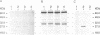Abstract
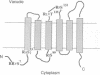
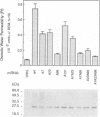
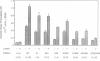
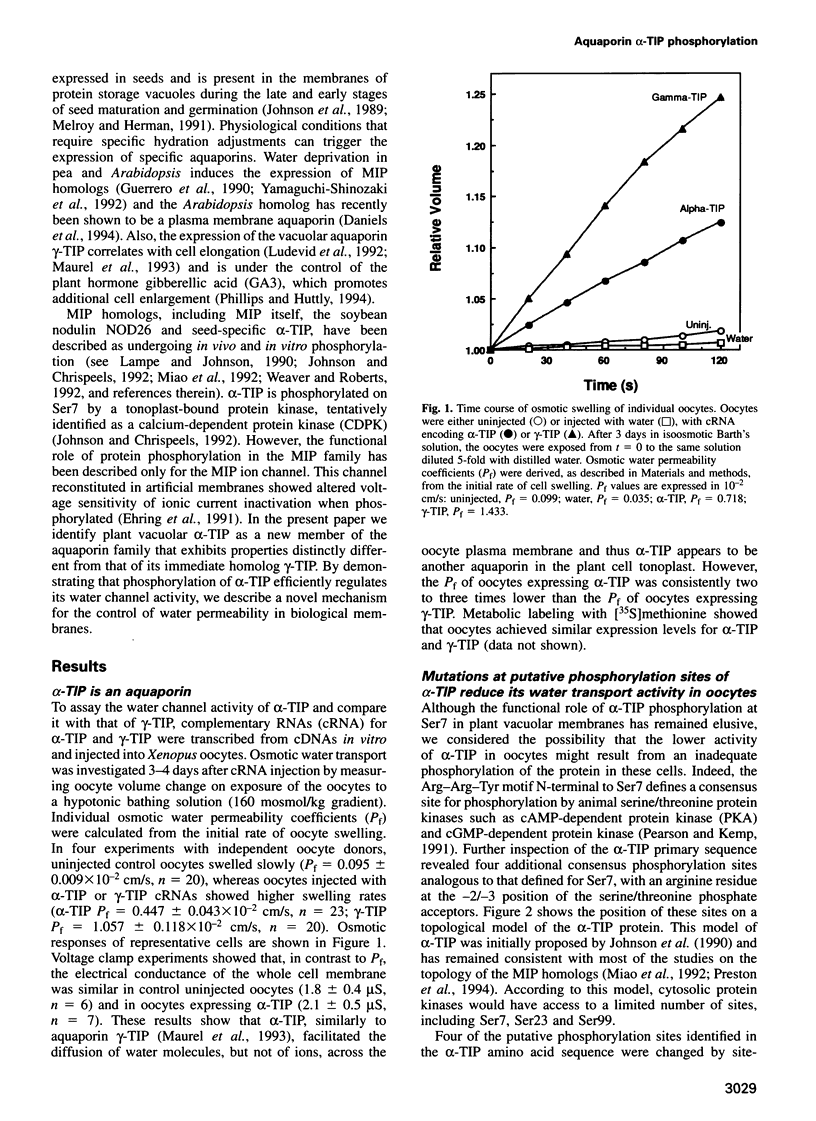
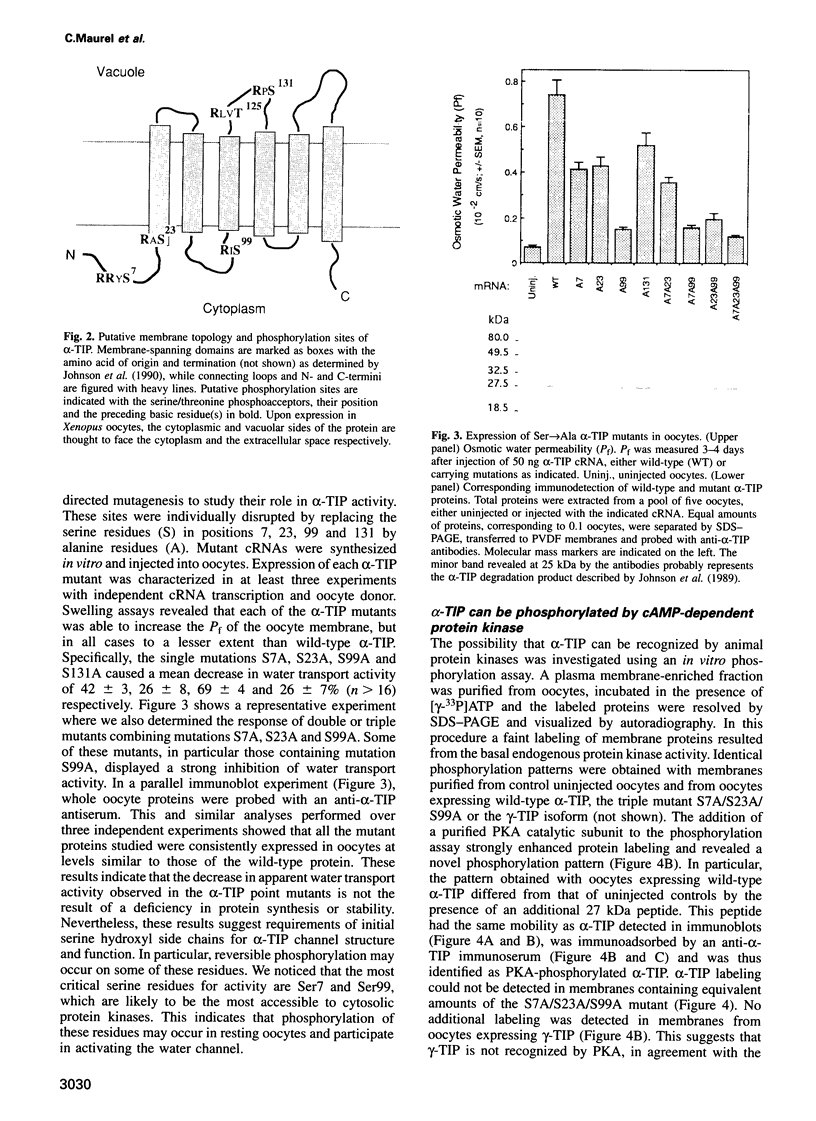
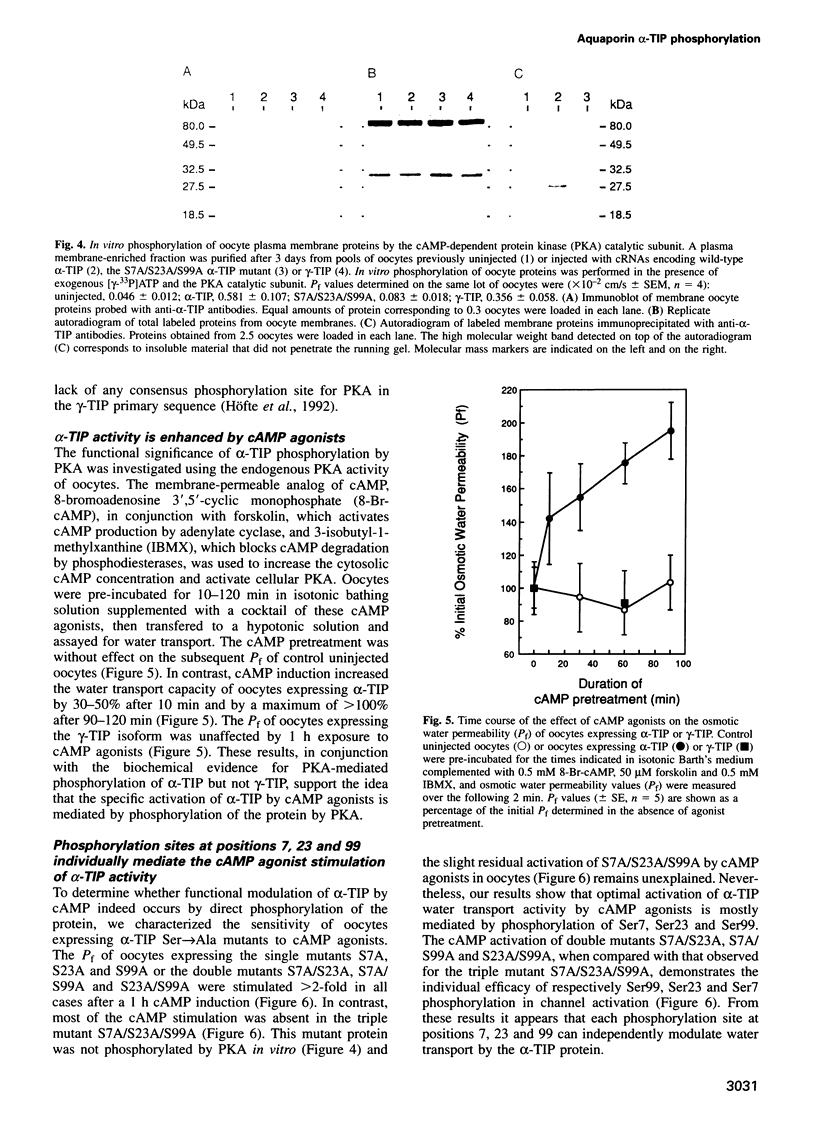
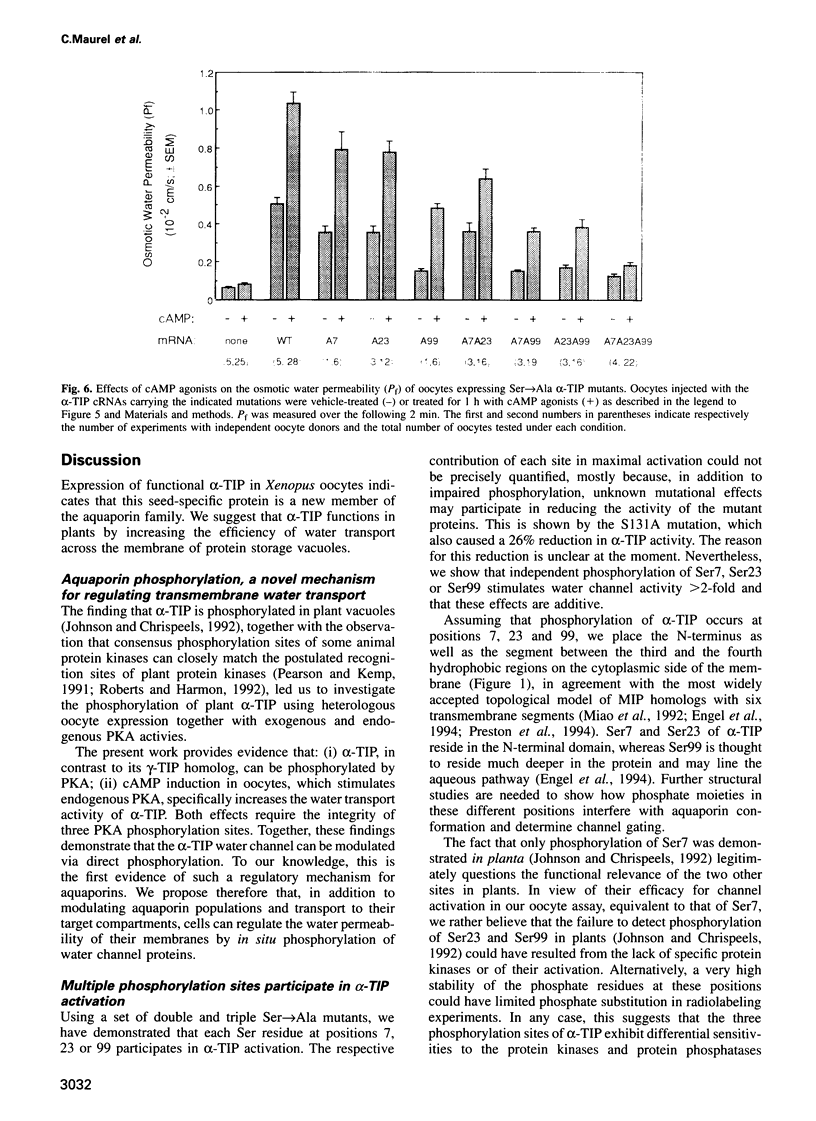
The vacuolar membrane protein alpha-TIP is a seed-specific protein of the Major Intrinsic Protein family. Expression of alpha-TIP in Xenopus oocytes conferred a 4- to 8-fold increase in the osmotic water permeability (Pf) of the oocyte plasma membrane, showing that alpha-TIP forms water channels and is thus a new aquaporin. alpha-TIP has three putative phosphorylation sites on the cytoplasmic side of the membrane (Ser7, Ser23 and Ser99), one of which (Ser7) has been shown to be phosphorylated. We present several lines of evidence that the activity of this aquaporin is regulated by phosphorylation. First, mutation of the putative phosphorylation sites in alpha-TIP (Ser7Ala, Ser23Ala and Ser99Ala) reduced the apparent water transport activity of alpha-TIP in oocytes, suggesting that phosphorylation of alpha-TIP occurs in the oocytes and participates in the control of water channel activity. Second, exposure of oocytes to the cAMP agonists 8-bromoadenosine 3',5'-cyclic monophosphate, forskolin and 3-isobutyl-1-methylxanthine, which stimulate endogenous protein kinase A (PKA), increased the water transport activity of alpha-TIP by 80-100% after 60 min. That the protein can be phosphorylated by PKA was demonstrated by phosphorylating alpha-TIP in isolated oocyte membranes with the bovine PKA catalytic subunit. Third, the integrity of the three sites at positions 7, 23 and 99 was necessary for the cAMP-dependent increase in the Pf of oocytes expressing alpha-TIP, as well as for in vitro phosphorylation of alpha-TIP. These findings demonstrate that the alpha-TIP water channel can be modulated via phosphorylation of Ser7, Ser23 and Ser99.(ABSTRACT TRUNCATED AT 250 WORDS)
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alexandre J., Lassalles J. P. Hydrostatic and osmotic pressure activated channel in plant vacuole. Biophys J. 1991 Dec;60(6):1326–1336. doi: 10.1016/S0006-3495(91)82170-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderberg R. J., Walker-Simmons M. K. Isolation of a wheat cDNA clone for an abscisic acid-inducible transcript with homology to protein kinases. Proc Natl Acad Sci U S A. 1992 Nov 1;89(21):10183–10187. doi: 10.1073/pnas.89.21.10183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brewster J. L., de Valoir T., Dwyer N. D., Winter E., Gustin M. C. An osmosensing signal transduction pathway in yeast. Science. 1993 Mar 19;259(5102):1760–1763. doi: 10.1126/science.7681220. [DOI] [PubMed] [Google Scholar]
- Chrispeels M. J., Maurel C. Aquaporins: the molecular basis of facilitated water movement through living plant cells? Plant Physiol. 1994 May;105(1):9–13. doi: 10.1104/pp.105.1.9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daniels M. J., Mirkov T. E., Chrispeels M. J. The plasma membrane of Arabidopsis thaliana contains a mercury-insensitive aquaporin that is a homolog of the tonoplast water channel protein TIP. Plant Physiol. 1994 Dec;106(4):1325–1333. doi: 10.1104/pp.106.4.1325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DiGiovanni S. R., Nielsen S., Christensen E. I., Knepper M. A. Regulation of collecting duct water channel expression by vasopressin in Brattleboro rat. Proc Natl Acad Sci U S A. 1994 Sep 13;91(19):8984–8988. doi: 10.1073/pnas.91.19.8984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Echevarria M., Frindt G., Preston G. M., Milovanovic S., Agre P., Fischbarg J., Windhager E. E. Expression of multiple water channel activities in Xenopus oocytes injected with mRNA from rat kidney. J Gen Physiol. 1993 Jun;101(6):827–841. doi: 10.1085/jgp.101.6.827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ehring G. R., Lagos N., Zampighi G. A., Hall J. E. Phosphorylation modulates the voltage dependence of channels reconstituted from the major intrinsic protein of lens fiber membranes. J Membr Biol. 1992 Feb;126(1):75–88. doi: 10.1007/BF00233462. [DOI] [PubMed] [Google Scholar]
- Fushimi K., Uchida S., Hara Y., Hirata Y., Marumo F., Sasaki S. Cloning and expression of apical membrane water channel of rat kidney collecting tubule. Nature. 1993 Feb 11;361(6412):549–552. doi: 10.1038/361549a0. [DOI] [PubMed] [Google Scholar]
- Galcheva-Gargova Z., Dérijard B., Wu I. H., Davis R. J. An osmosensing signal transduction pathway in mammalian cells. Science. 1994 Aug 5;265(5173):806–808. doi: 10.1126/science.8047888. [DOI] [PubMed] [Google Scholar]
- Guerrero F. D., Jones J. T., Mullet J. E. Turgor-responsive gene transcription and RNA levels increase rapidly when pea shoots are wilted. Sequence and expression of three inducible genes. Plant Mol Biol. 1990 Jul;15(1):11–26. doi: 10.1007/BF00017720. [DOI] [PubMed] [Google Scholar]
- Harris H. W., Jr, Strange K., Zeidel M. L. Current understanding of the cellular biology and molecular structure of the antidiuretic hormone-stimulated water transport pathway. J Clin Invest. 1991 Jul;88(1):1–8. doi: 10.1172/JCI115263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Homblé F., Véry A. A. Coupling of water and potassium ions in K channels of the tonoplast of Chara. Biophys J. 1992 Oct;63(4):996–999. doi: 10.1016/S0006-3495(92)81666-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Höfte H., Hubbard L., Reizer J., Ludevid D., Herman E. M., Chrispeels M. J. Vegetative and Seed-Specific Forms of Tonoplast Intrinsic Protein in the Vacuolar Membrane of Arabidopsis thaliana. Plant Physiol. 1992 Jun;99(2):561–570. doi: 10.1104/pp.99.2.561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson K. D., Chrispeels M. J. Tonoplast-bound protein kinase phosphorylates tonoplast intrinsic protein. Plant Physiol. 1992 Dec;100(4):1787–1795. doi: 10.1104/pp.100.4.1787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson K. D., Herman E. M., Chrispeels M. J. An abundant, highly conserved tonoplast protein in seeds. Plant Physiol. 1989 Nov;91(3):1006–1013. doi: 10.1104/pp.91.3.1006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson K. D., Höfte H., Chrispeels M. J. An intrinsic tonoplast protein of protein storage vacuoles in seeds is structurally related to a bacterial solute transporter (GIpF). Plant Cell. 1990 Jun;2(6):525–532. doi: 10.1105/tpc.2.6.525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurosaki F., Nishi A. Stimulation of calcium influx and calcium cascade by cyclic AMP in cultured carrot cells. Arch Biochem Biophys. 1993 Apr;302(1):144–151. doi: 10.1006/abbi.1993.1192. [DOI] [PubMed] [Google Scholar]
- Lampe P. D., Johnson R. G. Amino acid sequence of in vivo phosphorylation sites in the main intrinsic protein (MIP) of lens membranes. Eur J Biochem. 1990 Dec 12;194(2):541–547. doi: 10.1111/j.1432-1033.1990.tb15650.x. [DOI] [PubMed] [Google Scholar]
- Li W., Luan S., Schreiber S. L., Assmann S. M. Cyclic AMP stimulates K+ channel activity in mesophyll cells of Vicia faba L. Plant Physiol. 1994 Nov;106(3):957–961. doi: 10.1104/pp.106.3.957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ludevid D., Höfte H., Himelblau E., Chrispeels M. J. The Expression Pattern of the Tonoplast Intrinsic Protein gamma-TIP in Arabidopsis thaliana Is Correlated with Cell Enlargement. Plant Physiol. 1992 Dec;100(4):1633–1639. doi: 10.1104/pp.100.4.1633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maeshima M., Hara-Nishimura I., Takeuchi Y., Nishimura M. Accumulation of Vacuolar H+-Pyrophosphatase and H+-ATPase during Reformation of the Central Vacuole in Germinating Pumpkin Seeds. Plant Physiol. 1994 Sep;106(1):61–69. doi: 10.1104/pp.106.1.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maurel C., Reizer J., Schroeder J. I., Chrispeels M. J., Saier M. H., Jr Functional characterization of the Escherichia coli glycerol facilitator, GlpF, in Xenopus oocytes. J Biol Chem. 1994 Apr 22;269(16):11869–11872. [PubMed] [Google Scholar]
- Maurel C., Reizer J., Schroeder J. I., Chrispeels M. J. The vacuolar membrane protein gamma-TIP creates water specific channels in Xenopus oocytes. EMBO J. 1993 Jun;12(6):2241–2247. doi: 10.1002/j.1460-2075.1993.tb05877.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miao G. H., Hong Z., Verma D. P. Topology and phosphorylation of soybean nodulin-26, an intrinsic protein of the peribacteroid membrane. J Cell Biol. 1992 Jul;118(2):481–490. doi: 10.1083/jcb.118.2.481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen S., DiGiovanni S. R., Christensen E. I., Knepper M. A., Harris H. W. Cellular and subcellular immunolocalization of vasopressin-regulated water channel in rat kidney. Proc Natl Acad Sci U S A. 1993 Dec 15;90(24):11663–11667. doi: 10.1073/pnas.90.24.11663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearson R. B., Kemp B. E. Protein kinase phosphorylation site sequences and consensus specificity motifs: tabulations. Methods Enzymol. 1991;200:62–81. doi: 10.1016/0076-6879(91)00127-i. [DOI] [PubMed] [Google Scholar]
- Phillips A. L., Huttly A. K. Cloning of two gibberellin-regulated cDNAs from Arabidopsis thaliana by subtractive hybridization: expression of the tonoplast water channel, gamma-TIP, is increased by GA3. Plant Mol Biol. 1994 Feb;24(4):603–615. doi: 10.1007/BF00023557. [DOI] [PubMed] [Google Scholar]
- Preston G. M., Carroll T. P., Guggino W. B., Agre P. Appearance of water channels in Xenopus oocytes expressing red cell CHIP28 protein. Science. 1992 Apr 17;256(5055):385–387. doi: 10.1126/science.256.5055.385. [DOI] [PubMed] [Google Scholar]
- Preston G. M., Jung J. S., Guggino W. B., Agre P. Membrane topology of aquaporin CHIP. Analysis of functional epitope-scanning mutants by vectorial proteolysis. J Biol Chem. 1994 Jan 21;269(3):1668–1673. [PubMed] [Google Scholar]
- Stanssens P., Opsomer C., McKeown Y. M., Kramer W., Zabeau M., Fritz H. J. Efficient oligonucleotide-directed construction of mutations in expression vectors by the gapped duplex DNA method using alternating selectable markers. Nucleic Acids Res. 1989 Jun 26;17(12):4441–4454. doi: 10.1093/nar/17.12.4441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Urao T., Katagiri T., Mizoguchi T., Yamaguchi-Shinozaki K., Hayashida N., Shinozaki K. Two genes that encode Ca(2+)-dependent protein kinases are induced by drought and high-salt stresses in Arabidopsis thaliana. Mol Gen Genet. 1994 Aug 15;244(4):331–340. doi: 10.1007/BF00286684. [DOI] [PubMed] [Google Scholar]
- Weaver C. D., Roberts D. M. Determination of the site of phosphorylation of nodulin 26 by the calcium-dependent protein kinase from soybean nodules. Biochemistry. 1992 Sep 22;31(37):8954–8959. doi: 10.1021/bi00152a035. [DOI] [PubMed] [Google Scholar]