Abstract
An urban epidemic of dengue in Senegal during 2009 affected 196 persons and included 5 cases of dengue hemorrhagic fever and 1 fatal case of dengue shock syndrome. Dengue virus serotype 3 was identified from all patients, and Aedes aegypti mosquitoes were identified as the primary vector of the virus.
Keywords: dengue, outbreak, Senegal, epidemic, dengue fever, dengue virus, viruses, DENV-3 genotype 3
Dengue is an arboviral disease transmitted by Aedes spp. mosquitoes and caused by 4 serotypes of dengue virus (DENV): DENV-1, DENV-2, DENV-3, and DENV-4. DENV belongs to the family Flaviviridae, genus Flavivirus (1). More than 2.5 billion persons worldwide are considered at risk for dengue (2); the number of dengue infections per year has been estimated at 390 million, although only 96 million are symptomatic (3).
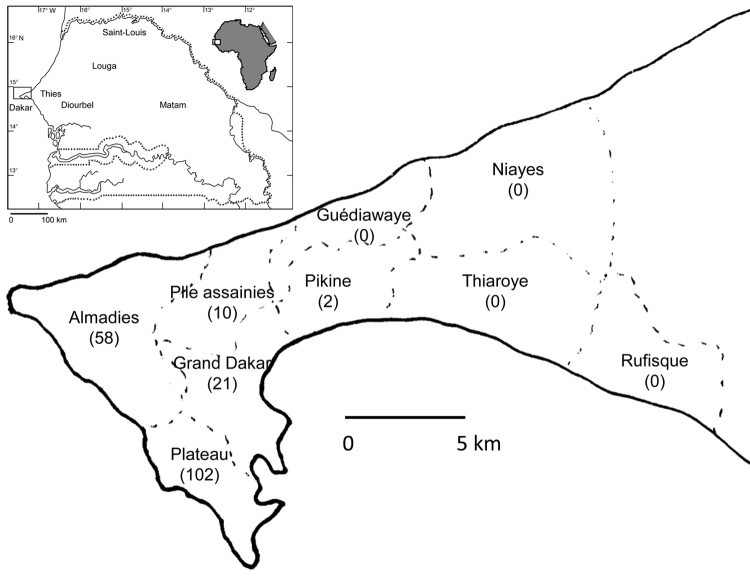
Most dengue infections occur in urban areas in tropical and subtropical regions, but imported cases have been reported in nontropical regions. During October 2009, imported DENV-3 infections were diagnosed in Turin, Italy (4), and Marseille, France (5), from patients returning from the Louga and Thies regions in Senegal (Figure 1). DENV-specific IgM and/or RNA were also detected in 5 persons living in Dakar, Senegal, who were suspected to have dengue. We report results from investigation of the 2009 dengue epidemic in Senegal.
Figure 1.
Geographic distribution of patients with confirmed dengue in the region of Dakar, Senegal. Number of patients is shown in parentheses. Inset shows location of Dakar in Senegal and in Africa.
The Study
During October 2009–January 2010, a total of 696 serum samples were collected from persons in Senegal who were suspected to have dengue. A suspected dengue case was defined as fever and >2 of the following: myalgia, arthralgia, headache, and rash. Most samples were collected in Dakar (n = 606) and Thies (n = 87) (Table, Appendix). In Dakar, samples were collected from 9 neighborhoods: Plateau (n = 202), Almadies (n = 157), Grand Dakar (n = 117), Parcelles Assainies (n = 82), Pikine (n = 13), Guediawaye (n = 15), Niayes (n = 5), Thiaroye (n = 10), and Rufisque (n = 5) (Figure 1). Samples were tested by real-time reverse transcription PCR and ELISA for virus genome and IgM, respectively.
Table. Epidemiologic and biological characteristics of suspected and confirmed dengue cases, Senegal, 2009*.
| Characteristic | Dakar |
Thies | Louga | Other† | Total | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pateau | Almadies | Grand Dakar | Parcelle | Pikine | Guediawaye | Niayes | Thiaroye | Rufisque | |||||
| Suspected cases | 202 | 157 | 117 | 82 | 13 | 15 | 5 | 10 | 5 | 87 | 1‡ | 3 | 696 |
| Patient sex | |||||||||||||
| M | 91 | 60 | 56 | 44 | 8 | 10 | 2 | 6 | 2 | 38 | 1‡ | 3 | 320 |
| F | 111 | 97 | 61 | 38 | 5 | 5 | 3 | 4 | 3 | 49 | 0 | 0 | 376 |
| Patient age, y, median (range) | 30 (1–76) | 29 (2–79) | 38 (5–93) | 28 (3–60) | 25 (9–55) | 29 (20–35) | 33 (11–56) | 40 (8–78) | 45 (26–69) | 30 (6–69) | 32 | 53 | |
| Health facility visited | |||||||||||||
| Private laboratory | 144 | 125 | 64 | 40 | 2 | 0 | 5 | 0 | 5 | 0 | 1‡ | 0 | 385 |
| Private clinic | 23 | 10 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 34 |
| Public health | 35 | 22 | 53 | 42 | 10 | 15 | 0 | 10 | 0 | 87 | 0 | 3 | 277 |
| Patient nationality | |||||||||||||
| Senegalese | 96 | 98 | 89 | 82 | 13 | 15 | 5 | 10 | 5 | 87 | 1 | 3 | 504 |
| Lebanese | 86 | 11 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 98 |
| French | 13 | 33 | 6 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 52 |
| Lusophone | 5 | 14 | 20 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 39 |
| Chinese |
2 |
1 |
1 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
0 |
4 |
| Confirmed cases | 120 | 30 | 27 | 14 | 2 | 0 | 0 | 0 | 0 | 3 | 1‡ | 0 | 196 |
| Patient sex | |||||||||||||
| M | 56 | 12 | 12 | 8 | 0 | 0 | 0 | 0 | 0 | 1 | 1‡ | 0 | 89 |
| F | 64 | 18 | 15 | 6 | 2 | 0 | 0 | 0 | 0 | 2 | 0 | 0 | 107 |
| Patient age, y, median (range) | 32 (1–70) | 26 (6–42) | 44 (18–93) | 26 (15–37) | 18 (16–20) | NA | NA | NA | NA | 42 (20–57) | NA | NA | |
| WHO disease classification | |||||||||||||
| DF | 115 | 29 | 27 | 14 | 2 | 0 | 0 | 0 | 0 | 3 | 1‡ | 0 | 190 |
| DHF | 4 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 5 |
| DSS | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Laboratory testing conducted | 47 | 10 | 9 | NA | NA | NA | NA | NA | NA | NA | NA | NA | 66 |
| Leukopenia | 34 | 7 | 5 | NA | NA | NA | NA | NA | NA | NA | NA | NA | 46 |
| Thrombocytopenia | 32 | 5 | 4 | NA | NA | NA | NA | NA | NA | NA | NA | NA | 41 |
| Patient hospitalized | 16 | 10 | 5 | NA | NA | NA | NA | NA | NA | NA | NA | NA | 31 |
| Patient nationality | |||||||||||||
| Senegalese | 46 | 14 | 12 | 14 | 2 | 0 | 0 | 0 | 0 | 2 | 1‡ | 0 | 90 |
| Lebanese | 63 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 65 |
| Lusophone | 3 | 5 | 12 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 20 |
| French | 7 | 9 | 3 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 20 |
| Chinese | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
*Values are no. patients except as indicated. NA, not available; WHO, World Health Organization; DF, dengue fever; DHF, dengue hemorrhagic fever; DSS, dengue shock syndrome. †Diourbel, Saint Louis, and Matam. ‡Case identified and diagnosed in Italy.
In conjunction with human testing, mosquito sampling was performed during December 2009 in households with confirmed dengue cases, and entomologic risk indexes (i.e., Breteau and container indices [6]) were evaluated. DENV isolation and identification was performed on homogenized mosquitoes and human serum samples injected into mosquito cell lines, as described (7).
Partial DENV envelope protein coding regions were amplified by reverse transcription PCR (8), purified after electrophoresis from agarose gels, and sequenced for dengue serotype identification and phylogenetic analyses. A total of 196 (28.2%) suspected dengue cases were confirmed as dengue. Among confirmed case-patients, 45.9% (90/196), 33.0% (65/196), 10.0% (20/196), 10.2% (20/196), and 0.05% (1/196) belonged to the Senegalese, Lebanese, Lusophone, French, and Chinese communities, respectively; the Lusophone community includes persons from Cape Verde and Guinea-Bissau who live in Senegal. The ratio of confirmed to suspected cases was 17.8% (90/504), 66.3% (65/98), 51.2% (20/39), 38.4% (20/52), and 25.0% (1/4) for the Senegalese, Lebanese, Lusophone, French, and Chinese communities, respectively.
Among the 196 confirmed case-patients, 31 were hospitalized; 5 were found to have dengue hemorrhagic fever (DHF), and 1 died of dengue shock syndrome. The M:F sex ratio for the case-patients was 0.83 (89 male, 107 female), and the median age was 31 years (range 1–93).
The fatal case was in a 71-year-old Lebanese woman hospitalized in a private clinic in Dakar on October 30 with a 4-day history of fever; she had severe thrombocytopenia and elevated transaminase levels. Despite treatment (voluven and adrenaline), she died of cardiac arrest. Disseminated intravascular coagulation was probably responsible for the hemorrhagic syndrome.
Of the 5 other patients (4 Lebanese and 1 Senegalese) who had hemorrhagic manifestations, all had fever, epistaxis, and melena (5/5) associated with thrombocytopenia (platelet count 50–90 × 109/L) and leukopenia (leukocyte count 2.5–3.5 × 109 cells/L); 4 reported headache, and 3 reported myalgia/arthralgia. All 5 patients were hospitalized and received transfusions of fresh frozen plasma, platelet concentrates, and other supportive treatments. Dengue-specific IgM and/or RNA were detected in serum samples collected 2 or 8 days after the onset of symptoms. All patients recovered and were discharged from the hospital after 8 or 10 days.
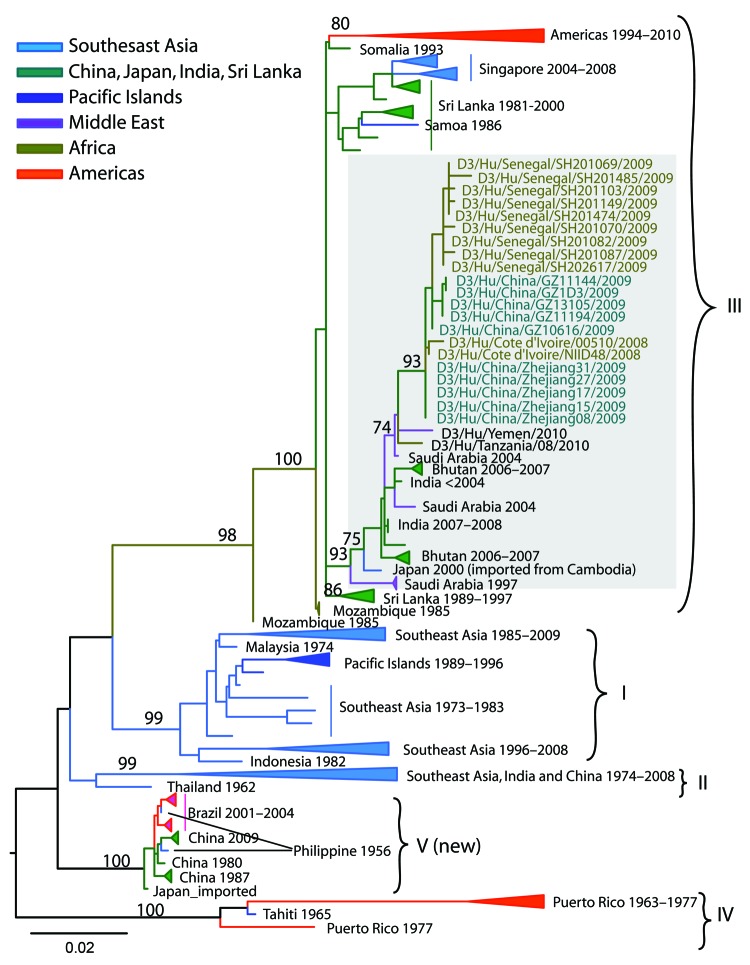
Using mosquito continuous cell lines, we recovered 49 DENV-3 isolates from confirmed case-patients. Phylogenetic analysis of sequences from mosquito and human samples revealed that DENV-3 genotype III closely related to isolates circulating in Côte d’Ivoire (2008) and China (2009) was circulating during the Senegal outbreak (Figure 2). Dates of illness onset of confirmed cases indicated that the outbreak started in late September 2009, peaked in mid-November, and declined in mid-December.
Figure 2.
Maximum-likelihood phylogenetic tree of dengue virus serotype 3 (D3) sequences from Senegal compared with other sequences. The tree was constructed on the basis of an 885-bp segment of the envelope protein gene. Bootstrap values >70 are labeled next to the node. Sequences from different geographic areas are shown by different colors. Gray shading indicates sequences from Senegal and closely related strains. Scale bar indicates nucleotide substitutions per site.
The entomologic investigation found high epidemic risk in all localities infested with DENV vectors. The Breteau index ranged from 6.6 to 195.2 in Dakar, 1.6 to 32.7 in Louga, and 1.1 to 14.9 in Thies, whereas the container indices ranged from 15 to 63.2 in Dakar, 5.3 to 15.2 in Thies, and 14.3 to 64.2 in Louga. A total of 5,730 mosquitoes were collected; these belonged to 8 species: Aedes aegypti, Anopheles gambiae, Culex quinquefasciatus, Cx. tigripes, Cx. tritaeniorhynchus, Cx. antennatus, Cx. ethiopicus, and Cx. nebulosus. Most mosquitoes were collected as adults, but 1,675 emerged from larvae that were collected in the field. Cx quinquefasciatus mosquitoes predominated, but Ae. aegypti mosquitoes, the only dengue vector collected, were found in all sites. DENV-3 was detected from 3 pools of mosquitoes collected in 2 neighborhoods of Dakar: Plateau (2 pools) and Parcelles Assainies (1 pool).
Conclusions
An epidemic of DENV-3 occurred in Senegal during September–December 2009; of the 196 laboratory-confirmed cases, most (193) occurred in Dakar. This finding could indicate that transmission rates were higher in this urban area but may have been the result of bias in sample collection; dengue surveillance was less active at health facilities in in other regions, which provided only 277 (39%) of the 696 samples collected from persons who were suspected to have dengue. Fever, headache, myalgia, vomiting, thrombocytopenia, and leukopenia were the most frequent signs and symptoms among patients with confirmed dengue, as described (9).
The proportion of DHF cases in this outbreak seemed to be high at 3% when compared with previous reports in the Americas from the 1980s through 2007, in which DHF rates ranged from 1.3% to 2.4% (10). However, our sample size was limited and lacks confirmatory power.
The percentage of confirmed dengue cases among suspected cases in different communities showed that persons in Senegalese communities were significantly less affected than those in Leebanese (χ2 = 98.3, df = 1; p<0.0001), Lusophone (χ2 = 23.3, df = 1; p<0.0001), and French (χ2 = 11.3, df = 1; p<0.001) communities. Moreover, 5 (83.3%) of the 6 cases with hemorrhagic manifestations occurred in the Lebanese community, which suggests that disease severity might be associated with community exposure.
Ae. aegypti mosquitoes were the most likely vector of DENV-3 transmission during this epidemic, given their association with DENV and the overlap of their distribution and abundance with the locations of dengue confirmed cases. Ae. aegypti mosquitoes are known to be a competent vector for DENV in West Africa (11) and could maintain DENV-3 through vertical transmission in Senegal, as described (12). The absence of suspected or confirmed dengue cases in Louga, despite the high density of Ae. aegypti mosquitoes, suggests that the first reported case-patient contracted the DENV infection elsewhere, possibly in Dakar, which he had visited several times.
The phylogenetic analysis of DENV-3 strains isolated during the outbreak suggests that they belong to genotype III and are closely related to DENV-3 isolated from Côte d’Ivoire and China in 2008 and 2009, respectively (13). Hence, the strain responsible for this outbreak may have been introduced into Senegal by travelers from Asia or from Côte d’Ivoire.
Our findings suggest that increased urban dengue activity is plausible in Senegal. Given ongoing population growth, explosive urbanization, infrastructure building, and international travel, dengue surveillance and preparedness should be reinforced. Furthermore, phylogenetic studies incorporating more DENV-3 strains would shed light on the origins of this DENV-3 outbreak.
Acknowledgments
We thank Magueye Ndiaye, Modou Diagne, Carlos Fortez, Moussa Dia, and Oumar Ndiaye for excellent technical assistance with laboratory diagnosis and field investigations. We also thank the authorities and the field agents of the Ministry of Health of Senegal for facilitating the investigation of this outbreak.
This work was supported by grants from the Institut Pasteur de Dakar, Senegal; the Ministry of Health of Senegal; and the US National Institutes of Health (grant AI069145).
Biography
Dr Ousmane Faye is a virologist and specialist in acarology who works at the Arbovirus and Viral Haemorrhagic Fever Unit, Institut Pasteur de Dakar, Senegal. His primary research interests include the vectorial transmission of arboviruses and viruses causing hemorrhagic fevers.
Footnotes
Suggested citation for this article: Faye Ou, Ba Y, Faye Om, Talla C, Diallo D, Chen R, et al. Urban epidemic of dengue virus serotype 3 infection, Senegal, 2009. Emerg Infect Dis [Internet]. 2014 Mar [date cited]. http://dx.doi.org/10.3201/eid2003.121885
References
- 1.Lindenbach B, Thiel H, Rice C. Flaviviridae: the virus and their replication. In: Knipe DM, Howley PM, editors. Fields virology, 5th ed. Philadelphia: Lippincott, Williams & Wilkins; 2007. p. 1101–52. [Google Scholar]
- 2.World Health Organization. Dengue: guidelines for diagnosis, treatment, prevention, and control. Geneva. Organization. 2009;•••:174. [PubMed] [Google Scholar]
- 3.Bhatt S, Gething PW, Brady OJ, Messina JP, Farlow AW, Moyes CL, et al. The global distribution and burden of dengue. Nature. 2013;496:504–7 . 10.1038/nature12060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Nisii C, Carletti F, Castilletti C, Bordi L, Meschi S, Selleri M, et al. A case of dengue type 3 virus infection imported from Africa to Italy, October 2009. Euro Surveill. 2010;15:19487 . [PubMed] [Google Scholar]
- 5.Franco L, Di Caro A, Carletti F, Vapalahti O, Renaudat C, Zeller H, et al. Recent expansion of dengue virus serotype 3 in West Africa. Euro Surveill. 2010;15:19490 . [PubMed] [Google Scholar]
- 6.Lok CK. Methods and indices used in the surveillance of dengue vectors. Mosquito Borne Diseases Bulletin. 1985;1:79–81. [Google Scholar]
- 7.Digoutte JP, Calvo-Wilson MA, Mondo M, Traore-Lamizana M, Adam F. Continuous cell lines and immune ascitic fluid pools in arbovirus detection. Res Virol. 1992;143:417–22 . 10.1016/S0923-2516(06)80135-4 [DOI] [PubMed] [Google Scholar]
- 8.Gaunt MW, Sall AA, De Lamballerie X, Falconar AI, Dzhivanian TI, Gould AG. Phylogenetic relationships of flaviviruses correlate with their epidemiology, disease association and biogeography. J Gen Virol. 2001;82:1867–76 . [DOI] [PubMed] [Google Scholar]
- 9.Aggarwal A, Chandra J, Aneja S, Patwari AK, Dutta AK. An epidemic of dengue hemorrhagic fever and dengue shock syndrome in children in Delhi. Indian Pediatr. 1998;35:727–32 . [PubMed] [Google Scholar]
- 10.San Martín JL, Brathwaite O, Zambrano B, Solórzano JO, Bouckenooghe A, Dayan GH, et al. The epidemiology of dengue in the Americas over the last three decades: a worrisome reality. Am J Trop Med Hyg. 2010;82:128–35 . 10.4269/ajtmh.2010.09-0346 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Vazeille M, Yébakima A, Lourenço-de-Oliveira R, Andriamahefazafy B, Correira A, Rodrigues JM, et al. Oral receptivity of Aedes aegypti from Cape Verde for yellow fever, dengue, and chikungunya viruses. Vector Borne Zoonotic Dis. 2013;13:37–40 . 10.1089/vbz.2012.0982 [DOI] [PubMed] [Google Scholar]
- 12.Joshi V, Mourya DT, Sharma RC. Persistence of dengue-3 virus through transovarial transmission passage in successive generations of Aedes aegypti mosquitoes. Am J Trop Med Hyg. 2002;67:158–61 . [DOI] [PubMed] [Google Scholar]
- 13.Sun J, Lin J, Yan J, Fan W, Lu L, Lv H, et al. Dengue virus serotype 3 subtype III, Zhejiang Province, China. Emerg Infect Dis. 2011;17:321–3 . 10.3201/eid1702.100396 [DOI] [PMC free article] [PubMed] [Google Scholar]