Abstract
Background
Vitamin D deficiency during pregnancy has been associated with increased risk of complications and adverse perinatal outcomes. We evaluated seasonal variation of 25(OH)D among pregnant women, focusing on patterns and determinants of variation.
Methods
Data came from three cohort studies in the US that included 2,583 non-Hispanic Black and White women having prenatal 25(OH)D concentrations determined. Fourier time series and generalised linear models were used to estimate the magnitude of 25(OH)D seasonality. We modelled seasonal variability using a stationary cosinor model to estimate the phase shift, peak-trough difference, and annual mean of 25(OH)D.
Results
We observed a peak for 25(OH)D in summer, a nadir in winter, and a phase of 8 months, which resulted from fluctuations in 25(OH)D3 rather than 25(OH)D2. After adjustment for covariates, the annual mean concentrations and estimated peak–trough difference of 25(OH)D among Black women were 19.8 ng/mL (95% CI 18.9, 20.5), and 5.8 ng/mL (95% CI 4.7, 6.7), and for non-Hispanic White women, 33.0 ng/mL (95% CI 32.6, 33.4) and 7.4 ng/mL (95% CI 6.0, 8.9).
Conclusions
Non-Hispanic Black women had lower average 25(OH)D concentrations throughout the year and smaller seasonal variation levels than non-Hispanic White women. This study's confirmation of 25(OH)D seasonality over a calendar year, has the potential to enhance public health interventions targeted to improve maternal and perinatal outcomes.
Keywords: 25-hydroxyvitamin D, vitamin D, pregnant women, season, epidemiology
Vitamin D, a nutrient and a prohormone, is essential for normal absorption of calcium from the intestine. There are two mains forms: cholecalciferol (D3) and ergocalciferol (D2). The D3 form is obtained from synthesis in the skin with exposure to ultraviolet radiation (UVR) and from dietary sources such as oily fish and supplements. D2 is essentially only available from supplements as it is synthetically derived from plant-based sources. Both forms are metabolized in the liver to circulating 25-hydroxyvitamin D [25(OH)D2 and 25(OH)D3, herein denoted as 25(OH)D unless specified].1-4 Serum 25(OH)D concentrations, the active metabolite used to assess vitamin D status, can vary substantially within individuals and across populations over the calendar year and are highest at the end of summer and lowest at the end of winter.5 This variation is likely due to seasonal differences in sunlight intensity during summer when individuals receive more UVR exposure.6 In addition to regulating calcium metabolism, vitamin D helps maintain normal concentrations of blood glucose, regulates hormonal secretions and controls cellular growth and differentiation.7
Vitamin D deficiency during pregnancy has received increased attention due to its association with increased risk of medical complications during pregnancy and other adverse perinatal outcomes including preeclampsia,8, 9 gestational diabetes,10, 11 and increased caesarian sections.12 Studies have shown that maternal antepartum 25(OH)D concentration level is a strong predictor of newborn vitamin D status;13, 14 infants of mothers with deficient vitamin D concentrations experience depleted vitamin D concentrations in utero and are born with low vitamin D stores that must carry them through the first few months of life.13, 15
Seasonal and racial disparities of 25(OH)D serum concentrations have been described in specific subgroups of the general US population.16 Blacks synthesize less cutaneous vitamin D than other racial groups.17, 18 Studies have found racial disparities of 25(OH)D among pregnant women, with Black pregnant women having lower concentrations compared to white pregnant women.19 While a clear seasonal pattern in 25(OH)D was observed for White pregnant women, seasonal variation was not observed for Black pregnant women, in part due to limited sample size.20 Increasing our understanding of seasonal variability of vitamin D among pregnant women could have a tremendous public health impact as it can inform clinical vitamin D supplementation strategies and behavior recommendations for pregnant women of all races.
Therefore, we aimed to evaluate and describe seasonal variability of vitamin D focusing on patterns and determinants of variation among non-Hispanic Black and White pregnant women by geographical and maternal sociodemographic characteristics. We also aimed to examine the extent to which seasonal changes of 25(OH)D are principally due to the variability of vitamin D2 or D3.
Methods
Data and study settings
Data used for this research were drawn from three prospective cohort studies of pregnant women; each of the studies included a questionnaire, medical records and archived serum samples. Participating cohorts included the Omega study, the Pregnancy, Infection and Nutrition (PIN) study, and the Pregnancy Outcomes and Community Health (POUCH) study. Detailed descriptions of the cohorts are published.21-23 Briefly, investigators in the Omega study (1996–2008) prospectively followed pregnant women attending prenatal care clinics affiliated with the Swedish Medical Center and Tacoma General Hospital in Seattle and Tacoma, Washington, respectively. The Omega study was designed to assess the influence of maternal diet, physical activity and other lifestyle characteristics on the incidence of preeclampsia, gestational diabetes mellitus and other adverse pregnancy outcomes. In the PIN study pregnant women were recruited from selected prenatal care clinics in North Carolina over three study waves: PIN1 (1995–2005), PIN2 (1999–2001) and PIN3 (2000–2005). The PIN study was designed to investigate the role of infections, stress, physical activity and nutrition on preterm delivery. Investigators of the POUCH study (1998–2004) prospectively followed pregnant women recruited at the time of maternal serum alpha-fetoprotein (MSAFP) screening during prenatal visits from 52 select clinics in five Michigan communities. POUCH was designed to investigate infectious, maternal vascular disease, and stress pathways to preterm delivery.
In all three cohorts, participants were invited to provide blood samples and participate in an in-person interview at enrollment. Collected maternal blood samples were frozen at -70 degrees Celsius and stored until analysis. Our study, included data from women who were enrolled in a cohort at less than 29 weeks of gestation and who completed enrollment and had a single initial blood draw prior to 29 weeks of gestation. The present study was restricted to singleton pregnancies among non-Hispanic Black and White women sampled as part of a multi-cohort, nested case-control study.24 Maternal medical records were abstracted and we included women who had information on 25(OH)D serum concentration ascertained with the pregnancy week at draw and date of draw. After these restrictions, this study's final sample size included 2,583 pregnant women who delivered preterm (870) or at term (1713).
Ethical approval for each study was granted by the Institutional Review Boards (IRB) at the respective institution (Swedish Medical Center and Tacoma General Hospital for Omega, University of North Carolina at Chapel Hill for PIN, and Michigan State University, Michigan Department of Community Health and participating community hospitals for POUCH). All participants provided written informed consent.
Laboratory measurements
Serum aliquots were shipped to a single laboratory, participating in the Vitamin D Quality Assessment Scheme (DEQAS, www.deqas.org), for assay of total serum 25OHD (25OHD2 and 25OHD3) by liquid chromatography-tandem mass spectrometry (LC-MS/MS).25 Samples from each cohort were assayed together with separate runs by cohort. Before analysis, serum samples were derivatized with 4-phenyl-1,2,4-triazoline-3,5-dione (PTAD) to improve sensitivity of assays at low concentration of analyte.26 Structural epimers were not assayed. The lower limits of detection for 25(OH)D3 and 25(OH)D2 were 0.6 and 1.1 ng/mL, respectively. For quality control purposes, we used standard reference material 972 levels 1–4 from the U.S. National Institute of Standards and Technology (NIST).27 The mean coefficients of variation (CV) for 25OHD3 were 9.5% at 12.3 ng/mL and 8.3% at 70.7 ng/mL. For 25(OH)D2, the mean CVs were 28.8% at 2.8 ng/mL and 10.7% at 25.4 ng/mL.
We assessed the heterogeneity between and within cohort-specific estimates for 25(OH)D and used the DerSimonian and Laird Q statistic to test 25(OH)D heterogeneity between studies. The Q statistic was consistent with homogeneity across the studies, thus indicating that the data could be combined across cohorts (P = 0.11). Nevertheless, due to the small number of studies (n=3) and consequently low statistical power of the tests, we visually examined the overlap of cohort-specific mean 25(OH)D and confidence intervals in a forest plot. These data visualization exercises confirmed the likelihood of homogeneity of 25(OH)D mean concentrations across the three cohorts. 28, 29
Covariates
We limited covariate assessment to those similarly defined in all three cohorts. These include self-reported maternal race and Hispanic origin (non-Hispanic Black and non-Hispanic White), maternal age at blood draw, educational level (high school or less, post-high school), and prepregnancy body mass index (BMI). Prepregnancy BMI was calculated from maternal height via interview and self-reported pre-pregnancy weight; BMI scores were categorized as: <25 (normal), 25–30 (overweight) and ≥30 (obese) kg/m2.30 We created a variable aggregating months into the four seasons of the northern hemisphere at the blood sample draw date (winter: December to February, spring: March to May, summer: June to August and fall: September to November).
Statistical analysis
First, we graphically assessed the presence of seasonality in the distribution of the observed serum concentrations of 25(OH)D using a time plot and a lowess adjustment based on the data.31 After identifying a seasonal pattern, we developed an observational Fourier analysis to determine the highest frequency (or dominant period) of 25(OH)D serum concentrations over time. We used a time series periodogram to compute the highest frequency to cover a seasonal cycle in the observed serum concentrations of 25(OH)D.32
Second, we described the pooled and stratified covariates in analysis by study site, presenting counts and percentages for categorical variables. To describe 25(OH)D seasonality, we used data reduction.33 We then grouped the data into four seasons and determined the mean and standard deviation of 25(OH)D serum concentrations presented by study site and maternal race.
Third, we assessed the monthly variability of 25(OH)D over a period of 12 months by fitting a generalized linear model with robust standard error estimation, assuming a normal distribution of the 25(OH)D distribution and, an identity link.34 We then used the Dickey-Fuller test to confirm the stationary of 25(OH)D serum concentrations over time (i.e., peak-trough differences remaining constant over time) to develop a stationary cosinor models.33, 35, 36
In cosinor models, the dependent variable 25(OH)D was modeled as a sine wave characterized by a phase shift (i.e., location of peak and trough concentrations on the time axis), and amplitude (i.e., maximum variation of the sine wave from its mean height). The time (t) variable (month) was transformed as cosine(t) and sine(t), which were then fit as predictors of 25(OH)D. The resulting coefficients of the cosine(t) and sine(t) were then transformed to derive the total peak–trough difference (which is equal to twice the amplitude) and the marginal annual mean (i.e., the intercept term of the model). Adjustment for other covariates effects on the yearly mean of 25(OH)D is accomplished by adding terms to the regression model.16, 33 We adjusted derived marginal annual means of 25(OH)D and peak-trough differences for the main effects of maternal age, race and study site. In adjusted cosinor models, we centered study site and maternal age and race at their study means, so that the reported annual mean of 25(OH)D concentration was standardized to the mean covariate values of the study population.16 We used the Delta method to derive standard errors and confidence intervals of the derived adjusted means and peak-trough differences.37 Finally, we explored the sensitivity of our estimates to alternative model specifications. All analyses were completed using Stata v.12.1.
Results
Women in the Omega study were older more likely to be non-Hispanic White (96.4%), to have had higher educational attainment levels (95.5% post-high school), and lower prepregnancy BMI (73.1% under 25) compared with women in the PIN and POUCH studies. There were only 27 non-Hispanic Black women in Omega study, and their sociodemographic and lifestyle characteristics differed as compared with their counterparts in the PIN and POUCH cohort. For example, non-Hispanic Black women in Omega study were older, better educated and had lower prepregnancy BMI as compared with their counterparts in the PIN and POUCH studies, respectively.
The frequency distribution of the season of blood collection did not differ considerably across the three study cohorts (Table 1).
Table 1. Maternal Sociodemographic Characteristics, Gestational Trimester of Blood Draw and Body Mass Index of Pregnant Women at by Site and Pooled Results, 1996-2008 (n= 2,583).
| Variables | OMEGA, Seattle (N=754), n(%) | PIN, North Carolina (N=992), n(%) | POUCH, Michigan (N=837), n(%) | Pooled (N=2,583), n(%) |
|---|---|---|---|---|
| Maternal Age | ||||
| 15 to 24 | 34(4.5) | 339(34.2) | 364(43.5) | 737(28.5) |
| 25 to 34 | 451(59.8) | 497(50.1) | 410(49.0) | 1,358(52.6) |
| ≥35 | 269(35.7) | 156(15.7) | 63(7.5) | 488(18.9) |
| Race | ||||
| Non-Hispanic Black | 27(3.6) | 350(35.3) | 272(32.5) | 649(25.1) |
| Non-Hispanic White | 727(96.4) | 642(64.7) | 565(67.5) | 1,934(74.9) |
| Maternal Education | ||||
| High school or less | 33(4.5) | 367(37.0) | 392(46.8) | 792(30.8) |
| Post-High school | 703(95.5) | 625(63.0) | 445(53.1) | 1,773(69.2) |
| Pre-pregnancy BMI (kg/m2) | ||||
| <25(Normal) | 551(73.1) | 554(56.2) | 425(50.8) | 1,530(59.4) |
| 25 to 30 (Overweight) | 138(18.3) | 195(19.8) | 177(21.1) | 510(19.8) |
| >30 (Obese) | 65(8.6) | 236(24.0) | 235(28.1) | 536(20.8) |
| Season | ||||
| Winter (December to February) | 173(22.9) | 211(21.3) | 209(25.0) | 593(23.0) |
| Spring (March to May) | 187(24.8) | 343(34.6) | 214(25.6) | 744(28.8) |
| Summer (June to August) | 178(23.6) | 208(21.0) | 229(27.3) | 615(23.8) |
| Fall (September to November) | 216(28.6) | 230(23.1) | 185(22.1) | 631(24.4) |
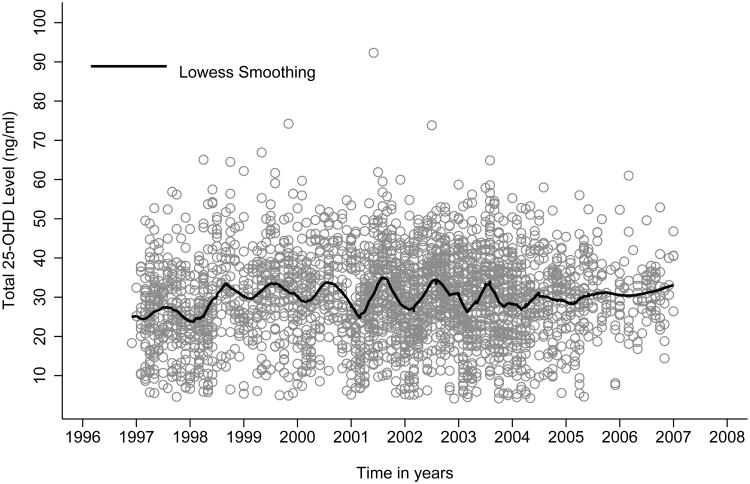
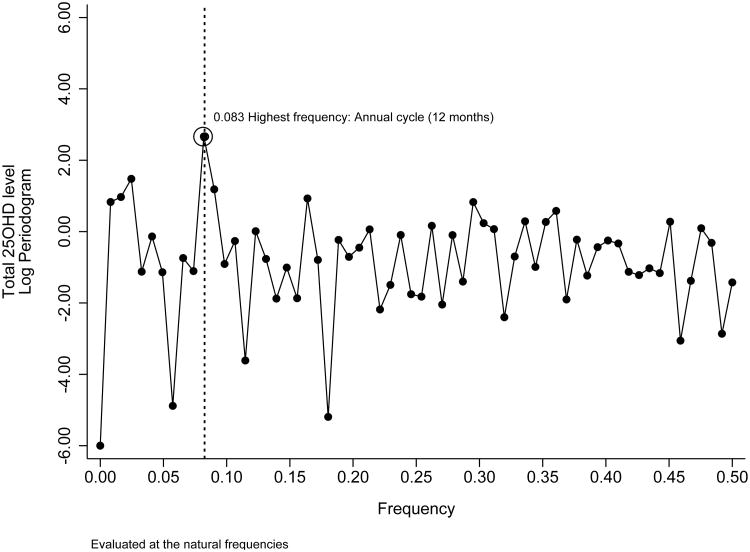
The time plot indicates the presence of a seasonal pattern with a sinusoidal distribution of 25(OH)D serum concentrations over time (Figure 1). The Fourier series analysis based on the periodogram shows a peak at a frequency of 0.083, which corresponds with the highest frequency observed in the data. This peak reflects a cycle of 12 months and confirms the presence of a seasonal pattern as illustrated in the time plot (Figure 2). The Dickey-Fuller test further confirms the stationary of 25OHD concentrations over time (P <0.001) with constant seasonal peak-trough differences during the period in analysis.
Figure 1. Time line and Lowess Smoothing of 25(OH)D serum concentrations, 1996-2008 (n = 2,583).
Figure 2. Peridogram and highest frequency of 25(OH)D serum concentrations, 1996-2008 (n = 2,583).
Observed annual mean of 25(OH)D serum concentrations by season show a peak at the end of the summer and a nadir in winter. Compared with non-Hispanic Whites, non-Hispanic Black women have lower 25(OH)D serum concentrations during all four seasons, although the variability of serum concentration is higher among non-Hispanic White women. In comparing non-Hispanic Black pregnant women at the three study sites, women at the PIN site show the lowest annual serum 25(OH)D concentration while women in the Omega study shows the highest 25(OH)D concentrations. Among non-Hispanic White women, the Omega participants also show the lowest serum concentrations compared with the two other study sites (Table 2).
Table 2. Mean (ng/mL) and Standard Deviation of 25(OH)D Serum Concentrations by Seasons, Site and Race, 1996-2008 (n= 2,583).
| Non-Hispanic Black (n=649), Mean(Standard Deviation) | Non-Hispanic White (n=1934), Mean(standard deviation) | |||||||
|---|---|---|---|---|---|---|---|---|
|
|
|
|||||||
| OMEGA, Seattle (n=27) | PIN, North Carolina (n=350) | POUCH, Michigan (n=272) | Total (n=649) | OMEGA, Seattle (n=727) | PIN, North Carolina (n=642) | POUCH, Michigan (n=565) | Total (n=1934) | |
| Winter (Dec to Feb) | 24.6(6.9) | 17.5(8.6) | 17.7(9.2) | 18.1(8.9) | 29.7(8.4) | 29.4(9.9) | 34.6(10.9) | 31.2(10.1) |
| Spring (Mar to May) | 27.6(6.7) | 18.0(8.8) | 18.5(8.2) | 18.7(8.7) | 29.4(8.9) | 30.8(9.4) | 33.5(10.3) | 31.0(9.7) |
| Summer (Jun to Aug) | 36.5(4.5) | 21.6(8.5) | 24.8(10.4) | 23.6(9.8) | 33.4(8.6) | 35.0(10.8) | 39.3(9.5) | 35.9(9.9) |
| Fall (Sep to Nov) | 22.5(6.6) | 19.4(9.8) | 22.5(8.9) | 20.7(9.5) | 31.9(7.7) | 33.0(8.8) | 36.7(10.6) | 33.5(9.2) |
| Annual | 26.8(7.3) | 19.0(9.0) | 20.9(9.6) | 20.2(9.4) | 31.2(8.6) | 31.9(9.9) | 36.1(10.5) | 32.8(9.9) |
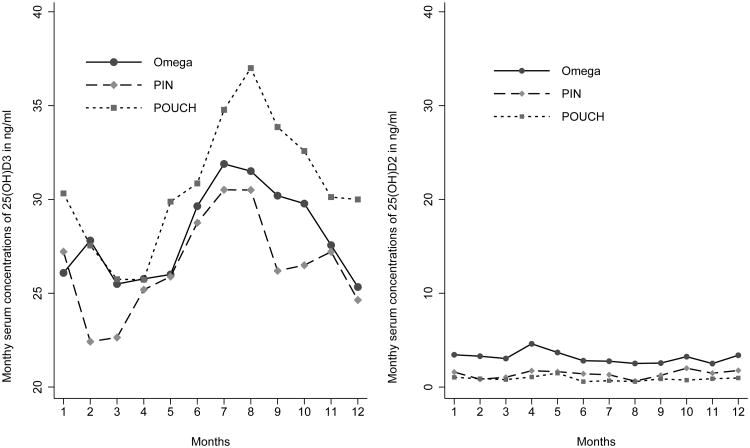
As illustrated in Figure 3, monthly differences of 25(OH)D show a clear seasonal pattern compatible with a sinusoidal wave ranging from 16.1% in August (peak) to -14.1% in March (trough) (Table 3). This sinusoidal variability is principally due to changes in 25(OH)D3 concentrations over the calendar year as 25(OH)D2 concentrations remain stable over time (Figure 3).
Figure 3. Observed monthly means of 25(OH)D2 and D3 serum concentrations by site, 1996-2008 (n = 2,583).
Table 3. Observed Monthly Means (ng/mL), Standard Deviations and Fitted Differences of 25(OH)D Serum Concentrations, 1996-2008 (n= 2,583).
| Month | N | Mean | Standard deviation | Absolute difference [95% CI] | Relative difference (%) |
|---|---|---|---|---|---|
| January | 221 | 29.6 | 11.0 | Reference | Reference |
| February | 202 | 26.9 | 11.5 | −2.68 [−4.83, −0.54] | −9.1 |
| March | 233 | 25.5 | 10.8 | −4.17 [−6.17, −2.16] | −14.1 |
| April | 270 | 27.5 | 11.1 | −2.11 [−4.07, −0.14] | −7.1 |
| May | 241 | 28.8 | 10.7 | −0.80 [−2.79, 1.18] | −2.7 |
| June | 207 | 30.8 | 11.2 | 1.14 [−0.97, 3.25] | 3.8 |
| July | 191 | 33.6 | 10.9 | 4.01 [1.89, 6.13] | 13.5 |
| August | 215 | 34.4 | 11.1 | 4.76 [2.68, 6.84] | 16.0 |
| September | 197 | 31.0 | 10.2 | 1.40 [−0.63, 3.44] | 4.7 |
| October | 232 | 31.1 | 11.3 | 1.49 [−0.57, 3.54] | 5.0 |
| November | 202 | 29.6 | 10.8 | −0.04[−2.08, 1.99] | −0.1 |
| December | 172 | 28.2 | 10.9 | −1.41[−3.60, 0.77] | −4.8 |
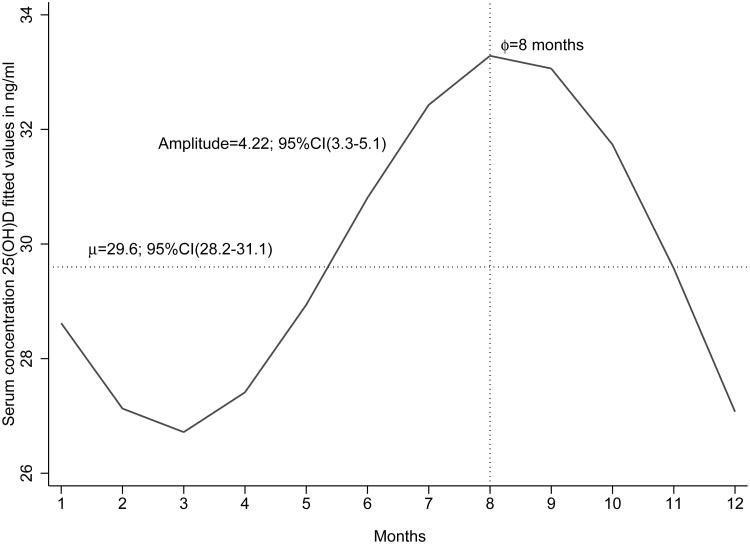
From the unadjusted stationary cosinor model, the derived annual mean of 25(OH)D serum concentration is 29.6 ng/mL (95%CI: 28.2-31.1), and the estimated amplitude is 4.2 ng/mL (95% CI 3.3, 5.1) with a phase of 8 months (Figure 4).
Figure 4. Trigonometric stationary cosinor fitted annual distribution of 25(OH)D serum concentrations, 1996-2008 (n = 2,583).
Regarding the amplitude of seasonal variation, non-Hispanic White race, women aged between 25 to 34 years, normal BMI, and post-high school education show greater peak–trough differences in 25(OH)D concentrations (sine wave amplitude). The PIN study has the lowest annual peak-trough and mean serum concentrations of 25(OH)D, while the POUCH study the highest (Table 4).
Table 4. Crude and Adjusted Annual Means and Mean Peak-trough in 25(OH)D Serum Concentrations, 1996-2008 (2,583).
| Variables | Unadjusted 25(OH)D Annual mean, (ng/mL) | 95%CI | Adjusted 25(OH)D Annual mean, (ng/mL)* | 95%CI | Mean Peak-Trough 25(OH)D difference, (ng/mL)* | 95%CI |
|---|---|---|---|---|---|---|
| Maternal Age | ||||||
| 15 to 24 | 27.7 | 27.1, 28.4 | 28.6 | 27.9, 29.4 | 5.8 | 4.8, 6.8 |
| 25 to 34 | 29.9 | 29.4, 30.3 | 30.3 | 29.7, 30.8 | 7.4 | 6.0, 8.8 |
| ≥35 | 31.9 | 31.2, 32.8 | 29.4 | 28.7, 30.2 | 6.6 | 5.0, 8.1 |
| P for difference | <0.001 | 0.01 | 0.03 | |||
| Race | ||||||
| Non-Hispanic Black | 20.2 | 19.5, 21.0 | 19.8 | 18.9, 20.5 | 5.8 | 4.7, 6.7 |
| Non-Hispanic White | 32.8 | 32.4, 33,2 | 33.0 | 32.6, 33.4 | 7.4 | 6.0, 8.9 |
| P for difference | <0.001 | <0.001 | 0.03 | |||
| Site | ||||||
| OMEGA (Seattle) | 30.8 | 30.0, 31.6 | 28.0 | 27.1, 28.5 | 5.7 | 4.7, 6.8 |
| PIN (North Carolina) | 27.5 | 26.8, 28.2 | 28.8 | 28.2, 29.4 | 6.8 | 5.3, 8.2 |
| POUCH (Michigan) | 31.2 | 30.4, 31.9 | 32.3 | 31.6, 33.0 | 10.2 | 8.7, 11.7 |
| P for difference | 0.001 | <0.001 | <0.001 | |||
| Maternal Education | ||||||
| High school or less | 26.7 | 26.0, 27.5 | 28.6 | 27.8, 29.4 | 5.9 | 4.8, 6.9 |
| Post-high school | 30.9 | 30.4, 31.5 | 30.1 | 29.6, 30.6 | 7.4 | 5.9, 8.7 |
| P for difference | <0.001 | <0.001 | 0.04 | |||
| Pre-pregnancy BMI (kg/m2) | ||||||
| <25 (Normal) | 31.4 | 30.8, 31.9 | 30.9 | 30.4, 31.4 | 5.9 | 4.8, 6.9 |
| 25 to 30 (Overweight) | 29.4 | 28.5, 30.3 | 28.9 | 28.5, 29.3 | 4.5 | 3.1, 5.9 |
| >30 (Obese) | 25.1 | 24.2, 26.0 | 26.8 | 26.1, 27.6 | 1.6 | 0.2, 3.0 |
| P for difference | <0.001 | <0.001 | <0.001 |
Models were adjusted for the main effect of maternal age, race and study site. Annual means were centered to reflect study population values for maternal age, race and study site.
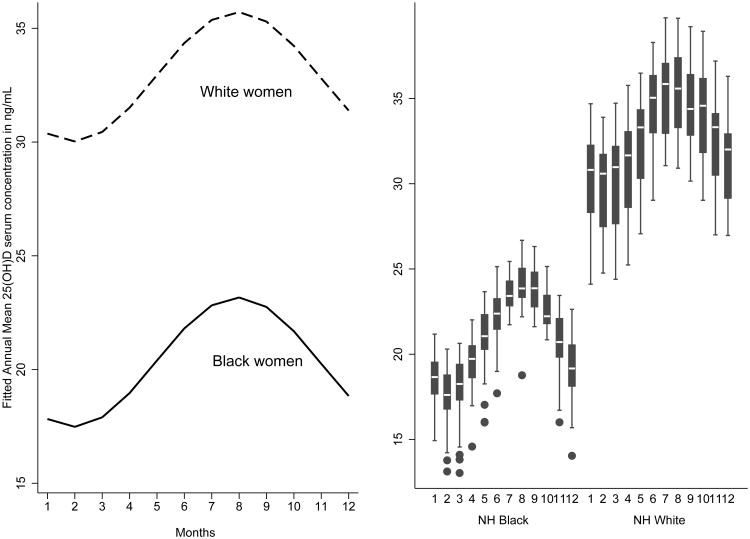
As indicated in Figure 5, regardless of maternal age and study site, non-Hispanic Black women have lower annual mean 25(OH)D serum concentrations (with concentrations <20 ng/mL from November thru April) (Figure 5).
Figure 5. Trigonometric stationary cosinor fitted annual adjusted distribution of 25(OH)D serum concentrations by race, 1996-2008 (n = 2,583).
Lastly, given that non-Hispanic Black women from the Omega study differed from those in the PIN and POUCH studies, we completed sensitivity analyses after excluding Omega study (n= 27) participants. Findings from these sensitivity analyses were similar to those from our primary analyses. For example, after adjustment for covariates, the annual mean concentrations of 25(OH)D among non-Hispanic Black women were 19.2 ng/mL in the restricted analysis (19.8 ng/mL in the primary analysis). The corresponding value for estimated peak–trough differences of 25(OH)D among non-Hispanic Black women in the restricted cohort was 5.9 ng/mL (5.8 ng/mL in the primary analysis).
Comments
In this study of pregnant women, we documented a seasonal pattern of 25(OH)D serum concentrations. Regardless of maternal age and study site, pregnant non-Hispanic Black women have lower mean 25(OH)D concentrations throughout the calendar year and lower mean peak-trough differences than pregnant non-Hispanic White women. Seasonality in 25(OH)D varies by educational attainment and prepregnancy obesity. Further, seasonal variability of 25(OH)D was due to 25(OH)D3, but not of 25(OH)D2. As the source of 25(OHD)D2 is primarily supplements, and not UV radiation, the lack of seasonal variability in this form is not surprising.
To the best of our knowledge this is the largest study to evaluate seasonal variation of 25(OH)D concentration among pregnant U.S. women and the first study to reveal a seasonal pattern of 25(OH)D serum concentrations among U.S. non-Hispanic Black pregnant women. Our study utilized rigorous analytic approaches that consider gestational age at collection time.
Our study results are also consistent with prior studies reporting seasonal variation of serum 25(OH)D concentrations. Shoben et al.'s study of community-based older adults demonstrated a sinusoudal variation of serum 25(OH)D concentrations thoughout the calender year.16 Similarly, Bolland et al. found that seasonal variation of 25(OH)D serum concentrations significantly affects the diagnosis of vitamin D sufficiency, which requires seasonally adjusted thresholds.38 Collectively the results of our study and those of others, show that consideration of seasonality variation is necessary to develop assesment of vitamin D deficency and vitamin D supplementation strategies.
A substantial body of evidence shows that Black women synthesize less cutaneous vitamin D compared to White women.17, 18 The skin pigment melanin absorbs UVR photons and can reduce 25(OH)D3 synthesis.18 Consequently, many African Americans are at higher risk of vitamin D deficiency. Our study results provide the first clear, objective evidence of seasonal variation of serum 25(OH)D concentrations among pregnant non-Hispanic Black U.S. women. We are aware of only one published study that evaluates patterns of serum 25(OH)D seasonal variation in Black and Whites during pregnancy. In their study of 200 Black and 200 White women living in Pittsburgh, Pennsylvania, Bodnar et al. reported seasonality of vitamin D concentrations among White pregnant women but not among Black women (ethnicity not specified).20 The authors speculate that Black women are unable to accumulate adequate concentrations of vitamin D when sunlight increases in summer, based on their findings of no significant variability of 25(OH)D serum concentrations over the astronomical calendar year. However, their study was limited by a small sample size.
Our results show that the variability of 25(OH)D among non-Hispanic Black pregnant women follows a clear seasonal pattern, which is likely due to differences in exposure to UVR.7, 33 This pattern of seasonality is characterized by higher serum concentrations of 25(OH)D during summer months (June to August) and lower concentrations in winter months (December through February), when less UVR reaches the earth, minimizing the quantity of vitamin D that can be synthesized by the skin.39,40
On the other hand, differences in sunlight exposition and skin pigmentation among pregnant women might not be the only factors that explain racial differences of 25(OH)D serum concentrations. Other factors, could explain racial 25(OH)D inequalities, such as obesity, diet, educational attainment, smoking status, and/or socioeconomic status.41-43
One limitation of the present study is the reduced availability of determinants of variation in 25(OH)D serum concentrations in our data, such as diminished sunlight exposure.44 Regardless of latitude and seasonality, diminished sunlight exposure could be affected by social conventions, sunscreen use, air pollution levels (e.g., urban vs. suburban) and degree of regular outdoor physical activity.45, 46 Hence, if factors associated with diminished sunlight exposure had been considered in our analysis, the observed racial differences in vitamin D concentrations may have been attenuated. In addition, differences found among non-Hispanic White women, with the highest concentrations of 25(OH)D in Michigan, which is much further north than North Carolina, may be explained by diminished sunlight exposure due to time spent outdoors or due variations in use of sunscreen or clothing options and styles. Future studies will benefit from including assessment of these and other behavioral factors that may account for unexpected variations in 25OHD concentrations according to study site.
Other limitations include lack of data on dietary or supplemental sources of vitamin D and smoking status. Vitamin D supplementation during pregnancy is usually limited to prenatal care vitamins or fortified foods. Although we do not have information on prenatal Vitamin D use, no changes have been reported for the current U.S. vitamin D supplementation guidelines of 400 IU daily for pregnant women.47 Thus, our results should not have been affected by a time-period effect regarding vitamin D use. Moreover, having run the samples of each cohort separately in the laboratory might have induced potential batch run differences… Finally, our annual estimates of 25(OH)D variability cannot assess the specific distribution of seasonal vitamin D variation for individuals. To validate our vitamin D variability estimation at a population level we would have needed repeated samples of 25(OH)D for women at an individual level.
Recent studies have shown that vitamin D supplementation of 4,000 IU/day during pregnancy improved maternal vitamin D status throughout pregnancy and improved vitamin D status at birth.48,49 However, assessing vitamin D sufficiency may require seasonally adjusted thresholds based on collection time.38 Thus, as our study indicates, increased knowledge about the patterns and determinants of variability of 25(OH)D among pregnant women could serve to better inform clinical-care decision making so as to prevent adverse maternal and perinatal outcomes caused by vitamin D deficiency.
In summary, this study's confirmation of 25(OH)D seasonality over the calendar year in a population of pregnant U.S. women, can enhance public health interventions targeted to improve maternal and perinatal outcomes. Vitamin D supplements and behavior recommendations to maximize skin production, balancing the risk to sun exposure, should be recommended during pregnancy with consideration of racial subgroups of pregnant women who are at higher risk of vitamin D deficiency, specifically dark-skinned pregnant women.
Acknowledgments
The research reported in this article was supported by several awards: CDC 200-2008-27956-12; OMEGA: NICHD R01-HD-32562; PIN: NIH http://www.cpc.unc.edu/projects/pin/funding; POUCH: NIH R01 HD-034543, R01 HD-34543, March of Dimes Foundation 20-FY98-0697 through 20-FY04-37, Thrasher Research Foundation grant 02816-7, CDC U01 DP000143-01.
Footnotes
Disclaimer: The findings and conclusions in this report are those of the authors and do not necessarily represent the official position of the Centers for Disease Control and Prevention.
References
- 1.Cannell JJ, Hollis BW, Zasloff M, Heaney RP. Diagnosis and treatment of vitamin D deficiency. Expert Opinion in Pharmacotherapy. 2008;9(1):107–118. doi: 10.1517/14656566.9.1.107. [DOI] [PubMed] [Google Scholar]
- 2.Holick MF. Vitamin D: A millenium perspective. Journal of Cellular Biochemistry. 2003;88(2):296–307. doi: 10.1002/jcb.10338. [DOI] [PubMed] [Google Scholar]
- 3.Holick MF. Vitamin D deficiency. New England Journal of Medicine. 2007;357(3):266–281. doi: 10.1056/NEJMra070553. [DOI] [PubMed] [Google Scholar]
- 4.Holick MF. Deficiency of sunlight and vitamin D. BMJ. 2008;336(7657):1318–1319. doi: 10.1136/bmj.39581.411424.80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fraser DR. The physiological economy of vitamin D. Lancet. 1983;1(8331):969–972. doi: 10.1016/s0140-6736(83)92090-1. [DOI] [PubMed] [Google Scholar]
- 6.Rajakumar K, Greenspan SL, Thomas SB, et al. SOLAR ultraviolet radiation and vitamin D: a historical perspective. American Journal of Public Health. 2007;97(10):1746–1754. doi: 10.2105/AJPH.2006.091736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.DeLuca HF. Overview of general physiologic features and functions of vitamin D. American Journal of Clinical Nutrition. 2004;80(6 Suppl):1689S–1696S. doi: 10.1093/ajcn/80.6.1689S. [DOI] [PubMed] [Google Scholar]
- 8.Aghajafari F, Nagulesapillai T, Ronksley PE, Tough SC, O'Beirne M, Rabi DM. Association between maternal serum 25-hydroxyvitamin D level and pregnancy and neonatal outcomes: systematic review and meta-analysis of observational studies. BMJ. 2013;346:f1169. doi: 10.1136/bmj.f1169. [DOI] [PubMed] [Google Scholar]
- 9.Bodnar LM, Catov JM, Simhan HN, Holick FM, Powers RW, Roberts JM. Maternal vitamin D deficiency increases the risk of preeclampsia. Journal of Clinical Endocrinology and Metabolism. 2007;92(9):3517–3522. doi: 10.1210/jc.2007-0718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhang C, Qiu C, Hu FB, David RM, van Dam RM, Bralley A, et al. Maternal plasma 25-hydroxyvitamin D concentrations and the risk for gestational diabetes mellitus. PLoS One. 2008;3(11):e3753. doi: 10.1371/journal.pone.0003753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Burris HH, Rifas-Shiman SL, Kleinman K, Litonjua AA, Huh SY, Rich-Edwards JW, et al. Vitamin D deficiency in pregnancy and gestational diabetes mellitus. Amerian Journal of Obstetric and Gynecology. 2012;207(3):182 e181–188. doi: 10.1016/j.ajog.2012.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Merewood A, Mehta SD, Chen TC, Bauchner H, Holick MF. Association Between Vitamin D Deficiency and Primary Cesarean Section. Journal of Clinical Endocrinology and Metabolism. 2009;94(3):940–955. doi: 10.1210/jc.2008-1217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gale CR, Robinson SM, Harvey NC, Javaid MK, Jiang B, Martyn CN, et al. Maternal vitamin D status during pregnancy and child outcomes. European Journal of Clinical Nutrition. 2008;62(1):68–77. doi: 10.1038/sj.ejcn.1602680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hollis BW, Pittard WB. Evaluation of the total fetomaternal vitamin D relationships at term: evidence for racial differences. Journal of Clinical Endocrinology and Metabolism. 1984;59(4):652–657. doi: 10.1210/jcem-59-4-652. [DOI] [PubMed] [Google Scholar]
- 15.Zeghoud F, Vervel C, Guillozo H, Walrant-Debray O, Boutignon H, Garabedian M. Subclinical vitamin D deficiency in neonates: definition and response to vitamin D supplements. The American Journal of Clinical Nutrition. 1997;65(3):771–778. doi: 10.1093/ajcn/65.3.771. [DOI] [PubMed] [Google Scholar]
- 16.Shoben AB, Kestenbaum B, Levin G, Hoofnagle AN, Psaty BM, Siscovick DS, et al. Seasonal variation in 25-hydroxyvitamin D concentrations in the cardiovascular health study. American Journal of Epidemiology. 2011;174(12):1363–1372. doi: 10.1093/aje/kwr258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Loomis WF. Skin-pigment regulation of vitamin-D biosynthesis in man. Science. 1967;157(3788):501–506. doi: 10.1126/science.157.3788.501. [DOI] [PubMed] [Google Scholar]
- 18.Clemens TL, Adams JS, Henderson SL, Holick MF. Increased skin pigment reduces the capacity of skin to synthesise vitamin D3. Lancet. 1982;1(8263):74–76. doi: 10.1016/s0140-6736(82)90214-8. [DOI] [PubMed] [Google Scholar]
- 19.Ginde AA, Sullivan AF, Mansbach JM, Camargo CA. Vitamin D insufficiency in pregnant and nonpregnant women of childbearing age in the United States. American Journal of Obstettric and Gynecology. 2010;202(5):436 e431–438. doi: 10.1016/j.ajog.2009.11.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bodnar LM, Simhan HN, Powers RW, Frank MP, Cooperstein E, Roberts JM. High prevalence of vitamin D insufficiency in black and white pregnant women residing in the northern United States and their neonates. The Journal of Nutrition. 2007;137(2):447–452. doi: 10.1093/jn/137.2.447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Savitz DA, Dole N, Williams J, Thorp JM, McDonald T, Carter AC, et al. Determinants of participation in an epidemiological study of preterm delivery. Paediatric and Perinatal Epidemiology. 1999;13(1):114–125. doi: 10.1046/j.1365-3016.1999.00156.x. [DOI] [PubMed] [Google Scholar]
- 22.Holzman C, Bullen B, Fisher R, Paneth N, Reuss L. Pregnancy outcomes and community health: the POUCH study of preterm delivery. Paediatric and Perinatal Epidemiology. 2001;15(2):136–158. doi: 10.1046/j.1365-3016.2001.00014.x. [DOI] [PubMed] [Google Scholar]
- 23.Williams MA, Frederick IO, Qiu C, Meryman LJ, King IB, Walsh SW, et al. Maternal erythrocyte omega-3 and omega-6 fatty acids, and plasma lipid concentrations, are associated with habitual dietary fish consumption in early pregnancy. Clinical Biochemistry. 2006;39(11):1063–1070. doi: 10.1016/j.clinbiochem.2006.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ferre CD, Holzman C, Siega-Riz AM, Enquobahrie D, Williams MA, Dole N, et al. 140th Annual Meeting of the American Public Health Association. San Francisco, CA: American Public Health Association; 2012. Maternal vitamin D status and preterm birth. [Google Scholar]
- 25.Chen H, McCoy LF, Schleicher RL, Pfeiffer CM. Measurement of 25-hydroxyvitamin D3 (25OHD3) and 25-hydroxyvitamin D2 (25OHD2) in human serum using liquid chromatography-tandem mass spectrometry and its comparison to a radioimmunoassay method. Clinical Chimica Acta. 2008;391:6–12. doi: 10.1016/j.cca.2008.01.017. [DOI] [PubMed] [Google Scholar]
- 26.Aronov PA, Hall LM, Dettmer K, Stephensen CB, Hammock BD. Metabolic profiling of major vitamin D metabolites using Diels-Alder derivatization and ultra-performance liquid chromatography-tandem mass spectrometry. Analytical and Bioanalytical Chemistry. 2008;391:1917–1930. doi: 10.1007/s00216-008-2095-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Phinney KW, Bedner M, Tai SS, Vamathevan VV, Sander LC, Sharpless KE, et al. Development and certification of a standard reference material for vitamin D metabolites in human serum. Analytical Chemistry. 2012;84:956–962. doi: 10.1021/ac202047n. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Berlin JA, Laird NM, Sacks HS, Chalmers TC. A comparison of statistical methods for combining event rates from clinical trials. Statistics in Medicine. 1989;8(2):141–151. doi: 10.1002/sim.4780080202. [DOI] [PubMed] [Google Scholar]
- 29.Kirkwood BR, Sterne JAC. Essential medical statistics. 2nd. Malden, Mass.: Blackwell Science; 2003. [Google Scholar]
- 30.Park S, Sappenfield WM, Bish C, Salihu H, Goodman D, Bensyl DM. Assessment of the Institute of Medicine recommendations for weight gain during pregnancy: Florida, 2004-2007. Maternal and Child Health Journal. 2011;15(3):289–301. doi: 10.1007/s10995-010-0596-5. [DOI] [PubMed] [Google Scholar]
- 31.Borkowf CB, Albert PS, Abnet CC. Using lowess to remove systematic trends over time in predictor variables prior to logistic regression with quantile categories. Statistic in Medicine. 2003;22(9):1477–1493. doi: 10.1002/sim.1507. [DOI] [PubMed] [Google Scholar]
- 32.Bloomfield P. Fourier analysis of time series: an introduction. 2nd. New York; Chichester: Wiley; 2000. [Google Scholar]
- 33.Barnett AG, Dobson AJ. Analysing seasonal health data. Berlin; London: Springer; 2010. [Google Scholar]
- 34.Hardin JW, Hilbe J. Generalized linear models and extensions. 3rd. College Station, Tex.: Stata Press; 2012. [Google Scholar]
- 35.Dickey DA, Fuller WA. Distribution of the estimators for autoregressive time series with a unit root. Journal of the American Statistics Association. 1979;74:427–431. [Google Scholar]
- 36.Mikulich SK, Zerbe GO, Jones RH, Crowley TJ. Comparing linear and nonlinear mixed model approaches to cosinor analysis. Statistics in Medicine. 2003;22(20):3195–3211. doi: 10.1002/sim.1560. [DOI] [PubMed] [Google Scholar]
- 37.Rice JA. Mathematical statistics and data analysis. Monterey, Calif.: Brooks/Cole Pub. Co.; 1988. [Google Scholar]
- 38.Bolland MJ, Grey AB, Ames RW, Mason BH, Horne AM, Gamble GD, et al. The effects of seasonal variation of 25-hydroxyvitamin D and fat mass on a diagnosis of vitamin D sufficiency. The American Journal of Clinical Nutrition. 2007;86(4):959–964. doi: 10.1093/ajcn/86.4.959. [DOI] [PubMed] [Google Scholar]
- 39.Webb AR, Kline L, Holick MF. Influence of season and latitude on the cutaneous synthesis of vitamin D3: exposure to winter sunlight in Boston and Edmonton will not promote vitamin D3 synthesis in human skin. Journal of Clinical Endocrinology and Metabolism. 1988;67(2):373–378. doi: 10.1210/jcem-67-2-373. [DOI] [PubMed] [Google Scholar]
- 40.Webb AR, Holick MF. The role of sunlight in the cutaneous production of vitamin D3. Annual Review of Nutrition. 1988;8:375–399. doi: 10.1146/annurev.nu.08.070188.002111. [DOI] [PubMed] [Google Scholar]
- 41.Bodnar LM, Catov JM, Roberts JM, Simhan HN. Prepregnancy obesity predicts poor vitamin D status in mothers and their neonates. The Journal of Nutrition. 2007;137(11):2437–2442. doi: 10.1093/jn/137.11.2437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.van den Berg G, van Eijsden M, Vrijkotte TG, Gemke RJ. Suboptimal maternal vitamin D status and low education level as determinants of small-for-gestational-age birth weight. European Journal of Nutrition. 2013;52(1):273–279. doi: 10.1007/s00394-012-0327-3. [DOI] [PubMed] [Google Scholar]
- 43.Vahamiko S, Isolauri E, Poussa T, Laitinen K. The impact of dietary counselling during pregnancy on vitamin intake and status of women and their children. International Journal of Food Science and Nutrition. 2013;64(5):551–560. doi: 10.3109/09637486.2013.766153. [DOI] [PubMed] [Google Scholar]
- 44.De-Regil LM, Palacios C, Ansary A, Kulier R, Pena-Rosas JP. Vitamin D supplementation for women during pregnancy. Cochrane Database of Systematic Reviews. 2012;2:CD008873. doi: 10.1002/14651858.CD008873.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Holick MF, Chen TC, Lu Z, Sauter E. Vitamin D and skin physiology: a D-lightful story. Journal of Bone and Mineral Research. 2007;22(2):V28–33. doi: 10.1359/jbmr.07s211. [DOI] [PubMed] [Google Scholar]
- 46.Maghbooli Z, Hossein-Nezhad A, Shafaei AR, Karimi F, Madani FS, Larijani B. Vitamin D status in mothers and their newborns in Iran. BMC Pregnancy and Childbirth. 2007;7:1. doi: 10.1186/1471-2393-7-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Institute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D. Institute of Medicine; U.S: 2011. Committee to Review Dietary Reference Intakes for Vitamin D and Calcium Food and Nutrition Board. [Google Scholar]
- 48.Wagner CL, McNeil RB, Johnson DD, Hulsey TC, Ebeling M, Robison C, et al. Health characteristics and outcomes of two randomized vitamin D supplementation trials during pregnancy: A combined analysis. The Journal of Steroid Biochemistry and Molecular Biology. 2013;136:313–320. doi: 10.1016/j.jsbmb.2013.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wagner CL, McNeil R, Hamilton SA, Winkler J, Rodriguez Cook C, Warner G, et al. A randomized trial of vitamin D supplementation in 2 community health center networks in South Carolina. American Journal of Obstetric and Gynecology. 2013;208:137 e131–113. doi: 10.1016/j.ajog.2012.10.888. [DOI] [PMC free article] [PubMed] [Google Scholar]