Abstract
Although most chemotherapeutic drugs have the potential to exert cardiotoxicity, these drugs have been chosen for use in cancer treatment because survival and curability benefits outweigh the risk of these complications. Anthracyclines, for example, are a powerful class of chemotherapeutic agents; however, their use is restricted by dose-related cardiotoxicity. Experimental evidence strongly supports the role of reactive oxygen species in this process, suggesting that antioxidants may be effective in protecting the heart from toxicity. Clinical use of antioxidants to protect the heart during anthracycline chemotherapy has been controversial due to the potential for reduced cytotoxic efficacy toward cancer cells. Results from randomized clinical trials addressing whether antioxidants either reduce the incidence of clinical heart failure among patients undergoing anthracycline-based chemotherapy or reduce the response rates to anthracycline-based chemotherapy have been unclear. While anthracyclines are by far the most well-studied antitumor agents with cardiotoxic properties, evidence now shows that reactive oxygen species may play roles in cardiotoxicity induced by other chemotherapeutic agents such as cyclophosphamide, cisplatin, 5-fluorouracil, and trastuzumab. Thus, in the new era of combination therapy and long-term survival of cancer patients, the use of antioxidants to support cancer therapy should be revisited.
Keywords: antioxidants, cancer, cardioprotection, cardiotoxicity, chemotherapy, free radicals, reactive oxygen species
The era of cardio-oncology
In the modern era of the 21st century, substantial progress in the understanding of cancer has resulted in improved early diagnosis, therapy, and survival of patients with many types of this disease. Therapeutic strategies now involve targeted drugs and combinations of multiple different agents, and the intervention is often used at the adjuvant stage. As patient survival increases and such new strategies are used, the secondary and latent effects of cancer chemotherapeutic agents have become more and more of concern. In particular, cardiotoxicity is a serious life-threatening complication and has been known to occur in response to treatment of patients with most, if not all of antitumor drugs (Table 1). Nevertheless, these drugs have been used for cancer treatment because the survival and curability benefits outweigh the risk of cardiovascular complications. Although acute cardiotoxicity is a concern, at the time of chemotherapeutic treatment, serious latent consequences can also manifest many years later. Thus, it has become clear that patients should be managed by a team of oncologists and cardiologists, not only to cure cancer, but also to reduce toxicity from therapy including cardiotoxicity. The concept of cardio-oncology has been developed to foster interactions of these two clinical disciplines to better care for patients who receive cancer therapy and also to foster research for the development of optimal strategies to treat cancer while reducing cardiovascular events (Minotti et al., 2010; Albini et al., 2010).
Table 1.
Partial list of cancer chemotherapeutic agents that are suspected to cause cardiotoxicity
| Anthracyclines [Lefrak et al., 1973; Shan et al., 1996] |
| Taxanes (paclitaxel, docetaxel) [Perotti et al., 2003] |
| 5-Fluorouracil [Alter et al., 2006; Lamberti et al., 2012] |
| Nitrogen mustard alkylating agents (cyclophosphamide, ifosfamide, melphalan) [Santos et al., 1971; Gottdiener et al., 1981; Kandylis et al., 1989; Zver et al., 2008; Feliz et al., 2011] |
| Cisplatin [Pai & Nahata, 2000] |
| Trastuzumab [Keefe, 2002; Seidman et al., 2002] |
| Fludarabin [Ritchie et al., 2001] |
| Mitomycin [Buzdar et al., 1978; Verweij et al., 1988] |
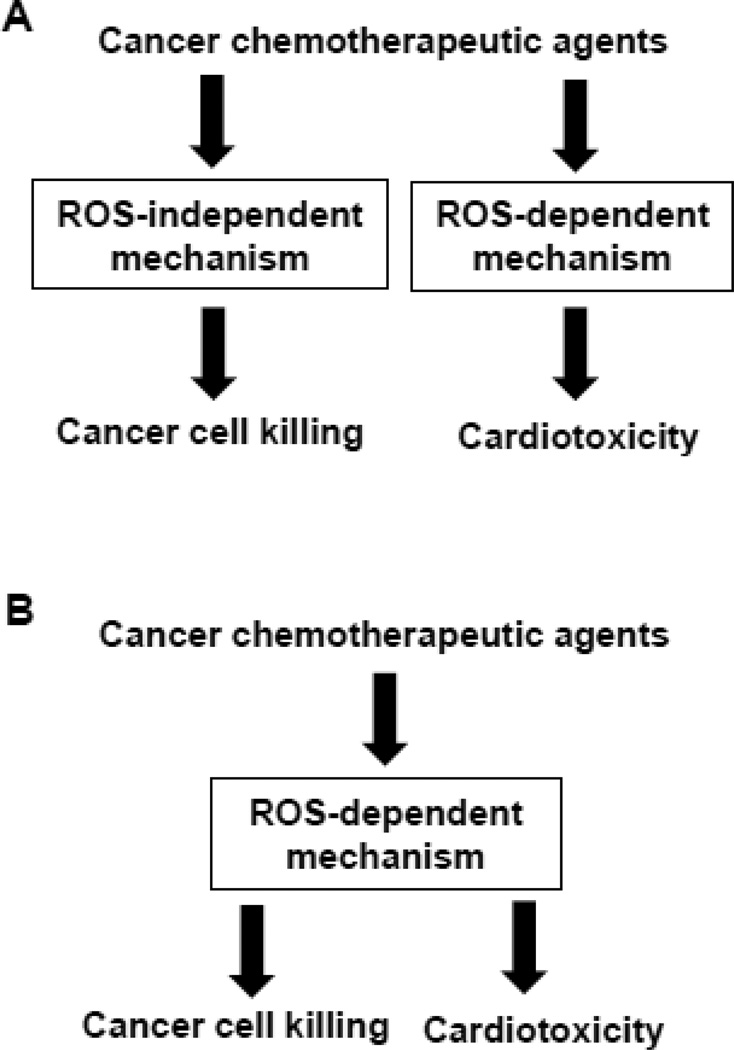
Numerous experimental results show that reactive oxygen species (ROS) mediate mechanisms of cardiotoxicity induced by various cancer chemotherapeutic agents, implying that concomitant administration of antioxidants may provide some relief. Cancer cell killing by these agents may utilize ROS-independent mechanisms (Fig. 1A), implying that antioxidants may reduce cardiotoxicity without affecting the efficacy to kill cancer cells. However, whether antioxidants are useful for preventing cardiotoxicity during treatment with cancer therapeutic agents, particularly anthracyclines, has generated serious controversy, as ROS may also be involved in cancer cell killing (Fig. 1B). Unfortunately, with the exception of dexrazoxane, information on whether patients undergoing cancer chemotherapy and/or radiation therapy should receive either dietary or pharmacologic antioxidants is quite limited. Whether patients should be given antioxidants during cancer treatment is unclear. In addition, whether patients who have completed cancer treatment should receive antioxidants to prevent cardiotoxicity, which may occur years after the treatment, has not been addressed. Combination therapies, in particular with anthracyclines and trastuzumab, have also gained attention for serious concerns regarding cardiotoxicity (Albini et al., 2010). Whether antioxidants are effective in attenuating cardiotoxicity induced by trastuzumab or other cancer treatments, such as cisplatin and thoracic irradiation is unclear. The present work revisits the issue on the role of antioxidant therapy in cardiotoxicity induced by cancer therapy in the era of cardio-oncology. There are clear needs for more research on the effects of antioxidants on cardiotoxicity induced by various antitumor drugs both at basic and clinical levels, and we hope that this work will foster such effort to help develop optimal therapeutic strategies to treat cancer.
Fig. 1.
Possible roles of ROS in cancer killing and cardiotoxic effects of cancer chemotherapeutic agents
Definitions of ROS and antioxidants
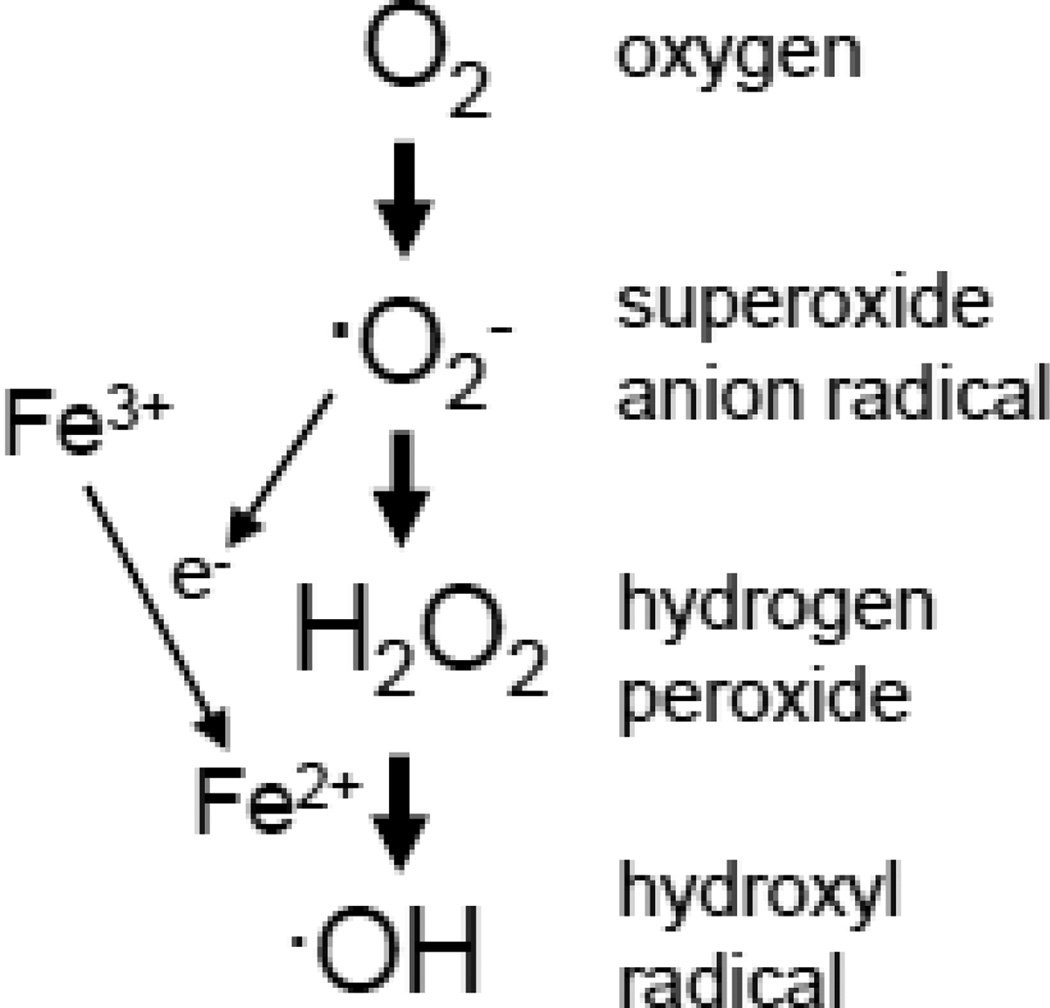
While ROS can broadly be defined as any reactive molecules containing oxygen, in this article we define ROS more strictly to include superoxide anion radical (·O2−), hydrogen peroxide (H2O2), and hydroxyl radical (HO·) as depicted in Figure 2. One-electron reduction of molecular oxygen (O2) produces ·O2− through various enzymatic and non-enzymatic mechanisms. Superoxide, once produced, can undergo a disproportionation reaction, in which one molecule of ·O2− reacts with another ·O2− molecule, leading to the formation of hydrogen peroxide at physiologic pH. This reaction is called dismutation and can be catalyzed by the class of enzymes called superoxide dismutases. A common target of H2O2 is cysteine thiolates. Hydrogen peroxide also serves as an electron source for the Fenton reaction in which reduced iron catalyzes the formation of HO·. Hydroxyl radicals are among the most potent ROS and can initiate lipid peroxidation, promote DNA oxidation and induce protein oxidation, such as protein carbonylation. Superoxide and other reductants can donate an electron to oxidized iron to promote the Fenton reaction. Superoxide can also react with nitric oxide, thereby forming peroxynitrite. This would reduce the availability of nitric oxide and produce a potent oxidant peroynitrite (Halliwell & Gutteridge, 1985).
Fig. 2. Definition of reactive oxygen species (ROS).
In this article, ROS refer to superoxide anion radical (·O2−), hydrogen peroxide (H2O2), and hydroxyl radical (OH). One-electron reduction of O2 forms ·O2−. H2O2 is formed by two ·O2− molecules interacting with each other in a reaction called dismutation. OH can be generated by the Fenton reaction that is catalyzed by ferrous iron (Fe2+), which can be produced by the reduction of ferric iron (Fe3+) by ·O2− or other reductants.
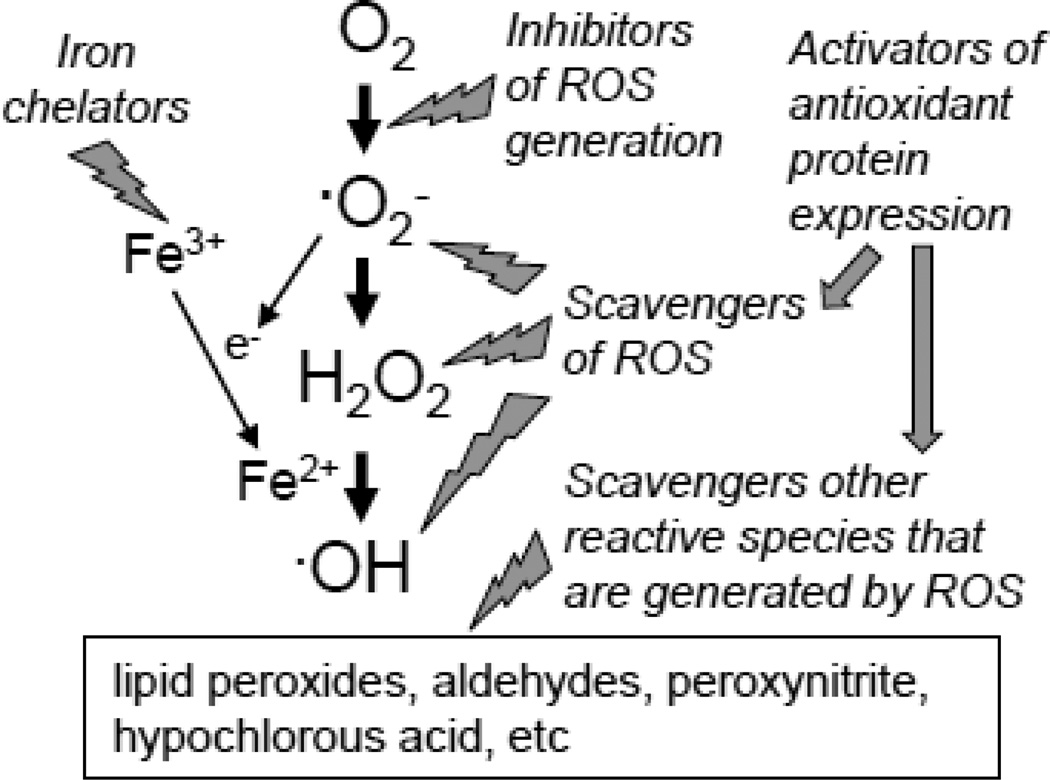
In this article, we define the term “antioxidant” broadly as any molecules that are capable of inhibiting the formation and actions of ROS. This definition could include (i) inhibitors of ROS producing processes; (ii) scavengers of ROS themselves; (iii) scavengers of other reactive substances that may be produced in response to the actions and reactions of ROS; (iv) metal chelators; and (v) molecules that promote the expression of antioxidant proteins (Fig. 3). The inhibitors of ROS producing processes could include those that inhibit ROS producing oxidases, such as NAD(P)H oxidase, xanthine oxidase, and monoamine oxidase. Many molecules have been shown to scavenge ROS. These include the scavengers of the highly reactive HO·, which will react with any organic compounds with very high kinetic rate constants. Antioxidants may also scavenge and eliminate other reactive species that can be formed in response to ROS, such as lipid peroxides, other aldehydes, peroxynitrite, and hypochlorous acid. Chelators of metals that promote the Fenton reaction, and in particular iron chelators can inhibit the formation of HO·. Finally, molecules that are capable of acting as nucleophiles and promoting gene expression of enzymes/proteins such as superoxide dismutase, catalase, glutathione peroxidase, glutathione reductase, thioredoxin, thioredoxin reductase, glutaredoxin and peroxyredoxin, which increase the endogenous antioxidant defense system, have been recently recognized as potent mechanisms against ROS damage. Moreover, the transcription factor Nrf2 often regulates these processes (Maher & Yamamoto, 2010).
Fig. 3. Definition of antioxidants.
In the present work, the term ‘antioxidants’ refers broadly to any molecules that have the capacity to inhibit the generation and action of ROS. These include (i) the inhibitors of oxidases (such as NADPH oxidase, xanthine oxidase, and monoamine oxidase), which may produce ·O2− or H2O2; (ii) small molecule and enzymatic scavengers of ROS; (iii) scavengers of other reactive species that are produced by ROS, such as lipid peroxides, other aldehydes, peroxynitrite, and hypochlorous acid; (iv) iron chelators that inhibit the Fenton reaction; and (v) molecules that act as nucleophiles to promote the expression of proteins with antioxidant capacities through mechanisms such as involvement of Nrf2.
Mechanisms of cardiotoxicity by various antitumor drugs
In the modern era of combination therapy, it is important to evaluate the effects of multiple antitumor drugs that are capable of promoting cardiotoxicity. While anthracyclines are by far the most well-studied antitumor agents with regard to cardiotoxicity, experimental results suggest that other chemotherapeutic agents, such as cyclophosphamide, cisplatin, 5-fluorouracil, and trastuzumab also exert cardiotoxicity through ROS. Here, we review the literature, which supports the hypothesis that antitumor drugs promote cardiotoxicity through the formation of ROS. Therefore, antioxidants may protect the heart.
Anthracyclines
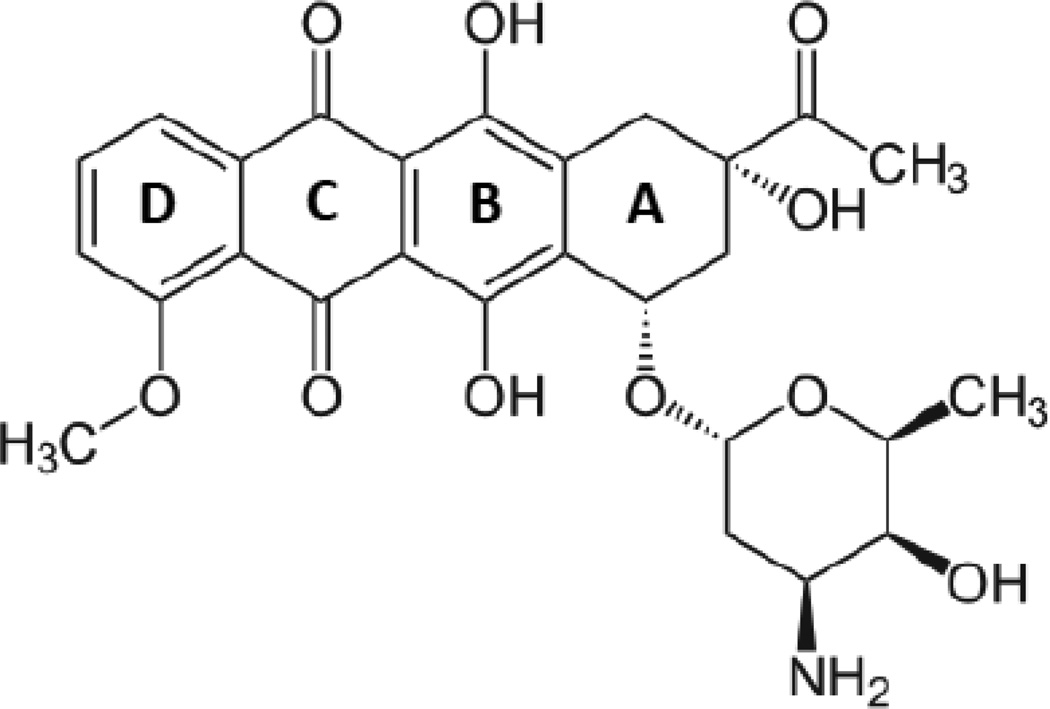
Anthracycline antibiotics derived from the bacteria Streptomyces peucetius var. caesius are widely used in cancer chemotherapy. Anthracyclines are composed of a tetracyclic ring structure attached to the sugar daunosamine and also have quinone and hydroquinone moieties, which allow them to act as electron donating and accepting agents (Fig. 4). There are two main compounds in this class, doxorubicin and daunorubicin, whose chemical structures differ only by a single hydroxyl group, but whose indications are unique. Doxorubicin and its related compound epirubicin are used for the treatment of solid tumors, such as breast adenocarcinoma or sarcomas, whereas daunorubicin and its related compound idarubicin are largely used for the treatment of acute leukemias (Chabner et al., 2011). Anthracyclines are known to be one of the most effective chemotherapy agents, which have been found to be therapeutically superior to other first generation cancer chemotherapeutic agents, such as cyclophosphamide, fluorouracil, and methotrexate in the treatment of early stage breast cancer (EBCTCG, 2005).
Fig. 4. Structure of daunorubicin (the prototypical anthracycline).
Anthracyclines contain a characteristic tetracyclic ring structure (A–D) and daunosamine glycoside (conjugated to A).
The therapeutic use of anthracyclines is limited due to the induction of debilitating cardiomyopathy and congestive heart failure. Acute cardiotoxicity and cardiac side effects that follow soon after induction therapy with anthracyclines can include ST and T wave changes, tachycardia, cardiac arrhythmias, reduced ejection fraction, hypotension, and increased levels of Troponin enzymes (Lipshultz et al., 2004). These effects are generally considered reversible (Singal et al., 1997). Chronic cardiac side effects can include dilated cardiomyopathy and left ventricular failure (Chabner et al., 2011). These effects are considered irreversible and generally lead to a poor prognosis (Singal et al., 1997). Early post-mortem studies of patients treated with large doses of anthracyclines describe cardiac specimens as having degenerating myocardial cells, a decreased overall number of cells, and mitochondrial swelling (Lefrak et al., 1973). Cardiac cell toxicity was found to be related to the cumulative dose of anthracyclines used. In a retrospective study of 399 patient records, heart failure reportedly occurred in 4% of patients who received 500 – 550 mg/m2 of doxorubicin, 18% of patients who received 551 to 600 mg/m2, and 36% of patients who received a dose greater than 601 mg/m2 (Lefrak et al., 1973). Limitations on cumulative lifetime doses of anthracyclines were subsequently set and patients now rarely ever exceed a lifetime dose of 550 mg/m2 of surface area.
It is widely accepted that the oxidative stress placed on myocardial cells is likely the major contributing factor to the cardiotoxic effect. Anthracyclines have a high affinity for the phospholipid cardiolipin, which is concentrated in mitochondrial inner membranes. Because cardiac myocytes are relatively rich in mitochondria, anthracyclines accumulate in cardiac cells at higher concentrations than other cells of the body (Goormaghtigh et al., 1990). It is likely that in addition to compound accrual, additional cardiomyocyte-specific metabolism is involved in anthracycline cardiotoxicity. The quinone moiety of anthracycline undergoes redox cycling either in the presence of certain enzymes or in the presence of iron to produce ROS (Fig. 5). This oxidative stress leads to cardiac myocyte damage and dilated cardiomyopathy.
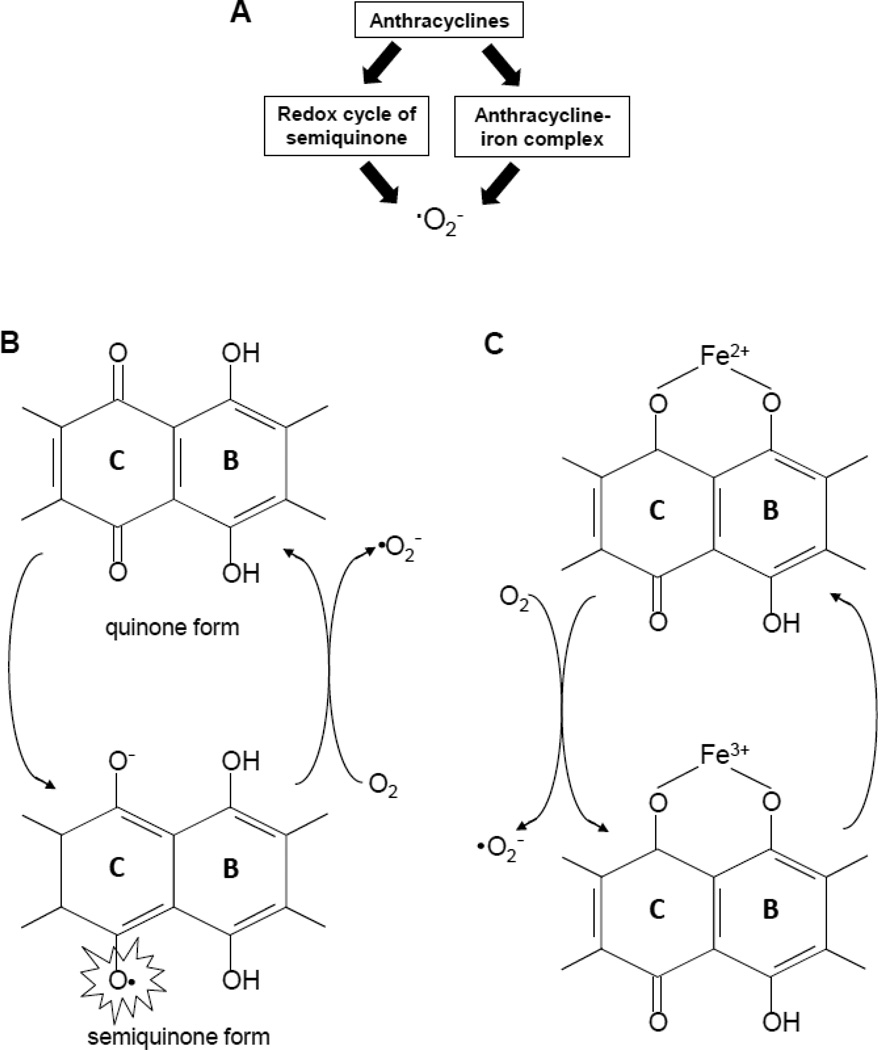
Fig. 5. Mechanisms of ROS generation from anthracyclines.
(A) Two major hypotheses include redox cycling of semiquinone and the formation of anthracycline-iron complex, both of which promote one-electron reduction of O2 to form ·O2−. (B) The redox cycling mechanism involves the reduction of the ring C to a semiquinone free radical by flavoprotein oxidoreductases. The semiquinone form is oxidized back to the quinone form in the presence of O2 forming ·O2− in the process. (C) Anthracycline-Fe3+ complex can be formed on rings C and B, thereby reducing to anthracycline-Fe2+, which in turn reduces O2 to form ·O2−.
There are two major hypotheses for the mechanism of anthracycline-mediated production of ROS: (i) one-electron reduction of O2 to form ·O2− through redox cycling of semiquinone and (ii) one-electron reduction of O2 to form ·O2− by the anthracycline-iron complex (Fig. 5A). In the cellular environment, the ring C of anthracyclines can be reduced to a semiquinone free radical by several flavoprotein oxidoreductases. Under normoxic conditions, the seminquinone free radical reduces O2 to form ·O2− (Fig. 5B). Through this catalytic redox cycling mechanism, a small amount of anthracyclines can produce abundant ·O2− The anthracycline-oxidized iron (Fe3+) complex can be reduced by NADH cytochrome P450 reductase or thiols of cysteine or glutathione. The anthracycline-reduced iron (Fe2+) complex reduces O2 to form ·O2− (Fig. 5C). Produced ·O2− can then form other ROS and reactive species as described above, thereby leading to cellular damage.
In vitro and animal models have been conducted to evaluate the role of incorporating antioxidants with anthracyclines in the hopes of reducing oxidative damage to cardiac cells. Early studies evaluated the effectiveness of common antioxidants found in the diet, which are known to prevent cellular oxidative damage. Many animal models showed a reduction in anthracycline cardiotoxicity by co-administering compounds such as vitamin E (Herman et al., 1983; Geetha et al., 1990; Lubawy et al., 1979; Myers et al., 1976; Milei et al., 1986), vitamin C (Shimpo et al., 1991), vitamin A (Tesoriere et al., 1994), coenzyme Q (Judy et al., 1984; Berrazzoli et al., 1975; Cortes et al., 1978), and flavonoids (Van Acker et al., 1997; Husken et al., 1995). Other pharmaceutical compounds with known antioxidant properties, such as probucol (Kumar et al., 2001; Iliskovic et al., 1999; Singal et al., 1995), carvedilol (Oliveira et al., 2004; Matsui et al., 1999), and dantrolene (Buyukokuroglu et al., 2004) have also been found to protect cardiac damage in animal models.
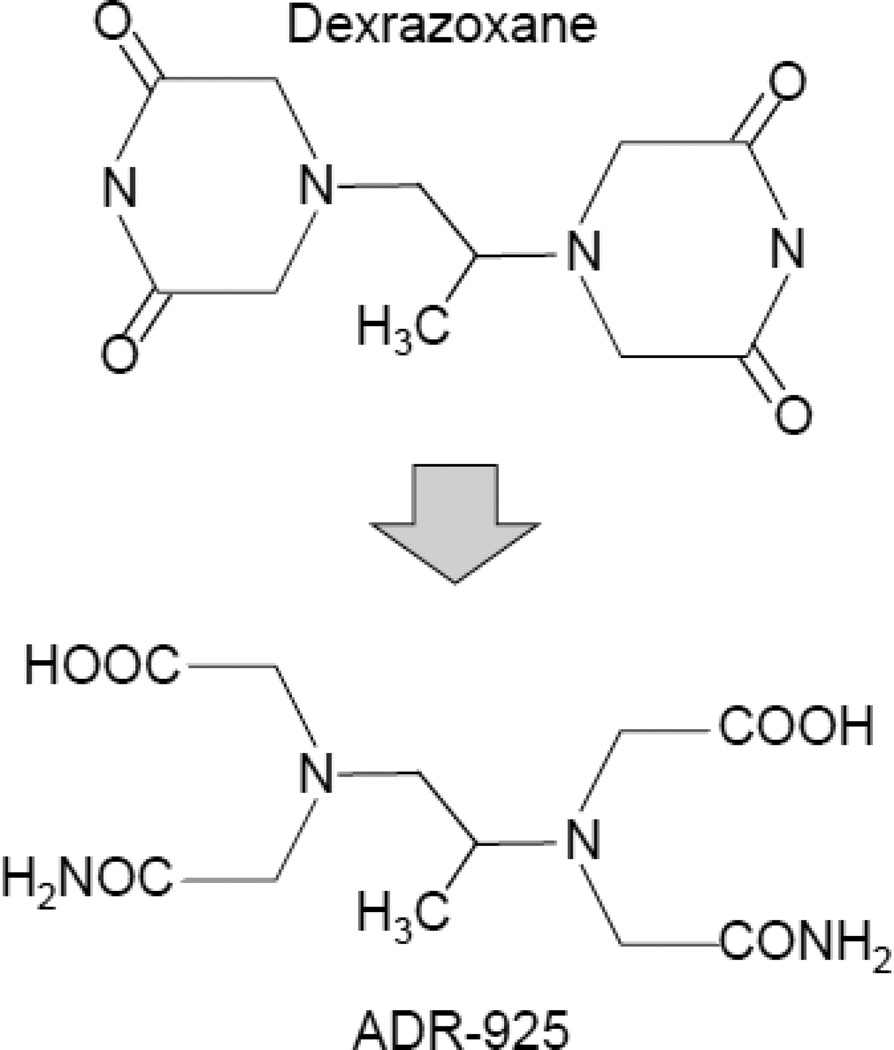
Much attention has been placed on the novel compound ICRF-187, which was later patented as dexrazoxane. Dexrazoxane can be hydrolyzed to an active metabolite (ADR-925) that is an iron chelate (Fig. 6), thereby decreasing the amount of iron available to bind with anthracyclines and thus inhibiting the formation of ROS. Early preclinical studies of ICRF-187 showed significantly reduced myocardial cell damage in animals co-administered dexrazoxane plus doxorubicin compared to doxorubicin alone (Herman et al., 1979; Herman et al., 1981). Human studies on this drug will be discussed later in this article.
Fig. 6. Structures of dexrazoxane and its active iron-chelating metabolite.
Dexrazoxane (ICRF-187) once administered is hydrolyzed to become its activate metabolite (ADR-925) that can chelate iron.
Trastuzumab (Herceptin)
Cell surface tyrosine kinase receptor HER2 (also known as HER2/neu and ErbB2) functions as an oncogene in some malignancies, including breast, lung, and endometrial cancer (Slamon et al., 1989; Roskoski et al., 2004). Thus, the inhibition of this signaling pathway is considered as an effective means for cancer treatment. Trastuzumab (Herceptin, produced by Genentech, CA), a monoclonal antibody that binds to HER2, was approved by the FDA in 1998 for treating breast cancer and has been shown to improve survival of patients (Slamon et al., 2001). Trastuzumab in combination with other chemotherapeutic agents, such as doxorubicin, increases both survival and response rate compared to trastuzumab alone (Nahta & Esteva, 2003).
Although a combination therapy with doxorubicin and trastuzumab has significantly reduced the morbidity and mortality in breast cancer patients, cardiotoxicity remains as a serious concern. While cardiotoxicity by doxorubicin treatment is expected, the risk for developing cardiac dysfunction is higher when trastuzumab is used as adjuvant therapy with doxorubicin (Wadhwa et al., 2009). Thus, agents that are capable of preventing cardiotoxicity in the setting of combination therapies would be useful.
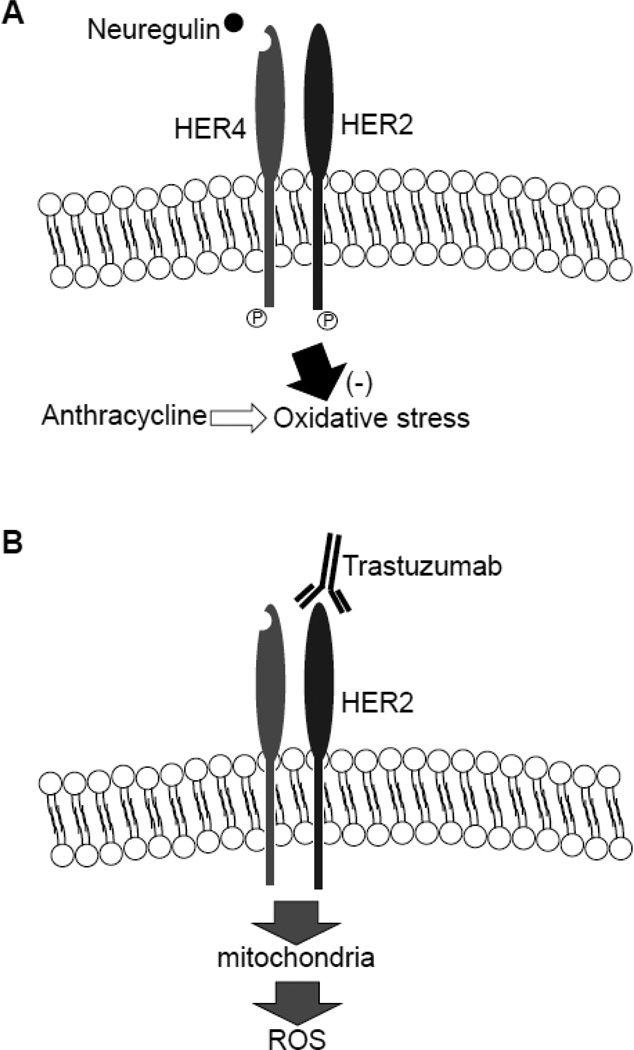
The question that is addressed in this review article is whether ROS are involved and if antioxidants may be useful for protecting the heart. It has been reported that activation of the HER2 receptor by neuregulin-1 reduces anthracycline-induced oxidative stress in adult rat cardiomyocytes (Timolati et al., 2006) (Fig. 7A). Therefore, the benefit of trastuzumab antagonism of neuregulin-1-HER2 signaling in cancer cells may be offset by decreasing the ability of cardiomyocytes to withstand ROS. Gordon et al. (2009) reported that the blockade of the neuregulin-1-ErbB2 receptor interaction induced cardiomyocyte death through mitochondrial ROS production (Fig. 7B). These results suggest that antioxidants may be useful for protecting the heart against toxicity induced by trastuzumab. Consistently, a murine study demonstrated that probucol attenuates cardiotoxicity induced by a combination of doxorubicin and trastuzumab (Walker et al., 2011). In rats, melatonin (which is capable of scavenging reactive species such as HO, H2O2, hypochlorous acid, and peroxynitrite), mercaptoethylguanidine (a compound that can inhibit inducible nitric oxide synthase and scavenges peroxinitrite) and 1400W (an inhibitor of inducible nitric oxide synthase) were all found to attenuate cardiotoxicity induced by a combination of doxorubicin and trastuzumab (Ozturk et al., 2011).
Fig. 7. Mechanism of HER2 receptor-regulation of oxidative stress in the heart.
(A) The activation of the HER2 receptor by neuregulin-1 reduces anthracycline-induced oxidative stress. (B) Inhibition of the HER2 receptor by trastuzumab produces mitochondrial ROS.
Taxanes
Taxanes produced by the plants of the genus Taxus (yews), including paclitaxel (Taxol) and docetaxel (Taxotere) interfere with microtubule dynamics and are widely used as antitumor agents. A combination of doxorubicin and paclitaxel has been shown to be effective in treating breast cancer. While paclitaxel alone does not induced cardiotoxicity, taxanes and anthracyclines appear to synergize toxicity (Perotti et al., 2003). This synergy is thought to be due to the ability of taxanes to stimulate an enzymatic conversion of doxorubicin to its secondary alcohol metabolite doxorubicinol (Salvatorelli et al., 2006). However, the levels of doxorubicin-derived ROS are not segmented by taxanes in human myocardial strips (Salvatorelli et al., 2007). In adult rat ventricular myocytes, a minor increase in ROS production by paclitaxel or by anti-ErbB2, as monitored by measuring dichlorofluorescein (DCF) fluorescence, was observed. However, no additive or synergistic effects by the two drugs were observed (Pentassuglia et al., 2007).
Cyclophosphamide
Cyclophosphamide is a nitrogen mustard-alkylating agent, which adds an alkyl group to DNA, and has been widely used in the treatment of chronic and acute leukemia, multiple myeloma, and lymphomas (Mythili et al., 2007). Cyclophosphamide-induced cardiotoxicity was found also to be attenuated by antioxidants, including alpha-lipoic acid (Mythili et al., 2006) and probucol (Asiri, 2010), in rats. A murine study demonstrated that while vitamin E has protective effects against doxorubicin-induced cardiotoxicity, this antioxidants did not provide protection against cardiotoxicity induced by a combination of doxorubicin and cyclophosphamide (Bjelogrlic et al., 2005). These results highlight the importance of understanding the effects of cardio-protectants in the setting of combination therapy.
Cisplatin
Cisplatin is the first member of a class of platinum-containing anti-cancer drugs. The platinum complexes kill cancer cells by crosslinking DNA. Ciplatin has broadly been used for the treatment of testicular, ovarian, cervical, bladder and lung cancers. Cisplatin has been associated with cardiotoxicity as well as other side effects, such as nephrotoxicity (Pay & Nahata, 2000). Various animal studies have suggested that compounds with antioxidant properties, such as resveratrol (Wang et al., 2009), propionyl-L-carnitine (AL-Majed et al., 2006), α-lipoic acid (El-Awady et al., 2011; Hussein et al., 2012), and proanthocyanidin (Yousef et al., 2009), attenuate cisplatin-induced cardiotoxicity.
5-Fluorouracil
5-Fluorouracil, a thymidylate synthase inhibitor, can exert cardiac side effects with an incidence of 1.2–7.6% (Alter et al., 2006). Recently, Lamberti et al. (2012) reported that 5-Fluorouracil induces apoptosis of rat cardiac cell line through the generation of ROS. In rabbits, an antioxidant probucol was also found to protect 5-fluorouracil-induced endothelial damage (Kinhult et al., 2003). In guinea pigs, 5-fluorouracil was found to decrease the activities of cardiac superoxide dismutase and glutathione peroxidase, while it increased the catalase activity (Durak et al., 2000). Interestingly, in rats, 5-fluorouracil was found to inhibit doxorubicin-induced cardiotoxicity by reducing the levels of ROS (Stathopoulos et al., 1998).
ROS and mechanisms of antitumor actions
In regard to mechanisms of cardiotoxicity that involve ROS, antioxidants that have the ability to inhibit the generation and actions of ROS are expected to be useful in protecting the heart during cancer chemotherapy. However, ROS may also be involved in the mechanism of antitumor drugs to kill cancer cells. Thus, it has been generally speculated that the use of antioxidants may reduce the efficacy of these drugs to treat cancer, but the question remains as to whether ROS contribute to the anti-neoplastic effect of chemotherapeutic agents.
The mechanism by which anthracyclines exhibit anti-tumor effects is likely multifactorial. These agents can interact directly with DNA by intercalating themselves within the planar ring structure and thereby mechanically block the process of DNA replication and RNA synthesis (DuVernay et al., 1979). The intercalated anthracycline can also inhibit topoisomerase II, leading to apoptosis (Chabner et al., 2011). It has been suggested that tumor cells may become resistant to anthracyclines because of overexpression or altered expression of topoisomerase II (O’Malley et al., 2009).
The degree to which ROS contribute to the anti-neoplastic effects of anthracyclines has been a source of controversy. Similar to the case in the heart, ROS could be generated by anthracyclines through either enzymatic or non-enzymatic pathways. In the presence of enzymes such as NADH dehydrogenase, cytochrome P450 reductases, or nitric oxide synthase, the quinone moiety of anthracyclines will be reduced to a semiquinone, which in turn can donate an election to molecular O2 to form ·O2− The conversion of the quinine portion of anthracyclines can also be catalyzed non-enzymatically in the presence of iron through the formation of an anthracycline-iron complex that undergoes oxidation-reduction cycling and creates ·O2− as a byproduct (Serrano et al., 1999). The accumulation of intracellular ROS has been shown to result in DNA strand degradation and breakage (Bates et al., 1982; Eliot et al., 1988). However, many of the in vitro studies used greater intracellular concentrations of anthracyclines than those clinically utilized. While some studies have concluded that ROS do not contribute to anti-tumor effects of anthracyclines (Keizer et al., 1990), it remains unclear if the clinical doses of anthracycline chemotherapy creates a sufficient amount of oxidative stress to be toxic to tumor cells (Gewirtz, 1999).
Given the possibility that ROS formation may also contribute to the anti-neoplastic qualities of anthracyclines, early studies were also conducted to evaluate the efficacy of anthracyclines as a chemotherapy regimen when co-administered with antioxidant compounds. In most cases, the direct addition of antioxidants such as N-acetylcysteine (Myers et al., 1977; Myers et al., 1983), glutathione (Yoda et al., 1986), vitamin C (Shimpo et al., 1991; Cervantes et al., 1988), probucol (Siveski-Iliskovic et al., 1994), ICRF-187 (Woodman et al., 1975) and carvedilol (Oliveira et al., 2004) to the cell culture did not reduce the anti-neoplastic efficacy of anthracyclines in a variety of tumor cells. This has been considered strong evidence indicating that ROS accumulation and oxidative stress is not the major mechanism by which anthracyclines kill cancer cells. Despite reports that the inhibition of ErbB2 produces ROS in cancer cells (Dogan et al., 2011; Aird et al., 2012), the use of antioxidants during trastuzumab therapy may be supported by the observations that a vitamin E analog can enhance the antitumor activity of trastuzumab (Hahn et al., 2011).
The heart may be more susceptible to ROS damage due to of relatively low levels of antioxidant enzymes and because antitumor agents further reduce the levels of antioxidant enzymes. Cardiac muscle has been reported to contain 150 times less catalase and 4 times less superoxide dismutase then liver (Doroshow et al., 1980). Doxorubicin also decreases glutathione peroxidase and manganese superoxide dismutase (Li & Singal, 2000).
Human clinical trials using antioxidants to protect against cardiotoxicity
Some human clinical trials have been conducted to test the hypothesis that co-administration of antioxidants with cancer chemotherapeutic agents protect the heart without reducing the efficacy of chemotherapy. These studies are summarized below. However, most of these studies were underpowered, and larger multi-center trials must be designed to provide a sufficient number of participants to effectively evaluate if there is a benefit to these antioxidants. Furthermore, most of these studies examined the effects of anthracycline alone, and thus information on combination therapies is lacking.
N-Acetylcysteine
N-Acetylcysteine is a precursor of glutathione and its thiol moiety has been shown to reduce biological oxidants. Myers et al. (1983) reported the results of a randomized controlled trial to evaluate combination chemotherapy with oral N-acetylcysteine. This study had a total enrollment of 54 adult patients with solid tumors; 30 in the control group who received 75 mg/m2 of doxorubicin, and 24 in the intervention group who received 5.5 mg/m2 of N-acetylcysteine together with doxorubicin. Five patients in the intervention group refused to continue taking N-acetylcysteine secondary to nausea. After a 30 month follow-up, three patients in each group developed clinical heart failure (12.5% vs. 10%, P = 0.77). No data on the tumor response rate between groups were collected.
Amifostine
Amifostine is an organic thiophosphate prodrug that is dephosphorylated by alkaline phosphatases in vivo to an active thiol-containing metabolite with antioxidant properties. A randomized controlled trial was conducted by Gallegos-Castorena et al. (2007) to evaluate combination chemotherapy with amifostine infusions. Twenty-eight pediatric patients with untreated osteosarcoma were enrolled; 13 in the control group received combination chemotherapy with cisplatin and doxorubicin 75 mg/m2, and 15 in the intervention group received 740 mg/m2 amifostine infusion prior to chemotherapy infusion. At follow-up, two patients in the control group developed clinical heart failure compared to zero patients in the intervention group (P = 0.175). No data on the tumor response rate between groups were collected.
L-Carnitine
L-carnitine facilitates the transport of fatty acids into the mitochondrial matrix and can play a role in abating myocardial lipid peroxidation. Waldner et al. (2006) conducted a randomized controlled trial to evaluate combination chemotherapy with L-carnitine infusions. Forty adult patients with non-Hodgkin Lymphomas were randomly assigned to a study group; 20 in the control group received six cycles of CHOP therapy that included doxorubicin 50 mg/m2, and 20 in the intervention group received an additional infusion of 3 g L-carnitine before chemotherapy. No patients in the control or intervention groups developed heart failure and there was no significant difference in the left ventricular ejection fraction according to echocardiography following completion of six chemotherapy cycles. Additionally, there was no difference in the quality of life between the two study groups according to a standardized questionnaire. No data on the tumor response rate between groups were collected.
Carvedilol
Carvedilol is a cardioselective beta-blocker with antioxidant properties. A randomized controlled trial has been conducted evaluating combination chemotherapy with oral carvedilol (Kalay et al., 2006). Fifty adult patients with newly diagnosed solid tumors were enrolled; 25 in the control group received anthracycline based chemotherapy and 25 in the intervention group received 12.5 mg oral carvedilol during and following chemotherapy. One patient in the control group and no patients in the intervention group developed clinical heart failure (P = 1). Five patients in the control group and one patient in the intervention group developed subclinical heart failure as defined by echocardiography results (P = 0.13). No data on the tumor response rate between groups were collected.
Coenzyme Q10
Iarussi et al. (1994) reported results of a randomized controlled trial evaluating combination chemotherapy with oral coenzyme Q10. Twenty pediatric patients with either solid tumor or leukemia were enrolled; 10 patients in the control group received doxorubicin and 10 patients in the intervention group received 100 mg oral coenzyme Q10 twice daily during chemotherapy. No patients developed clinical or subclinical heart failure in either the control or intervention groups. No data on the tumor response rate between groups were collected.
Dexrazoxane
Seven randomized controlled trials have been conducted evaluating combination chemotherapy with oral dexrazoxane (Lipshultz et al., 2004; Lopez et al., 1998; Marty et al., 2006; Schwartz et al., 2009; Speyer et al., 1992; Swain et al., 1997; Venturini et al., 1996). Data from these studies were recently pooled for a meta-analysis through a Cochrane review (Van Dalen et al., 2011). Among these studies, a total of 1561 patients were enrolled; 792 were assigned to a control group receiving anthracycline-based chemotherapy and 769 patients were assigned to the intervention group receiving IV infusion of dexrazoxane at a varying dose of 10:1 or 20:1 ratio of study drug to doxorubicin. Sixty-nine patients in the control groups developed clinical heart failure compared with 11 patients in the intervention group (P < 0.00001, RR 0.18, 95% CI 0.10–0.32).
The Cochrane group was also able to pool data on tumor response rates from five trials (Lopez 1998; Marty 2006; Speyer et al., 1992; Swain et al., 1997; Venturini et al., 1996). These studies used standardized WHO criteria to evaluate clinical response to chemotherapy. Among 518 patients in the control groups, 260 reported complete or partial response to chemotherapy, and among 503 patients in the intervention groups with dexrazoxane, 223 patients reported complete and partial responses (P = 0.08). Additionally, when survival data was pooled from three trials (Marty et al., 2006; Speyer et al., 1992; Swain et al., 1997), no significant difference between the control or intervention groups was found in terms of overall or progression-free survival.
Melatonin
Lissoni et al. (1999) conducted a human study to investigate the effects of daily oral administration of melatonin on clinical outcomes of chemotherapeutic agents given to 250 patients with various cancers. Chemotherapeutic agents consisted of cisplatin plus etoposide or gemcitabine alone for lung cancer, doxorubicin alone, mitoxantrone alone or paclitaxel alone for breast cancer, 5-fluorouracil plus folinic acid for gastrointestinal tumors, and 5-fluorouracil plus cisplatin for head and neck cancer. After 1-year follow-up, the survival rate was found to be significantly higher in patients who were concomitantly treated with melatonin with therapeutic agents. The concomitant administration of melatonin also significantly reduced the frequency of side effects, including cardiotoxicity.
Limitations of existing human data
Cardiotoxicity of anthracycline-based chemotherapy remains a significant challenge and is the reason why lifetime cumulative dosing of these drugs has been limited. To date, the only compound to successfully reduce the incidence of heart failure has been the iron chelate dexrazoxane. However, despite improved clinical pictures of these patients and improved quality of life scores, controlled trials have not been able to show a survival benefit for patients co-treated with dexrazoxane. The vast majority of deaths in these trials were due to complications associated with the progression of cancer, such as pulmonary emboli and sepsis. Based on early investigations and more recent clinical trials, it is clear that anthracyclines are one of the most effective induction chemotherapies and that their anti-neoplastic cytotoxicity is improves with increased dosing (EBCTCG, 2005).
Future controlled trials with dexrazoxane are needed to compare larger doses of anthracycline chemotherapy plus dexrazoxane compared to current doses of anthracycline chemotherapy plus dexrazoxane. At this point, dosing of anthracyclines is limited to less that 550 mg/m2 because early investigations showed that dosing above this amount increases the risk of clinical heart failure to as high as 18% (Lefrak et al., 1973). However, given that trials showed dexrazoxane significantly reduces the risk of clinical heart failure, that increased doses of anthracyclines contribute to increased anti-neoplastic cytotoxicity, and that survival seems to be related more to the complications of progressive cancer rather than heart failure, increasing the cumulative dose of anthracyclines to above 550 mg/m2 plus dexrazoxane may increase the incidence of heart failure, but would also be a more effective chemotherapy regimen and increase overall survival.
Dexrazoxane trials should also be extended to examine its effects on other antitumor drugs, particularly when they are used in combination with agents such as anthracyclines. In the modern era, it is critical to understand the effects of dexrazoxane on patients who have completed chemotherapy to assess its long-term effects. This may be a challenge, given the side effects and discomfort that dexazoxane exerts on patients. In this regard, the use of antioxidants with fewer side effects would be an attractive approach.
Randomized controlled trials on other common compounds with antioxidant properties have been limited. None of the trials that examined the effectiveness of L-carnitine, amifostine, carvedilol, or coenzyme Q10 on anthracyclines showed a reduction in heart failure. However, all of these studies were underpowered. Larger multi-center trials must be designed to provide a sufficient number of participants to effectively evaluate if these antioxidant compounds provide a benefit. In addition to suffering from low statistical power, the randomized clinical trial investigating N-acetylcysteine was also hindered by poor patient compliance. Five patients out of twenty-four patients in the intervention group could not tolerate ingesting the study drug secondary to nausea. Thus, future, large randomized clinical trials may investigate an IV infusion of N-acetylcysteine as a method of delivering this antioxidant.
It is especially notable that no randomized clinical trials have been published as yet evaluating probucol as a possible cardioprotectant. This drug has fallen out of favor as a lipid lowering agent due to undesirable decreases in high density lipoprotein. However, it has been shown to have strong antioxidant properties in vitro (Mao et al., 1991). Animal models have also shown it to be effective at preventing the development of anthracycline-induced cardiomyopathy while not affecting the anti-neoplastic toxicity of anthracyclines (Siveski-Iliskovic et al., 1994). In addition, probucol was found to attenuate cardiotoxicity induced by a combination of doxorubicin and trastuzumab in mice (Walker et al., 2011).
An important consideration for antioxidant intervention is the latency period during the development of anthracycline cardiotoxicity. Therefore, it may be possible to uncouple initial anti-neoplastic toxicity from secondary progression of damage to the heart through a post-anthracycline treatment regimen of putative cardioprotective antioxidants. Moreover, trials should be designed to measure plasma levels of antioxidants to account for individual variability in absorption and metabolism during this phase. In this regard, responses may be stratified based on the effective concentrations of antioxidant in plasma, rather than solely on whether a subject had ingested the compound relative to control.
Conclusions
Recent success of cancer chemotherapy with combination therapy has raised the old argument on whether cancer patients should receive dietary and/or pharmacologic antioxidants. Whether antioxidants adversely affect the cancer cell killing function of chemotherapeutic agents while exerting cardioprotective effects in humans remain unclear. A critical question that still remains is if patients who have completed chemotherapy should receive antioxidants. The answer to this question will remain unknown until well-designed randomized clinical trials are conducted.
References
- 1.Aird KM, Allensworth JL, Batinic-Haberle I, Lyerly HK, Dewhirst MW, Devi GR. ErbB1/2 tyrosine kinase inhibitor mediates oxidative stress-induced apoptosis in inflammatory breast cancer cells. Breast Cancer Res Treat. 2012;132:109–119. doi: 10.1007/s10549-011-1568-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Al-Majed AA, Sayed-Ahmed MM, Al-Yahya AA, Aleisa AM, Al-Rejaie SS, Al-Shabanah OA. Propionyl-L-carnitine prevents the progression of cisplatin-induced cardiomyopathy in a carnitine-depleted rat model. Pharmacol Res. 2006;53:278–286. doi: 10.1016/j.phrs.2005.12.005. [DOI] [PubMed] [Google Scholar]
- 3.Albini A, Pennesi G, Donatelli F, Cammarota R, De Flora S, Noonan DM. Cardiotoxicity of anticancer drugs: The need for Cardio-Onclology and Cardio-Oncological Prevention. J. Natl. Cancer Inst. 2010;102:14–25. doi: 10.1093/jnci/djp440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alter P, Herzum M, Soufi M, Schaefer JR, Maisch B. Cardiotoxicity of 5-fluorouracil. Cardiovasc. Hematol. Agents Med. Chem. 2006;4:1–5. doi: 10.2174/187152506775268785. [DOI] [PubMed] [Google Scholar]
- 5.Asiri YA. Probucol attenuates cyclophosphamide-induced oxidative apoptosis, p53 and Bax signal expression in rat cardiac tissues. Oxid. Med. Cell Longev. 2010;3:308–316. doi: 10.4161/oxim.3.5.13107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bates DA, Winterbourn CC. Deoxyribose breakdown by the adriamycin semiquinone and H2O2: Evidence of hydroxyl radical participation. FEBS Lett. 1982;145:137–142. doi: 10.1016/0014-5793(82)81222-2. [DOI] [PubMed] [Google Scholar]
- 7.Berrazzoli C, Sala L, Tosana MG. Antagonistic action of ubiquinone on the experimental cardiotoxicity of adriamycin in the isolated rabbit heart. Int. Res. Commun. Syst. Med. Sci. 1975;3:342–367. [Google Scholar]
- 8.Bjelogrlic SK, Radic J, Jovic V, Radulovic S. Activity of d,l-alpha-tocopherol (vitamin E) against cardiotoxicity induced by doxorubicin and doxorubicin with cyclophosphamide in mice. Basic Clin. Pharmacol. Toxicol. 2005;97:311–319. doi: 10.1111/j.1742-7843.2005.pto_166.x. [DOI] [PubMed] [Google Scholar]
- 9.Buyukokuroglu ME, Taysi S, Buyukavci M, Bakan E. Prevention of acute adriamycin cardiotoxicity by dantrolene in rats. Hum. Exp. Toxicol. 2004;23:251–256. doi: 10.1191/0960327104ht443oa. [DOI] [PubMed] [Google Scholar]
- 10.Buzdar AU, Legha SS, Tashima CK, Hortobagyi GN, Yap HY, Krutchik AN, Luna MA, Blumenschein GR. Adriamycin and mitomycin C: possible synergistic cardiotoxicity. Cancer Treat. Rep. 1978;62:1005–1008. [PubMed] [Google Scholar]
- 11.Cervantes A, Pinedo HM, Lankelma J, Schuurhuis CJ. The role of oxygen-derived free radicals in the cytotoxicity of doxorubicin in multidrug resistant and sensitive human ovarian cancer cells. Cancer Lett. 1988;41:169–177. doi: 10.1016/0304-3835(88)90113-9. [DOI] [PubMed] [Google Scholar]
- 12.Chabner BA, Bertino J, Cleary J, Ortiz T, Lane A, Supko JG, Ryan D. Chapter 61. Cytotoxic Agents. In: Brunton LL, Chabner BA, Knollmann BC, editors. Goodman & Gilman's The Pharmacological Basis of Therapeutics. 12nd ed. New York: McGraw-Hill; 2011. [Google Scholar]
- 13.Cortes EP, Gupta M, Chou C, Amin VC, Folker K. Adriamycin cardiotoxicity: early detection by systolic time interval and possible prevention by coenzyme. Q. Cancer Treat. Rep. 1978;62:887–892. [PubMed] [Google Scholar]
- 14.Dogan I, Cumaoglu A, Aricioglu A, Ekmekci A. Inhibition of ErbB2 by herceptin reduces viability and survival, induces apoptosis and oxidative stress in Calu-3 cell line. Mol. Cell. Biochem. 2011;347:41–51. doi: 10.1007/s11010-010-0610-7. [DOI] [PubMed] [Google Scholar]
- 15.Doroshow JH, Locker GY, Myers CE. Enzymatic defenses of the mouse heart against reactive oxygen metabolites: alterations produced by doxorubicin. J. Clin. Invest. 1980;65:128–135. doi: 10.1172/JCI109642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Durak I, Karaayvaz M, Kavutcu M, Cimen MY, Kaçmaz M, Büyükkoçak S, Oztürk HS. Reduced antioxidant defense capacity in myocardial tissue from guinea pigs treated with 5-fluorouracil. J. Toxicol. Environ. Health A. 2000;59:585–589. doi: 10.1080/009841000156709. [DOI] [PubMed] [Google Scholar]
- 17.DuVernay VH, Jr, Pachter JA, Crooke ST. Deoxyribonucleic acid binding studies on several new anthracycline antitumor antibiotics. Sequence preference and structure activity relationships of marcellomycin and its analogues as compared to adriamycin. Biochemistry. 1979;18:4024–4030. doi: 10.1021/bi00585a028. [DOI] [PubMed] [Google Scholar]
- 18.Early Breast Cancer Trials Collaborative Group (EBCTCG) Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: An overview of the randomized trials. Lancet. 2005;365:1687–1717. doi: 10.1016/S0140-6736(05)66544-0. [DOI] [PubMed] [Google Scholar]
- 19.El-Awady el-SE, Moustafa YM, Abo-Elmatty DM, Radwan A. Cisplatin-induced cardiotoxicity: Mechanisms and cardioprotective strategies. Eur. J. Pharmacol. 2011;650:335–341. doi: 10.1016/j.ejphar.2010.09.085. [DOI] [PubMed] [Google Scholar]
- 20.Eliot H, Gianni L, Myers C. Oxidative destruction of DNA by the adriamycin-iron complex. Biochemistry. 1984;23:928–936. doi: 10.1021/bi00300a021. [DOI] [PubMed] [Google Scholar]
- 21.Feliz V, Saiyad S, Ramarao SM, Khan H, Leonelli F, Guglin M. Melphalan-induced supraventricular tachycardia: incidence and risk factors. Clin Cardiol. 2011;34:356–359. doi: 10.1002/clc.20904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Galetta F, Franzoni F, Cervetti G, Cecconi N, Carpi A, Petrini M, Santoro G. Effect of epirubicin-based chemotherapy and dexrazoxane supplementation on QT dispersion in non-Hodgkin lymphoma patients. Biomed. Pharmacother. 2005;59:541–544. doi: 10.1016/j.biopha.2004.12.003. [DOI] [PubMed] [Google Scholar]
- 23.Gallegos-Castorena S, Martínez-Avalos A, Mohar-Betancourt A, Guerrero-Avendaño G, Zapata-Tarrés M, Medina-Sansón A. Toxicity prevention with amifostine in pediatric osteosarcoma patients treated with cisplatin and doxorubicin. Pediatr. Hematol. Oncol. 2007;24:403–408. doi: 10.1080/08880010701451244. [DOI] [PubMed] [Google Scholar]
- 24.Geetha A, Sankar R, Marar T, Devi CS. Alpha-tocopherol reduces doxorubicin-induced toxicity in ratshistological and biochemical evidences. Ind. J. Physiol. Pharmacol. 1990;34:94–100. [PubMed] [Google Scholar]
- 25.Gewirtz DA. A critical evaluation of the mechanisms of action proposed for the antitumor effects of the anthracycline antibiotics adriamycin and daunorubicin. Biochem. Pharmacol. 1999;57:727–741. doi: 10.1016/s0006-2952(98)00307-4. [DOI] [PubMed] [Google Scholar]
- 26.Goormaghtigh E, Huart P, Brasseur R, Ruysschaert JM. Structure of the adriamycincardiolipin complex. Role in mitochondrial toxicity. Biophys. Chem. 1990;35:247–257. doi: 10.1016/0301-4622(90)80012-v. [DOI] [PubMed] [Google Scholar]
- 27.Gordon LI, Burke MA, Singh AT, Prachand S, Lieberman ED, Sun L, Naik TJ, Prasad SV, Ardehali H. Blockade of the erbB2 receptor induces cardiomyocyte death through mitochondrial and reactive oxygen species-dependent pathways. J. Biol. Chem. 2009;284:2080–2087. doi: 10.1074/jbc.M804570200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gottdiener JS, Appelbaum FR, Ferrans VJ, Deisseroth A, Ziegler J. Cardiotoxicity associated with high-dose cyclophosphamide therapy. Arch. Intern. Med. 1981;141:758–763. [PubMed] [Google Scholar]
- 29.Hahn T, Bradley-Dunlop DJ, Hurley LH, Von-Hoff D, Gately S, Mary DL, Lu H, Penichet ML, Besselsen DG, Cole BB, Meeuwsen T, Walker E, Akporiaye ET. The vitamin E analog, alpha-tocopheryloxyacetic acid enhances the anti-tumor activity of trastuzumab against HER2/neu-expressing breast cancer. BMC Cancer. 2011;11:471. doi: 10.1186/1471-2407-11-471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Halliwell B, Gutteridge J. Free Radicals in Biology and Medicine. Oxford: Oxford University Press; 1985. [Google Scholar]
- 31.Herman E, Ardalan B, Bier C, Waravdekar V. Reduction of daunorubicin lethality and myocardial cellular alterations by pretreatment with ICRF-187 in Syrian golden hamsters. Cancer Treat. Rep. 1979;63:89–92. [PubMed] [Google Scholar]
- 32.Herman EH, Ferrans VJ. Reduction of chronic doxorubicin cardiotoxicity in dogs by pretreatment with (+/−)-1,2-bis(3,5-dioxopiperazinyl-1-yl)propane (ICRF-187) Cancer Res. 1981;41:3436–3440. [PubMed] [Google Scholar]
- 33.Herman EH, Ferrans VJ. Influence of vitamin E and ICRF-187 on chronic doxorubicin cardiotoxicity in miniature swine. Lab. Invest. 1983;49:69–77. [PubMed] [Google Scholar]
- 34.Husken BC, de Jong J, Beekman B, Onderwater RC, Van der Vijgh WJ, Bast A. Modulation of the in vitro cardiotoxicity of doxorubicin by flavonoids. Cancer Chemother. Pharmacol. 1995;37:55–62. doi: 10.1007/BF00685629. [DOI] [PubMed] [Google Scholar]
- 35.Hussein A, Ahmed AA, Shouman SA, Sharawy S. Ameliorating effect of DL-α-lipoic acid against cisplatin-induced nephrotoxicity and cardiotoxicity in experimental animals. Drug Discov. Ther. 2012;6:147–156. [PubMed] [Google Scholar]
- 36.Iarussi D, Auricchio U, Agretto A, Murano A, Giuliano M, Casale F, Indolfi P, Iacono A. Protective effect of coenzyme Q10 on anthracyclines cardiotoxicity: control study in children with acute lymphoblastic leukemia and non-hodgkin lymphoma. Mol. Aspects Med. 1994;15:s207–s212. doi: 10.1016/0098-2997(94)90030-2. [DOI] [PubMed] [Google Scholar]
- 37.Iliskovic N, Hasinoff BB, Malisza KL, Li T, Danelisen I, Singal PK. Mechanism of beneficial effects of probucol in adriamycin cardiomyopathy. Mol. Cell. Biochem. 1999;196:43–49. [PubMed] [Google Scholar]
- 38.Judy WV, Hall JM, Dugan W, Toth PD, Folkers K. Coenzyme Q10 reduction of adriamycin cardiotoxicity. In: Folkers K, Yamamura Y, editors. Biomedical and Clinical Aspects of Coenzyme Q. vol. 4. Amsterdam: Elsevier; 1984. pp. 231–240. [Google Scholar]
- 39.Kalay N, Basar E, Ozdogru I, Er O, Cetinkaya Y, Dogan A, Inanc T, Oguzhan A, Eryol NK, Topsakal R, Ergin A. Protective effects of carvedilol against anthracycline-induced cardiomyopathy. J. Am. Coll. Cardiol. 2006;48:2258–2262. doi: 10.1016/j.jacc.2006.07.052. [DOI] [PubMed] [Google Scholar]
- 40.Kandylis K, Vassilomanolakis M, Tsoussis S, Efremidis AP. Ifosfamide cardiotoxicity in humans. Cancer Chemother. Pharmacol. 1989;24:395–396. doi: 10.1007/BF00257451. [DOI] [PubMed] [Google Scholar]
- 41.Keefe DL. Trastuzumab-associated cardiotoxicity. Cancer. 2002;95:1592–1600. doi: 10.1002/cncr.10854. [DOI] [PubMed] [Google Scholar]
- 42.Kesavan Shan MDA, AMichael Lincoff MD, James B, Young MD. Anthracycline-Induced Cardiotoxicity. Ann. Intern. Med. 1996;125:47–58. doi: 10.7326/0003-4819-125-1-199607010-00008. [DOI] [PubMed] [Google Scholar]
- 43.Lefrak EA, Pitha J, Rosenheim S, Gottlieb JA. A clinicopathologic analysis of adriamycin cardiotoxicity. Cancer. 1973;32:302–314. doi: 10.1002/1097-0142(197308)32:2<302::aid-cncr2820320205>3.0.co;2-2. [DOI] [PubMed] [Google Scholar]
- 44.Keizer HG, Pinedo HM, Schuurhuis GJ, Joenje H. Doxorubicin (adriamycin): A critical review of free radical dependent mechanisms of cytotoxicity. Pharmacol. Ther. 1990;47:219–231. doi: 10.1016/0163-7258(90)90088-j. [DOI] [PubMed] [Google Scholar]
- 45.Kinhult S, Albertsson M, Eskilsson J, Cwikiel M. Effects of probucol on endothelial damage by 5-fluorouracil. Acta Oncol. 2003;42:304–308. doi: 10.1080/02841860310004409. [DOI] [PubMed] [Google Scholar]
- 46.Kraft J, Grille W, Appelt M, Hossfeld DK, Eichelbaum M, Koslowski B, Quabeck K, Kuse R, Büchner T, Hiddemann W, et al. Effects of verapamil on anthracycline induced cardiomyopathy: preliminary results of a prospective multicenter trial. Haematol. Blood Transfus. 1990;33:566–570. doi: 10.1007/978-3-642-74643-7_103. [DOI] [PubMed] [Google Scholar]
- 47.Kumar D, Kirshenbaum LA, Li T, Danelisen I, Singal PK. Apoptosis in adriamycin cardiomyopathy and its modulation by probucol. Antioxid. Redox Signal. 2001;3:135–145. doi: 10.1089/152308601750100641. [DOI] [PubMed] [Google Scholar]
- 48.Lamberti M, Porto S, Marra M, Zappavigna S, Grimaldi A, Feola D, Pesce D, Naviglio S, Spina A, Sannolo N, Caraglia M. 5-Fluorouracil induces apoptosis in rat cardiocytes through intracellular oxidative stress. J. Exp. Clin. Cancer Res. 2012;31:60. doi: 10.1186/1756-9966-31-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lefrak EA, Pitha J, Rosenheim S, Gottlieb JA. A clinicopathologic analysis of adriamycin cardiotoxicity. Cancer. 1973;32:302–314. doi: 10.1002/1097-0142(197308)32:2<302::aid-cncr2820320205>3.0.co;2-2. [DOI] [PubMed] [Google Scholar]
- 50.Li T, Singal PK. Adriamycin-induced early changes in myocardial antioxidant enzymes and their modulation by probucol. Circulation. 2000;102:2105–2110. doi: 10.1161/01.cir.102.17.2105. [DOI] [PubMed] [Google Scholar]
- 51.Lipshultz SE, Rifai N, Dalton VM, Levy DE, Silverman LB, Lipsitz SR, Colan SD, Asselin BL, Barr RD, Clavell LA, Hurwitz CA, Moghrabi A, Samson Y, Schorin MA, Gelber RD, Sallan SE. The effect of dexrazoxane on myocardial injury in doxorubicin-treated children with acute lymphoblastic leukemia. N. Engl. J. Med. 2004;351:145–153. doi: 10.1056/NEJMoa035153. [DOI] [PubMed] [Google Scholar]
- 52.Lipshultz SE, Scully RE, Lipsitz SR, Sallan SE, Silverman LB, Miller TL, Barry EV, Asselin BL, Athale U, Clavell LA, Larsen E, Moghrabi A, Samson Y, Michon B, Schorin MA, Cohen HJ, Neuberg DS, Orav EJ, Colan SD. Assessment of dexrazoxane as a cardioprotectant in doxorubicin-treated children with high-risk acute lymphoblastic leukaemia: long-term follow-up of a prospective, randomised, multicentre trial. Lancet Oncol. 2010;11:950–961. doi: 10.1016/S1470-2045(10)70204-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lissoni P, Barni S, Mandalà M, Ardizzoia A, Paolorossi F, Vaghi M, Longarini R, Malugani F, Tancini G. Decreased toxicity and increased efficacy of cancer chemotherapy using the pineal hormone melatonin in metastatic solid tumour patients with poor clinical status. Eur. J. Cancer. 1999;35:1688–1692. doi: 10.1016/s0959-8049(99)00159-8. [DOI] [PubMed] [Google Scholar]
- 54.Lopez M, Vici P, Di Lauro K, Conti F, Paoletti G, Ferraironi A, Sciuto R, Giannarelli D, Maini CL. Randomized prospective clinical trial of high-dose epirubicin and dexrazoxane in patients with advanced breast cancer and soft tissue sarcomas. J. Clin. Oncol. 1998;16:86–92. doi: 10.1200/JCO.1998.16.1.86. [DOI] [PubMed] [Google Scholar]
- 55.Lubawy WC, Whaley J, Hurley LH. Coenzyme Q10 or alpha-tocopherol reduces the acute toxicity of anthramycin in mice. Res. Commun. Chem. Pathol. Pharmacol. 1979;24:401–404. [PubMed] [Google Scholar]
- 56.Maher J, Yamamoto M. The rise of antioxidant signaling--the evolution and hormetic actions of Nrf2. Toxicol. Appl. Pharmacol. 2010;244:4–15. doi: 10.1016/j.taap.2010.01.011. [DOI] [PubMed] [Google Scholar]
- 57.Mao SJ, Yates MT, Rechtin AE, Jackson RL, Van Sickle WA. Antioxidant activity of probucol and its analogues in hypercholesterolemic Watanabe rabbits. J. Med. Chem. 1991;34:298–302. doi: 10.1021/jm00105a046. [DOI] [PubMed] [Google Scholar]
- 58.Marty M, Espie M, Llombart A, Monnier A, Rapoport BL, Stahalova V. Multicenter randomized phase III study of the cardioprotective effect of dexrazoxane (Cardioxane) in advanced/metastatic breast cancer patients treated with anthracycline-based chemotherapy. Ann. Oncol. 2006;17:614–622. doi: 10.1093/annonc/mdj134. [DOI] [PubMed] [Google Scholar]
- 59.Matsui H, Morishima I, Numaguchi Y, Toki Y, Okumura K, Hayakawa T. Protective effects of carvedilol against doxorubcin-induced cardiomyopathy in rats. Life Sci. 1999;65:1265–1274. doi: 10.1016/s0024-3205(99)00362-8. [DOI] [PubMed] [Google Scholar]
- 60.Milei J, Boveris A, Llesuy S, Molina HA, Storino R, Ortega D, Milei SE. Amelioration of adriamycin- induced cardiotoxicity in rabbits by prenylamine and vitamins A and E. Am. Heart J. 1986;111:95–102. doi: 10.1016/0002-8703(86)90559-4. [DOI] [PubMed] [Google Scholar]
- 61.Minotti G, Salvatorelli E, Menna P. Pharmacological foundations of cardio-oncology. J. Pharmacol. Exp. Ther. 2010;334:2–8. doi: 10.1124/jpet.110.165860. [DOI] [PubMed] [Google Scholar]
- 62.Myers CE, Bonow R, Palmeri S, Jenkins J, Corden C, Locke C, Doroshow J, Epstein SA. A randomized controlled trial assessing the prevention of doxorubicin cardiomyopathy by N-acetylcysteine. Semin. Oncol. 1983;10:53–55. [PubMed] [Google Scholar]
- 63.Myers CE, McGuire WP, Liss KH, Ifrim I, Grotzinger K, Young RC. Adriamycin: The role of lipid peroxidation in cardiac toxicity and tumor response. Science. 1977;197:165–167. doi: 10.1126/science.877547. [DOI] [PubMed] [Google Scholar]
- 64.Myers CE, Mcguire W, Young R. Adriamycin: amelioration of toxicity by α-tocopherol. Cancer Treat. Rep. 1976;60:961–962. [PubMed] [Google Scholar]
- 65.Mythili Y, Sudharsan PT, Amudha G, Varalakshmi P. Effect of DL-alpha-lipoic acid on cyclophosphamide induced lysosomal changes in oxidative cardiotoxicity. Life Sci. 2007;80:1993–1998. doi: 10.1016/j.lfs.2007.02.040. [DOI] [PubMed] [Google Scholar]
- 66.Mythili Y, Sudharsan PT, Sudhahar V, Varalakshmi P. Protective effect of DL-alpha-lipoic acid on cyclophosphamide induced hyperlipidemic cardiomyopathy. Eur. J. Pharmacol. 2006;543:92–96. doi: 10.1016/j.ejphar.2006.06.007. [DOI] [PubMed] [Google Scholar]
- 67.Nhata R, Esteva FJ. HER-2-targeted therapy - lessons learned and future directions. Clin. Cancer Res. 2003;9:5078–5084. [PubMed] [Google Scholar]
- 68.Oliveira PJ, Bjork JA, Santos MS, Leino RL, Froberg MK, Moreno AJ, Wallace KB. Carvedilol-mediated antioxidant protection against doxorubicin-induced cardiac mitochondrial toxicity. Toxicol. Appl. Pharmacol. 2004;200:159–168. doi: 10.1016/j.taap.2004.04.005. [DOI] [PubMed] [Google Scholar]
- 69.O'Malley FP, Chia S, Tu D, Shepherd LE, Levine MN, Bramwell VH, Andrulis IL, Pritchard KI. Topoisomerase II alpha and responsiveness of breast cancer to adjuvant chemotherapy. J. Natl. Cancer Inst. 2009;101:644–650. doi: 10.1093/jnci/djp067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ozturk M, Ozler M, Kurt YG, Ozturk B, Uysal B, Ersoz N, Yasar M, Demirbas S, Kurt B, Acikel C, Oztas Y, Arpaci F, Topal T, Ozet A, Ataergin S, Kuzhan O, Oter S, Korkmaz A. Efficacy of melatonin, mercaptoehylguanidine and 1400W in doxorubicin- and trastuzumab-induced cardiotoxicity. J. Pineal Res. 2011;50:89–96. doi: 10.1111/j.1600-079X.2010.00818.x. [DOI] [PubMed] [Google Scholar]
- 71.Pai VB, Nahata MC. Cardiotoxicity of chemotherapeutic agents: incidence, treatment and prevention. Drug Saf. 2000;22:263–302. doi: 10.2165/00002018-200022040-00002. [DOI] [PubMed] [Google Scholar]
- 72.Pentassuglia L, Timolati F, Seifriz F, Abudukadier K, Suter TM, Zuppinger C. Inhibition of ErbB2/neuregulin signaling augments paclitaxel-induced cardiotoxicity in adult ventricular myocytes. Exp. Cell Res. 2007;313:1588–1601. doi: 10.1016/j.yexcr.2007.02.007. [DOI] [PubMed] [Google Scholar]
- 73.Perotti S, Cresta G, Grasselli G, Capri G, Minotti G, Gianni L. Cardiotoxic effects of anthracycline-taxane combinations. Expert Opin. Drug Saf. 2003;2:59–71. doi: 10.1517/14740338.2.1.59. [DOI] [PubMed] [Google Scholar]
- 74.Ritchie DS, Seymour JF, Roberts AW, Szer J, Grigg AP. Acute left ventricular failure following melphalan and fludarabine conditioning. Bone Marrow Transplant. 2001;28:101–103. doi: 10.1038/sj.bmt.1703098. [DOI] [PubMed] [Google Scholar]
- 75.Roskoski R., Jr The ErbB/HER receptor protein-tyrosine kinases and cancer. Biochem. Biophys. Res. Commun. 2004;319:1–11. doi: 10.1016/j.bbrc.2004.04.150. [DOI] [PubMed] [Google Scholar]
- 76.Salvatorelli E, Menna P, Cascegna S, Liberi G, Calafiore A, Gianni L, Minotti G. Paclitaxel and docetaxel stimulation of doxorubicinol formation in the human heart: implications for cardiotoxicity of doxorubicin-taxane chemotherapies. J. Pharmacol. Exp. Ther. 2006;318:424–433. doi: 10.1124/jpet.106.103846. [DOI] [PubMed] [Google Scholar]
- 77.Salvatorelli E, Menna P, Gianni L, Minotti G. Defective taxane stimulation of epirubicinol formation in the human heart: insight into the cardiac tolerability of epirubicin-taxane chemotherapies. J. Pharmacol. Exp. Ther. 2007;320:790–800. doi: 10.1124/jpet.106.116160. [DOI] [PubMed] [Google Scholar]
- 78.Santos GW, Sensenbrenner LL, Burke PJ, Colvin M, Owens AH, Jr, Bias WB, Slavin RE. Marrow transplanation in man following cyclophosphamide. Transplant. Proc. 1971;3:400–404. [PubMed] [Google Scholar]
- 79.Schwartz CL, Constine LS, Villaluna D, London WB, Hutchison RE, Sposto R, Lipshultz SE, Turner CS, deAlarcon PA, Chauvenet A. A risk-adapted, response-based approach using ABVE-PC for children and adolescents with intermediate and high-risk Hodgkin lymphoma: the results of P9425. Blood. 2009;114:2051–2059. doi: 10.1182/blood-2008-10-184143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Seidman A, Hudis C, Pierri MK, Shak S, Paton V, Ashby M, Murphy M, Stewart SJ, Keefe D. Cardiac dysfunction in the trastuzumab clinical trials experience. J Clin Oncol. 2002;20:1215–1221. doi: 10.1200/JCO.2002.20.5.1215. [DOI] [PubMed] [Google Scholar]
- 81.Serrano J, Palmeira CM, Kuehl DW, Wallace KB. Cardioselective and cumulative oxidation of mitochondrial DNA following subchronic doxorubicin administration. Biochim. Biophys. Acta. 1999;1411:201–205. doi: 10.1016/s0005-2728(99)00011-0. [DOI] [PubMed] [Google Scholar]
- 82.Shimpo K, Nagatsu T, Yamada K, Sato T, Niimi H, Shamoto M, Takeuchi T, Umezawa H, Fujita K. Ascorbic acid and adriamycin toxicity. Am. J. Clin. Nutr. 1991;54:1298S–l301S. doi: 10.1093/ajcn/54.6.1298s. [DOI] [PubMed] [Google Scholar]
- 83.Singal PK, Iliskovic N, Li T, Kumar D. Adriamycin cardiomyopathy: pathophysiology and prevention. FASEB J. 1997;11:931–936. doi: 10.1096/fasebj.11.12.9337145. [DOI] [PubMed] [Google Scholar]
- 84.Singal PK, Siveski-Iliskovic N, Hill M, Thomas TP, Li T. Combination therapy with probucol prevents adriamycin-induced cardiomyopathy. J. Mol. Cell. Cardiol. 1995;27:1055–1063. doi: 10.1016/0022-2828(95)90074-8. [DOI] [PubMed] [Google Scholar]
- 85.Siveski-Iliskovic N, Kaul N, Singal PK. Probucol promotes endogenous antioxidants and provides protection against adriamycin-induced cardiomyopathy in rats. Circulation. 1994;89:2829–2835. doi: 10.1161/01.cir.89.6.2829. [DOI] [PubMed] [Google Scholar]
- 86.Slamon DJ, Godolphin W, Jones LA, Holt JA, Wong SG, Keith DE, Levin WJ, Stuart SG, Udove J, Ullrich A. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science. 1989;244:707–712. doi: 10.1126/science.2470152. [DOI] [PubMed] [Google Scholar]
- 87.Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V, Bajamonde A, Fleming T, Eiermann W, Wolter J, Pegram M, Baselga J, Norton L. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N. Engl. J. Med. 2001;344:783–792. doi: 10.1056/NEJM200103153441101. [DOI] [PubMed] [Google Scholar]
- 88.Speyer JL, Green MD, Zeleniuch-Jacquotte A, Wernz JC, Rey M, Sanger J, Kramer E, Ferrans V, Hochster H, Meyers M, et al. ICRF-187 permits longer treatment with doxorubicin in women with breast cancer. J. Clin. Oncol. 1992;10:117–127. doi: 10.1200/JCO.1992.10.1.117. [DOI] [PubMed] [Google Scholar]
- 89.Stathopoulos GP, Malamos NA, Dontas I, Deliconstantinos G, Perrea-Kotsareli D, Karayannacos PE. Inhibition of adriamycin cardiotoxicity by 5-fluorouracil: a potential free oxygen radical scavenger. Anticancer Res. 1998;18:4387–4392. [PubMed] [Google Scholar]
- 90.Swain SM, Whaley FS, Gerber MC, Weisberg S, York M, Spicer D, Jones SE, Wadler S, Desai A, Vogel C, Speyer J, Mittelman A, Reddy S, Pendergrass K, Velez-Garcia E, Ewer MS, Bianchine JR, Gams RA. Cardioprotection with dexrazoxane for doxorubicin-containing therapy in advanced breast cancer. J. Clin. Oncol. 1997;15:1318–1332. doi: 10.1200/JCO.1997.15.4.1318. [DOI] [PubMed] [Google Scholar]
- 91.Tesoriere L, Ciaccio M, Valenza M, Bongiorno A, Maresi E, Albiero R, Livrea MA. Effect of vitamin-A administration on resistance of rat-heart against doxorubi- cin-induced cardiotoxicity and lethality. J. Pharm. Exp. Ther. 1994;269:430–436. [PubMed] [Google Scholar]
- 92.Timolati F, Ott D, Pentassuglia L, Giraud MN, Perriard JC, Suter TM, Zuppinger C. Neuregulin-1 beta attenuates doxorubicin-induced alterations of excitation-contraction coupling and reduces oxidative stress in adult rat cardiomyocytes. J. Mol. Cell Cardiol. 2006;41:845–854. doi: 10.1016/j.yjmcc.2006.08.002. [DOI] [PubMed] [Google Scholar]
- 93.van Acker SA, Boven E, Kuiper K, van der Berg DJ, Grimbergen JA, Kramer K, Bast A, van der Vijgh WJ. Monohydroxyethylrutoside, a dose-dependent cardioprotective agent, does not affect the antitumor activity of doxorubicin. Clin. Cancer Res. 1997;3:1747–1754. [PubMed] [Google Scholar]
- 94.van Dalen EC, Caron HN, Dickinson HO, Kremer LC. Cardioprotective interventions for cancer patients receiving anthracyclines. Cochrane Database Syst. Rev. 2011;6:CD003917. doi: 10.1002/14651858.CD003917.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Venturini M, Michelotti A, Del Mastro L, Gallo L, Carnino F, Garrone O, Tibaldi C, Molea N, Bellina RC, Pronzato P, Cyrus P, Vinke J, Testore F, Guelfi M, Lionetto R, Bruzzi P, Conte PF, Rosso R. Multicenter randomized controlled clinical trial to evaluate cardioprotection of dexrazoxane versus no cardioprotection in women receiving epirubicin chemotherapy for advanced breast cancer. J. Clin. Oncol. 1996;14:3112–3120. doi: 10.1200/JCO.1996.14.12.3112. [DOI] [PubMed] [Google Scholar]
- 96.Verweij J, Funke-Küpper AJ, Teule GJ, Pinedo HM. A prospective study on the dose dependency of cardiotoxicity induced by mitomycin C. Med. Oncol. Tumor Pharmacother. 1988;5:159–163. doi: 10.1007/BF02986439. [DOI] [PubMed] [Google Scholar]
- 97.Wadhwa D, Fallah-Rad N, Grenier D, Krahn M, Fang T, Ahmadie R, Walker JR, Lister D, Arora RC, Barac I, Morris A, Jassal DS. Trastuzumab mediated cardiotoxicity in the setting of the adjuvant chemotherapy for breast cancer: A retrospective study. Breast Cancer Res. Treat. 2009;117:357–364. doi: 10.1007/s10549-008-0260-6. [DOI] [PubMed] [Google Scholar]
- 98.Wagdi PH, Rouvinez G, Fluri M, Aeschbacher B, Thoni A, Schefer H, Meier B. Cardioprotection in chemo- and radiotherapy of malignant diseases--an echocardiographic pilot study. Praxis. 1995;84:1220–1223. [PubMed] [Google Scholar]
- 99.Waldner R, Laschan C, Lohninger A, Gessner M, Tuchler H, Huemer M, Spiegel W, Karlic H. Effects of doxorubicin-containing chemotherapy and a combination with L-carnitine on oxidative metabolism in patients with non-Hodgkin lymphoma. J. Cancer Res. Clin. Oncol. 2006;132:121–128. doi: 10.1007/s00432-005-0054-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Walker JR, Sharma A, Lytwyn M, Bohonis S, Thliveris J, Singal PK, Jassal DS. The cardioprotective role of probucol against anthracycline and trastuzumab-mediated cardiotoxicity. J. Am. Soc. Echocardiogr. 2011;24:699–705. doi: 10.1016/j.echo.2011.01.018. [DOI] [PubMed] [Google Scholar]
- 101.Wang J, He D, Zhang Q, Han Y, Jin S, Qi F. Resveratrol protects against Cisplatininduced cardiotoxicity by alleviating oxidative damage. Cancer Biother. Radiopharm. 2009;24:675–680. doi: 10.1089/cbr.2009.0679. [DOI] [PubMed] [Google Scholar]
- 102.Wexler LH, Andrich MP, Venzon D, Berg SL, Weaver-McClure L, Chen CC, Dilsizian V, Avila N, Jarosinski P, Balis FM, Poplack DG, Horowitz ME. Randomized trial of the cardioprotective agent ICRF-187 in pediatric sarcoma patients treated with doxorubicin. J. Clin. Oncol. 1996;14:362–372. doi: 10.1200/JCO.1996.14.2.362. [DOI] [PubMed] [Google Scholar]
- 103.Woodman RJ, Cysyk RL, Kline I, Gang M, Venditti JM. Enhancement of the effectiveness of daunorubicin (NSC-82151) or adriamycin (NSC-123127) against early mouse L1210 leukemia with ICRF-159 (NSC-129943) Cancer Chemother. Rep. 1975;59:689–695. [PubMed] [Google Scholar]
- 104.Yoda Y, Nakazawa M, Abe T, Kawakami Z. Prevention of doxorubicin myocardial toxicity in mice by reduced glutathione. Cancer Res. 1986;46:2551–2556. [PubMed] [Google Scholar]
- 105.Yousef MI, Saad AA, El-Shennawy LK. Protective effect of grape seed proanthocyanidin extract against oxidative stress induced by cisplatin in rats. Food Chem. Toxicol. 2009;47:1176–1183. doi: 10.1016/j.fct.2009.02.007. [DOI] [PubMed] [Google Scholar]
- 106.Zver S, Zadnik V, Cernelc P, Kozelj M. Cardiac toxicity of high-dose cyclophosphamide and melphalan in patients with multiple myeloma treated with tandem autologous hematopoietic stem cell transplantation. Int. J. Hematol. 2008;88:227–236. doi: 10.1007/s12185-008-0112-5. [DOI] [PubMed] [Google Scholar]