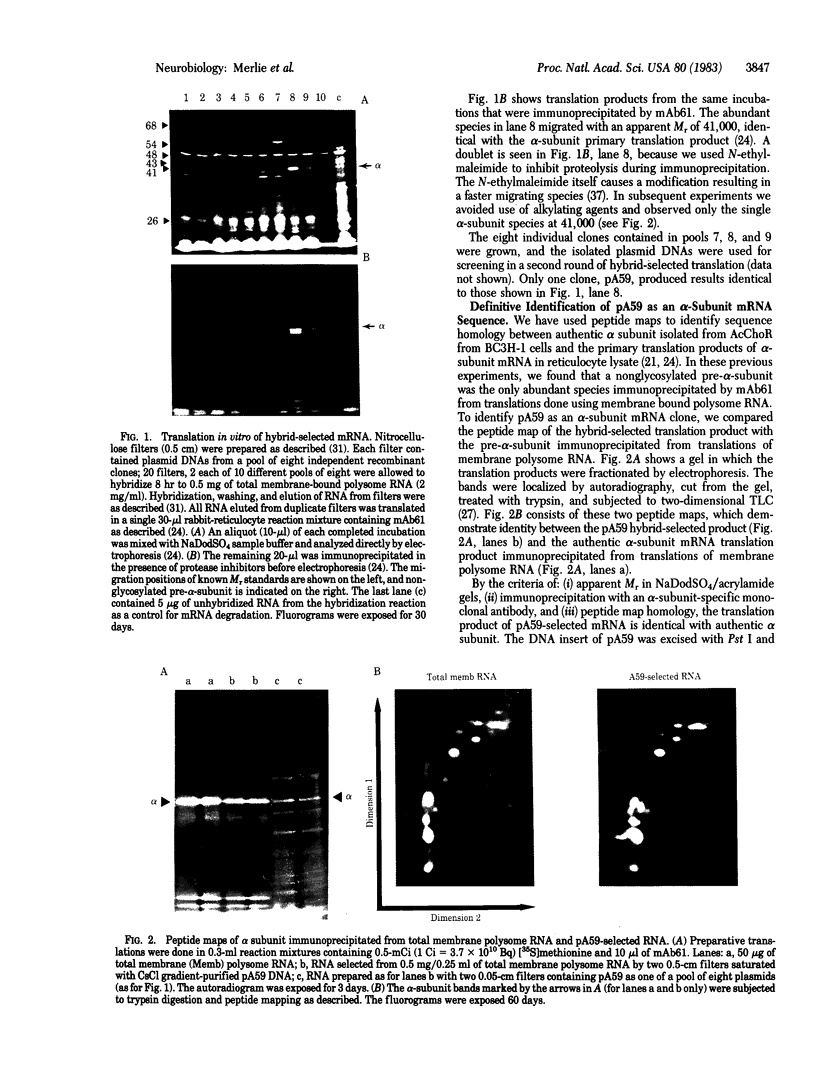
Abstract
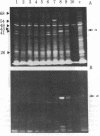
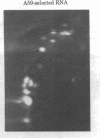
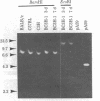
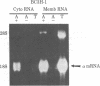
Sequences from a gene coding for mouse acetylcholine receptor alpha subunit have been inserted into a recombinant plasmid and cloned in Escherichia coli. mRNAs for acetylcholine receptors occur in low abundance in vertebrate muscle. To clone the mouse alpha-subunit cDNA, we made use of (i) a cell line, BC3H-1, that overproduces the alpha-subunit mRNA and (ii) a polysome fractionation procedure that results in enrichment of alpha-subunit mRNA. Polyadenylylated RNA was used to construct a cDNA library of 750 recombinant clones. Acetylcholine receptor-specific sequences were identified by hybrid-selected translation, followed by monoclonal antibody precipitation and peptide mapping of the translation product. One clone (pA59) that fit these criteria was found in the first 80 isolates. It had a 700-base-pair insert that was excised with Pst I. Blot-hybridization experiments with nick-translated pA59 DNA showed that BC3H-1 cells contain 100-1,000 times more alpha-subunit mRNA than does newborn or adult mouse muscle. Blot hybridization of restriction digests of mouse liver DNA revealed that pA59 is homologous to a very small number (probably one) of genomic sequences.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson D. J., Blobel G. In vitro synthesis, glycosylation, and membrane insertion of the four subunits of Torpedo acetylcholine receptor. Proc Natl Acad Sci U S A. 1981 Sep;78(9):5598–5602. doi: 10.1073/pnas.78.9.5598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ballivet M., Patrick J., Lee J., Heinemann S. Molecular cloning of cDNA coding for the gamma subunit of Torpedo acetylcholine receptor. Proc Natl Acad Sci U S A. 1982 Jul;79(14):4466–4470. doi: 10.1073/pnas.79.14.4466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bevan S., Steinbach J. H. The distribution of alpha-bungarotoxin binding sites of mammalian skeletal muscle developing in vivo. J Physiol. 1977 May;267(1):195–213. doi: 10.1113/jphysiol.1977.sp011808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boulter J., Patrick J. Purification of an acetylcholine receptor from a nonfusing muscle cell line. Biochemistry. 1977 Nov 1;16(22):4900–4908. doi: 10.1021/bi00641a025. [DOI] [PubMed] [Google Scholar]
- Buell G. N., Wickens M. P., Payvar F., Schimke R. T. Synthesis of full length cDNAs from four partially purified oviduct mRNAs. J Biol Chem. 1978 Apr 10;253(7):2471–2482. [PubMed] [Google Scholar]
- Claudio T., Ballivet M., Patrick J., Heinemann S. Nucleotide and deduced amino acid sequences of Torpedo californica acetylcholine receptor gamma subunit. Proc Natl Acad Sci U S A. 1983 Feb;80(4):1111–1115. doi: 10.1073/pnas.80.4.1111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conti-Tronconi B. M., Hunkapiller M. W., Lindstrom J. M., Raftery M. A. Amino acid sequence homology between "alpha" subunits from Torpedo and Electrophorus acetylcholine receptor. Biochem Biophys Res Commun. 1982 May 31;106(2):312–318. doi: 10.1016/0006-291x(82)91111-1. [DOI] [PubMed] [Google Scholar]
- Conti-Tronconi B. M., Raftery M. A. The nicotinic cholinergic receptor: correlation of molecular structure with functional properties. Annu Rev Biochem. 1982;51:491–530. doi: 10.1146/annurev.bi.51.070182.002423. [DOI] [PubMed] [Google Scholar]
- Davis R. W., Thomas M., Cameron J., St John T. P., Scherer S., Padgett R. A. Rapid DNA isolations for enzymatic and hybridization analysis. Methods Enzymol. 1980;65(1):404–411. doi: 10.1016/s0076-6879(80)65051-4. [DOI] [PubMed] [Google Scholar]
- Einarson B., Gullick W., Conti-Tronconi B., Ellisman M., Lindstrom J. Subunit composition of bovine muscle acetylcholine receptor. Biochemistry. 1982 Oct 12;21(21):5295–5302. doi: 10.1021/bi00264a027. [DOI] [PubMed] [Google Scholar]
- Fambrough D. M. Control of acetylcholine receptors in skeletal muscle. Physiol Rev. 1979 Jan;59(1):165–227. doi: 10.1152/physrev.1979.59.1.165. [DOI] [PubMed] [Google Scholar]
- Giraudat J., Devillers-Thiery A., Auffray C., Rougeon F., Changeux J. P. Identification of a cDNA clone coding for the acetylcholine binding subunit of Torpedo marmorata acetylcholine receptor. EMBO J. 1982;1(6):713–717. doi: 10.1002/j.1460-2075.1982.tb01235.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodman H. M., MacDonald R. J. Cloning of hormone genes from a mixture of cDNA molecules. Methods Enzymol. 1979;68:75–90. doi: 10.1016/0076-6879(79)68007-2. [DOI] [PubMed] [Google Scholar]
- Gotti C., Conti-Tronconi B. M., Raftery M. A. Mammalian muscle acetylcholine receptor purification and characterization. Biochemistry. 1982 Jun 22;21(13):3148–3154. doi: 10.1021/bi00256a018. [DOI] [PubMed] [Google Scholar]
- Hall Z. W., Reiness C. G. Electrical stimulation of denervated muscles reduces incorporation of methionine into the ACh receptor. Nature. 1977 Aug 18;268(5621):655–657. doi: 10.1038/268655a0. [DOI] [PubMed] [Google Scholar]
- Lehrach H., Diamond D., Wozney J. M., Boedtker H. RNA molecular weight determinations by gel electrophoresis under denaturing conditions, a critical reexamination. Biochemistry. 1977 Oct 18;16(21):4743–4751. doi: 10.1021/bi00640a033. [DOI] [PubMed] [Google Scholar]
- Lindstrom J., Walter B., Einarson B. Immunochemical similarities between subunits of acetylcholine receptors from Torpedo, Electrophorus, and mammalian muscle. Biochemistry. 1979 Oct 16;18(21):4470–4480. doi: 10.1021/bi00588a004. [DOI] [PubMed] [Google Scholar]
- McKeown M., Firtel R. A. Evidence for sub-families of actin genes in Dictyostelium as determined by comparisons of 3' end sequences. J Mol Biol. 1981 Oct 5;151(4):593–606. doi: 10.1016/0022-2836(81)90425-3. [DOI] [PubMed] [Google Scholar]
- Merlie J. P., Buckingham M. E., Whalen R. G. Molecular aspects of myogenesis. Curr Top Dev Biol. 1977;11:61–114. doi: 10.1016/s0070-2153(08)60743-7. [DOI] [PubMed] [Google Scholar]
- Merlie J. P., Changeux J. P., Gros F. Skeletal muscle acetylcholine receptor. Purification, characterization, and turnover in muscle cell cultures. J Biol Chem. 1978 Apr 25;253(8):2882–2891. [PubMed] [Google Scholar]
- Merlie J. P., Hofler J. G., Sebbane R. Acetylcholine receptor synthesis from membrane polysomes. J Biol Chem. 1981 Jul 10;256(13):6995–6999. [PubMed] [Google Scholar]
- Merlie J. P., Sebbane R. Acetylcholine receptor subunits transit a precursor pool before acquiring alpha-bungarotoxin binding activity. J Biol Chem. 1981 Apr 25;256(8):3605–3608. [PubMed] [Google Scholar]
- Merlie J. P., Sebbane R., Tzartos S., Lindstrom J. Inhibition of glycosylation with tunicamycin blocks assembly of newly synthesized acetylcholine receptor subunits in muscle cells. J Biol Chem. 1982 Mar 10;257(5):2694–2701. [PubMed] [Google Scholar]
- Morley B. J., Kemp G. E. Characterization of a putative nicotinic acetylcholine receptor in mammalian brain. Brain Res. 1981 Aug;228(1):81–104. doi: 10.1016/0165-0173(81)90013-8. [DOI] [PubMed] [Google Scholar]
- Noda M., Takahashi H., Tanabe T., Toyosato M., Furutani Y., Hirose T., Asai M., Inayama S., Miyata T., Numa S. Primary structure of alpha-subunit precursor of Torpedo californica acetylcholine receptor deduced from cDNA sequence. Nature. 1982 Oct 28;299(5886):793–797. doi: 10.1038/299793a0. [DOI] [PubMed] [Google Scholar]
- Noda M., Takahashi H., Tanabe T., Toyosato M., Kikyotani S., Hirose T., Asai M., Takashima H., Inayama S., Miyata T. Primary structures of beta- and delta-subunit precursors of Torpedo californica acetylcholine receptor deduced from cDNA sequences. Nature. 1983 Jan 20;301(5897):251–255. doi: 10.1038/301251a0. [DOI] [PubMed] [Google Scholar]
- Palva E. T., Hirst T. R., Hardy S. J., Holmgren J., Randall L. Synthesis of a precursor to the B subunit of heat-labile enterotoxin in Escherichia coli. J Bacteriol. 1981 Apr;146(1):325–330. doi: 10.1128/jb.146.1.325-330.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parnes J. R., Velan B., Felsenfeld A., Ramanathan L., Ferrini U., Appella E., Seidman J. G. Mouse beta 2-microglobulin cDNA clones: a screening procedure for cDNA clones corresponding to rare mRNAs. Proc Natl Acad Sci U S A. 1981 Apr;78(4):2253–2257. doi: 10.1073/pnas.78.4.2253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patrick J., McMillan J., Wolfson H., O'Brien J. C. Acetylcholine receptor metabolism in a nonfusing muscle cell line. J Biol Chem. 1977 Mar 25;252(6):2143–2153. [PubMed] [Google Scholar]
- Pumplin D. W., Fambrough D. M. Turnover of acetylcholine receptors in skeletal muscle. Annu Rev Physiol. 1982;44:319–335. doi: 10.1146/annurev.ph.44.030182.001535. [DOI] [PubMed] [Google Scholar]
- Raftery M. A., Hunkapiller M. W., Strader C. D., Hood L. E. Acetylcholine receptor: complex of homologous subunits. Science. 1980 Jun 27;208(4451):1454–1456. doi: 10.1126/science.7384786. [DOI] [PubMed] [Google Scholar]
- Rigby P. W., Dieckmann M., Rhodes C., Berg P. Labeling deoxyribonucleic acid to high specific activity in vitro by nick translation with DNA polymerase I. J Mol Biol. 1977 Jun 15;113(1):237–251. doi: 10.1016/0022-2836(77)90052-3. [DOI] [PubMed] [Google Scholar]
- Schubert D., Harris A. J., Devine C. E., Heinemann S. Characterization of a unique muscle cell line. J Cell Biol. 1974 May;61(2):398–413. doi: 10.1083/jcb.61.2.398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sebbane R., Clokey G., Merlie J. P., Tzartos S., Lindstrom J. Characterization of the mRNA for mouse muscle acetylcholine receptor alpha subunit by quantitative translation in vitro. J Biol Chem. 1983 Mar 10;258(5):3294–3303. [PubMed] [Google Scholar]
- Sine S., Taylor P. Functional consequences of agonist-mediated state transitions in the cholinergic receptor. Studies in cultured muscle cells. J Biol Chem. 1979 May 10;254(9):3315–3325. [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Sumikawa K., Houghton M., Smith J. C., Bell L., Richards B. M., Barnard E. A. The molecular cloning and characterisation of cDNA coding for the alpha subunit of the acetylcholine receptor. Nucleic Acids Res. 1982 Oct 11;10(19):5809–5822. doi: 10.1093/nar/10.19.5809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tzartos S. J., Rand D. E., Einarson B. L., Lindstrom J. M. Mapping of surface structures of electrophorus acetylcholine receptor using monoclonal antibodies. J Biol Chem. 1981 Aug 25;256(16):8635–8645. [PubMed] [Google Scholar]
- Villa-Komaroff L., Efstratiadis A., Broome S., Lomedico P., Tizard R., Naber S. P., Chick W. L., Gilbert W. A bacterial clone synthesizing proinsulin. Proc Natl Acad Sci U S A. 1978 Aug;75(8):3727–3731. doi: 10.1073/pnas.75.8.3727. [DOI] [PMC free article] [PubMed] [Google Scholar]