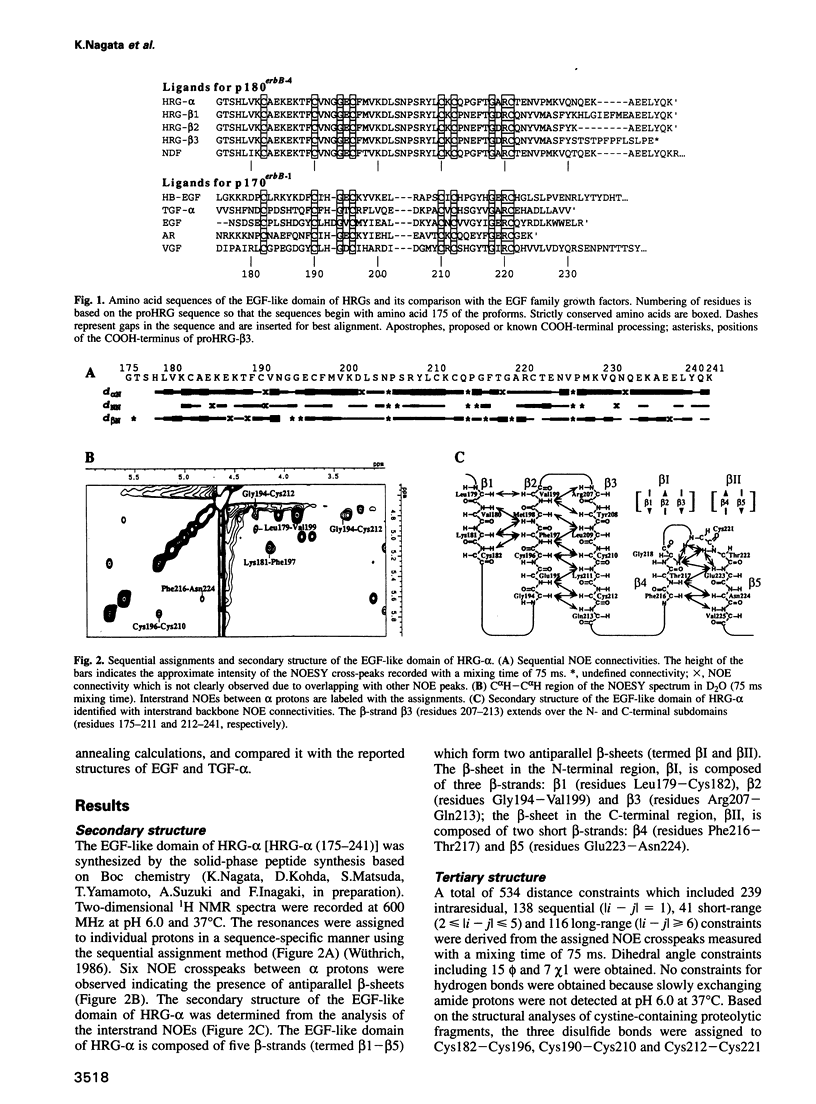
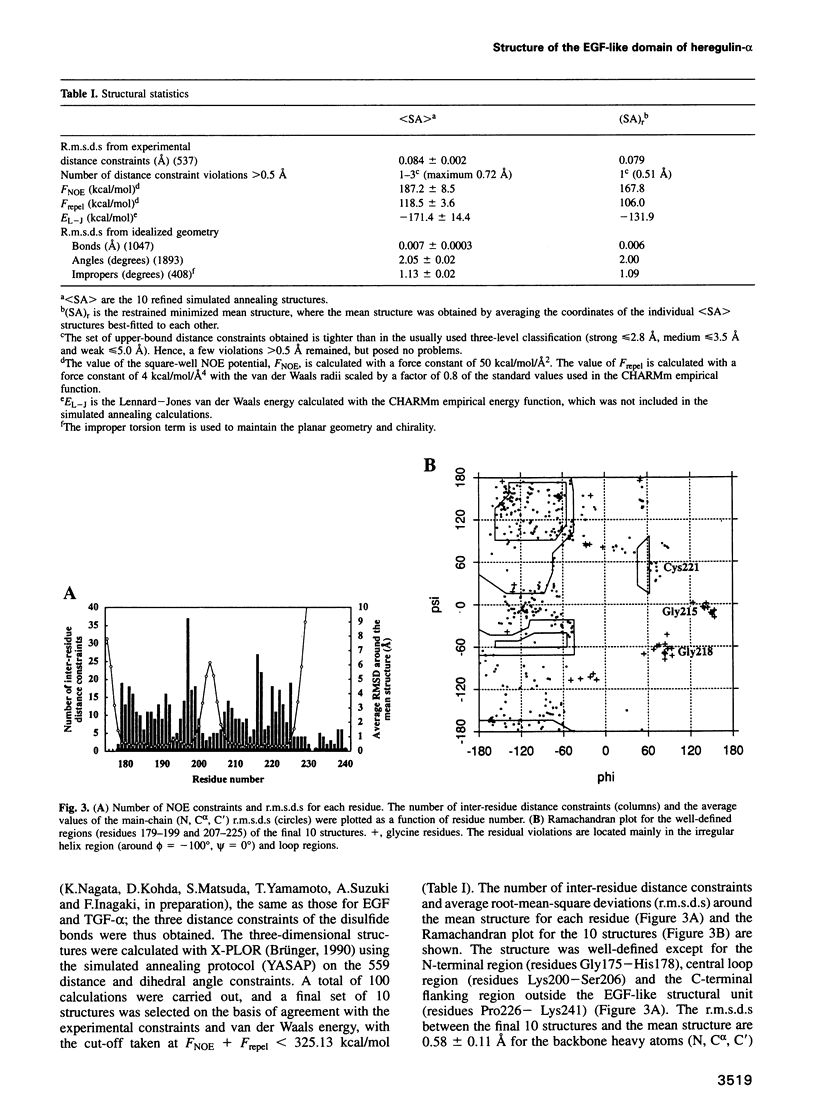
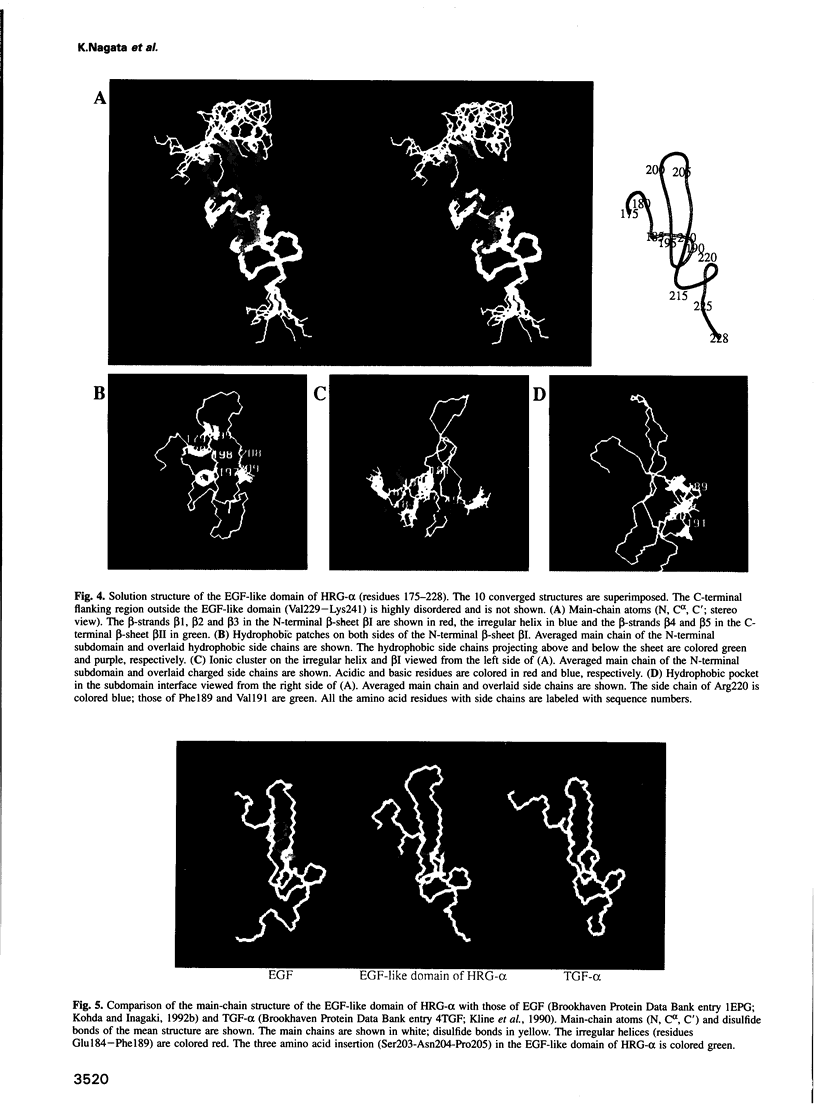
Abstract
p185erbB-2 and p180erbB-4 are epidermal growth factor (EGF) receptor-like tyrosine kinases, whose co-expression is observed in many breast carcinomas. Heregulins (HRGs), which contain an immunoglobulin unit and an EGF-like domain, bind to p180erbB-4 and activate p180erbB-4 and p185erbB-2 through transphosphorylation or receptor heterodimerization. The EGF-like domain is sufficient for the activation. Despite the sequence similarity, no cross activity is seen between the p180erbB-4 ligands (HRGs) and the p170erbB-1 ligands [EGF and transforming growth factor (TGF)-alpha]. To investigate the structural basis of receptor specificity, we have determined the solution structure of the EGF-like domain of HRG-alpha by two-dimensional 1H nuclear magnetic resonance spectroscopy and simulated annealing calculations. Though its main-chain fold is similar to those of EGF and TGF-alpha, distinctive structural features are observed on the molecular surface including an ionic cluster and hydrophobic patches, which afford HRG-alpha the specific affinity for p180erbB-4. The structure should provide a basis for the structure-activity relationship of HRGs and for the design of drugs which prevent progression of breast cancer.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bargmann C. I., Hung M. C., Weinberg R. A. The neu oncogene encodes an epidermal growth factor receptor-related protein. Nature. 1986 Jan 16;319(6050):226–230. doi: 10.1038/319226a0. [DOI] [PubMed] [Google Scholar]
- Blomquist M. C., Hunt L. T., Barker W. C. Vaccinia virus 19-kilodalton protein: relationship to several mammalian proteins, including two growth factors. Proc Natl Acad Sci U S A. 1984 Dec;81(23):7363–7367. doi: 10.1073/pnas.81.23.7363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campion S. R., Matsunami R. K., Engler D. A., Niyogi S. K. Biochemical properties of site-directed mutants of human epidermal growth factor: importance of solvent-exposed hydrophobic residues of the amino-terminal domain in receptor binding. Biochemistry. 1990 Oct 23;29(42):9988–9993. doi: 10.1021/bi00494a032. [DOI] [PubMed] [Google Scholar]
- Carpenter G., Cohen S. Epidermal growth factor. J Biol Chem. 1990 May 15;265(14):7709–7712. [PubMed] [Google Scholar]
- Carpenter G., Cohen S. Epidermal growth factor. Annu Rev Biochem. 1979;48:193–216. doi: 10.1146/annurev.bi.48.070179.001205. [DOI] [PubMed] [Google Scholar]
- Clore G. M., Brünger A. T., Karplus M., Gronenborn A. M. Application of molecular dynamics with interproton distance restraints to three-dimensional protein structure determination. A model study of crambin. J Mol Biol. 1986 Oct 5;191(3):523–551. doi: 10.1016/0022-2836(86)90146-4. [DOI] [PubMed] [Google Scholar]
- Derynck R., Roberts A. B., Winkler M. E., Chen E. Y., Goeddel D. V. Human transforming growth factor-alpha: precursor structure and expression in E. coli. Cell. 1984 Aug;38(1):287–297. doi: 10.1016/0092-8674(84)90550-6. [DOI] [PubMed] [Google Scholar]
- Dudgeon T. J., Cooke R. M., Baron M., Campbell I. D., Edwards R. M., Fallon A. Structure-function analysis of epidermal growth factor: site directed mutagenesis and nuclear magnetic resonance. FEBS Lett. 1990 Feb 26;261(2):392–396. doi: 10.1016/0014-5793(90)80600-n. [DOI] [PubMed] [Google Scholar]
- Engler D. A., Montelione G. T., Niyogi S. K. Human epidermal growth factor. Distinct roles of tyrosine 37 and arginine 41 in receptor binding as determined by site-directed mutagenesis and nuclear magnetic resonance spectroscopy. FEBS Lett. 1990 Oct 1;271(1-2):47–50. doi: 10.1016/0014-5793(90)80368-s. [DOI] [PubMed] [Google Scholar]
- Falls D. L., Rosen K. M., Corfas G., Lane W. S., Fischbach G. D. ARIA, a protein that stimulates acetylcholine receptor synthesis, is a member of the neu ligand family. Cell. 1993 Mar 12;72(5):801–815. doi: 10.1016/0092-8674(93)90407-h. [DOI] [PubMed] [Google Scholar]
- Harvey T. S., Wilkinson A. J., Tappin M. J., Cooke R. M., Campbell I. D. The solution structure of human transforming growth factor alpha. Eur J Biochem. 1991 Jun 15;198(3):555–562. doi: 10.1111/j.1432-1033.1991.tb16050.x. [DOI] [PubMed] [Google Scholar]
- Hatanaka H., Oka M., Kohda D., Tate S., Suda A., Tamiya N., Inagaki F. Tertiary structure of erabutoxin b in aqueous solution as elucidated by two-dimensional nuclear magnetic resonance. J Mol Biol. 1994 Jul 8;240(2):155–166. doi: 10.1006/jmbi.1994.1429. [DOI] [PubMed] [Google Scholar]
- Higashiyama S., Abraham J. A., Miller J., Fiddes J. C., Klagsbrun M. A heparin-binding growth factor secreted by macrophage-like cells that is related to EGF. Science. 1991 Feb 22;251(4996):936–939. doi: 10.1126/science.1840698. [DOI] [PubMed] [Google Scholar]
- Holmes W. E., Sliwkowski M. X., Akita R. W., Henzel W. J., Lee J., Park J. W., Yansura D., Abadi N., Raab H., Lewis G. D. Identification of heregulin, a specific activator of p185erbB2. Science. 1992 May 22;256(5060):1205–1210. doi: 10.1126/science.256.5060.1205. [DOI] [PubMed] [Google Scholar]
- Hommel U., Dudgeon T. J., Fallon A., Edwards R. M., Campbell I. D. Structure-function relationships in human epidermal growth factor studied by site-directed mutagenesis and 1H NMR. Biochemistry. 1991 Sep 10;30(36):8891–8898. doi: 10.1021/bi00100a024. [DOI] [PubMed] [Google Scholar]
- Hommel U., Harvey T. S., Driscoll P. C., Campbell I. D. Human epidermal growth factor. High resolution solution structure and comparison with human transforming growth factor alpha. J Mol Biol. 1992 Sep 5;227(1):271–282. doi: 10.1016/0022-2836(92)90697-i. [DOI] [PubMed] [Google Scholar]
- Kline T. P., Brown F. K., Brown S. C., Jeffs P. W., Kopple K. D., Mueller L. Solution structures of human transforming growth factor alpha derived from 1H NMR data. Biochemistry. 1990 Aug 28;29(34):7805–7813. doi: 10.1021/bi00486a005. [DOI] [PubMed] [Google Scholar]
- Kohda D., Hatanaka H., Odaka M., Mandiyan V., Ullrich A., Schlessinger J., Inagaki F. Solution structure of the SH3 domain of phospholipase C-gamma. Cell. 1993 Mar 26;72(6):953–960. doi: 10.1016/0092-8674(93)90583-c. [DOI] [PubMed] [Google Scholar]
- Kohda D., Inagaki F. Structure of epidermal growth factor bound to perdeuterated dodecylphosphocholine micelles determined by two-dimensional NMR and simulated annealing calculations. Biochemistry. 1992 Jan 28;31(3):677–685. doi: 10.1021/bi00118a007. [DOI] [PubMed] [Google Scholar]
- Kohda D., Inagaki F. Three-dimensional nuclear magnetic resonance structures of mouse epidermal growth factor in acidic and physiological pH solutions. Biochemistry. 1992 Dec 1;31(47):11928–11939. doi: 10.1021/bi00162a036. [DOI] [PubMed] [Google Scholar]
- Koide H., Muto Y., Kasai H., Hoshi K., Takusari H., Kohri K., Takahashi S., Sasaki T., Tsukumo K., Miyake T. Recognition of an antiparallel beta-sheet structure of human epidermal growth factor by its receptor. Site-directed mutagenesis studies of Ala-30 and Asn-32. FEBS Lett. 1992 May 4;302(1):39–42. doi: 10.1016/0014-5793(92)80279-p. [DOI] [PubMed] [Google Scholar]
- Koide H., Muto Y., Kasai H., Kohri K., Hoshi K., Takahashi S., Tsukumo K., Sasaki T., Oka T., Miyake T. A site-directed mutagenesis study on the role of isoleucine-23 of human epidermal growth factor in the receptor binding. Biochim Biophys Acta. 1992 Apr 17;1120(3):257–261. doi: 10.1016/0167-4838(92)90245-9. [DOI] [PubMed] [Google Scholar]
- Marchionni M. A., Goodearl A. D., Chen M. S., Bermingham-McDonogh O., Kirk C., Hendricks M., Danehy F., Misumi D., Sudhalter J., Kobayashi K. Glial growth factors are alternatively spliced erbB2 ligands expressed in the nervous system. Nature. 1993 Mar 25;362(6418):312–318. doi: 10.1038/362312a0. [DOI] [PubMed] [Google Scholar]
- Montelione G. T., Wüthrich K., Burgess A. W., Nice E. C., Wagner G., Gibson K. D., Scheraga H. A. Solution structure of murine epidermal growth factor determined by NMR spectroscopy and refined by energy minimization with restraints. Biochemistry. 1992 Jan 14;31(1):236–249. doi: 10.1021/bi00116a033. [DOI] [PubMed] [Google Scholar]
- Moy F. J., Li Y. C., Rauenbuehler P., Winkler M. E., Scheraga H. A., Montelione G. T. Solution structure of human type-alpha transforming growth factor determined by heteronuclear NMR spectroscopy and refined by energy minimization with restraints. Biochemistry. 1993 Jul 27;32(29):7334–7353. doi: 10.1021/bi00080a003. [DOI] [PubMed] [Google Scholar]
- Moy F. J., Scheraga H. A., Liu J. F., Wu R., Montelione G. T. Conformational characterization of a single-site mutant of murine epidermal growth factor (EGF) by 1H NMR provides evidence that leucine-47 is involved in the interactions with the EGF receptor. Proc Natl Acad Sci U S A. 1989 Dec;86(24):9836–9840. doi: 10.1073/pnas.86.24.9836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peles E., Ben-Levy R., Tzahar E., Liu N., Wen D., Yarden Y. Cell-type specific interaction of Neu differentiation factor (NDF/heregulin) with Neu/HER-2 suggests complex ligand-receptor relationships. EMBO J. 1993 Mar;12(3):961–971. doi: 10.1002/j.1460-2075.1993.tb05737.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plowman G. D., Culouscou J. M., Whitney G. S., Green J. M., Carlton G. W., Foy L., Neubauer M. G., Shoyab M. Ligand-specific activation of HER4/p180erbB4, a fourth member of the epidermal growth factor receptor family. Proc Natl Acad Sci U S A. 1993 Mar 1;90(5):1746–1750. doi: 10.1073/pnas.90.5.1746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plowman G. D., Green J. M., Culouscou J. M., Carlton G. W., Rothwell V. M., Buckley S. Heregulin induces tyrosine phosphorylation of HER4/p180erbB4. Nature. 1993 Dec 2;366(6454):473–475. doi: 10.1038/366473a0. [DOI] [PubMed] [Google Scholar]
- Plowman G. D., Whitney G. S., Neubauer M. G., Green J. M., McDonald V. L., Todaro G. J., Shoyab M. Molecular cloning and expression of an additional epidermal growth factor receptor-related gene. Proc Natl Acad Sci U S A. 1990 Jul;87(13):4905–4909. doi: 10.1073/pnas.87.13.4905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rance M., Sørensen O. W., Bodenhausen G., Wagner G., Ernst R. R., Wüthrich K. Improved spectral resolution in cosy 1H NMR spectra of proteins via double quantum filtering. Biochem Biophys Res Commun. 1983 Dec 16;117(2):479–485. doi: 10.1016/0006-291x(83)91225-1. [DOI] [PubMed] [Google Scholar]
- Schumacher R., Soos M. A., Schlessinger J., Brandenburg D., Siddle K., Ullrich A. Signaling-competent receptor chimeras allow mapping of major insulin receptor binding domain determinants. J Biol Chem. 1993 Jan 15;268(2):1087–1094. [PubMed] [Google Scholar]
- Shoyab M., Plowman G. D., McDonald V. L., Bradley J. G., Todaro G. J. Structure and function of human amphiregulin: a member of the epidermal growth factor family. Science. 1989 Feb 24;243(4894 Pt 1):1074–1076. doi: 10.1126/science.2466334. [DOI] [PubMed] [Google Scholar]
- Ullrich A., Coussens L., Hayflick J. S., Dull T. J., Gray A., Tam A. W., Lee J., Yarden Y., Libermann T. A., Schlessinger J. Human epidermal growth factor receptor cDNA sequence and aberrant expression of the amplified gene in A431 epidermoid carcinoma cells. 1984 May 31-Jun 6Nature. 309(5967):418–425. doi: 10.1038/309418a0. [DOI] [PubMed] [Google Scholar]
- Wagner G., Braun W., Havel T. F., Schaumann T., Go N., Wüthrich K. Protein structures in solution by nuclear magnetic resonance and distance geometry. The polypeptide fold of the basic pancreatic trypsin inhibitor determined using two different algorithms, DISGEO and DISMAN. J Mol Biol. 1987 Aug 5;196(3):611–639. doi: 10.1016/0022-2836(87)90037-4. [DOI] [PubMed] [Google Scholar]
- Wen D., Peles E., Cupples R., Suggs S. V., Bacus S. S., Luo Y., Trail G., Hu S., Silbiger S. M., Levy R. B. Neu differentiation factor: a transmembrane glycoprotein containing an EGF domain and an immunoglobulin homology unit. Cell. 1992 May 1;69(3):559–572. doi: 10.1016/0092-8674(92)90456-m. [DOI] [PubMed] [Google Scholar]
- Yamamoto T., Ikawa S., Akiyama T., Semba K., Nomura N., Miyajima N., Saito T., Toyoshima K. Similarity of protein encoded by the human c-erb-B-2 gene to epidermal growth factor receptor. Nature. 1986 Jan 16;319(6050):230–234. doi: 10.1038/319230a0. [DOI] [PubMed] [Google Scholar]