Abstract
Periventricular leukomalacia (PVL) is a common white matter lesion affecting the neonatal brain. PVL is closely associated with cerebral palsy (CP) and characterized by increase in the number of astrocytes, which can be detected by positivity for glial fibrillary acidic protein (GFAP). Change in myelin basic protein (MBP) is an early sign of white matter abnormality. Maternal or placental infection can damage the neonatal brain. In the present study, we investigated the effects of treadmill walking exercise on GFAP and MBP expressions in rats with maternal lipopolysaccharide (LPS)-induced PVL. Immunohistochemistry was performed for the detection of GFAP and MBP. The present results showed that intracervical maternal LPS injection during pregnancy increased GFAP expression in the striatum and decreased MBP expression in the corpus callosum of rats. The results also showed that treadmill walking exercise suppressed GFAP expression and enhanced MBP expression in the brains of rats with maternal LPS-induced PVL. The present study revealed that treadmill walking exercise is effective for the suppressing astrogliosis and hypomyelination associated with PVL. Here in this study, we showed that treadmill walking exercise may be effective therapeutic strategy for alleviating the detrimental effects of CP.
Keywords: Periventricular leukomalacia, Cerebral palsy, Glial fibrillary acidic protein, Myelin basic protein, Treadmill walking exercise
INTRODUCTION
Approximately 10% of newborns are born prematurely, and severe disturbances in mental development and perinatal brain injuries have been observed in premature infants (Yesilirmak et al., 2007). More than 10% of these children have adverse neurological outcomes, including cognitive, motor, and behavioral deficits (Wozniak et al., 2004). Periventricular leukomalacia (PVL), a common white matter lesion in neonatal brain, is closely associated with cerebral palsy (CP) (Cai et al., 2000). The exact mechanisms underlying PVL have not been clarified; however, it has been proposed that maternal or placental infection may damage the neonatal brain (Adams-Chapman and Stoll, 2006).
Astroglia and myelin are important components of white matter. Reactive astrogliosis, typified by astrocyte proliferation and/or astrocytic hypertrophy, is a common phenomenon in the central nerve system (CNS) following brain trauma or inflammation (Maxwell et al., 1990). Direct injection of cytokines into the neonatal mouse brain increased reactive astrogliosis, indicating that pro-inflammatory cytokines induce astrogliosis (Balasingam et al., 1994). Clinical studies have provided evidences that PVL is closely associated with an increased number of astrocytes in white matter lesions of the neonatal brain (Deguchi et al., 1997; Obonai et al., 1997). Astrogliosis can be detected by positivity for glial fibrillary acidic protein (GFAP), an intermediate filament protein specific to astrocytes (Deguchi et al., 1997; Obonai et al., 1997). Myelin basic protein (MBP) is the most abundant protein in the myelin sheath (Kurumatani et al., 1998), and a change in the content of MBP is an early sign of white matter abnormality.
Maternal LPS administration increases the number of GFAP-positive astrocytes, decreases MBP expressions and alters microglia immunoreactivity in the brain of offspring (Cai et al., 2000). CP is caused by damage to the white matter, particularly in the periven tricular region, of the developing brain (Bell and Hallenbeck, 2002). PVL is strongly correlated with the development of CP (Bejar et al., 1992). Toso et al. (2005) reported that LPS exposure during pregnancy induce inflammatory model of CP in offspring.
It is well known that exercise induces several physiological and biochemical changes in the brain (Chaouloff, 1997; Sutoo and Akiyama, 1996). Running is known to be an important positive stimulus for neurogenesis (Lee et al., 2013; Ra et al., 2002). Physical exercise is also known to enhance neuronal plasticity and alter the transcription levels of various genes, in a manner associated with increased neuronal activity and synaptic remodeling (Tong et al., 2001). Molteni et al. (2002) reported that voluntary physical activity involving the use of a running wheel increases the expressions of hippocampal genes that are associated with neuronal plasticity. In addition, physical exercise has been reported to upregulate the levels of various neuropeptides and neurotrophins, such as neurotensin, substance P, and brain-derived neurotrophic factor (BDNF) mRNA, in the rat hippocampus (Bucinskaite et al., 1996; Kim et al., 2013; Russo-Neustadt et al., 1999).
In the present study, we investigated whether treadmill walking would have a protective effect on astrogliosis and hypomyelination in rats with PVL. The animal model of PVL was induced by maternal intracervical injection of LPS. In the present study, immunohistochemistry was performed for the detection of GFAP in the striatum and MBP in the corpus callosum.
MATERIALS AND METHODS
Animals and treatments
This study was performed in accordance with the guidelines of the National Institutes of Health (NIH) and the Korean Academy of Medical Sciences. Female Sprague-Dawley rats (180±10 g, 8 weeks old, n=20) were allowed to mate with male rats for a period of 24 h. The female rats were then individually housed in plastic home cages under controlled temperature (20±2°C) and a light –dark cycle consisting of 12 h of light and 12 h of darkness (lights on from 07:00 to 19:00 h). Food and water were made available ad libitum.
The pregnant rats were divided into two groups: the sham group and the LPS-treated group (n=4 in each group). On the 9th, 13th, and 17th day of pregnancy, the pregnant rats in the LPS-treated group received intracervical injections of 1.0 mg/kg LPS (from Escherichia coli, serotype 055:B5, Sigma Chemical Co., St. Louis, MO, USA) suspended in pyrogen-free saline (PFS), and the pregnant rats in the sham group were treated with PFS. After birth, the neonatal rats were divided into three groups: the sham group, the LPS-treated group, and the LPS-treated and exercise group (n=4 in each group).
Exercise protocol
Eight weeks after birth, the rats in the exercise group were forced to walk on a motorized treadmill for 30 min three times a week for 4 weeks. The exercise load consisted of walking at the speed of 8 meters/min at an inclination of 0°.
Tissue preparation
The animals were sacrificed immediately after the completion of the last exercise. The rats were weighed and given an overdose of Zoletil 50® (10 mg/kg i.p.; Vibac Laboratories, Carros, France). After a complete lack of response was observed, the rats were transcardially perfused with 50 mM phosphate-buffered saline (PBS) and subsequently fixed with freshly prepared 100 mM phosphate buffer (PB, pH 7.4) containing 4% paraformaldehyde. The brains were removed and fixed in the same fixative overnight and then transferred to a 30% sucrose solution for cryoprotection. Serial coronal sections of 40-μm thickness were obtained using a freezing microtome (Leica, Nussloch, Germany).
GFAP and MBP immunohistochemistry
An average of six sections within the striatum spanning from Bregma 1.20 mm to 0.96 mm and six sections within the corpus callosum spanning from Bregma 0.84 mm to 0.60 mm were obtained from each brain. Tissue sections were incubated overnight with rabbit anti-GFAP antibody or anti-MBP antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA) at a dilution of 1:500.
The sections were then incubated with anti-rabbit secondary antibody (1:200, Vector Laboratories, Burlingame, CA, USA). The sections were subsequently incubated with avidin- biotin-peroxidase complex (1:100, Vector Laboratories) for 1 h at room temperature. Immunoreactivity was visualized by incubating the sections in a solution containing 0.02% 3, 3′-diaminobenzidine tetrahydrochloride (DAB) and 0.03% H2O2 in 50 mM Tris-HCl (pH 7.6) for approximately 5 min. The sections were then washed three times with PBS and mounted onto gelatin-coated slides. The slides were air-dried overnight at room temperature, and coverslips were mounted using Permount® (Fisher Scientific, New Jersey, NJ, USA).
Data analysis
The intensities of GFAP and MBP staining were quantitatively assessed using a microdensitometrical method based on optical density (the mean gray scale) with the use of an image analyzer (Multiscan, Fullerton, CA, USA). Before starting the image analysis, the light source was adjusted to the brightness that generated the best possible contrast between immunopositive and immunonegative cells.
Statistically significant differences were determined by one-way analysis of variance (ANOVA) followed by Duncan’s Post-hoc analysis. The results were expressed as the mean±standard error of the mean (SEM). Differences were considered significant at P<0.05.
RESULTS
Effects of treadmill walking exercise on body weight in rats with maternal endotoxin-induced PVL
Eight weeks after birth, the mean body weight of the rats was 297±3.85 g in the sham group, 246±24.33 g in the LPS-treated group, and 299±2.00 g in the LPS-treated and exercise group. After 4 weeks of exercise, the mean body weight of the rats was 394±4.60 g in the sham group, 347±7.54 g in the LPS-treated group, 342±7.50 g in the LPS-treated and exercise group. There was no significant difference in body weight between the groups.
Effect of treadmill walking exercise on GFAP expression in the striatum of rats with maternal endotoxin-induced PVL
Photomicrographs of GFAP expression in the striatum are presented in Fig. 1. The optical density of the GFAP-positive cells in the striatum was 56.89±0.89/mm2 in the sham group, 60.29±0.36/mm2 in the LPS-treated group, and 57.77±0.14/mm2 in the LPS-treated and exercise group (Fig. 2). The results of the present study revealed that GFAP expression in the striatum was significantly increased in the LPS-treated group and that treadmill exercise suppressed the LPS-induced increase in GFAP expression.
Fig. 1.

Photomicrographs of glial fibrillary acidic protein (GFAP)-positive cells in the striatum. The scale bar represents 250 μm.
Fig. 2.
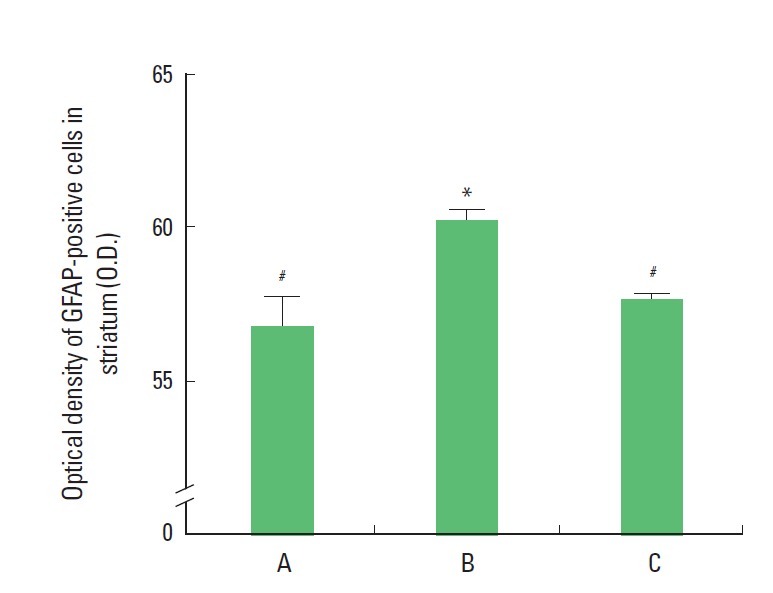
Effect of treadmill walking exercise on the expression of glial fibrillary acidic protein (GFAP) in the striatum of rats with maternal endotoxin-induced periventricular leukomalacia (PVL). (A) Sham group, (B) lipopolysaccharide (LPS)-treated group, (C) LPS-treated and exercise group. The data are presented as the mean± SEM. *Represents P< 0.05 compared to the sham group. #P< 0.05 compared to the LPS-treated group.
Effect of treadmill walking exercise on MBP expression in the corpus callosum of rats with maternal endotoxin-induced PVL
Photomicrographs of MBP expression in the corpus callosum are presented in Fig. 3. The optical density of the MBP-positive cells in the corpus callosum was 106.70±3.49/mm2 in the sham group, 93.30±2.00/mm2 in the LPS-treated group, and 103.12 ±2.16/mm2 in the LPS-treated and exercise group (Fig. 4). The results of the present study showed that the expression of MBP in the corpus callosum was significantly suppressed in the LPS-treated group and that treadmill exercise alleviated the LPS-induced suppression of MBP expression.
Fig. 3.
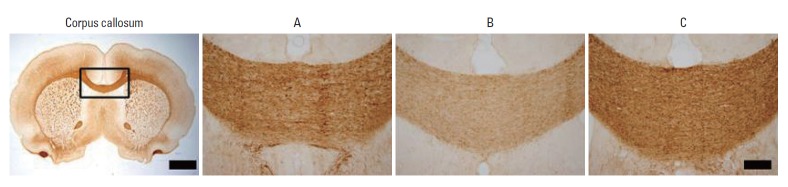
Photomicrographs of myelin basic protein (MBP)-positive cells in the corpus callosum. The scale bar represents 250 μm.
Fig. 4.
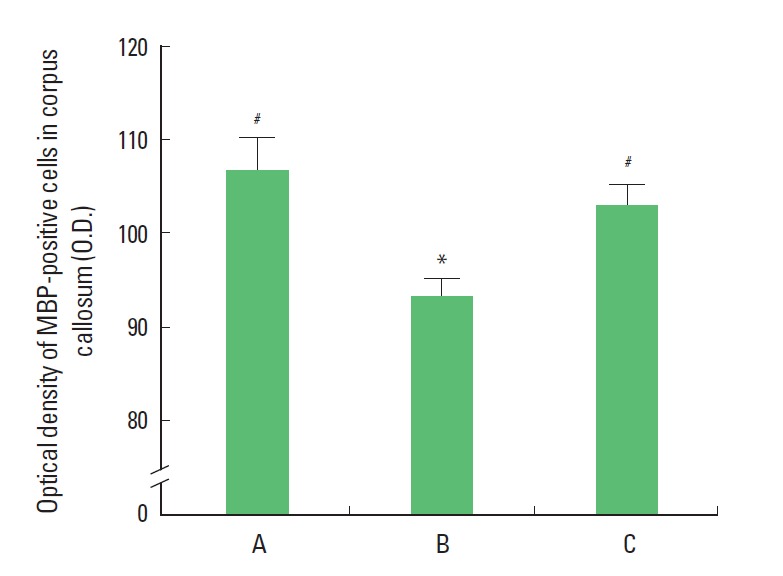
Effect of treadmill walking exercise on the expression of myelin basic protein (MBP) in the corpus callosum of rats with maternal endotoxin-induced periventricular leukomalacia (PVL). (A) Sham group, (B) lipopolysaccharide (LPS)-treated group, (C) LPS-treated and exercise group. The data are presented as the mean± SEM. *Represents P< 0.05 compared to the sham group. #P< 0.05 compared to the LPS-treated group.
DISCUSSION
Maternal infection as well as increased levels of pro-inflammatory cytokines in amniotic fluid and fetal blood is associated with preterm birth and neonatal neurological disorders (Goepfert et al., 2004; Zlokovic et al., 2005). White matter injury, especially such as PVL, is a common form of prenatal brain injury occurring in the immature babies, which results in life-long disorders of movement, posture, and cognition. The pathogenesis of PVL is multi-factorial and likely involves in ischemia-reperfusion, formation of reactive oxygen and nitrogen species, glutamate exitotoxicity, impairment of myelination due to loss of oligodendrocytes, apoptotic cell death, microglial activation, and maternal or fetal infection (Adams-Chapman, 2006; Leviton and Dammann, 2004). PVL is known as the major antecedent disorder developing to CP (Yesilirmak et al., 2007). Several animal models have been established to study the mechanisms of PVL and to search the therapeutic potential of candidate agents (Adams-Chapman, 2006). One of the widely used animal models for PVL can be achieved by administration of LPS to pregnant animals (Cai et al., 2004; Hagberg et al., 2002).
In the present results, intracervical injection of LPS into the maternal rats induced astrogliosis in the striatum of rats that was represented by enhanced expression of GFAP in the striatum. Many studies have shown that PVL is closely associated with the increased number of GFAP-positive astrocytes in the white matter lesion of infant brains (Deguchi et al., 1997; Obonai et al., 1997). Therefore, the increased GFAP content in the neonatal rat brain after maternal LPS administration is an indication of potential role of cytokines in mediating maternal infection and induction of PVL in newborn infants.
In the present results, intracervical injection of LPS into the maternal rats induced hypomyelination in the corpus callosum of rats, that was represented by suppressed expression of MBP in the corpus callosum. MBP is the most abundant protein in the myelin sheath. One possible reason for the reduced MBP staining is a reduction MBP production by the oligodendrocyte or a reduction in the number of oligodendrocytes after maternal LPS treatment. Reduced myelination and oligodendrocytes are concomitantly observed in the infant brains with PVL (Wang et al., 2006).
In the present results, we investigated the effects of treadmill walking exercise on astrogliosis and hypomyelination in rats with PVL. Treadmill walking exercise suppressed LPS-induced astrogliosis in the striatum and alleviated LPS-induced hypomyelination in the corpus callosum of the rats with PVL. The present study revealed that treadmill walking exercise is effective for the suppressing astrogliosis and hypomyelination associated with PVL. In the present study, we showed that treadmill walking exercise may be effective therapeutic strategy for alleviating the detrimental effects of CP.
Acknowledgments
This work was supported by the research grant of the Jeju National University in 2011.
Footnotes
CONFLICT OF INEREST
No potential conflict of interest relevant to this article was reported.
REFERENCES
- Adams-Chapman I. Neurodevelopmental outcome of the late preterm infant. Clin Perinatol. 2006;33:947–964. doi: 10.1016/j.clp.2006.09.004. [DOI] [PubMed] [Google Scholar]
- Adams-Chapman I, Stoll BJ. Neonatal infection and long-term neurodevelopmental outcome in the preterm infant. Curr Opin Infect Dis. 2006;19:290–297. doi: 10.1097/01.qco.0000224825.57976.87. [DOI] [PubMed] [Google Scholar]
- Balasingam V, Tejada-Berges T, Wright E, Bouckova R, Yong VW. Reactive astrogliosis in the neonatal mouse brain and its modulation by cytokines. J Neurosci. 1994;14:846–856. doi: 10.1523/JNEUROSCI.14-02-00846.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bejar RF, Vaucher YE, Benirschke K, Berry CC. Postnatal white matter necrosis in preterm infants. J Perinatol. 1992;12:2–8. [PubMed] [Google Scholar]
- Bell MJ, Hallenbeck JM. Effects of intrauterine inflammation on developing rat brain. J Neurosci Res. 2002;70:570–579. doi: 10.1002/jnr.10423. [DOI] [PubMed] [Google Scholar]
- Bucinskaite V, Theodorsson E, Crumpton K, Stenfors C, Ekblom A, Lundeberg T. Effects of repeated sensory stimulation (electro-acupuncture) and physical exercise (running) on open-field behaviour and concentrations of neuropeptides in the hippocampus in WKY and SHR rats. Eur J Neurosci. 1996;8:382–387. doi: 10.1111/j.1460-9568.1996.tb01221.x. [DOI] [PubMed] [Google Scholar]
- Cai Z, Pan ZL, Pang Y, Evans OB, Rhodes PG. Cytokine induction in fetal rat brains and brain injury in neonatal rats after maternal lipopolysaccharide administration. Pediatr Res. 2000;47:64–72. doi: 10.1203/00006450-200001000-00013. [DOI] [PubMed] [Google Scholar]
- Chaouloff F. Effects of acute physical exercise on central serotonergic systems. Med Sci Sports Exerc. 1997;29:58–62. doi: 10.1097/00005768-199701000-00009. [DOI] [PubMed] [Google Scholar]
- Deguchi K, Oguchi K, Takashima S. Characteristic neuropathology of leukomalacia in extremely low birth weight infants. Pediatr Neurol. 1997;16:296–300. doi: 10.1016/s0887-8994(97)00041-6. [DOI] [PubMed] [Google Scholar]
- Goepfert AR, Andrew WW, Carlo W, Ramsev PS, Cliver SP, Goldenberg RL, et al. Umbilical cord plasma interleukin-6 concentrations in preterm infants and risk of neonatal morbidity. Am J Obstet Gynecol. 2004;191:1375–1381. doi: 10.1016/j.ajog.2004.06.086. [DOI] [PubMed] [Google Scholar]
- Hagberg H, Peebles D, Mallard C. Models of white matter injury: Comparison of infectious, hypoxic-ischemic, and excitotoxic insults. Ment Retard Dev Disabil Res Rev. 2002;8:30–38. doi: 10.1002/mrdd.10007. [DOI] [PubMed] [Google Scholar]
- Kim SE, Ko IG, Park CY, Shin MS, Kim CJ, Jee YS. Treadmill and wheel exercise alleviate lipopolysaccharide-induced short-term memory impairment by enhancing neuronal maturation in rats. Mol Med Rep. 2013;7:31–36. doi: 10.3892/mmr.2012.1160. [DOI] [PubMed] [Google Scholar]
- Lee SW, Kim YS, Jun TW, Seo JH, Kim K, Shin MS, Kim CJ. The impact of duration of one bout treadmill exercise on cell proliferation and central fatigue in rats. J Exerc Rehabil. 2013;9:463–469. doi: 10.12965/jer.130069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurumatani T, Kudo T, Ikura Y, Takeda M. White matter changes in the gerbil brain under chronic cerebral hypoperfusion. Stroke. 1998;29:1058–1062. doi: 10.1161/01.str.29.5.1058. [DOI] [PubMed] [Google Scholar]
- Leviton A, Dammann O. Coagulation, inflammation, and the risk of neonatal white matter damage. Pediatr Res. 2004;55:541–545. doi: 10.1203/01.PDR.0000121197.24154.82. [DOI] [PubMed] [Google Scholar]
- Maxwell WL, Follows R, Ashhurst DE, Berry M. The response of the cerebral hemisphere of the rat to injury. Philos Trans R Soc Lond B Biol Sci. 1990;328:501–513. doi: 10.1098/rstb.1990.0122. [DOI] [PubMed] [Google Scholar]
- Molteni R, Ying Z, Gomez-Pinilla F. Differential effects of acute and chronic exercise on plasticity-related genes in the rat hippocampus revealed by microarray. Eur J Neurosci. 2002;16:1107–1116. doi: 10.1046/j.1460-9568.2002.02158.x. [DOI] [PubMed] [Google Scholar]
- Obonai T, Asanuma M, Mizuta R, Horie H, Tanaka J, Takashima S. Evidence of brain ischemia in early neonatal sudden death syndrome. Neuropediatrics. 1997;28:145–148. doi: 10.1055/s-2007-973691. [DOI] [PubMed] [Google Scholar]
- Ra SM, Kim H, Jang MH, Shin MC, Lee TH, Lim BV, Kim CJ, Kim EH, Kim KM, Kim SS. Treadmill running and swimming increase cell proliferation in the hippocampal dentate gyrus of rats. Neurosci Lett. 2002;333:123–126. doi: 10.1016/s0304-3940(02)01031-5. [DOI] [PubMed] [Google Scholar]
- Russo-Neustadt A, Beard RC, Cotman CW. Exercise, antidepressant medications, and enhanced brain derived neurotrophic factor expression. Neuropsychopharmacology. 1999;21:679–682. doi: 10.1016/S0893-133X(99)00059-7. [DOI] [PubMed] [Google Scholar]
- Sutoo DE, Akiyama K. The mechanism by which exercise modifies brain function. Physiol Behav. 1996;60:177–181. doi: 10.1016/0031-9384(96)00011-x. [DOI] [PubMed] [Google Scholar]
- Tong L, Shen H, Perreau VM, Balazs R, Cotman CW. Effects of exercise on gene-expression profile in the rat hippocampus. Neurobiol Dis. 2001;8:1046–1056. doi: 10.1006/nbdi.2001.0427. [DOI] [PubMed] [Google Scholar]
- Toso L, Poggi S, Park J, Einat H, Roberson R, Dunlap V, et al. Inflammatory-mediated model of cerebral palsy with developmental sequelae. Am J Obstet Gynecol. 2005;193:933–941. doi: 10.1016/j.ajog.2005.05.072. [DOI] [PubMed] [Google Scholar]
- Wang X, Rousset CI, Hagberg H, Mallard C. Lipopolysaccharide-induced inflammation and perinatal brain injury. Semin Fetal Neonatal Med. 2006;11:343–353. doi: 10.1016/j.siny.2006.04.002. [DOI] [PubMed] [Google Scholar]
- Wozniak DF, Hartman RE, Boyle MP, Vogt SK, Brooks AR, Tenkova T, Young C, Olney JW, Muglia LJ. Apoptotic neurodegeneration induced by ethanol in neonatal mice is associated with profound learning/memory deficits in juveniles followed by progressive functional recovery in adults. Neurobiol Dis. 2004;17:403–414. doi: 10.1016/j.nbd.2004.08.006. [DOI] [PubMed] [Google Scholar]
- Yesilirmak DC, Kumral A, Baskin H, Ergur BU, Aykan S, Genc S, Genc K, Yilmaz O, Tugyan K, Giray O, Duman N, Ozkan H. Activated protein C reduces endotoxin-induced white matter injury in the developing rat brain. Brain Res. 2007;1164:14–23. doi: 10.1016/j.brainres.2007.04.083. [DOI] [PubMed] [Google Scholar]
- Zlokovic BV, Zhang C, Liu D, Fernandez J, Griffin JH, Chopp M. Functional recovery after embolic stroke in rodents by activated protein C. Ann Neurol. 2005;58:474–477. doi: 10.1002/ana.20602. [DOI] [PubMed] [Google Scholar]


