Abstract
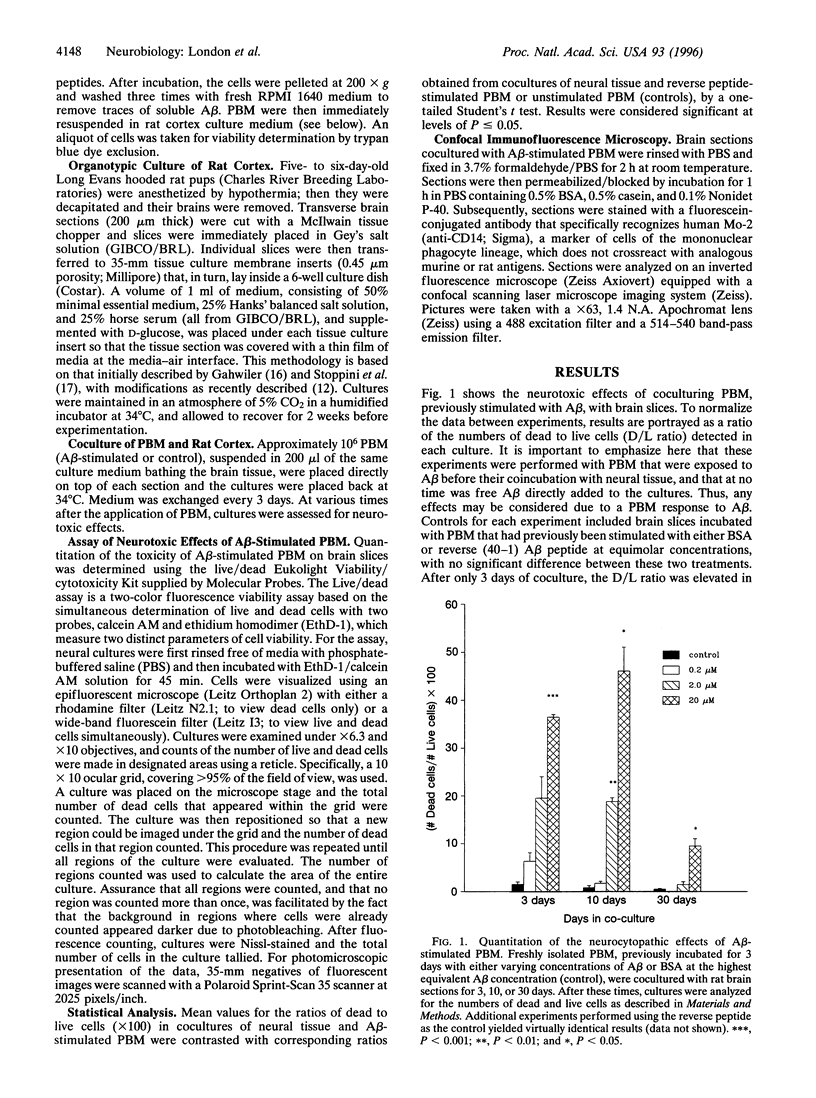
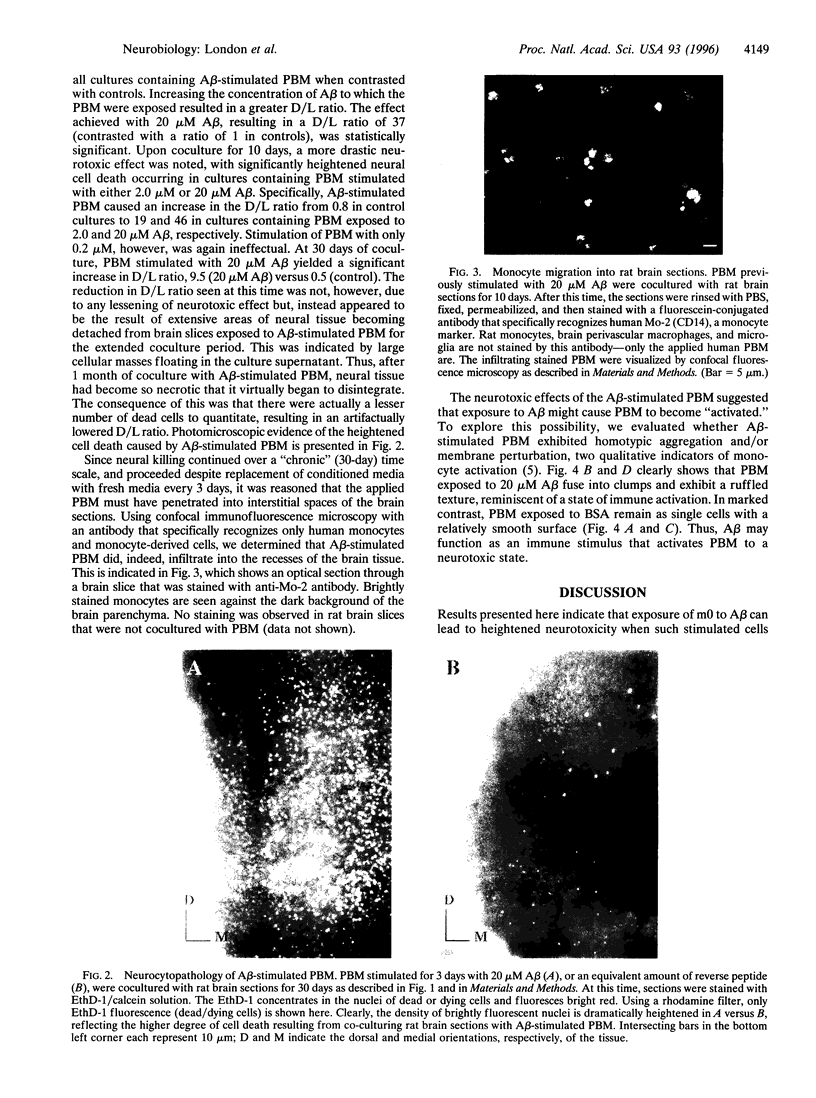
Growing evidence indicates that cells of the mononuclear phagocyte lineage, which includes peripheral blood monocytes (PBM) and tissue macrophages, participate in a variety of neurodestructive events and may play a pivotal role in neurodegenerative conditions such as Alzheimer disease. The present study sought to determine whether exposure of PBM to beta-amyloid peptide (A beta), the major protein of the amyloid fibrils that accumulate in the brain in Alzheimer disease, could induce cytopathic activity in these cells upon their subsequent incubation with neural tissue. PBM were incubated with A beta for 3 days, centrifuged and washed to remove traces of cell-free A beta, and then applied to organotypic cultures of rat brain for varying periods of time. By using a cell-viability assay to quantitate neurocytopathic effect, an increase in the ratio of dead to live cells was detected in cultures containing A beta-stimulated PBM versus control PBM (stimulated with either bovine serum albumin or reverse A beta peptide) as early as 3 days after coculture. The ratio of dead to live cells increased further by 10 days of coculture. By 30 days of coculture, the dead to live cell ratio remained elevated, and the intensity of neurocytopathic effect was such that large areas of brain mass dissociated from the cultures. These results indicate that stimulation of PBM with A beta significantly heightens their neurocytopathic activity and highlight the possibility that inflammatory reactions in the brain play a role in the neurodegeneration that accompanies Alzheimer disease.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Araujo D. M., Cotman C. W. Beta-amyloid stimulates glial cells in vitro to produce growth factors that accumulate in senile plaques in Alzheimer's disease. Brain Res. 1992 Jan 8;569(1):141–145. doi: 10.1016/0006-8993(92)90380-r. [DOI] [PubMed] [Google Scholar]
- Behl C., Davis J. B., Lesley R., Schubert D. Hydrogen peroxide mediates amyloid beta protein toxicity. Cell. 1994 Jun 17;77(6):817–827. doi: 10.1016/0092-8674(94)90131-7. [DOI] [PubMed] [Google Scholar]
- Brugg B., Dubreuil Y. L., Huber G., Wollman E. E., Delhaye-Bouchaud N., Mariani J. Inflammatory processes induce beta-amyloid precursor protein changes in mouse brain. Proc Natl Acad Sci U S A. 1995 Mar 28;92(7):3032–3035. doi: 10.1073/pnas.92.7.3032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis J. B., McMurray H. F., Schubert D. The amyloid beta-protein of Alzheimer's disease is chemotactic for mononuclear phagocytes. Biochem Biophys Res Commun. 1992 Dec 15;189(2):1096–1100. doi: 10.1016/0006-291x(92)92317-q. [DOI] [PubMed] [Google Scholar]
- Denholm E. M., Wolber F. M. A simple method for the purification of human peripheral blood monocytes. A substitute for Sepracell-MN. J Immunol Methods. 1991 Nov 22;144(2):247–251. doi: 10.1016/0022-1759(91)90092-t. [DOI] [PubMed] [Google Scholar]
- Dickson D. W., Lee S. C., Mattiace L. A., Yen S. H., Brosnan C. Microglia and cytokines in neurological disease, with special reference to AIDS and Alzheimer's disease. Glia. 1993 Jan;7(1):75–83. doi: 10.1002/glia.440070113. [DOI] [PubMed] [Google Scholar]
- Ebly E. M., Parhad I. M., Hogan D. B., Fung T. S. Prevalence and types of dementia in the very old: results from the Canadian Study of Health and Aging. Neurology. 1994 Sep;44(9):1593–1600. doi: 10.1212/wnl.44.9.1593. [DOI] [PubMed] [Google Scholar]
- Eddleston M., Mucke L. Molecular profile of reactive astrocytes--implications for their role in neurologic disease. Neuroscience. 1993 May;54(1):15–36. doi: 10.1016/0306-4522(93)90380-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frackowiak J., Wisniewski H. M., Wegiel J., Merz G. S., Iqbal K., Wang K. C. Ultrastructure of the microglia that phagocytose amyloid and the microglia that produce beta-amyloid fibrils. Acta Neuropathol. 1992;84(3):225–233. doi: 10.1007/BF00227813. [DOI] [PubMed] [Google Scholar]
- Games D., Adams D., Alessandrini R., Barbour R., Berthelette P., Blackwell C., Carr T., Clemens J., Donaldson T., Gillespie F. Alzheimer-type neuropathology in transgenic mice overexpressing V717F beta-amyloid precursor protein. Nature. 1995 Feb 9;373(6514):523–527. doi: 10.1038/373523a0. [DOI] [PubMed] [Google Scholar]
- Genis P., Jett M., Bernton E. W., Boyle T., Gelbard H. A., Dzenko K., Keane R. W., Resnick L., Mizrachi Y., Volsky D. J. Cytokines and arachidonic metabolites produced during human immunodeficiency virus (HIV)-infected macrophage-astroglia interactions: implications for the neuropathogenesis of HIV disease. J Exp Med. 1992 Dec 1;176(6):1703–1718. doi: 10.1084/jem.176.6.1703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giordano T., Pan J. B., Monteggia L. M., Holzman T. F., Snyder S. W., Krafft G., Ghanbari H., Kowall N. W. Similarities between beta amyloid peptides 1-40 and 40-1: effects on aggregation, toxicity in vitro, and injection in young and aged rats. Exp Neurol. 1994 Feb;125(2):175–182. doi: 10.1006/exnr.1994.1022. [DOI] [PubMed] [Google Scholar]
- Giulian D., Vaca K., Corpuz M. Brain glia release factors with opposing actions upon neuronal survival. J Neurosci. 1993 Jan;13(1):29–37. doi: 10.1523/JNEUROSCI.13-01-00029.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giulian D., Vaca K., Noonan C. A. Secretion of neurotoxins by mononuclear phagocytes infected with HIV-1. Science. 1990 Dec 14;250(4987):1593–1596. doi: 10.1126/science.2148832. [DOI] [PubMed] [Google Scholar]
- Gähwiler B. H. Organotypic cultures of neural tissue. Trends Neurosci. 1988 Nov;11(11):484–489. doi: 10.1016/0166-2236(88)90007-0. [DOI] [PubMed] [Google Scholar]
- Hu S., Sheng W. S., Peterson P. K., Chao C. C. Cytokine modulation of murine microglial cell superoxide production. Glia. 1995 Jan;13(1):45–50. doi: 10.1002/glia.440130106. [DOI] [PubMed] [Google Scholar]
- Itagaki S., McGeer P. L., Akiyama H., Zhu S., Selkoe D. Relationship of microglia and astrocytes to amyloid deposits of Alzheimer disease. J Neuroimmunol. 1989 Oct;24(3):173–182. doi: 10.1016/0165-5728(89)90115-x. [DOI] [PubMed] [Google Scholar]
- Joachim C. L., Selkoe D. J. The seminal role of beta-amyloid in the pathogenesis of Alzheimer disease. Alzheimer Dis Assoc Disord. 1992 Spring;6(1):7–34. doi: 10.1097/00002093-199205000-00003. [DOI] [PubMed] [Google Scholar]
- Klegeris A., Walker D. G., McGeer P. L. Activation of macrophages by Alzheimer beta amyloid peptide. Biochem Biophys Res Commun. 1994 Mar 15;199(2):984–991. doi: 10.1006/bbrc.1994.1326. [DOI] [PubMed] [Google Scholar]
- Korotzer A. R., Pike C. J., Cotman C. W. beta-Amyloid peptides induce degeneration of cultured rat microglia. Brain Res. 1993 Oct 8;624(1-2):121–125. doi: 10.1016/0006-8993(93)90068-x. [DOI] [PubMed] [Google Scholar]
- LaFerla F. M., Tinkle B. T., Bieberich C. J., Haudenschild C. C., Jay G. The Alzheimer's A beta peptide induces neurodegeneration and apoptotic cell death in transgenic mice. Nat Genet. 1995 Jan;9(1):21–30. doi: 10.1038/ng0195-21. [DOI] [PubMed] [Google Scholar]
- Mattson M. P., Barger S. W., Cheng B., Lieberburg I., Smith-Swintosky V. L., Rydel R. E. beta-Amyloid precursor protein metabolites and loss of neuronal Ca2+ homeostasis in Alzheimer's disease. Trends Neurosci. 1993 Oct;16(10):409–414. doi: 10.1016/0166-2236(93)90009-b. [DOI] [PubMed] [Google Scholar]
- McGeer P. L., Kawamata T., Walker D. G., Akiyama H., Tooyama I., McGeer E. G. Microglia in degenerative neurological disease. Glia. 1993 Jan;7(1):84–92. doi: 10.1002/glia.440070114. [DOI] [PubMed] [Google Scholar]
- Peterson P. K., Hu S., Anderson W. R., Chao C. C. Nitric oxide production and neurotoxicity mediated by activated microglia from human versus mouse brain. J Infect Dis. 1994 Aug;170(2):457–460. doi: 10.1093/infdis/170.2.457. [DOI] [PubMed] [Google Scholar]
- Pike C. J., Walencewicz-Wasserman A. J., Kosmoski J., Cribbs D. H., Glabe C. G., Cotman C. W. Structure-activity analyses of beta-amyloid peptides: contributions of the beta 25-35 region to aggregation and neurotoxicity. J Neurochem. 1995 Jan;64(1):253–265. doi: 10.1046/j.1471-4159.1995.64010253.x. [DOI] [PubMed] [Google Scholar]
- Pike C. J., Walencewicz A. J., Glabe C. G., Cotman C. W. In vitro aging of beta-amyloid protein causes peptide aggregation and neurotoxicity. Brain Res. 1991 Nov 1;563(1-2):311–314. doi: 10.1016/0006-8993(91)91553-d. [DOI] [PubMed] [Google Scholar]
- Pulliam L., Herndier B. G., Tang N. M., McGrath M. S. Human immunodeficiency virus-infected macrophages produce soluble factors that cause histological and neurochemical alterations in cultured human brains. J Clin Invest. 1991 Feb;87(2):503–512. doi: 10.1172/JCI115024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogers J., Kirby L. C., Hempelman S. R., Berry D. L., McGeer P. L., Kaszniak A. W., Zalinski J., Cofield M., Mansukhani L., Willson P. Clinical trial of indomethacin in Alzheimer's disease. Neurology. 1993 Aug;43(8):1609–1611. doi: 10.1212/wnl.43.8.1609. [DOI] [PubMed] [Google Scholar]
- Shaffer L. M., Dority M. D., Gupta-Bansal R., Frederickson R. C., Younkin S. G., Brunden K. R. Amyloid beta protein (A beta) removal by neuroglial cells in culture. Neurobiol Aging. 1995 Sep-Oct;16(5):737–745. doi: 10.1016/0197-4580(95)00055-j. [DOI] [PubMed] [Google Scholar]
- Stanley L. C., Mrak R. E., Woody R. C., Perrot L. J., Zhang S., Marshak D. R., Nelson S. J., Griffin W. S. Glial cytokines as neuropathogenic factors in HIV infection: pathogenic similarities to Alzheimer's disease. J Neuropathol Exp Neurol. 1994 May;53(3):231–238. doi: 10.1097/00005072-199405000-00003. [DOI] [PubMed] [Google Scholar]
- Stoppini L., Buchs P. A., Muller D. A simple method for organotypic cultures of nervous tissue. J Neurosci Methods. 1991 Apr;37(2):173–182. doi: 10.1016/0165-0270(91)90128-m. [DOI] [PubMed] [Google Scholar]
- Tardieu M., Héry C., Peudenier S., Boespflug O., Montagnier L. Human immunodeficiency virus type 1-infected monocytic cells can destroy human neural cells after cell-to-cell adhesion. Ann Neurol. 1992 Jul;32(1):11–17. doi: 10.1002/ana.410320104. [DOI] [PubMed] [Google Scholar]
- Théry Clotilde, Chamak Brigitte, Mallat Michel. Cytotoxic Effect of Brain Macrophages on Developing Neurons. Eur J Neurosci. 1991 Oct;3(11):1155–1164. doi: 10.1111/j.1460-9568.1991.tb00050.x. [DOI] [PubMed] [Google Scholar]
- Wahl S. M., Allen J. B., McCartney-Francis N., Morganti-Kossmann M. C., Kossmann T., Ellingsworth L., Mai U. E., Mergenhagen S. E., Orenstein J. M. Macrophage- and astrocyte-derived transforming growth factor beta as a mediator of central nervous system dysfunction in acquired immune deficiency syndrome. J Exp Med. 1991 Apr 1;173(4):981–991. doi: 10.1084/jem.173.4.981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wisniewski H. M., Barcikowska M., Kida E. Phagocytosis of beta/A4 amyloid fibrils of the neuritic neocortical plaques. Acta Neuropathol. 1991;81(5):588–590. doi: 10.1007/BF00310142. [DOI] [PubMed] [Google Scholar]
- Yankner B. A., Duffy L. K., Kirschner D. A. Neurotrophic and neurotoxic effects of amyloid beta protein: reversal by tachykinin neuropeptides. Science. 1990 Oct 12;250(4978):279–282. doi: 10.1126/science.2218531. [DOI] [PubMed] [Google Scholar]