmetric cell division does not implicate actomyosin as a major player in asymmetric protein localization. Consistent with this, the weak actin inhibitor cytochalasin D does not inhibit the process, although it can prevent cytokinesis 36. The model also might explain why asymmetric segregation of aPKC alone is sufficient to generate different fates even when Numb and Miranda are inherited by both daughter cells in mutants with altered spindle orientation 84. As both proteins need to be membrane bound to carry out their functions (Numb acts on endocytic vesicles and Miranda acts by recruiting other proteins to the cortex), they can be inhibited by aPKC phosphorylation in one of the two daughter cells. Therefore, it is the ratio between aPKC and basal determinants that ultimately determines the fate of each daughter cell 84.
The telophase pathway: microtubule–cortex interactions
Numb and Miranda still segregate asymmetrically in mutants in which the asymmetric localization of pro- and metaphase is completely abolished 15,69,70. This is due to a second pathway for asymmetric determinant localization that acts in ana- and telophase of the cell cycle (recently reviewed in 85). In contrast to the Prophase pathway, the telophase pathway is sensitive to microtubule-depolymerizing drugs or to mutations affecting astral microtubules 85,86 (Fig. 2b). In wild-type D. melanogaster, the pathway is not required for asymmetric protein localization in metaphase, as disruption of microtubules has no effect 36. In inscuteable mutants, however, in which Par-3–Par-6–aPKC is delocalized in interphase 43,44 and mitotic spindles are no longer oriented along the apical basal axis, the microtubule-dependent pathway is responsible for Pins and Gαi accumulation over one of the two spindle poles in mitosis and asymmetric segregation of determinants so that cell fate specification occurs normally in a large subset of neuroblasts 86. The microtubule-dependent pathway for neuroblast polarization depends on the Pins binding partner Discs large (Dlg), a membrane-associated guanylate kinase (MAGUK) that also plays a part in Numb and Miranda localization in wild-type embryos. It also requires the kinesin Khc-73, which localizes to microtubule plus-ends and can bind to Dlg. These observations have suggested a model in which Khc73 transported on astral microtubules is responsible for the accumulation of Dlg and Pins over one spindle pole. Pins then recruits the microtubule-binding protein Mud, and this mutual microtubule–cortex interaction stabilizes spindle orientation.
Although this model is attractive and consistent with all data, several key questions remain. For example, it is unknown how Dlg and Pins establish the localization of basal determinants when the Par proteins are not asymmetric. Furthermore, the phenotype of dlg mutants is not entirely consistent with the model: basal determinants do not localize in metaphase in these mutants but the rescue of asymmetric cell division in telophase still occurs 69,70. Therefore, Dlg is clearly required for the telophase pathway when other regulators are missing, but its function in the telophase pathway can be replaced when the rest of the machinery is intact. Clearly, the precise molecular function of the telophase pathway still needs to be defined.
Asymmetric protein segregation in C. elegans
The mechanisms that are thought to operate in C. elegans asymmetric cell division are remarkably similar to D. melanogaster, even though the segregating determinants Pie-1 and Mex-5 localize asymmetrically in the cytoplasm and not at the cortex. The basis for their asymmetric localization is both regulated protein degradation and a reaction–diffusion mechanism in which asymmetry is established through different ratios of slowly and rapidly diffusing isoforms in the anterior and posterior halves.
Protein degradation contributes to Pie-1 asymmetry in late-stage embryos, but not in the zygote 87-89. Pie-1 degradation during these late cycles is mediated by the SOCS box protein ZIF-1 87, which interacts with the CCCH Zn-fingers of Pie-1 and also binds to a ubiquitin ligase complex containing Elongin C, the Cullin Cul-2 and the E2 enzyme ubc-5. Together, these proteins degrade Pie-1 in somatic cells and thereby restrict its expression to the germline. Interestingly, Mex-5 activates ZIF-1 and is also required for restricting Pie-1 to the germline. This degradation mechanism nicely explains the antagonistic expression of Mex-5 and Pie-1 in later embryos.
In the zygote, the asymmetric localization of Pie-1 and Mex-5 is thought to be established through a reaction–diffusion mechanism 88,90 (Fig. 2c). This mechanism is used to describe chemical reactions and involves two substances that can be converted into each other by a chemical reaction and move in space with different kinetics. The mechanism was initially applied to biology by Turing 91 and it is now well established that reaction–diffusion mechanisms are responsible for pattern generation in many biological systems 92. In this case, the two substances can be differentially modified forms of a protein or a free and a complex-associated type. FRAP and FCS experiments have shown that both Pie-1 and Mex-5 exist as rapidly and slowly diffusing isoforms 88,90. The ratio between these isoforms is different in the anterior and posterior parts of the zygote, with more slowly diffusing Pie-1 localized posteriorly and more slowly diffusing Mex-5 anteriorly. In both cases, mathematical modelling of the protein distributions resulting from the measured diffusion coefficients predicts the observed asymmetric protein distributions.
So how are the apparent differences in cytoplasmic mobility established? Mex-5 needs to be phosphorylated by Par-1 to localize asymmetrically 89. Par-1 is concentrated posteriorly and can locally modify the association of Mex-5 with the actin cytoskeleton and change Mex-5 mobility. This explains the actin dependence of Mex-5 asymmetry, although the asymmetric movement of the actin meshwork itself adds an additional complication. For Pie-1, differential association with posterior P granules was proposed 88. As the P granules segregate asymmetrically in a Par-protein dependent manner and therefore this would explain Pie-1 asymmetry although biochemical evidence for this is still missing.
Thus, differential association with membranes or other cellular components rather than directional transport establishes the asymmetric localization of cell fate determinants both in D. melanogaster and in C. elegans.
A new role for the centrosome
10 years ago, microtubules were thought not to have a role during asymmetric cell division in D. melanogaster 18,67. Now, it is clear that microtubules play an important role in the telophase pathway. In addition, microtubule-dependent cortical interactions are integral to maintain polarity over many divisions.
D. melanogaster neuroblasts repeatedly divide along the apical-basal axis. Real-time analysis of spindle orientation has revealed that the mitotic spindle is established parallel to the embryonic surface but then rotates by 90 ° into its final vertical position 93 (Fig. 3). It was thought that both centrosomes organize microtubule asters simultaneously at the onset of mitosis and set up a bipolar mitotic spindle in prophase.
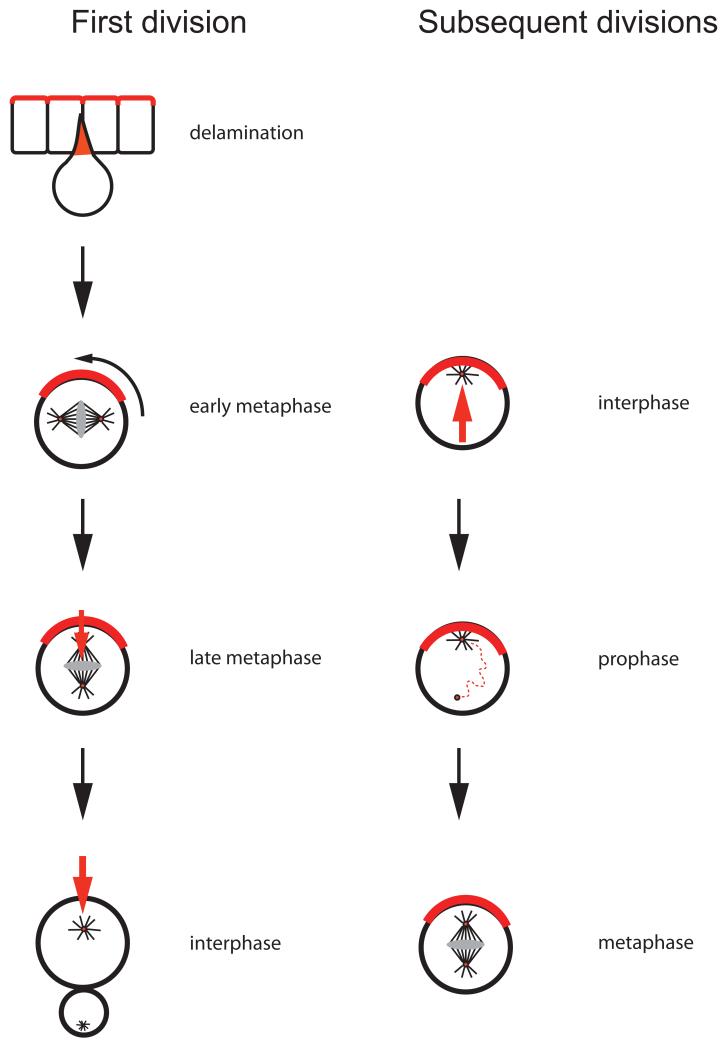
Figure 3. Par-proteins and centrosomes establish cortical polarity in Drosophila neuroblasts.
During the first neuroblast cycle, Par proteins are inherited from the apical cortex of the overlying epithelium. Through a series of adaptor proteins, they recruit Mud (red), which forms cortical attachment sites for astral microtubules and thereby induces spindle rotation into an apical–basal orientation. During subsequent cell cycles, centrosomes are responsible for maintaining cortical polarity in interphase. Cortical Pins, in turn, is required for maintaining the apical centrosome while the sister centriole migrates through the cytoplasm. On the basal side, this centriole recruits pericentriolar material to form a bipolar spindle in the proper apical-basal orientation.
More recently, it became clear that this mechanism only applies to the first division of embryonic neuroblasts. During subsequent divisions, the apical position of the neuroblast centrosome that results from the previous cytokinesis is maintained throughout interphase 94,95. After centriole duplication, the daughter centriole is initially devoid of pericentriolar material when it migrates to the basal side of the neuroblast. Only in prophase, shortly before nuclear envelope breakdown, a second microtubule aster appears. As a result, the mitotic spindle is already set up in its final, vertical orientation and does not rotate substantially in metaphase. Thus, contrary to what was thought before, the orientation of most neuroblast divisions is established very early in the cell cycle.
The orientation of the spindle across several neuroblast divisions is maintained by crosstalk between the centrosome and apical proteins. In pins mutants, the apical aster loses its microtubule-nucleating activity and starts to migrate basally, resulting in two identical centrioles and random spindle orientation 95. This suggests that apical proteins maintain the apical position of the centrosome in interphase. However, the positioning of apical proteins can also be instructed by the centrosome itself. When microtubules are transiently inactivated, the apical centrosome assumes a random position and induces the localized accumulation of Par proteins at its new position 96. This symmetry breaking property of the neuroblast centrosome is strikingly similar to C. elegans where it is the sperm-centrosome that breaks symmetry and establishes Par-domains. In contrast to D. melanogaster, however, the centrosome removes rather than attracts Par-3-Par-6-aPKC in the C. elegans zygote.
One important implication of these new findings is that the sister centrioles are not identical in neuroblasts and could therefore be involved in maintaining asymmetric cell division. In yeast, it has been demonstrated that the newly born centriole (called spindle pole body in yeast) is always inherited by the bud and never by the mother cell. During the asymmetric divisions of D. melanogaster testes, the mother centriole remains anchored at the stem cell niche and is always inherited by the daughter cell, which retains the self-renewal capacity. These observations have raised speculations about centrioles having fate determining properties 97. For example, during brain development in vertebrates, the mother centriole is preferentially inherited by the progenitor cell 98. In this case, removal of ninein, a protein that ensures this inheritance pattern, causes both randomization of centriole inheritance and a defect in progenitor cell maintenance. Although this is just a correlation, this finding indicates that centrosome asymmetry might contribute to asymmetric cell division in vertebrate cells.
Specifying daughter cell sizes
Besides cell fate, the daughter cells of D. melanogaster neuroblasts and the C. elegans zygote are different in size. Identification of the mechanisms through which this asymmetry is established has revealed an exciting role for heterotrimeric G-proteins in mediating microtubule cortex interactions (Fig. 4). Although the involvement of G-proteins was already clear ten years ago, how they interact with microtubules and establish cell asymmetry was discovered only recently.
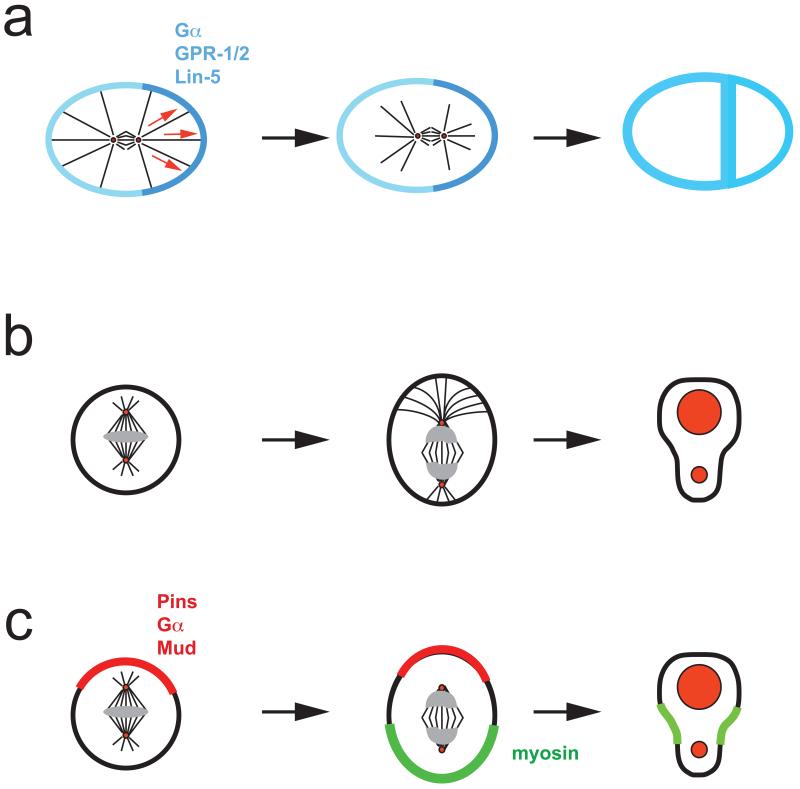
Figure 4. Three ways to generate different daughter cell sizes.
a ∣ In the C. elegans zygote, heterotrimeric G-protein α-subunits and the GoLoco proteins GPR-1 and GPR-2 recruit the dynein binding protein Lin-5 to the cortex to facilitate microtubule-cell cortex interactions. Higher concentration of GPR-1/2 on the posterior side, results in a net pulling force and posterior spindle displacement.
b ∣ In Drosophila neuroblasts, the apical microtubule aster is larger resulting in spindle displacement towards the basal and asymmetric cleavage.
c ∣ In neuroblasts, the apical Pins–Gα–Mud complex (red) induces a basal shift of cortical myosin,(green) resulting in basal displacement of the cleavage furrow. This pathway can generate asymmetric daughter cell sizes even in the absence of a mitotic spindle.
In C. elegans, size asymmetry during the first division is due to an asymmetric displacement of the mitotic spindle towards the posterior end of the cell (Fig. 4a). This is thought to be due to increased pulling forces exerted on the spindle at the posterior end mediated by heterotrimeric G-proteins and their binding partners, the C. elegans Pins homologs GPR-1 and GPR-2 99,100 (reviewed in 49). GPR-1 and GPR-2 carry a GoLoco domain that binds to the GDP-bound form of the G-protein α-subunit. Gα–GPR recruits Lin-5 101, the C. elegans homologue of the human microtubule-binding protein NuMA, the microtubule minus-end directed motor dynein and its subunit Lis-1 102. These proteins form a complex in the cytoplasm and are recruited to the plasma membrane by binding to Gα. At the membrane, this complex can form an attachment site for the plus ends of astral microtubules, thereby exerting pulling force on the mitotic spindle. As the concentration of GPR-1 and GPR-2 is higher at the posterior cortex, the mitotic spindle is pulled towards this end.
It is likely that the mechanism identified in C. elegans applies to both Drosophila and mammals, as all components of the system are conserved and their subcellular localization and biochemical interactions are similar to what is found in worms. In Drosophila neuroblasts, however, the mechanisms regulating daughter cell sizes are different from C. elegans. First, the mitotic spindle itself is asymmetric in shape with a larger apical and a much smaller basal microtubule aster (Fig. 2b). Second, recent experiments suggest that the site of cytokinesis is not only determined by the spindle but that a second, cortical pathway exists 77 (Fig. 2c). Evidence for this pathway comes from live imaging experiments showing that the cleavage furrow proteins Anillin, Pavarotti (a D. melanogaster guanidine nucleotide exchange factor for Rho) and myosin accumulate in the basal side of the cell before the mitotic spindle becomes asymmetric, and this is mediated by the apical Pins–Gα–Mud complex. Surprisingly, this cortical asymmetry as well as the resulting asymmetric cleavage furrow can even be established when spindle formation is blocked by microtubuledepolymerizing drugs, and the resulting checkpoint arrest is overcome by a mutation in a kinetochore protein. In mutants affecting spindle orientation but not cortical polarity, the cortical polarity pathway and the classical-spindle induced pathway are both active, resulting in the formation of anucleate lobes of cytoplasm that are cleaved from the mother cell. These surprising recent findings that shake the dogma for how cytokinesis is established will certainly spark new insights into his important process. As myosin asymmetry has recently also been described in C. elegans, the new mechanism is conserved and might well occur in higher organisms as well 103.
Asymmetric division in tumour formation
The connection between asymmetric cell division and tumorigenesis has been one of the most surprising and important findings in the field during the past 10 years. Furthermore, studies in mammals have identified a link between tumorigenesis and dysregulated asymmetric cell division of stem cells.
Tumorigenesis in D. melanogaster
Genetic screens for brain tumour formation in D. melanogaster carried out in the 1970s revealed an involvement for lgl, dlg, lgd, brat and l(3)mbt 104,105. In D. melanogaster mutants for any of these genes neuroblasts fail to differentiate, leading to tumour-like overproliferation. Upon transplantation, the tumours continue to grow, undergo metastasis and become aneuploid.
The identification of Lgl and Dlg as key regulators of asymmetric cell division 69,70 and of Brat as a segregating determinant 31,33 suggested that those tumours actually arise from defects in asymmetric cell division. Indeed, transplantable tumours also form in mitotic neuroblast clones mutant for numb or prospero 20,32 or upon overexpression of activated aPKC 106. Subsequent analysis showed that tumours can also occur in mutants for the mitotic kinases Aurora A 107,108 or Polo 35, on overactivation of Notch 108, in mutants affecting spindle orientation 46-48,84,109 or even when neuroblasts divide with an excess number of centrosomes 110,111. In all these cases, defects in asymmetric cell division are the root cause for tumour formation. Notably, however, mutations apical proteins such as aPKC or Pins have the opposite phenotype, resulting in fewer neuroblasts.
The simplest explanation for tumour formation is that defects in segregating determinants result in symmetric divisions, giving rise to two neuroblasts (Fig. 5a). The resulting exponential increase in neuroblast number would explain certain aspects of tumour formation, but not why tumour neuroblasts do not exit the cell cycle and continue to proliferate even in adult brains 32 or on transplantation 112. In addition, a detailed analysis of brat mutant clones showed that tumour formation does not simply involve a series of symmetric divisions 20. After an initial delay phase in which the Brat-inheriting cell fails to initiate correct marker expression and enters a prolonged cell cycle block, it divides and enters a second phase in which it proliferates rapidly and indefinitely. Thus, defects in asymmetric cell division cause the formation of tumour neuroblasts that lack the mechanisms responsible for cell cycle exit during pupal development.
Figure 5. Asymmetric cell division and tumour formation.
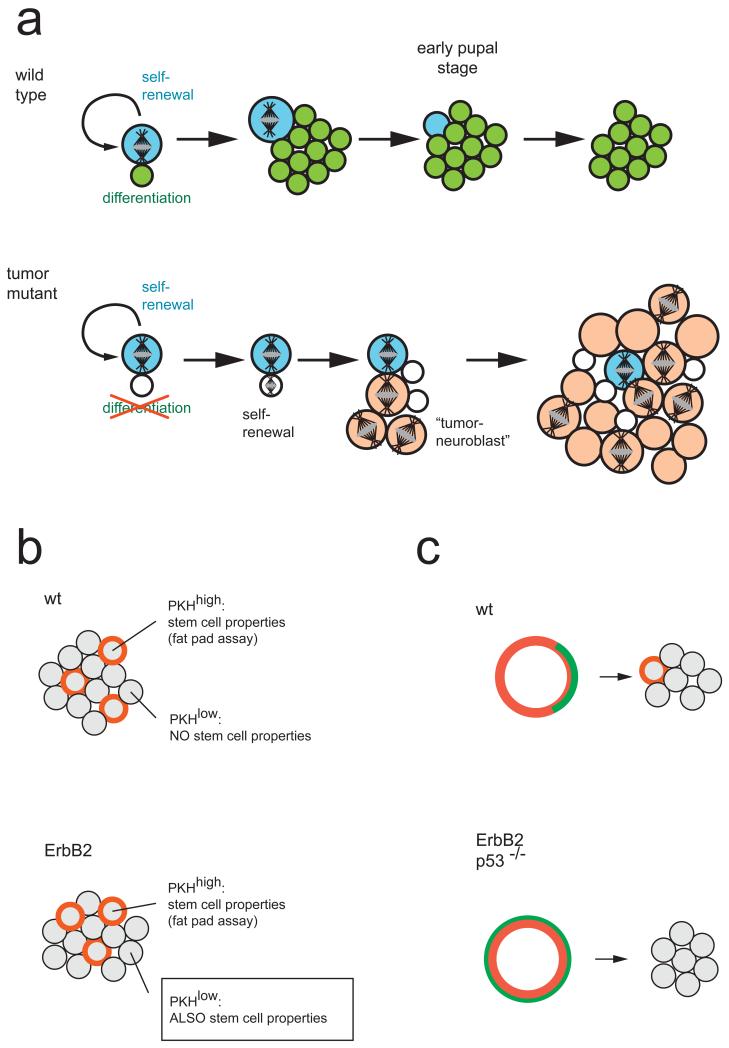
a ∣ Wild-type Drosophila neuroblasts generate one small self renewing (blue) and one large differentiating (green) daughter cell. The differentiating daughter cell exits the cell cycle after a terminal division (not shown). The neuroblast shrinks during pupal stages and undergoes apoptosis. In mutants affecting asymmetric cell division, the smaller daughter cell (white) cannot differentiate. After some time, it undergoes mitosis and reverts back to a tumour neuroblast (orange). These tumuor neuroblasts are abnormal because they do not exit the cell cycle during pupal stages. Whether or not the original neuroblast (blue) disappears or also continues to proliferate is unclear.
b ∣ Mammospheres grown from wild-type mammary gland tissue or ErbB2 mutant tumour tissue contain the same number of slowly proliferating (PKH-26 dye (red) retaining) cells. In wild-type tissue, only the cells retaining the dye can form secondary mammospheres, but in tumour tissue, mammospheres can be grown from any cell.
c ∣ PKH-26 dye-retaining cells can be purified by FACS sorting. In wild-type tissue, these cells localize Numb asymmetrically (green). When cultured, one dye-retaining cell remains, indicating that the initial division was asymmetric. When isolated from a ErbB2 tumour model or p53-mutant mice, PKH-26high cells do not divide asymmetrically and all daughter cells lose the dye, indicating that the initial division was symmetric.
Formation of tumour neuroblasts in mutants affecting asymmetric cell division can be explained by genetic or epigenetic defects or by the intrinsic properties of the growth control mechanism. In the genetic model, DNA mutations are responsible for immortalizing neuroblasts; however, although aneuploidy does occur in transplanted neuroblast tumours 104 and may be responsible for their metastatic behaviour 112, it has not been described in primary tumours. As mutations causing genome instability do not result in brain tumours 111, it is more likely that transcriptional and/or epigenetic changes alter the behaviour of the mutant neuroblasts. The transcriptional network governing self-renewal in neuroblasts needs to be reprogrammed towards a stable and irreversible differentiation state after asymmetric cell division. Defects in this process could create a new stable state in which the self-renewal program is active but the modules controlling the exit from proliferation are missing. For example, neuroblasts serially express different transcription factors at different developmental stages 113, and a reset of this developmental timer during each defective asymmetric division could explain immortalization. Finally, it is possible that the growth control mechanism acting in pupae can only deal with a limited number of neuroblasts, for example because a growth inhibitor is limiting or because neuroblasts secret an autocrine growth promnoting factor that competes with a systemic extrinsic factor. Several redundant mechanisms have been proposed to stop neuroblast proliferation in wild-type flies. In the abdomen of the ventral nerve chord, transient expression of the homeotic gene abdominal A eliminates neuroblasts by inducing apoptotic cell death 114. In the central brain, a decrease in Insulin and PI3 kinase signalling causes a reduction of neuroblast size followed by caspase-mediated cell death during the pupal stages of development 115. When caspase activation is prevented, neuroblast size is still reduced, and the cells are eliminated by a caspase-independent autophagic pathway that is regulated by the transcription factor Foxo. When both caspases and Foxo are inhibited, neuroblasts continue to proliferate and generate functional neurons even in adult flies. Surprisingly, however, this does not result in a tumour, indicating that both an increase in neuroblast number and inhibition of the elimination pathways contribute to tumour formation.
Clearly, identifying the molecular events connecting asymmetric cell division to cellular immortalization is one of the greatest new challenges in the field. This is particularly important because defects in asymmetric cell division are relevant for human tumorigenesis as well 116 (see below) 117,118 and may be part of the mechanisms that convert a normal mammalian stem cell into what is known as a cancer stem cell (box 1).
Box 1. Tumour stem cells.
The tumor stem cell hypothesis claims that tumors contain a rare population of cells that have stem cell properties and are the only tumor cells capable of generating all other cell types in the tumor. The hypothesis 116 is based on xenotransplantation experiments in which transplantation of human tumours into immunocompromized mice recapitulates the human tumour histology. It had long been known that only few cells in a tumour can initiate tumour formation in those transplantation experiments. In the 1990s, John Dick’s laboratory demonstrated for leukaemia that these cells express stem cell markers and that tumour formation involves a cellular hierarchy not unlike the one in normal haematopoiesis 138,139. These experiments formed the basis of the tumor stem cell hypothesis and soon after, similar experiments identified tumour stem cells in brain and mammary tumours 140,141 and in almost any other type of human cancer 142. Whether or not the formation of cancer stem cells is an intrinsic property of tumorigenesis is intensly debated 143. Opponents argue that they are simply a subtype of human tumour cells that adapt more easily to the environment of the mouse host. It is particularly controversial whether human tumours actually arise from stem cells. In mouse models, intestinal cancer can be induced by mutating the APC tumour suppressor in stem cells but not in non-stem cell types 144. In a mouse glioblastoma model, tumour formation coincides with the appearance of abnormal stem cell populations 145. These results suggest that DNA mutations in stem cells might be the initial event in those tumours. Experiments in Drosophila melanogaster, in which this hypothesis can be stringently tested might shed light on the mechanisms that cause stem cells to become malignant.
Tumorigenesis and mammalian stem cells
The ability to generate both self-renewing and differentiating daughter cells is a defining feature of any stem cell, and asymmetric cell division is one of the mechanisms used to establish this. Evidence for asymmetric cell division exists for stem cells in the developing brain 98,119, in muscles 120, the skin 121, the gut 122, mammary glands 117 and the haematopoietic system 118. Nevertheless, the mechanisms that guide these asymmetric cell divisions are generally not well understood (box 2). In fact, transferring our detailed understanding of the process from D. melanogaster and C. elegans to vertebrates has been much more challenging than expected. Almost all molecular players are conserved in vertebrates but they often act in distinct ways. Numb, for example, is polarized in neural progenitors, but this is because it regulates trafficking of E-cadherin at adherens junctions 123. Par proteins are apical, but unlike in neuroblasts, only few progenitor divisions are aligned along the axis of Par protein polarity 124. Nevertheless, building on the results from flies and worms, some exciting connections between asymmetric cell division and tumorigenesis have recently been identified.
Box 2. Asymmetric cell division in vertebrates.
Almost all the molecules regulating asymmetric cell division in D. melanogaster and C. elegans are conserved in vertebrates. Similarly to those in invertebrate model organisms, Par-3, Par-6 and aPKC act together to establish cell polarity in vertebrates 7. Pins, Gα and NuMA (the Lin-5/Mud homolog) regulate spindle orientation and microtubule-cortex association 132 and Numb controls endocytosis 133. Nevertheless, their function during asymmetric cell division in vertebrates is not clear.
Asymmetric cell division in neural progenitor cells is the best understood asymmetric cell division process in mammals 134. After an initial expansion phase by symmetric division, progenitors undergo asymmetric divisions, giving rise to two daughter cells: one progenitor cell and one cell that either differentiates into a neuron or becomes an intermediate progenitor, which forms two neurons after a terminal symmetric division. Progenitors are located in the apical side of the neuroepithelium, where Par proteins accumulate in the apical cortex. Unlike in D. melanogaster, however, the apical membrane domain in dividing progenitors is very narrow, and even slight twists of the cleavage plane lead to their asymmetric inheritance 124. Numb is expressed by the progenitors and concentrates on apical adherens junctions and on the basolateral plasma membrane. This has led to a model in which the asymmetric inheritance of PAR 3 during oblique divisions (divisions occuring at ~45° angle) inactivates Numb in one of the two daughter cells so that it no longer inhibits Notch and two daughters with unequal signalling levels are formed 135. As PAR 3 is a key factor promoting Numb phosphorylation by aPKC in D. melanogaster 76, it is possible that, in vertebrates, differential phosphorylation of Numb might be responsible for different activity in the two daughter cells. In addition to this Par protein-mediated asymmetry, the asymmetric inheritance of apical and basal processes 136, the polarized localization of the Brat homolog TRIM3 137 and extracellular signals might have a role in establishing asymmetry.
Stem cells from mouse mammary glands can grow into spherical cultures known as mammospheres that recapitulate the mammary morphogenic programme 125. The stem cells can be isolated following labelling with a lipophilic vital dye, as they retain the dye whereas dividing cells do not (Fig. 5b) 117. When purified from wild-type mammary glands, these stem cells divide asymmetrically and segregate Numb into one of their two daughter cells (Fig. 5c). In a mouse mammary tumour model, the number of stem cells is increased (Fig. 5b) and they divide symmetrically – Numb is no longer asymmetrically localized and both daughter cells behave identically in terms of dye dilution (Fig. 5c). Similar observations have been made in p53-mutant mice. As p53 degradation is regulated by Numb 126, it is possible that the asymmetric inheritance of Numb regulates p53 levels and restricts stem cell fate to only one of the two daughter cells. Consistent with this, Numb is a major tumour suppressor in breast cancer 127. Numb also acts in the haematopoietic system, where it can inhibit the progression of chronic myeloid leukaemia (CML). Cultured hematopoietic progenitors normally divide and segregate Numb asymmetrically, but their divisions become symmetric when the oncogene NUP98-HOXA9 is expressed 118. NUP98-HOXA9 induces the expression of Musashi 2, which in turn inhibits Numb, potentially triggering the enormous expansion of undifferentiated progenitors in advanced stage CML 128. Thus, the conserved connection with tumorigenesis establishes an unprecedented clinical relevance for research on asymmetric cell division.
Open questions and future challenges
The progress in our understanding of asymmetric cell division during the past 10 years has been enormous. We have learned that the phosphorylation of cell fate determinants by asymmetrically distributed kinases is the driving force for their asymmetric localization whereas polarized transport seems to have a minor role. It has become clear that microtubules, which were originally thought not to be involved in asymmetric cell division, mediate essential interactions between centrosomes and the cell cortex. These interactions maintain the polarity axis over multiple divisions and guide asymmetric protein localization during late mitosis. An exciting connection between asymmetric cell division and tumorigenesis has emerged in flies, mice and humans, which has given rise to major challenges, in part because of our still incomplete understanding of asymmetric cell division in vertebrates. In addition, newly emerging technologies lay the ground for a systems-level understanding of the process.
Although we have learned the basic principles of asymmetric determinant segregation, our understanding of the cell fate choices that are influenced by those determinants is limited. We know that Numb acts on Notch, that Prospero is a transcription factor and that Brat regulates post-transcriptional events, but how these factors cooperate to prevent self-renewal is unclear. In fact, we do not understand the transcriptional network that governs and maintains self-renewal in D. melanogaster neuroblasts. We also do not know how the initial bias on this network is stabilized over time resulting in a daughter cell that terminally exits proliferation. And in particular, we do not know how defects in fate specification result in the formation of misguided tumour-initiating cells.
The answers to these questions may come from the spectacular technological advances in the field. The establishment of genome-wide transgenic RNA interference libraries in flies 129 allows us to test gene functions at an unprecedented speed and on a near genome-wide level 130. In addition, the development of new sequencing technologies has opened new dimensions for genome-wide profiling of transcription, RNA splicing and chromatin association 131. It is likely that these technologies will establish D. melanogaster neuroblasts as one of the best model systems for the establishment and stabilization of cell fate choices and shed light on the mechanisms of stem cell-derived tumour formation. The potential clinical relevance of those findings will be a strong motivation to embark on those difficult tasks.
Supplementary Material
Suppl. Movie 1 ∣ Asymmetric segregation of cell fate determinats in mitosis.
The movie shows a dividing sensory organ precursor cell in the Drosophila notum. Histone-RFP (red) and Pon-GFP (green) are driven by neuralized-Gal4.
Suppl. Figure 1 ∣ Cell type specific markers in neuroblast lineages.
Expression of the markers Asense, Deadpan, Prospero and Elav distinguishes cell types in D. melanogaster larval neuroblast lineages. Deadpan is expressed in all self-renewing cells (Type I and type II neuroblasts and INPs) and its expression disappears from the small daughter cell immediately after division. Asense is expressed by type I but not type II neuroblasts and in INPs. Its expression persists for a while after asymmetric division. Prospero is expressed in the daughter cells of type I but not type II neuroblasts. It is also expressed in the small daughter cells of INPs and its expression persists in neurons for a while. Note that Prospero expression is weaker immediately after asymmetric division and becomes stronger in neurons. Elav protein is a marker for terminally differentiating neurons although Elav RNA is more widely expressed.
Glossary terms
- Neuroblast
D. melanogaster neural progenitor cell generating all neurons and glia cells in the brain
- Mushroom body
A mushroom shaped paired neuropil structure found in the Drosophila melanogaster brain that functions in learning and memory.
- Optic lobe
morphologically distinct part of the developing Drosophila brain forming the visual processing centers
- P-granules
Ribonucleoprotein particles that segregate with and mark all cells of the C. elegant germline
- Centrosome
Also called the microtubule-organizing centre (MTOC) or spindle pole, this structure nucleates microtubules and is important for signalling processes.
- Centriole
A small organelle consisting of two short, barrel-like arrays of microtubules that organize the centrosome and contributes to cytokinesis and cell-cycle progression.
Author biography
Juergen Knoblich has been a senior scientist at IMBA (the Institute of Molecular Biotechnology of the Austrian Academy of Sciences) in Vienna since 2004 and is currently deputy director of the institute. Before this, he has been a group leader at the neighbouring IMP (Institute of Molecular Pathology) from 1997 to 2003. He carried out his doctoral work with Christian Lehner at the Max Planck Institute in Tübingen and worked as a postdoc with Yuh Nung Jan at the University of California, San Francisco (UCSF). His laboratory uses both Drosophila and mouse models to study the biology of asymmetric cell division and tumorigenesis in stem cell lineages.
References
- 1.Conklin EG. The organization and cell-lineage of the ascidian egg. J. Acad. Nat. Sci. Philadelphia. 1905;13:1–119. [Google Scholar]
- 2.Rhyu MS, Jan LY, Jan YN. Asymmetric distribution of numb protein during division of the sensory organ precursor cell confers distinct fates to daughter cells. Cell. 1994;76:477–491. doi: 10.1016/0092-8674(94)90112-0. [DOI] [PubMed] [Google Scholar]
- 3.Spana EP, Kopczynski C, Goodman CS, Doe CQ. Asymmetric localization of numb autonomously determines sibling neuron identity in the Drosophila CNS. Development. 1995;121:3489–3494. doi: 10.1242/dev.121.11.3489. [DOI] [PubMed] [Google Scholar]
- 4.Uemura T, Shepherd S, Ackerman L, Jan LY, Jan YN. numb, a gene required in determination of cell fate during sensory organ formation in Drosophila embryos. Cell. 1989;58:349–360. doi: 10.1016/0092-8674(89)90849-0. [DOI] [PubMed] [Google Scholar]
- 5.Kemphues KJ, Priess JR, Morton DG, Cheng NS. Identification of genes required for cytoplasmic localization in early C. elegans embryos. Cell. 1988;52:311–320. doi: 10.1016/s0092-8674(88)80024-2. [DOI] [PubMed] [Google Scholar]
- 6.Ohno S. Intercellular junctions and cellular polarity: the PAR-aPKC complex, a conserved core cassette playing fundamental roles in cell polarity. Curr Opin Cell Biol. 2001;13:641–68. doi: 10.1016/s0955-0674(00)00264-7. [DOI] [PubMed] [Google Scholar]
- 7.Suzuki A, Ohno S. The PAR-aPKC system: lessons in polarity. J Cell Sci. 2006;119:979–987. doi: 10.1242/jcs.02898. [DOI] [PubMed] [Google Scholar]
- 8.Etemad-Moghadam B, Guo S, Kemphues KJ. Asymmetrically distributed PAR-3 protein contributes to cell polarity and spindle alignment in early C. elegans embryos. Cell. 1995;83:743–752. doi: 10.1016/0092-8674(95)90187-6. [DOI] [PubMed] [Google Scholar]
- 9.Hung TJ, Kemphues KJ. PAR-6 is a conserved PDZ domain-containing protein that colocalizes with PAR-3 in Caenorhabditis elegans embryos. Development. 1999;126:127–135. doi: 10.1242/dev.126.1.127. [DOI] [PubMed] [Google Scholar]
- 10.Tabuse Y, et al. Atypical protein kinase C cooperates with PAR-3 to establish embryonic polarity in Caenorhabditis elegans. Development. 1998;125:3607–3614. doi: 10.1242/dev.125.18.3607. [DOI] [PubMed] [Google Scholar]
- 11.Guo S, Kemphues KJ. par-1, a gene required for establishing polarity in C. elegans embryos, encodes a putative Ser/Thr kinase that is asymmetrically distributed. Cell. 1995;81:611–620. doi: 10.1016/0092-8674(95)90082-9. [DOI] [PubMed] [Google Scholar]
- 12.Boyd L, Guo S, Levitan D, Stinchcomb DT, Kemphues KJ. PAR-2 is asymmetrically distributed and promotes association of P granules and PAR-1 with the cortex in C. elegans embryos. Development. 1996;122:3075–3084. doi: 10.1242/dev.122.10.3075. [DOI] [PubMed] [Google Scholar]
- 13.Wodarz A, Ramrath A, Kuchinke U, Knust E. Bazooka provides an apical cue for Inscuteable localization in Drosophila neuroblasts. Nature. 1999;402:544–547. doi: 10.1038/990128. [DOI] [PubMed] [Google Scholar]
- 14.Schober M, Schaefer M, Knoblich JA. Bazooka recruits Inscuteable to orient asymmetric cell divisions in Drosophila neuroblasts. Nature. 1999;402:548–551. doi: 10.1038/990135. [DOI] [PubMed] [Google Scholar]
- 15.Petronczki M, Knoblich JA. DmPAR-6 directs epithelial polarity and asymmetric cell division of neuroblasts in Drosophila. Nat Cell Biol. 2001;3:43–49. doi: 10.1038/35050550. [DOI] [PubMed] [Google Scholar]
- 16.Wodarz A, Ramrath A, Grimm A, Knust E. Drosophila atypical protein kinase C associates with Bazooka and controls polarity of epithelia and neuroblasts. J Cell Biol. 2000;150:1361–1374. doi: 10.1083/jcb.150.6.1361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rolls MM, Albertson R, Shih HP, Lee CY, Doe CQ. Drosophila aPKC regulates cell polarity and cell proliferation in neuroblasts and epithelia. J Cell Biol. 2003;163:1089–1098. doi: 10.1083/jcb.200306079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Knoblich JA. Asymmetric cell division during animal development. Nat Rev Mol Cell Biol. 2001;2:11–20. doi: 10.1038/35048085. [DOI] [PubMed] [Google Scholar]
- 19.Ito K, Hotta Y. Proliferation pattern of postembryonic neuroblasts in the brain of Drosophila melanogaster. Dev Biol. 1992;149:134–148. doi: 10.1016/0012-1606(92)90270-q. [DOI] [PubMed] [Google Scholar]
- 20.Bowman SK, et al. The tumor suppressors Brat and Numb regulate transit-amplifying neuroblast lineages in Drosophila. Dev Cell. 2008;14:535–546. doi: 10.1016/j.devcel.2008.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Boone JQ, Doe CQ. Identification of Drosophila type II neuroblast lineages containing transit amplifying ganglion mother cells. Dev Neurobiol. 2008;68:1185–1195. doi: 10.1002/dneu.20648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bello BC, Izergina N, Caussinus E, Reichert H. Amplification of neural stem cell proliferation by intermediate progenitor cells in Drosophila brain development. Neural Develop. 2008;3:5. doi: 10.1186/1749-8104-3-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ito K, Awano W, Suzuki K, Hiromi Y, Yamamoto D. The Drosophila mushroom body is a quadruple structure of clonal units each of which contains a virtually identical set of neurones and glial cells. Development. 1997;124:761–771. doi: 10.1242/dev.124.4.761. [DOI] [PubMed] [Google Scholar]
- 24.Egger B, Boone JQ, Stevens NR, Brand AH, Doe CQ. Regulation of spindle orientation and neural stem cell fate in the Drosophila optic lobe. Neural Develop. 2007;2:1. doi: 10.1186/1749-8104-2-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Neumüller RA, Knoblich JA. Dividing cellular asymmetry: asymmetric cell division and its implications for stem cells and cancer. Genes Dev. 2009;23:2675–2699. doi: 10.1101/gad.1850809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Knoblich JA. Mechanisms of asymmetric stem cell division. Cell. 2008;132:583–597. doi: 10.1016/j.cell.2008.02.007. [DOI] [PubMed] [Google Scholar]
- 27.Wu PS, Egger B, Brand AH. Asymmetric stem cell division: Lessons from Drosophila. Semin Cell Dev Biol. 2008;19:283–293. doi: 10.1016/j.semcdb.2008.01.007. [DOI] [PubMed] [Google Scholar]
- 28.Doe CQ. Neural stem cells: balancing self-renewal with differentiation. Development. 2008;135:1575–1587. doi: 10.1242/dev.014977. [DOI] [PubMed] [Google Scholar]
- 29.Berdnik D, Torok T, Gonzalez-Gaitan M, Knoblich JA. The endocytic protein alpha-Adaptin is required for numb-mediated asymmetric cell division in Drosophila. Dev Cell. 2002;3:221–231. doi: 10.1016/s1534-5807(02)00215-0. [DOI] [PubMed] [Google Scholar]
- 30.Sonoda J, Wharton RP. Drosophila Brain Tumor is a translational repressor. Genes Dev. 2001;15:762–773. doi: 10.1101/gad.870801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lee CY, Wilkinson BD, Siegrist SE, Wharton RP, Doe CQ. Brat Is a Miranda Cargo Protein that Promotes Neuronal Differentiation and Inhibits Neuroblast Self-Renewal. Dev Cell. 2006;10:441–449. doi: 10.1016/j.devcel.2006.01.017. [DOI] [PubMed] [Google Scholar]
- 32.Bello B, Reichert H, Hirth F. The brain tumor gene negatively regulates neural progenitor cell proliferation in the larval central brain of Drosophila. Development. 2006;133:2639–2648. doi: 10.1242/dev.02429. [DOI] [PubMed] [Google Scholar]
- 33.Betschinger J, Mechtler K, Knoblich JA. Asymmetric segregation of the tumor suppressor brat regulates self-renewal in Drosophila neural stem cells. Cell. 2006;124:1241–1253. doi: 10.1016/j.cell.2006.01.038. [DOI] [PubMed] [Google Scholar]
- 34.Lu B, Rothenberg M, Jan LY, Jan YN. Partner of Numb colocalizes with Numb during mitosis and directs Numb asymmetric localization in Drosophila neural and muscle progenitors. Cell. 1998;95:225–235. doi: 10.1016/s0092-8674(00)81753-5. [DOI] [PubMed] [Google Scholar]
- 35.Wang H, Ouyang Y, Somers WG, Chia W, Lu B. Polo inhibits progenitor self-renewal and regulates Numb asymmetry by phosphorylating Pon. Nature. 2007;449:96–100. doi: 10.1038/nature06056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Knoblich JA, Jan LY, Jan YN. Asymmetric segregation of Numb and Prospero during cell division. Nature. 1995;377:624–627. doi: 10.1038/377624a0. [DOI] [PubMed] [Google Scholar]
- 37.Spana EP, Doe CQ. The prospero transcription factor is asymmetrically localized to the cell cortex during neuroblast mitosis in Drosophila. Development. 1995;121:3187–3195. doi: 10.1242/dev.121.10.3187. [DOI] [PubMed] [Google Scholar]
- 38.Shen CP, Jan LY, Jan YN. Miranda is required for the asymmetric localization of Prospero during mitosis in Drosophila. Cell. 1997;90:449–458. doi: 10.1016/s0092-8674(00)80505-x. [DOI] [PubMed] [Google Scholar]
- 39.Ikeshima-Kataoka H, Skeath JB, Nabeshima Y, Doe CQ, Matsuzaki F. Miranda directs Prospero to a daughter cell during Drosophila asymmetric divisions. Nature. 1997;390:625–629. doi: 10.1038/37641. [DOI] [PubMed] [Google Scholar]
- 40.Matsuzaki F, Ohshiro T, Ikeshima-Kataoka H, Izumi H. miranda localizes staufen and prospero asymmetrically in mitotic neuroblasts and epithelial cells in early Drosophila embryogenesis. Development. 1998;125:4089–4098. doi: 10.1242/dev.125.20.4089. [DOI] [PubMed] [Google Scholar]
- 41.Kraut R, Campos-Ortega JA. inscuteable, a neural precursor gene of Drosophila, encodes a candidate for a cytoskeleton adaptor protein. Dev Biol. 1996;174:65–81. doi: 10.1006/dbio.1996.0052. [DOI] [PubMed] [Google Scholar]
- 42.Kraut R, Chia W, Jan LY, Jan YN, Knoblich JA. Role of inscuteable in orienting asymmetric cell divisions in Drosophila. Nature. 1996;383:50–55. doi: 10.1038/383050a0. [DOI] [PubMed] [Google Scholar]
- 43.Schaefer M, Petronczki M, Dorner D, Forte M, Knoblich JA. Heterotrimeric G proteins direct two modes of asymmetric cell division in the Drosophila nervous system. Cell. 2001;107:183–194. doi: 10.1016/s0092-8674(01)00521-9. [DOI] [PubMed] [Google Scholar]
- 44.Schaefer M, Shevchenko A, Shevchenko A, Knoblich JA. A protein complex containing Inscuteable and the Galpha-binding protein Pins orients asymmetric cell divisions in Drosophila. Curr Biol. 2000;10:353–362. doi: 10.1016/s0960-9822(00)00401-2. [DOI] [PubMed] [Google Scholar]
- 45.Yu F, Morin X, Cai Y, Yang X, Chia W. Analysis of partner of inscuteable, a novel player of Drosophila asymmetric divisions, reveals two distinct steps in inscuteable apical localization. Cell. 2000;100:399–409. doi: 10.1016/s0092-8674(00)80676-5. [DOI] [PubMed] [Google Scholar]
- 46.Siller KH, Cabernard C, Doe CQ. The NuMA-related Mud protein binds Pins and regulates spindle orientation in Drosophila neuroblasts. Nat Cell Biol. 2006;8:594–600. doi: 10.1038/ncb1412. [DOI] [PubMed] [Google Scholar]
- 47.Izumi Y, Ohta N, Hisata K, Raabe T, Matsuzaki F. Drosophila Pins-binding protein Mud regulates spindle-polarity coupling and centrosome organization. Nat Cell Biol. 2006;8:586–593. doi: 10.1038/ncb1409. [DOI] [PubMed] [Google Scholar]
- 48.Bowman SK, Neumuller RA, Novatchkova M, Du Q, Knoblich JA. The Drosophila NuMA Homolog Mud Regulates Spindle Orientation in Asymmetric Cell Division. Dev Cell. 2006;10:731–742. doi: 10.1016/j.devcel.2006.05.005. [DOI] [PubMed] [Google Scholar]
- 49.Gonczy P. Mechanisms of asymmetric cell division: flies and worms pave the way. Nat Rev Mol Cell Biol. 2008;9:355–366. doi: 10.1038/nrm2388. [DOI] [PubMed] [Google Scholar]
- 50.Cowan CR, Hyman AA. Asymmetric Cell Division in C. elegans: Cortical Polarity and Spindle Positioning. Annu Rev Cell Dev Biol. 2004;20:427–453. doi: 10.1146/annurev.cellbio.19.111301.113823. [DOI] [PubMed] [Google Scholar]
- 51.Munro E, Nance J, Priess JR. Cortical Flows Powered by Asymmetrical Contraction Transport PAR Proteins to Establish and Maintain Anterior-Posterior Polarity in the Early C. elegans Embryo. Dev Cell. 2004;7:413–424. doi: 10.1016/j.devcel.2004.08.001. [DOI] [PubMed] [Google Scholar]
- 52.Cowan CR, Hyman AA. Centrosomes direct cell polarity independently of microtubule assembly in C. elegans embryos. Nature. 2004;431:92–96. doi: 10.1038/nature02825. [DOI] [PubMed] [Google Scholar]
- 53.Tsai MC, Ahringer J. Microtubules are involved in anterior-posterior axis formation in C. elegans embryos. J Cell Biol. 2007;179:397–402. doi: 10.1083/jcb.200708101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Jenkins N, Saam JR, Mango SE. CYK-4/GAP provides a localized cue to initiate anteroposterior polarity upon fertilization. Science. 2006;313:1298–1301. doi: 10.1126/science.1130291. [DOI] [PubMed] [Google Scholar]
- 55.Zonies S, Motegi F, Hao Y, Seydoux G. Symmetry breaking and polarization of the C. elegans zygote by the polarity protein PAR-2. Development. 2010;137:1669–1677. doi: 10.1242/dev.045823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hao Y, Boyd L, Seydoux G. Stabilization of Cell Polarity by the C. elegans RING Protein PAR-2. Dev Cell. 2006;10:199–208. doi: 10.1016/j.devcel.2005.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Gonczy P, Rose LS. Asymmetric cell division and axis formation in the embryo. WormBook. 2005:1–20. doi: 10.1895/wormbook.1.30.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Mello CC, et al. The PIE-1 protein and germline specification in C. elegans embryos. Nature. 1996;382:710–712. doi: 10.1038/382710a0. [DOI] [PubMed] [Google Scholar]
- 59.Zhang F, Barboric M, Blackwell TK, Peterlin BM. A model of repression: CTD analogs and PIE-1 inhibit transcriptional elongation by P-TEFb. Genes Dev. 2003;17:748–758. doi: 10.1101/gad.1068203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Mello CC, Draper BW, Krause M, Weintraub H, Priess JR. The pie-1 and mex-1 genes and maternal control of blastomere identity in early C. elegans embryos. Cell. 1992;7:10. doi: 10.1016/0092-8674(92)90542-k. [DOI] [PubMed] [Google Scholar]
- 61.Schubert CM, Lin R, de Vries CJ, Plasterk RH, Priess JR. MEX-5 and MEX-6 function to establish soma/germline asymmetry in early C. elegans embryos. Mol Cell. 2000;5:671–682. doi: 10.1016/s1097-2765(00)80246-4. [DOI] [PubMed] [Google Scholar]
- 62.Draper BW, Mello CC, Bowerman B, Hardin J, Priess JR. MEX-3 is a KH domain protein that regulates blastomere identity in early C. elegans embryos. Cell. 1996;87:205–216. doi: 10.1016/s0092-8674(00)81339-2. [DOI] [PubMed] [Google Scholar]
- 63.Cuenca AA, Schetter A, Aceto D, Kemphues K, Seydoux G. Polarization of the C. elegans zygote proceeds via distinct establishment and maintenance phases. Development. 2003;130:1255–1265. doi: 10.1242/dev.00284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Broadus J, Doe CQ. Extrinsic cues, intrinsic cues and microfilaments regulate asymmetric protein localization in Drosophila neuroblasts. Curr Biol. 1997;7:827–835. doi: 10.1016/s0960-9822(06)00370-8. [DOI] [PubMed] [Google Scholar]
- 65.Knoblich JA, Jan LY, Jan YN. The N terminus of the Drosophila Numb protein directs membrane association and actin-dependent asymmetric localization. Proc Natl Acad Sci U S A. 1997;94:13005–13010. doi: 10.1073/pnas.94.24.13005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Shen CP, et al. Miranda as a multidomain adapter linking apically localized Inscuteable and basally localized Staufen and Prospero during asymmetric cell division in Drosophila. Genes Dev. 1998;12:1837–1846. doi: 10.1101/gad.12.12.1837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Jan YN, Jan LY. Asymmetric cell division in the Drosophila nervous system. Nat Rev Neurosci. 2001;2:772–779. doi: 10.1038/35097516. [DOI] [PubMed] [Google Scholar]
- 68.Petritsch C, Tavosanis G, Turck CW, Jan LY, Jan YN. The Drosophila Myosin VI Jaguar Is Required for Basal Protein Targeting and Correct Spindle Orientation in Mitotic Neuroblasts. Dev Cell. 2003;4:273–281. doi: 10.1016/s1534-5807(03)00020-0. [DOI] [PubMed] [Google Scholar]
- 69.Ohshiro T, Yagami T, Zhang C, Matsuzaki F. Role of cortical tumour-suppressor proteins in asymmetric division of Drosophila neuroblast. Nature. 2000;408:593–56. doi: 10.1038/35046087. [DOI] [PubMed] [Google Scholar]
- 70.Peng CY, Manning L, Albertson R, Doe CQ. The tumour-suppressor genes lgl and dlg regulate basal protein targeting in Drosophila neuroblasts. Nature. 2000;408:596–600. doi: 10.1038/35046094. [DOI] [PubMed] [Google Scholar]
- 71.Strand D, et al. The Drosophila lethal(2)giant larvae tumor suppressor protein forms homo-oligomers and is associated with nonmuscle myosin II heavy chain. J Cell Biol. 1994;127:1361–1373. doi: 10.1083/jcb.127.5.1361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Strand D, Raska I, Mechler BM. The Drosophila lethal(2)giant larvae tumor suppressor protein is a component of the cytoskeleton. J Cell Biol. 1994;127:1345–1360. doi: 10.1083/jcb.127.5.1345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Betschinger J, Mechtler K, Knoblich JA. The Par complex directs asymmetric cell division by phosphorylating the cytoskeletal protein Lgl. Nature. 2003;422:326–330. doi: 10.1038/nature01486. [DOI] [PubMed] [Google Scholar]
- 74.Betschinger J, Eisenhaber F, Knoblich JA. Phosphorylation-induced autoinhibition regulates the cytoskeletal protein Lethal (2) giant larvae. Curr Biol. 2005;15:276–282. doi: 10.1016/j.cub.2005.01.012. [DOI] [PubMed] [Google Scholar]
- 75.Barros CS, Phelps CB, Brand AH. Drosophila Nonmuscle Myosin II Promotes the Asymmetric Segregation of Cell Fate Determinants by Cortical Exclusion Rather Than Active Transport. Dev Cell. 2003;5:829–840. doi: 10.1016/s1534-5807(03)00359-9. [DOI] [PubMed] [Google Scholar]
- 76.Wirtz-Peitz F, Nishimura T, Knoblich JA. Linking cell cycle to asymmetric division: Aurora-A phosphorylates the Par complex to regulate Numb localization. Cell. 2008;135:161–173. doi: 10.1016/j.cell.2008.07.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Cabernard C, Prehoda KE, Doe CQ. A spindle-independent cleavage furrow positioning pathway. Nature. 2010;467:91–94. doi: 10.1038/nature09334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Atwood SX, Prehoda KE. aPKC Phosphorylates Miranda to Polarize Fate Determinants during Neuroblast Asymmetric Cell Division. Curr Biol. 2009;19:723–729. doi: 10.1016/j.cub.2009.03.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lu B, Ackerman L, Jan LY, Jan YN. Modes of protein movement that lead to the asymmetric localization of partner of Numb during Drosophila neuroblast division. Mol Cell. 1999;4:883–891. doi: 10.1016/s1097-2765(00)80218-x. [DOI] [PubMed] [Google Scholar]
- 80.Mayer B, Emery G, Berdnik D, Wirtz-Peitz F, Knoblich JA. Quantitative Analysis of Protein Dynamics during Asymmetric Cell Division. Curr Biol. 2005;15:1847–1854. doi: 10.1016/j.cub.2005.08.067. [DOI] [PubMed] [Google Scholar]
- 81.Smith CA, et al. aPKC-mediated phosphorylation regulates asymmetric membrane localization of the cell fate determinant Numb. EMBO J. 2007;26:468–480. doi: 10.1038/sj.emboj.7601495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Yamanaka T, et al. Mammalian Lgl Forms a Protein Complex with PAR-6 and aPKC Independently of PAR-3 to Regulate Epithelial Cell Polarity. Curr Biol. 2003;13:734–743. doi: 10.1016/s0960-9822(03)00244-6. [DOI] [PubMed] [Google Scholar]
- 83.Skwarek LC, Garroni MK, Commisso C, Boulianne GL. Neuralized contains a phosphoinositide-binding motif required downstream of ubiquitination for delta endocytosis and notch signaling. Dev Cell. 2007;13:783–795. doi: 10.1016/j.devcel.2007.10.020. [DOI] [PubMed] [Google Scholar]
- 84.Cabernard C, Doe CQ. Apical/basal spindle orientation is required for neuroblast homeostasis and neuronal differentiation in Drosophila. Dev Cell. 2009;17:134–141. doi: 10.1016/j.devcel.2009.06.009. [DOI] [PubMed] [Google Scholar]
- 85.Siegrist SE, Doe CQ. Microtubule-induced cortical cell polarity. Genes Dev. 2007;21:483–496. doi: 10.1101/gad.1511207. [DOI] [PubMed] [Google Scholar]
- 86.Siegrist SE, Doe CQ. Microtubule-induced pins/galphai cortical polarity in Drosophila neuroblasts. Cell. 2005;123:1323–1335. doi: 10.1016/j.cell.2005.09.043. [DOI] [PubMed] [Google Scholar]
- 87.DeRenzo C, Reese KJ, Seydoux G. Exclusion of germ plasm proteins from somatic lineages by cullin-dependent degradation. Nature. 2003;424:685–689. doi: 10.1038/nature01887.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Daniels BR, Perkins EM, Dobrowsky TM, Sun SX, Wirtz D. Asymmetric enrichment of PIE-1 in the Caenorhabditis elegans zygote mediated by binary counterdiffusion. J Cell Biol. 2009;184:473–479. doi: 10.1083/jcb.200809077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Tenlen JR, Molk JN, London N, Page BD, Priess JR. MEX-5 asymmetry in one-cell C. elegans embryos requires PAR-4- and PAR-1-dependent phosphorylation. Development. 2008;135:3665–3675. doi: 10.1242/dev.027060. [DOI] [PubMed] [Google Scholar]
- 90.Daniels BR, Dobrowsky TM, Perkins EM, Sun SX, Wirtz D. MEX-5 enrichment in the C. elegans early embryo mediated by differential diffusion. Development. 2010;137:2579–2585. doi: 10.1242/dev.051326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Turing A. The chemical basis of morphogenesis. Phil. Trans R. Soc. Lond. 1952;B327:37–72. [Google Scholar]
- 92.Kondo S. The reaction-diffusion system: a mechanism for autonomous pattern formation in the animal skin. Genes Cells. 2002;7:535–541. doi: 10.1046/j.1365-2443.2002.00543.x. [DOI] [PubMed] [Google Scholar]
- 93.Kaltschmidt JA, Davidson CM, Brown NH, Brand AH. Rotation and asymmetry of the mitotic spindle direct asymmetric cell division in the developing central nervous system. Nat Cell Biol. 2000;2:7–12. doi: 10.1038/71323. [DOI] [PubMed] [Google Scholar]
- 94.Rebollo E, Roldan M, Gonzalez C. Spindle alignment is achieved without rotation after the first cell cycle in Drosophila embryonic neuroblasts. Development. 2009;136:3393–3397. doi: 10.1242/dev.041822. [DOI] [PubMed] [Google Scholar]
- 95.Rebollo E, et al. Functionally Unequal Centrosomes Drive Spindle Orientation in Asymmetrically Dividing Drosophila Neural Stem Cells. Dev Cell. 2007;12:467–474. doi: 10.1016/j.devcel.2007.01.021. [DOI] [PubMed] [Google Scholar]
- 96.Januschke J, Gonzalez C. The interphase microtubule aster is a determinant of asymmetric division orientation in Drosophila neuroblasts. J Cell Biol. 2010;188:693–706. doi: 10.1083/jcb.200905024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Spradling AC, Zheng Y. Developmental biology. The mother of all stem cells? Science. 2007;315:469–470. doi: 10.1126/science.1138237. [DOI] [PubMed] [Google Scholar]
- 98.Wang X, et al. Asymmetric centrosome inheritance maintains neural progenitors in the neocortex. Nature. 2009;461:947–955. doi: 10.1038/nature08435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Gotta M, Dong Y, Peterson YK, Lanier SM, Ahringer J. Asymmetrically distributed C. elegans homologs of AGS3/PINS control spindle position in the early embryo. Curr Biol. 2003;13:1029–1037. doi: 10.1016/s0960-9822(03)00371-3. [DOI] [PubMed] [Google Scholar]
- 100.Colombo K, et al. Translation of polarity cues into asymmetric spindle positioning in Caenorhabditis elegans embryos. Science. 2003;300:1957–1961. doi: 10.1126/science.1084146. [DOI] [PubMed] [Google Scholar]
- 101.Srinivasan DG, Fisk RM, Xu H, Van Den Heuvel S. A complex of LIN-5 and GPR proteins regulates G protein signaling and spindle function in C. elegans. Genes Dev. 2003 doi: 10.1101/gad.1081203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Nguyen-Ngoc T, Afshar K, Gonczy P. Coupling of cortical dynein and Galpha proteins mediates spindle positioning in Caenorhabditis elegans. Nat Cell Biol. 2007;9:1294–1302. doi: 10.1038/ncb1649. [DOI] [PubMed] [Google Scholar]
- 103.Ou G, Stuurman N, D’Ambrosio M, Vale RD. Polarized Myosin Produces Unequal-Size Daughters During Asymmetric Cell Division. Science. 2010 doi: 10.1126/science.1196112. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Gateff E. Malignant neoplasms of genetic origin in Drosophila melanogaster. Science. 1978;200:1448–1459. doi: 10.1126/science.96525. [DOI] [PubMed] [Google Scholar]
- 105.Gateff E. Tumor suppressor and overgrowth suppressor genes of Drosophila melanogaster: developmental aspects. Int J Dev Biol. 1994;38:565–590. [PubMed] [Google Scholar]
- 106.Lee CY, Robinson KJ, Doe CQ. Lgl, Pins and aPKC regulate neuroblast self-renewal versus differentiation. Nature. 2006;439:594–598. doi: 10.1038/nature04299. [DOI] [PubMed] [Google Scholar]
- 107.Lee CY, et al. Drosophila Aurora-A kinase inhibits neuroblast self-renewal by regulating aPKC/Numb cortical polarity and spindle orientation. Genes Dev. 2006;20:3464–3474. doi: 10.1101/gad.1489406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Wang H, et al. Aurora-A acts as a tumor suppressor and regulates self-renewal of Drosophila neuroblasts. Genes Dev. 2006;20:3453–3463. doi: 10.1101/gad.1487506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Kitajima A, Fuse N, Isshiki T, Matsuzaki F. Progenitor properties of symmetrically dividing Drosophila neuroblasts during embryonic and larval development. Dev Biol. 2010 doi: 10.1016/j.ydbio.2010.06.029. [DOI] [PubMed] [Google Scholar]
- 110.Basto R, et al. Centrosome amplification can initiate tumorigenesis in flies. Cell. 2008;133:1032–1042. doi: 10.1016/j.cell.2008.05.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Castellanos E, Dominguez P, Gonzalez C. Centrosome Dysfunction in Drosophila Neural Stem Cells Causes Tumors that Are Not due to Genome Instability. Curr Biol. 2008;18:1209–1214. doi: 10.1016/j.cub.2008.07.029. [DOI] [PubMed] [Google Scholar]
- 112.Caussinus E, Gonzalez C. Induction of tumor growth by altered stem-cell asymmetric division in Drosophila melanogaster. Nat Genet. 2005;37:1125–1129. doi: 10.1038/ng1632. [DOI] [PubMed] [Google Scholar]
- 113.Maurange C, Cheng L, Gould AP. Temporal transcription factors and their targets schedule the end of neural proliferation in Drosophila. Cell. 2008;133:891–902. doi: 10.1016/j.cell.2008.03.034. [DOI] [PubMed] [Google Scholar]
- 114.Bello BC, Hirth F, Gould AP. A pulse of the Drosophila Hox protein Abdominal-A schedules the end of neural proliferation via neuroblast apoptosis. Neuron. 2003;37:209–219. doi: 10.1016/s0896-6273(02)01181-9. [DOI] [PubMed] [Google Scholar]
- 115.Siegrist SE, Haque NS, Chen CH, Hay BA, Hariharan IK. Inactivation of both Foxo and reaper promotes long-term adult neurogenesis in Drosophila. Curr Biol. 2010;20:643–648. doi: 10.1016/j.cub.2010.01.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Reya T, Morrison SJ, Clarke MF, Weissman IL. Stem cells, cancer, and cancer stem cells. Nature. 2001;414:105–111. doi: 10.1038/35102167. [DOI] [PubMed] [Google Scholar]
- 117.Cicalese A, et al. The tumor suppressor p53 regulates polarity of self-renewing divisions in mammary stem cells. Cell. 2009;138:1083–1095. doi: 10.1016/j.cell.2009.06.048. [DOI] [PubMed] [Google Scholar]
- 118.Wu M, et al. Imaging Hematopoietic Precursor Division in Real Time. Cell Stem Cell. 2007;1:541–554. doi: 10.1016/j.stem.2007.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Costa MR, Wen G, Lepier A, Schroeder T, Gotz M. Par-complex proteins promote proliferative progenitor divisions in the developing mouse cerebral cortex. Development. 2008;135:11–22. doi: 10.1242/dev.009951. [DOI] [PubMed] [Google Scholar]
- 120.Shinin V, Gayraud-Morel B, Gomes D, Tajbakhsh S. Asymmetric division and cosegregation of template DNA strands in adult muscle satellite cells. Nat Cell Biol. 2006;8:677–682. doi: 10.1038/ncb1425. [DOI] [PubMed] [Google Scholar]
- 121.Lechler T, Fuchs E. Asymmetric cell divisions promote stratification and differentiation of mammalian skin. Nature. 2005;437:275–280. doi: 10.1038/nature03922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Quyn AJ, et al. Spindle orientation bias in gut epithelial stem cell compartments is lost in precancerous tissue. Cell Stem Cell. 2010;6:175–181. doi: 10.1016/j.stem.2009.12.007. [DOI] [PubMed] [Google Scholar]
- 123.Rasin MR, et al. Numb and Numbl are required for maintenance of cadherin-based adhesion and polarity of neural progenitors. Nat Neurosci. 2007;10:819–827. doi: 10.1038/nn1924. [DOI] [PubMed] [Google Scholar]
- 124.Kosodo Y, et al. Asymmetric distribution of the apical plasma membrane during neurogenic divisions of mammalian neuroepithelial cells. EMBO J. 2004;23:2314–2324. doi: 10.1038/sj.emboj.7600223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Dontu G, et al. In vitro propagation and transcriptional profiling of human mammary stem/progenitor cells. Genes Dev. 2003;17:1253–1270. doi: 10.1101/gad.1061803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Colaluca IN, et al. NUMB controls p53 tumour suppressor activity. Nature. 2008;451:76–80. doi: 10.1038/nature06412. [DOI] [PubMed] [Google Scholar]
- 127.Pece S, et al. Loss of negative regulation by Numb over Notch is relevant to human breast carcinogenesis. J Cell Biol. 2004;167:215–221. doi: 10.1083/jcb.200406140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Ito T, et al. Regulation of myeloid leukaemia by the cell-fate determinant Musashi. Nature. 2010;466:765–768. doi: 10.1038/nature09171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Dietzl G, et al. A genome-wide transgenic RNAi library for conditional gene inactivation in Drosophila. Nature. 2007;448:151–156. doi: 10.1038/nature05954. [DOI] [PubMed] [Google Scholar]
- 130.Mummery-Widmer JL, et al. Genome-wide analysis of Notch signalling in Drosophila by transgenic RNAi. Nature. 2009;458:987–992. doi: 10.1038/nature07936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Shendure J, Ji H. Next-generation DNA sequencing. Nat Biotechnol. 2008;26:1135–1145. doi: 10.1038/nbt1486. [DOI] [PubMed] [Google Scholar]
- 132.Siller KH, Doe CQ. Spindle orientation during asymmetric cell division. Nat Cell Biol. 2009;11:365–374. doi: 10.1038/ncb0409-365. [DOI] [PubMed] [Google Scholar]
- 133.Santolini E, et al. Numb is an endocytic protein. J Cell Biol. 2000;151:1345–152. doi: 10.1083/jcb.151.6.1345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Gotz M, Huttner WB. The cell biology of neurogenesis. Nat Rev Mol Cell Biol. 2005;6:777–788. doi: 10.1038/nrm1739. [DOI] [PubMed] [Google Scholar]
- 135.Bultje RS, et al. Mammalian Par3 regulates progenitor cell asymmetric division via notch signaling in the developing neocortex. Neuron. 2009;63:189–202. doi: 10.1016/j.neuron.2009.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Miyata T, Kawaguchi A, Okano H, Ogawa M. Asymmetric inheritance of radial glial fibers by cortical neurons. Neuron. 2001;31:727–741. doi: 10.1016/s0896-6273(01)00420-2. [DOI] [PubMed] [Google Scholar]
- 137.Schwamborn JC, Berezikov E, Knoblich JA. The TRIM-NHL protein TRIM32 activates microRNAs and prevents self-renewal in mouse neural progenitors. Cell. 2009;136:913–925. doi: 10.1016/j.cell.2008.12.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Lapidot T, et al. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature. 1994;367:645–648. doi: 10.1038/367645a0. [DOI] [PubMed] [Google Scholar]
- 139.Bonnet D, Dick JE. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med. 1997;3:730–737. doi: 10.1038/nm0797-730. [DOI] [PubMed] [Google Scholar]
- 140.Singh SK, et al. Identification of a cancer stem cell in human brain tumors. Cancer Res. 2003;63:5821–5828. [PubMed] [Google Scholar]
- 141.Dick JE. Breast cancer stem cells revealed. Proc Natl Acad Sci U S A. 2003;100:3547–3549. doi: 10.1073/pnas.0830967100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Lobo NA, Shimono Y, Qian D, Clarke MF. The Biology of Cancer Stem Cells. Annu Rev Cell Dev Biol. 2007;23:675–699. doi: 10.1146/annurev.cellbio.22.010305.104154. [DOI] [PubMed] [Google Scholar]
- 143.Rosen JM, Jordan CT. The increasing complexity of the cancer stem cell paradigm. Science. 2009;324:1670–1673. doi: 10.1126/science.1171837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Barker N, et al. Crypt stem cells as the cells-of-origin of intestinal cancer. Nature. 2009;457:608–611. doi: 10.1038/nature07602. [DOI] [PubMed] [Google Scholar]
- 145.Kwon CH, et al. Pten haploinsufficiency accelerates formation of high-grade astrocytomas. Cancer Res. 2008;68:3286–3294. doi: 10.1158/0008-5472.CAN-07-6867. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Suppl. Movie 1 ∣ Asymmetric segregation of cell fate determinats in mitosis.
The movie shows a dividing sensory organ precursor cell in the Drosophila notum. Histone-RFP (red) and Pon-GFP (green) are driven by neuralized-Gal4.
Suppl. Figure 1 ∣ Cell type specific markers in neuroblast lineages.
Expression of the markers Asense, Deadpan, Prospero and Elav distinguishes cell types in D. melanogaster larval neuroblast lineages. Deadpan is expressed in all self-renewing cells (Type I and type II neuroblasts and INPs) and its expression disappears from the small daughter cell immediately after division. Asense is expressed by type I but not type II neuroblasts and in INPs. Its expression persists for a while after asymmetric division. Prospero is expressed in the daughter cells of type I but not type II neuroblasts. It is also expressed in the small daughter cells of INPs and its expression persists in neurons for a while. Note that Prospero expression is weaker immediately after asymmetric division and becomes stronger in neurons. Elav protein is a marker for terminally differentiating neurons although Elav RNA is more widely expressed.