Abstract
Alcoholic liver disease (ALD) is a complex chronic disease and is associated with a spectrum of liver injury ranging from steatosis and steatohepatitis to fibrosis and cirrhosis. Since effective therapies for ALD are still limited, Chinese herbal medicine is thought to be an important and alternative approach. This review focuses on the current scientific evidence of ALD by ten Chinese Materia Medica (中藥 zhōng yào), including Salviae Miltiorrhizae Radix (丹參 dān shēn), Notoginseng Radix (三七 sān qī), Lycii Fructus (枸杞子 gǒu qǐ zǐ), Cnidii Fructus (蛇床子 shé chuáng zǐ), Gentianae Radix (龍膽 lóng dǎn), Puerariae Radix (葛根 gé gēn), Puerariae Flos (葛花 gé huā), Magnoliae Officinalis Cortex (厚朴 hòu pò), Platycodonis Radix (桔梗 jié gěng), and Trigonellae Semen (胡蘆巴 hú lú bā). Potential mechanisms of these herbal medicines in ALD are involved in amelioration of enhanced inflammation, reduction of hepatic oxidative stress and lipogenesis, and enhancement of intestinal permeability in alcohol-induced liver injury models in vitro and in vivo. Accordingly, the evidenced therapeutic potential suggests that these herbs are promising candidates for prevention and development of new drugs for ALD in the future.
Keywords: Alcoholic liver disease, Alcohol-induced liver injury, Herbal medicine, Chinese Materia Medica, Traditional Chinese medicine
Introduction
World Health Organization has reported that the harmful use of alcohol is one of the world's leading health risks. Approximately 4.5% of the global burden of diseases and injury can be attributable to alcohol, which results in approximately 2.5 million deaths each year (World Health Organization, 2011). Chronic or excessive alcohol consumption is one of the major causes of liver disease and injury. Since the liver is the largest human organ and metabolizes various substances that can damage the body, it is important to prevent damage to healthy liver or repair the damaged liver from alcoholic liver disease (ALD) for a healthy life (Chien et al., 2011).
ALD is a complex chronic disease and typically progresses through multiple stages of alcoholic steatosis, hepatitis and cirrhosis to end-stage liver disease (Haber et al., 2003; Seth et al., 2008). The major feature of ALD is the enhanced inflammation in liver during alcohol exposure. Dysregulated cytokine metabolism and activity such as tumor necrosis factor (TNF-α) are crucial to the initiation of alcohol-induced liver injury (An et al., 2012). In addition, hepatic oxidative stress is known to play a prime role in the pathogenesis of ALD (Bondy, 1992). Induction of cytochrome P450 2E1 (CYP2E1) by alcohol is also another key pathway since the pathological changes in early alcoholic liver injuries such as steatosis and steatohepatitis are associated with CYP2E1 levels (Stewart et al., 2001; Wu and Cederbaum, 2005). Moreover, alcohol can impair fat oxidation and stimulate lipogenesis by interfering with DNA binding and transcription-activating properties of peroxisome proliferator-activated receptor-α (PPARα) and activating sterol regulatory element-binding protein 1 (SREBP-1) (You et al., 2002; You and Crabb, 2004). Understanding the mechanism underlying alcohol-induced effects provides an array of potential candidate for novel new therapeutic targets to reverse ALD.
In traditional Chinese medicine (TCM), liver disease such as fibrosis is perceived to be caused by poor blood circulation, toxin accumulation, and energy deficiency. Accordingly, Chinese medicine used therapy to treat liver injury is mainly based on increasing blood circulation, eliminating toxins, resolving stasis, and enhancing body immunity (Feng et al., 2009). However, perspectives on evidence-based medicine nowadays are encouraging medical practitioners to be guided in their practice by available scientific evidence in terms of efficacy and safety for individual therapies (Fogden and Neuberger, 2003). In the present review, an attempt is made to survey and comprehensively compile the recent scientific evidence regarding Chinese Materia Medica (CMM) (中藥 zhōng yào) and ALD through PubMed database (http://www.ncbi.nlm.nih.gov/pubmed/). All CMMs shown in this review were selected due to their experimental evidences in vivo and/or in vitro.
Chinese herbal medicines against alcoholic liver disease
Salviae Miltiorrhizae Radix (丹參 dān shēn; the root of Salvia miltiorrhiza, Labiatae)
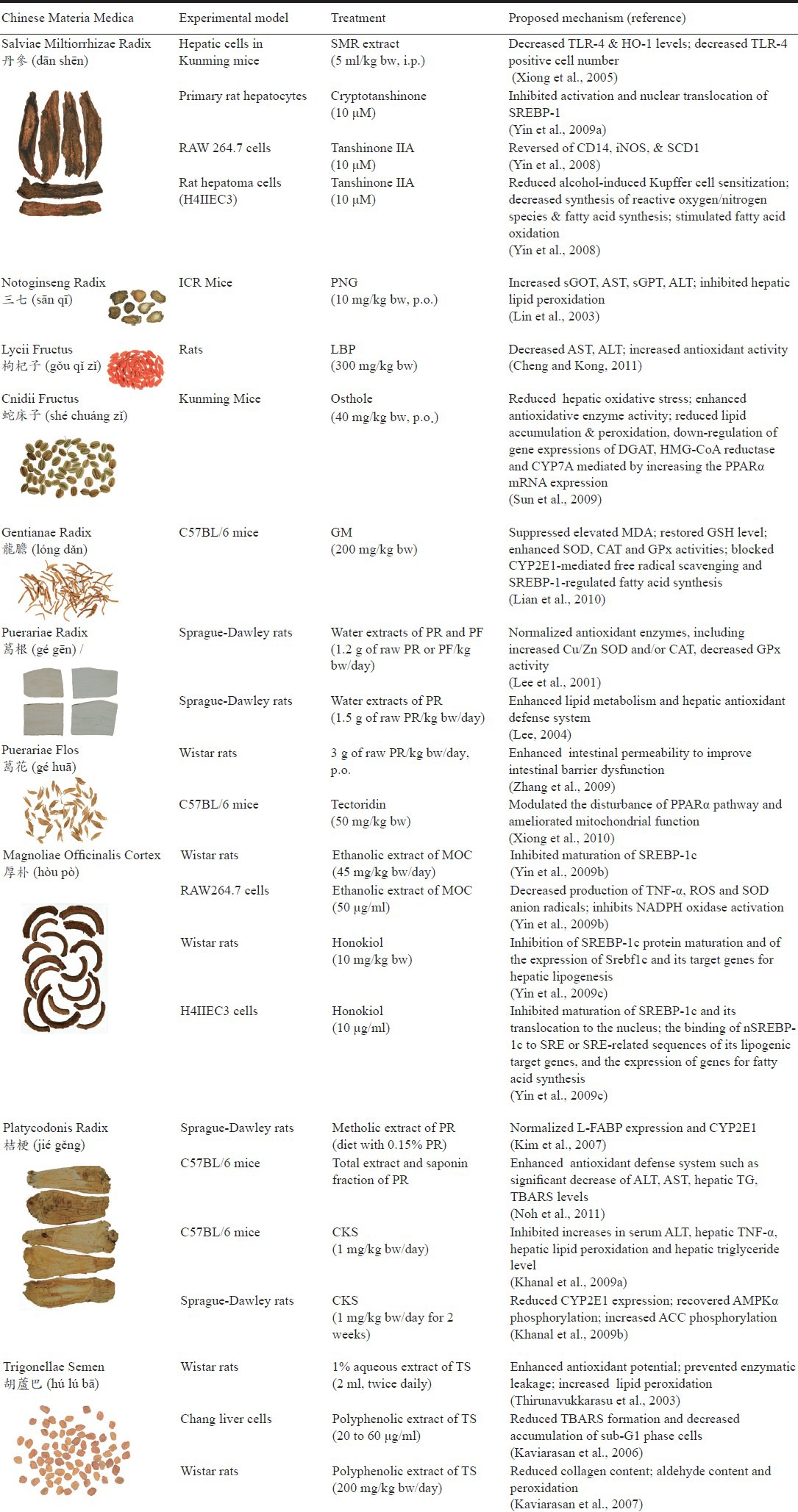
Salviae Miltiorrhizae Radix (SMR) has been used in the treatment of hypertension, coronary heart disease and myocardial infarction in TCM (Lei and Chiou, 1986; Sun et al., 2005; Wang et al., 1989). Also, it has been used widely to treat chronic liver disease. An in vivo study elucidated the protective effect of SMR on chronic alcoholic liver injury employing a mice model (Xiong et al., 2005). Administration of SMR (5 ml/kg bw, i.p.) could alleviate the alcohol-induced fatty degeneration and adiponecrosis of hepatic cells in Kunming mice. The protective mechanisms of SMR were probably due to the down-regulation of TLR-4 and HO-1 mRNA expressions and significant decrease of TLR-4 positive cell number in the animal study.
Cryptotanshinone and tanshinone IIA (Figure 1), diterpenes contained in SMR, were considered as active hepatoprotective components. In primary rat hepatocytes, 10 μM cryptotanshinone suppressed alcohol-induced lipid accumulation, and inhibited activation and nuclear translocation of sterol regulatory element-binding protein 1(SREBP-1) and the consequent transactivation of the target genes involved in fatty acid biosynthesis (Yin et al., 2009a). On the other hand, 10 μM tanshinone IIA protected RAW 264.7 cells from alcohol-induced production of superoxide radical, activation of NADPH oxidase, and subsequently death of the cells. Alcohol-induced expressions of CD14, iNOS, and SCD1 were completely reversed by tanshinone IIA. Also, tanshinone IIA effectively blocked alcohol-induced fat accumulation in rat hepatoma cells (H4IIEC3). Consequently, tanshinone IIA is the major active component of SMR to inhibit alcoholic liver disease by reducing alcohol-induced Kupffer cell sensitization, decreasing synthesis of reactive oxygen/nitrogen species and fatty acid synthesis, and stimulating fatty acid oxidation (Yin et al., 2008).
Figure 1.
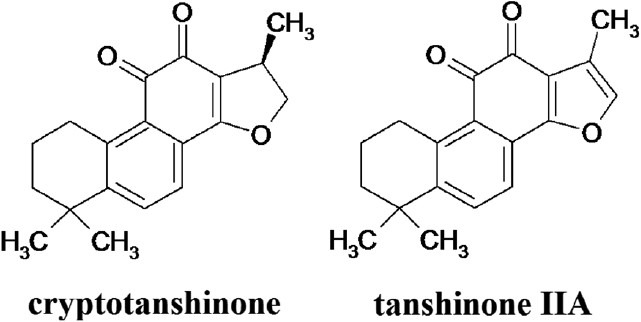
Structures of cryptotanshinone and tanshinone IIA from Salvia miltiorrhiza
Notoginseng Radix (三七 sān qī; the root of Panax notoginseng (Burk.) F.H. Chen, Araliaceae)
Notoginseng Radix is known in TCM to improve blood circulation and has been used to treat angina pectoris due to its property to increase coronary blood flow. Bioactivities associated with Panax notoginseng include anti-inflammatory, analgesic, antitumor, antihypertensive, anti-atherosclerotic, antithrombotic, neuroprotective and hepatoprotective effects (Ng, 2006; Pan et al., 2011). An animal study elucidated the hepatoprotective effects exerted by a hot water extract of P. notoginseng (PNG) in alcohol-fed ICR mice (Lin et al., 2003). PNG (10 mg/kg bw, p.o.) prevented the rise in serum glutamate–oxaloacetate transaminase (sGOT, AST) and glutamate–pyruvate transaminase (sGPT, ALT), and inhibited the hepatic lipid peroxidation (26% inhibition rate) against chronic alcohol-induced hepatotoxicity.
Lycii Fructus (枸杞子 gǒu qǐ zǐ; the fruit of Lycium barbarum, Solanaceae)
Lycii Fructus, commercially known as goji berry, has historically been used in TCM to improve eyesight and to strengthen the liver and kidney (Lee et al., 2012). The bioactivity of Lycii Fructus is mainly attributed to its polysaccharide complex (Figure 2), which consists of six monosaccharides (galactose, glucose, rhamnose, arabinose, mannose, and xylose). A recent in vivo study reported that Lycium barbarum polysaccharide (LBP) at a dose of 300 mg/kg bw significantly ameliorated liver injury by decreasing serum aspartate aminotransferase (AST) and alanine aminotransferase (ALT) activities and improving the antioxidant functions in alcohol-fed rats (Cheng and Kong, 2011). Moreover, LBP prevented the progression of alcohol-induced fatty liver by decreasing the levels of serum triglycerides, total cholesterol, and low density lipoprotein cholesterol, and by increasing high density lipoprotein cholesterol level.
Figure 2.

Structure of polysaccharide from Lycium barbarum
Cnidii Fructus (蛇床子 shé chuáng zǐ; the fruit of Cnidium monnieri (L.) Cusson, Apiaceae)
Cnidii Fructus has been used in the treatment of skin disease and gynecopathy in Chinese herbal medicine for many years (Lian, 2003). Osthole (Figure 3) is a coumarin isolated from Cnidii Fructus. Modern pharmacological studies reported that osthole prevented the development of hepatitis and hepatocellular carcinoma induced by anti-Fas antibody in mice (Okamoto et al., 2003; Okamoto et al., 2005). Recent studies showed that osthole (40 mg/kg bw, p.o.) dramatically decreased lipid accumulation to inhibit development of alcohol-induced fatty liver in Kunming mice (Sun et al., 2009). The potential mechanisms were related to reduction of hepatic oxidative stress, enhancement of antioxidative enzyme activity, reduction of lipid accumulation and peroxidation, and regulation the gene expressions of diacylglycerol acyltransferase (DGAT), 3-hydroxy-3-methylglutaryl-CoA (HMG-CoA) reductase and cholesterol 7α-hydroxylase (CYP7A) mediated by increasing the peroxisome proliferator-activated receptor-α (PPARα) mRNA expression (Sun et al., 2010; Zhang et al., 2011).
Figure 3.
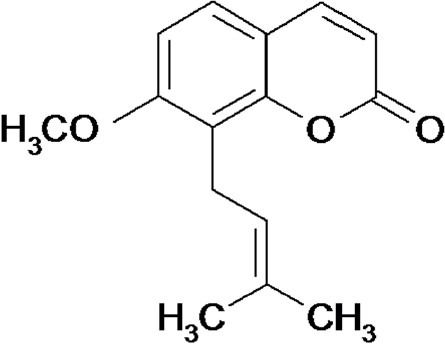
Structure of osthole from Cnidium monnieri
Gentianae Radix (龍膽 lóng dǎn; the root of Gentiana manshurica Kitagawa, Gentianaceae)
Gentiana manshurica Kitagawa is abundantly distributed in northeastern China and has been used traditionally as a folk remedy by Chinese people suffering from chronic liver disease (Wang et al., 2010). A recent in vivo study showed the protective effects of Gentianae Radix methanolic extract (GM) on acute alcohol-induced fatty liver (Lian et al., 2010). Treatment of GM (200 mg/kg bw) significantly reduced the increases in serum aspartate aminotransferase (AST) and alanine aminotransferase (ALT) levels, and the serum and hepatic triglyceride levels in ethanol-administered C57BL/6 mice. GM was also found to prevent alcohol-induced hepatic steatosis and necrosis by suppressing the elevation of malondialdehyde (MDA) levels, restoring the glutathione (GSH) levels, and enhancing the superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPx) activities. Moreover, GM efficaciously abrogated cytochrome P450 2E1 (CYP2E1) induction and significantly reduced the nuclear translocation of activating sterol regulatory element-binding protein 1 (SREBP-1) in alcohol-treated mice. In summary, GM possessed the ability to prevent alcohol-induced acute liver steatosis by blocking CYP2E1-mediated free radical scavenging effects and SREBP-1-regulated fatty acid synthesis.
Puerariae Radix (葛根 gé gēn; the root of Puerariae lobata, Leguminosae) and Puerariae Flos (葛花 gé huā; the flower of P. lobata)
Puerariae Radix (PR) and Puerariae Flos (PF) have been used in TCM to counteract problems associated with alcohol drinking and liver injury since ancient times (Keung and Vallee, 1998). The hepatoprotective effects of PR and PF in alcoholic liver damage have been experimentally demonstrated in animal studies (Lee et al., 2001). Water extracts of PR and PF (mixed with the diets based on 1.2 g of raw PR or PF/kg bw/ day) were reported to normalize antioxidant enzymes, including significant increase of the Cu/Zn superoxide dismutase (SOD) and/or catalase (CAT) activities and decrease of the glutathione peroxidase (GPx) activity, to the control level in the alcohol-treated Sprague-Dawley rats. Similarly, another animal study showed that PR water extract (mixed with the diets based on 1.5 g of raw PR/kg bw/day) alleviated the adverse effect of alcohol ingestion in Sprague-Dawley rats by enhancing the lipid metabolism as well as the hepatic antioxidant defense system (Lee, 2004). Moreover, a recent study showed that the inhibitory effect of PR extract (mixed with alcohol based on 3 g of raw PR/kg bw/day, p.o.) on alcohol-induced liver damage in Wistar rats was through enhancement of intestinal permeability to improve intestinal barrier dysfunction induced by alcohol (Zhang et al., 2009).
Tectoridin (Figure 4) is an isoflavone glycoside isolated from the flower of P. lobata and considered as an active component in liver protection. The hepatoprotective activity of tectoridin has been experimentally shown on tert-butyl hy peroxide-induced hepatotoxicity in mice (Lee et al., 2005). Recently, it was demonstrated that tectoridin prevented acute alcohol-induced liver steatosis in C57BL/6 mice (Xiong et al., 2010). Treatment of 50 mg/kg bwtectoridin significantly alleviated the over-production of thiobarbituric acid-reactive substance. Furthermore, tectoridin inhibited the reduction of peroxisome proliferator-activated receptor-α (PPARα) expression and its target genes, including medium-chain acyl-CoA dehydrogenase (MCAD), acyl-CoA oxidase (ACO) and cytochrome P450 4A (CYP 4A) at mRNA and enzyme activity levels. The possible mechanism of tectoridin to inhibit alcohol-induced hepatotoxicity was mainly by modulating the disturbance of PPARα pathway and ameliorating mitochondrial function.
Figure 4.
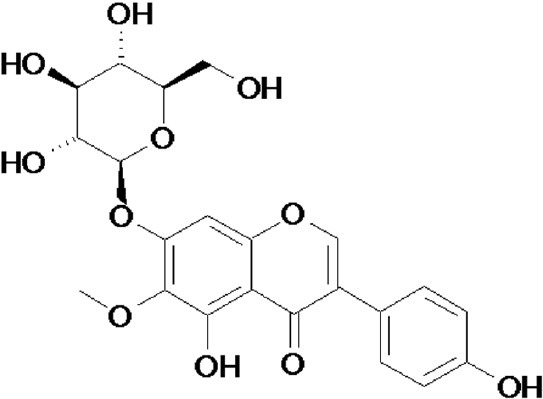
Structure of tectoridin from Puerariae lobata
Magnoliae Officinalis Cortex (厚朴 hòu pò; the stem bark of Magnolia officinalis Rehd. et Wols, Magnoliaceae)
Magnoliae Officinalis Cortex (MOC) is a traditional Chinese herbal medicine that has been used for treatment of gastrointestinal disorders, anxiety and allergic disease (Lee et al., 2011). A recent study showed that the ethanolic extract of MOC (50 μg/ml) was able to protect alcohol-induced cell death by decreasing the production of TNF-α, reactive oxygen species and superoxide anion radicals, and by inhibiting the activation of NADPH in RAW264.7 cells. In ethanol diet-fed animal model, Wistar rats treated with the ethanolic extract of MOC (45 mg/kg bw/day) completely reversed all the serum, hepatic parameters, and fatty liver changes by inhibiting maturation of activating sterol regulatory element-binding protein 1c (SREBP-1c) (Yin et al., 2009b).
Honokiol and magnolol (Figure 5) are isomers of biphenolic compounds isolated from MOC. In Lieber-DeCarli alcohol diet-fed Wistar rats, daily administration of honokiol (10 mg/kg bw) exerted efficacious abrogation of fat accumulation in the liver and completely reversed the effects on hepatotoxicity markers, including decreased hepatic glutathione and increased hepatic triglyceride and serum TNF-α. In addition, honokiol (10 μg/ml) reversed the production of lipid droplets in alcohol-treated H4IIEC3 cells. The investigation of mechanisms demonstrated that honokiol inhibited the maturation of SREBP-1c and its translocation to the nucleus, the binding of nSREBP-1c to SRE or SRE-related sequences of its lipogenic target genes, and the expression of genes for fatty acid synthesis. In contrast, magnolol had no effect on nSREBP-1c levels (Yin et al., 2009c).
Figure 5.
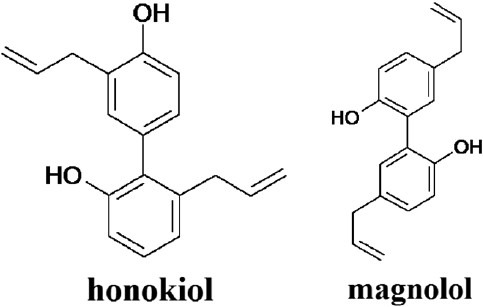
Structures of honokiol and magnolol from Magnolia officinalis
Platycodonis Radix (桔梗 jié gěng; the root of Platycodon grandiflorum A. DC, Campanulaceae)
Platycodonis Radix (PR) is used as a food additive and has been employed for treatment of inflammatory diseases and geriatric diseases such as bronchitis, asthma and pulmonary tuberculosis in TCM (Guo et al., 2007). The protective effects of PR on the liver have been demonstrated in rats with alcohol-induced hepatotoxicity. Administration of PR significantly prevented alcohol-induced elevation of serum and liver lipids by normalizing hepatic liver fatty acid binding protein (L-FABP) expression and cytochrome P450 2E1 (CYP2E1) activity in alcohol-treated Sprague-Dawley rats (Kim et al., 2007). A recent study also reported that PR (both the total extract and saponin fraction) had protective effects against alcohol-induced damage in vitro and in vivo (Noh et al., 2011). The heptoprotective mechanism of PR is by inhibition of lipid accumulation and peroxidation through the enhancement of the antioxidant defense system such as significant decrease of glutamic oxalacetic transaminase, glutamic pyruvic transaminase, hepatic triglyceride and thiobarbituric acid reactive substance levels.
Saponins (Figure 6) isolated from the PR (Changkil saponins, CKS) were reported to be the active components in hepatoprotective effects. To obtain saponin fraction, aqueous extract of Platycodonis Radix was subjected to column chromatography to remove saccharides and amino acids (Tada et al., 1975). In vivo studies showed that pretreatment with CKS (1 mg/kg bw/day for 7 days) prior to ethanol administration significantly prevented alcoholic liver injury in C57BL/6 mice by inhibiting the increases in serum alanine aminotransferase activity (ALT), hepatic TNF-α level, hepatic lipid peroxidation, and hepatic triglyceride level (Khanal et al., 2009a). In Lieber-DeCarli alcohol diet-fed Sprague–Dawley rats, the possible hepatoprotective mechanisms of CKS treatment (1 mg/kg bw/day for 2 weeks) in alcohol-induced liver damage were done through reduction of CYP2E1 expression, recovery of AMP-activated protein kinase-alpha (AMPKα) phosphorylation, and an increase in acetyl-CoA carboxylase phosphorylation (Khanal et al., 2009b).
Figure 6.
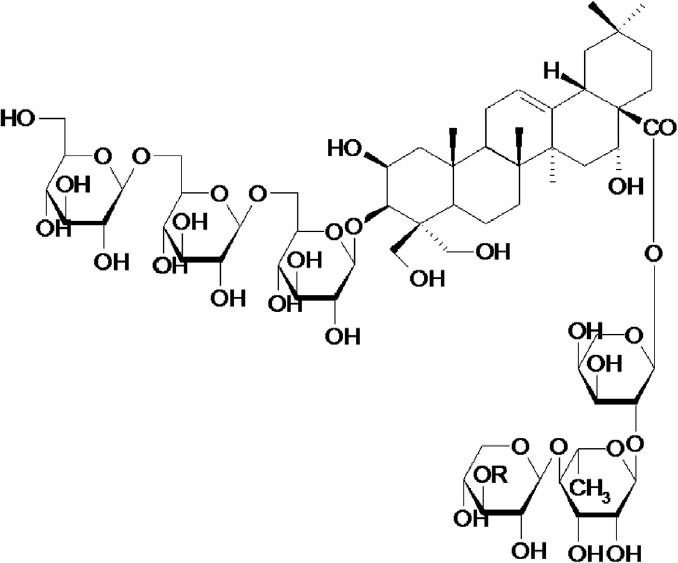
Structure of saponin from Platycodon grandiflorum
Trigonellae Semen (胡蘆巴 hú lú bā; the seed of Trigonella foenum-graecum, Leguminosea)
Trigonella foenum-graecum, also known as Fenugreek, is an annual herb and has a long history as both a culinary and medicinal herb. In Asia, seeds of this herb are commonly used as a spice in food preparations due to the strong aroma and flavor. Trigonellae Semen possesses restorative and nutritive properties and has been used in remedies for diabetes a n d hypercholesterolemia in Indian, Arabic and Chinese medicine. (Khosla et al., 1995; Sharma et al.,1990; Sharma et al., 1996). Aqueous extract (1%) of Trigonellae Semen (2 ml, twice daily) was reported to offer a significant protection against ethanol toxicity in Wistar rats by enhancement of the antioxidant potential and prevention of the enzymatic leakage and the rise in lipid peroxidation (Thirunavukkarasu et al., 2003).
Further studies indicated that the polyphenolic extract of Trigonellae Semen was considered cytoprotective during alcohol-induced liver damage in vitro and in vivo. Treatment of 20 to 60 μg/ml Trigonellae Semen polyphenol extract (FPEt) prevented alcohol-induced toxicity and apoptosis in human Chang liver cells by reducing thiobarbituric acid reactive substance formation and decreasing the accumulation of sub-G1 phase cells (Kaviarasan et al., 2006). Moreover, FPEt ameliorated the pathological liver changes induced by chronic alcohol feeding in Wistar rats. Administration of FPEt a dose of 200 mg/kg bw/day significantly improved lipid profile and reduced collagen content, aldehyde content and peroxidation (Kaviarasan et al.,2007). Furthermore, the protective effects of FPEt in alcoholic fatty liver diseases may involve in reducing the levels of lipid peroxidation products and protein carbonyl content, increasing the activities of antioxidant enzymes, and restoring the levels of thiol groups such as protein and non-protein thiols (Kaviarasan et al., 2008).
Conclusion
Herbal medicines have been used in China for more than 2,000 years. However, Chinese herbal medicines did not receive extensive analytical attention through western scientific methods until recent decades. In this review, ten herbal medicines traditionally used in Chinese Materia Medica have been experimentally demonstrated by modern in vitro and in vivo studies on alcohol-induced liver injury. As shown in Table 1, the potentially protective mechanisms of these herbs in alcoholic liver disease are involved in amelioration of enhanced inflammation, reduction of hepatic oxidative stress and lipogenesis, and enhancement of intestinal permeability. Hence, the evidenced therapeutic potential of these herbs suggests that Chinese herbal medicines may be good resources for prevention and development of new drugs for ALD in the future.
Table 1.
Hepatoprotective effect of Chinese herbal medicine against alcoholic liver disease
Acknowledgments
The authors gratefully acknowledge Prof. Yuan-Shiun Chang for providing the elegant photos of Chinese Materia Medica, and Mr. Eric Te-Yu Chen for his very kind editorial assistance.
References
- 1.An L., Wang X., Cederbaum A.I. Cytokines in alcoholic liver disease. Archives of Toxicology. 2012 doi: 10.1007/s00204-012-0814-6. DOI: 10.1007/s00204-012-0814-6. [DOI] [PubMed] [Google Scholar]
- 2.Bondy S.C. Ethanol toxicity and oxidative stress. Toxicology Letters. 1992;63:231–241. doi: 10.1016/0378-4274(92)90086-y. [DOI] [PubMed] [Google Scholar]
- 3.Cheng D., Kong H. The effect of Lycium barbarum polysaccharide on alcohol-induced oxidative stress in rats. Molecules. 2011;16:2542–2550. doi: 10.3390/molecules16032542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chien C.F., Wu Y.T., Tsai T.H. Biological analysis of herbal medicines used for the treatment of liver diseases. Biomedical Chromatography. 2011;25:21–38. doi: 10.1002/bmc.1568. [DOI] [PubMed] [Google Scholar]
- 5.Feng Y., Cheung K.F., Wang N., Liu P., Nagamatsu T., Tong Y. Chinese medicines as a resource for liver fibrosis treatment. Chinese Medicine. 2009;4:16. doi: 10.1186/1749-8546-4-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fogden E., Neuberger J. Alternative medicines and the liver. Liver International. 2003;23:213–220. doi: 10.1034/j.1600-0676.2003.00843.x. [DOI] [PubMed] [Google Scholar]
- 7.Guo L., Zhang C., Li L., Xiao Y.Q. Advances in studies on Platycodon grandiflorum (Chinese) China. Journal of Chinese Materia Medica. 2007;32:181–186. [PubMed] [Google Scholar]
- 8.Haber P.S., Warner R., Seth D., Gorrell M.D., Mc Caughan G.W. Pathogenesis and management of alcoholic hepatitis. Journal of Gastroenterology and Hepatology. 2003;18:1332–1344. doi: 10.1046/j.1440-1746.2003.03217.x. [DOI] [PubMed] [Google Scholar]
- 9.Kaviarasan S., Ramamurty N., Gunasekaran P., Varalakshmi E., Anuradha C.V. Fenugreek (Trigonella foenum-graecum) seed extract prevents ethanol-induced toxicity and apoptosis in Chang liver cells. Alcohol & Alcoholism. 2006;41:267–273. doi: 10.1093/alcalc/agl020. [DOI] [PubMed] [Google Scholar]
- 10.Kaviarasan S., Sundarapandiyan R., Anuradha C.V. Protective action of fenugreek (Trigonella foenum-graecum) seed polyphenols against alcohol-induced protein and lipid damage in rat liver Cell. Biology and Toxicology. 2008;24:391–400. doi: 10.1007/s10565-007-9050-x. [DOI] [PubMed] [Google Scholar]
- 11.Kaviarasan S., Viswanathan P., Anuradha C.V. Fenugreek seed (Trigonella foenum-graecum) polyphenols inhibit ethanol- induced collagen and lipid accumulation in rat liver. Cell Biology and Toxicology. 2007;23:373–383. doi: 10.1007/s10565-007-9000-7. [DOI] [PubMed] [Google Scholar]
- 12.Keung W.M., Vallee B.L. Kudzu root: an ancient Chinese source of modern antidipsotropic agents. Phytochemistry. 1998;47:499–506. doi: 10.1016/s0031-9422(97)00723-1. [DOI] [PubMed] [Google Scholar]
- 13.Khanal T., Choi J.H., Hwang Y.P., Chung Y.C., Jeong H.G. Saponins isolated from the root of Platycodon grandiflorum protect against acute ethanol-induced hepatotoxicity in mice. Food and Chemical Toxicology. 2009a;47:530–535. doi: 10.1016/j.fct.2008.12.009. [DOI] [PubMed] [Google Scholar]
- 14.Khanal T., Choi J.H., Hwang Y.P., Chung Y.C., Jeong H.G. Protective effects of saponins from the root of Platycodon grandiflorum against fatty liver in chronic ethanol feeding via the activation of AMP-dependent protein kinase. Food and Chemical Toxicology. 2009b;47:2749–2754. doi: 10.1016/j.fct.2009.08.006. [DOI] [PubMed] [Google Scholar]
- 15.Khosla P., Gupta D.D., Nagpal R.K. Effect of Trigonella foenum-graecum (Fenugreek) on blood glucose in normal and diabetic rats. Indian Journal of Physiology and Pharmacology. 1995;39:173–174. [PubMed] [Google Scholar]
- 16.Kim H.K., Kim D.S., Cho H.Y. Protective effects of Platycodi radix on alcohol-induced fatty liver. Bioscience Biotechnology and Biochemistry. 2007;71:1550–1552. doi: 10.1271/bbb.60523. [DOI] [PubMed] [Google Scholar]
- 17.Lee H.U., Bae E.A., Kim D.H. Hepatoprotective effect of tectoridin and tectorigenin on tert-butyl hyperoxide-induced liver injury. Journal of Pharmacological Sciences. 2005;97:541–544. doi: 10.1254/jphs.scz040467. [DOI] [PubMed] [Google Scholar]
- 18.Lee J.S. Supplementation of Pueraria radix water extract on changes of antioxidant enzymes and lipid profile in ethanol-treated rats. Clinica Chimica Acta. 2004;347:121–128. doi: 10.1016/j.cccn.2004.04.002. [DOI] [PubMed] [Google Scholar]
- 19.Lee K.H., Morris-Natschke S., Qian K., Dong Y., Yang X., Zhou T., Belding E., Wu S.F., Wada K., Akiyama T. Recent progress of research on herbal products used in traditional Chinese medicine: the herbs belonging to the Divine Husbandman's Herbal Foundation Canon (神農本草經 Shén Nóng Běn Cǎo Jīng) Journal of Traditional and Complementary Medicine. 2012;2:6–26. doi: 10.1016/s2225-4110(16)30066-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lee M.K., Cho S.Y., Jang J.Y., Cho M.S., Jeon S.M., Jang M.K., Kim M.J., Park Y.B. Effects of Puerariae Flos and Puerariae Radix extracts on antioxidant enzymes in ethanol-treated rats. The American Journal of Chinese Medicine. 2001;29:343–354. doi: 10.1142/S0192415X01000368. [DOI] [PubMed] [Google Scholar]
- 21.Lee Y.J., Lee Y.M., Lee C.K., Jung J.K., Han S.B., Hong J.T. Therapeutic applications of compounds in the Magnolia family. Pharmacology & Therapeutics. 2011;130:157–176. doi: 10.1016/j.pharmthera.2011.01.010. [DOI] [PubMed] [Google Scholar]
- 22.Lei X.L., Chiou G.C. Studies on cardiovascular actions of Salvia miltiorrhiza. The American Journal of Chinese Medicine. 1986;14:26–32. doi: 10.1142/S0192415X86000053. [DOI] [PubMed] [Google Scholar]
- 23.Lian L.H., Wu Y.L., Song S.Z., Wan Y., Xie W.X., Li X., Bai T., Ouyang B.Q., Nan J.X. Gentiana manshurica Kitagawa reverses acute alcohol-induced liver steatosis through blocking sterol regulatory element-binding protein-1 maturation. Journal of Agricultural and Food Chemistry. 2010;58:13013–13019. doi: 10.1021/jf103976y. [DOI] [PubMed] [Google Scholar]
- 24.Lian Q. Advances in the study of chemical constituents and pharmacological actions of Cnidium monnieri (L) Cusson (Chinese) Journal of Chinese Medicinal Materials. 2003;26:141–144. [PubMed] [Google Scholar]
- 25.Lin C.F., Wong K.L., Wu R.S., Huang T.C., Liu C.F. Protection by hot water extract of Panax notoginseng on chronicethanol-induced hepatotoxicity. Phytotherapy Research. 2003;17:1119–1122. doi: 10.1002/ptr.1329. [DOI] [PubMed] [Google Scholar]
- 26.Ng T.B. Pharmacological activity of sanchi ginseng (Panax notoginseng) The Journal of Pharmacy and Pharmacology. 2006;58:1007–1019. doi: 10.1211/jpp.58.8.0001. [DOI] [PubMed] [Google Scholar]
- 27.Noh J.R., Kim Y.H., Gang G.T., Hwang J.H., Kim S.K., Ryu S.Y., Kim Y.S., Lee H.S., Lee C.H. Hepatoprotective effect of Platycodon grandiflorum against chronic ethanol-induced oxidative stress in C57BL/6 mice. Annals of Nutrition & Metabolism. 2011;58:224–231. doi: 10.1159/000330117. [DOI] [PubMed] [Google Scholar]
- 28.Okamoto T., Kawasaki T., Hino O. Osthole prevents anti- Fas antibody-induced hepatitis in mice by affecting the caspase- 3-mediated apoptotic pathway. Biochemical Pharmacology. 2003;65:677–681. doi: 10.1016/s0006-2952(02)01606-4. [DOI] [PubMed] [Google Scholar]
- 29.Okamoto T., Kobayashi T., Yoshida S. Chemical aspects of coumarin compounds for the prevention of hepatocellular carcinomas Current medicinal chemistry. Anti-cancer Agents. 2005;5:47–51. doi: 10.2174/1568011053352622. [DOI] [PubMed] [Google Scholar]
- 30.Pan M.H., Chiou Y.S., Tsai M.L., Ho C.T. Anti-inflammatory activity of traditional Chinese medicinal herbs Journal of Traditional and Complementary. Medicine. 2011;1:8–24. doi: 10.1016/s2225-4110(16)30052-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Seth D., Hogg P.J., Gorrell M.D., Mc Caughan G.W., Haber P.S. Direct effects of alcohol on hepatic fibrinolytic balance: implications for alcoholic liver disease. Journal of Hepatology. 2008;48:614–627. doi: 10.1016/j.jhep.2007.12.015. [DOI] [PubMed] [Google Scholar]
- 32.Sharma R.D., Raghuram T.C., Rao N.S. Effect of fenugreek seeds on blood glucose and serum lipids in type I diabetes. European Journal of Clinical Nutrition. 1990;44:301–306. [PubMed] [Google Scholar]
- 33.Sharma R.D., Sarkar A., Hazra D.K., Misra B., Singh J.B., Maheshwari B.B., Sharma S.K. Hypolipidaemic effect of fenugreek seeds: a chronic study in non-insulin dependent diabetic patients. Phytotherapy Research. 1996;10:332–334. [Google Scholar]
- 34.Stewart S., Jones D., Day C.P. Alcoholic liver disease: new insights into mechanisms and preventative strategies. Trends in Molecular Medicine. 2001;7:408–413. doi: 10.1016/s1471-4914(01)02096-2. [DOI] [PubMed] [Google Scholar]
- 35.Sun F., Xie M.L., Xue J., Wang H.B. Osthol regulates hepatic PPAR alpha-mediated lipogenic gene expression in alcoholic fatty liver murine. Phytomedicine. 2010;17:669–673. doi: 10.1016/j.phymed.2009.10.021. [DOI] [PubMed] [Google Scholar]
- 36.Sun F., Xie M.L., Zhu L.J., Xue J., Gu Z.L. Inhibitory effect of osthole on alcohol-induced fatty liver in mice. Digestive and Liver Disease. 2009;41:127–133. doi: 10.1016/j.dld.2008.01.011. [DOI] [PubMed] [Google Scholar]
- 37.Sun J., Huang S.H., Tan B.K., Whiteman M., Zhu Y.C., Wu Y.J., Ng Y., Duan W., Zhu Y.Z. Effects of purified herbal extract of Salvia miltiorrhiza on ischemic rat myocardium after acute myocardial infarction. Life Sciences. 2005;76:2849–2860. doi: 10.1016/j.lfs.2004.11.016. [DOI] [PubMed] [Google Scholar]
- 38.Tada A., Kaneiwa Y., Shoji J., Shibata S. Studies on the saponins of the root of Platycodon grandiflorum A De Candolle I Isolation and the structure of platycodin-D. Chemical & Pharmaceutical Bulletin. 1975;23:2965–2972. doi: 10.1248/cpb.23.2965. [DOI] [PubMed] [Google Scholar]
- 39.Thirunavukkarasu V., Anuradha C.V., Viswanathan P. Protective effect of fenugreek (Trigonella foenum-graecum)seeds in experimental ethanol toxicity. Phytotherapy Research. 2003;17:737–743. doi: 10.1002/ptr.1198. [DOI] [PubMed] [Google Scholar]
- 40.Wang A.Y., Lian L.H., Jiang Y.Z., Wu Y.L., Nan J.X. Gentiana manshurica Kitagawa prevents acetaminophen-induced acute hepatic injury in mice via inhibiting JNK/ERK MAPK pathway. World Journal of Gastroenterology. 2010;16:384–391. doi: 10.3748/wjg.v16.i3.384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wang N., Luo H.W., Niwa M., Ji J. A new platelet aggregation inhibitor from Salvia miltiorrhiza. Planta Medica. 1989;55:390–391. doi: 10.1055/s-2006-962037. [DOI] [PubMed] [Google Scholar]
- 42.Switzerland: WHO Press; World Health Organization. 2011. Global Status Report on Alcohol and Health. [Google Scholar]
- 43.Wu D., Cederbaum A.I. Oxidative stress mediated toxicity exerted by ethanol-inducible CYP2E1. Toxicology and Applied Pharmacology. 2005;207:70–76. doi: 10.1016/j.taap.2005.01.057. [DOI] [PubMed] [Google Scholar]
- 44.Xiong Y., Yang Y., Yang J., Chai H., Li Y., Jia Z., Wang Z. Tectoridin an isoflavone glycoside from the flower of Pueraria lobata, prevents acute ethanol-induced liver steatosis in mice. Toxicology. 2010;276:64–72. doi: 10.1016/j.tox.2010.07.007. [DOI] [PubMed] [Google Scholar]
- 45.Xiong Z.B., Wu P., Huang Y.F. Protective mechanisms of radix Salviae miltiorrhizae against chronic alcoholic liver injury in mice (Chinese) Chinese. Journal of Integrated Traditional and Western Medicine. 2005;25:425–428. [PubMed] [Google Scholar]
- 46.Yin H.Q., Choi Y.J., Kim Y.C., Sohn D.H., Ryu S.Y., Lee B.H. Salvia miltiorrhiza Bunge and its active component cryptotanshinone protects primary cultured rat hepatocytes from acute ethanol-induced cytotoxicity and fatty infiltration. Food and Chemical Toxicology. 2009a;47:98–103. doi: 10.1016/j.fct.2008.10.018. [DOI] [PubMed] [Google Scholar]
- 47.Yin H.Q., Je Y.T., Kim Y.C., Shin Y.K., Sung S., Lee K., Jeong G.S., Lee B.H. Magnolia officinalis reverses alcoholic fatty liver by inhibiting the maturation of sterol regulatory element- binding protein-1c. Journal of Pharmacological Sciences. 2009b;109:486–495. doi: 10.1254/jphs.08182fp. [DOI] [PubMed] [Google Scholar]
- 48.Yin H.Q., Kim Y.S., Choi Y.J., Kim Y.C., Sohn D.H., Ryu S.Y., Lee B.H. Effects of tanshinone IIA on the hepatotoxicity and gene expression involved in alcoholic liver disease. Archives of Pharmacal Research. 2008;31:659–665. doi: 10.1007/s12272-001-1209-2. [DOI] [PubMed] [Google Scholar]
- 49.Yin H.Q., Kim Y.C., Chung Y.S., Shin Y.K., Lee B.H. Honokiol reverses alcoholic fatty liver by inhibiting the maturation of sterol regulatory element binding protein-1c and the expression of its downstream lipogenesis genes. Toxicology and Applied Pharmacology. 2009c;236:124–130. doi: 10.1016/j.taap.2008.12.030. [DOI] [PubMed] [Google Scholar]
- 50.You M., Crabb D.W. Recent advances in alcoholic liver disease II Minireview: molecular mechanisms of alcoholic fatty liver. American Journal of Physiology Gastrointestinal and Liver Physiology. 2004;287:G1–6. doi: 10.1152/ajpgi.00056.2004. [DOI] [PubMed] [Google Scholar]
- 51.You M., Fischer M., Deeg M.A., Crabb D.W. Ethanol induces fatty acid synthesis pathways by activation of sterol regulatory element-binding protein (SREBP) The Journal of Biological Chemistry. 2002;277:29342–29347. doi: 10.1074/jbc.M202411200. [DOI] [PubMed] [Google Scholar]
- 52.Zhang J., Xue J., Wang H., Zhang Y., Xie M. Osthole improves alcohol-induced fatty liver in mice by reduction of hepatic oxidative stress. Phytotherapy Research. 2011;25:638–643. doi: 10.1002/ptr.3315. [DOI] [PubMed] [Google Scholar]
- 53.Zhang R., Hu Y., Yuan J., Wu D. Effects of Puerariae Radix extract on the increasing intestinal permeability in rat with alcohol- induced liver injury. Journal of Ethnopharmacology. 2009;126:207–214. doi: 10.1016/j.jep.2009.08.044. [DOI] [PubMed] [Google Scholar]


