Abstract
Introduction
The diagnosis of acute pulmonary embolism (APE) in patients with chronic heart failure (CHF) remains a difficult task, despite the refinement of imaging techniques. The goal of this study was to assess the value of measuring tricuspid and mitral valve systolic annular velocities in CHF patients with suspected PE by tissue Doppler imaging (TDI).
Material and methods
The study included 75 patients with previously diagnosed CHF, admitted due to resting dyspnea, with a maximum tricuspid regurgitation pressure gradient (TRPG) of ≥ 35 mm Hg and positive D-dimer assay. Spiral computed tomography (sCT) was performed on all subjects to confirm APE. Acute pulmonary embolism was diagnosed in 35 patients (PE+), and excluded in 40 others (PE–). Tissue Doppler imaging was performed to measure maximum systolic lateral annular velocities in the mitral (SmLV) and tricuspid (SmRV) valves, as well as the SmRV/SmLV ratio.
Results
PE+ subjects were found to have higher SmLV than PE– subjects (6.0 cm/s (2.0–13.8 cm/s) vs. 4.2 cm/s (1.3–9.1 cm/s), p = 0.003). SmRV/SmLV ratios were 1.05 (0.50–2.50) and 1.56 (0.62–4.30), respectively (p < 0.0001). Areas under ROC curves for diagnosis of APE were 0.700 for SmLV and 0.789 for SmRV/SmLV. In multivariate logistic regression analysis, only SmRV/SmLV was statistically significant, with an odds ratio for APE of 6.26 (95% CI: 1.53–25.59; p = 0.009).
Conclusions
Tissue Doppler imaging of the lateral tricuspid and mitral annuli is a useful clinical tool that can help identify PE in CHF patients. Those patients who fulfill these criteria should be considered for further diagnostic studies to confirm PE.
Keywords: tissue Doppler imaging, pulmonary embolism, chronic heart failure
Introduction
Pulmonary embolism (PE) is the most severe form of venous thromboembolism (VTE), with an annual incidence of approx. 0.5–1 per 1000 of the population. The mortality rate of untreated PE is about 30%; when appropriate treatment is administered, this decreases to 5% [1]. Chronic heart failure (CHF) is a significant risk factor for PE. The current European Society of Cardiology guidelines include CHF in the category of moderate, patient-related predisposing factors. Patients with concurrent CHF and PE represent a diagnostic challenge, as the symptomatic similarity of the two conditions makes it difficult to differentiate between them. Patients with confirmed PE and CHF have a higher mortality rate than patients without CHF – 17% and 10%, respectively. Furthermore, PE is an independent risk factor for death or readmission in patients with CHF [2, 3].
Transthoracic echocardiography (TTE) is a non-invasive, easily available, and simple examination which can be performed even at the bedside. Signs of right ventricular (RV) overload are found in 25–50% of patients with confirmed PE, and are an indirect marker of PE. Since the presence of RV dysfunction in patients with a non-high risk for PE is associated with worse outcomes, the value of TTE in this group is prognostic, rather than diagnostic.
Although the role of echocardiography in the diagnosis of PE is limited, mainly due to its low sensitivity and specificity, we have previously demonstrated that measuring tricuspid (TAPSE) and mitral (MAPSE) annular plane systolic excursion can help identify patients with PE among those with CHF [4]. In addition to standard echocardiography, tissue Doppler echocardiography (TDE) can be performed to assess the myocardium. Tissue Doppler echocardiography allows for the evaluation of regional myocardial function by measuring the movement velocity of specific areas [5]. Damaged myocardium is characterized by decreased segmental wall velocity [6]. A similar change is observed in mitral and tricuspid valve systolic annular velocities, which decrease as a result of injury to the corresponding ventricles [7–12]. Patients with CHF frequently present with RV overload, which may suggest the presence of acute pulmonary embolism (APE). The preliminary data of our research evaluating the role of tissue Doppler imaging in PE in a limited number of patients were presented in our previous publication [13].
The goal of this study is to assess the value of measuring tricuspid and mitral valve systolic annular velocities by transthoracic tissue Doppler echocardiography in patients with chronic heart failure and suspected non-high risk of acute pulmonary embolism.
Material and methods
The study group consisted of 75 patients admitted due to exacerbation of previously diagnosed CHF, with resting dyspnea and signs of pulmonary congestion evidenced by chest X-ray.
The following inclusion criteria were applied: tricuspid regurgitation peak gradient (TRPG) ≥ 35 mm Hg by echocardiography, positive D-dimer assay by immuno-turbidimetric Liatest (> 0.5 µg/ml), and at least intermediate clinical probability of PE by Wells score [14].
Spiral computed tomography (sCT) to confirm PE was performed in all patients on the day of admission. Pulmonary embolism was diagnosed based on the presence of embolic material, i.e. at least one contrast enhancement defect in the pulmonary circulation, at the level of at least segmental arteries. In patients with a negative sCT for PE, compression ultrasound (CUS) of the proximal veins of the lower limb was performed. Compression ultrasound was performed using a linear transducer and following a four-point protocol. Incomplete venous compression under pressure applied by the transducer was considered indicative of thrombosis [15]. Subjects were divided into two groups, based on sCT results: subjects with PE confirmed by positive sCT (PE+), and subjects with PE excluded by negative sCT and negative CUS (PE–).
The following exclusion criteria were applied: acute coronary syndrome within 14 days before admission, anemia with a hemoglobin concentration below 11 g/dl, hyperthyroidism, mitral and/or tricuspid valve replacement, significant calcification of the mitral and/or tricuspid valve annuli, significant valvular defects and/or insufficiency, pericardial disease with pericardial fluid exceeding 10 mm in diastole, constrictive pericarditis, cardiac tamponade.
Echocardiographic examination
Standard echocardiography was performed in all patients using a 2.5 MHz transducer (System 5, Vingmed, General Electric) and a simultaneous ECG recording. End-diastolic dimension of the left ventricle (LVEDD) and the end-diastolic dimension of the right ventricle (RVEDD) were measured in the parasternal long-axis view. In the apical four-chamber view the tricuspid regurgitation peak velocity was measured with continuous wave Doppler and tricuspid value peak systolic gradient (TRPG) was calculated according to the simplified Bernoulli formula. The LV ejection fraction (LVEF) was measured according to the simplified Simpson's method. In order to assess the right ventricular function using one-dimensional M-mode echocardiography, the tricuspid annular peak systolic excursion (TAPSE) was measured [9, 16]. The subcostal view was used to assess inferior vena cava (IVC) diameter and respiratory dynamics.
Tissue Doppler echocardiography
Color tissue Doppler echocardiography with computer image analysis (Echopac 6.3, GE Vingmed) was performed using a 2.5 MHz transducer for all patients. Mitral valve velocity profile was recorded in the apical four-chamber view, by placing the Doppler volume over the lateral side of the valve, at the posterior leaflet. Systolic tricuspid valve lateral annular velocity was measured by an analogous method, over the anterior leaflet in the long-axis view of the right ventricle [7, 17, 18]. The following parameters were calculated: maximum systolic mitral valve lateral annular velocity (SmLV) and maximum systolic tricuspid valve lateral annular velocity (SmRV), and tricuspid-to-mitral valve systolic lateral annular velocity ratio (SmRV/SmLV). Additionally, the LV and RV myocardial performance indices (MPI) were calculated. All values are expressed as the mean of measurements in 3–5 consecutive cardiac cycles.
Treatment
All patients were on standard CHF treatment regimens prior to hospitalization, including angiotensin-converting enzyme inhibitors or angiotensin receptor blockers, β-blockers, and diuretics. All patients included in the study received anticoagulant therapy with unfractionated heparin (80 IU/kg intravenous bolus, followed by intravenous pump infusion at 18 IU/kg/h) until PE was confirmed or excluded. In patients with confirmed PE, unfractionated heparin was continued at the therapeutic dose for an average period of 5 days, and an oral anticoagulant (acenocoumarol) was introduced. Heparin was discontinued once the international normalized ratio (INR) was confirmed to be in the therapeutic range (2–3) on 2 successive days. In patients with a negative sCT and CUS, PE was excluded and therapeutic-range heparin was discontinued, and replaced by a prophylactic dose of 5000 IU administered subcutaneously every 12 h.
Statistical analysis
Values of parameters with a normal distribution are presented as mean ± standard deviation. Values with non-normal distributions are expressed as median and range. Study groups were compared using Student's t test or Mann-Whitney test, depending on the parameter distribution. Chi-squared (χ2) tests were used to compare qualitative variables across multiple groups.
Receiver operating characteristic (ROC) analysis curves were used to determine the optimal cutoff points for identifying patients with PE. Uni- and multivariate logistic regression analyses were performed to assess the diagnostic value of the studied parameters. The following parameters were evaluated by tissue Doppler echocardiography for all 75 subjects: SmLV and SmRV, and SmRV/SmLV, the MPI for both RV and LV and the RV MPI to LV MPI ratio (RV/LV MPI). Odds ratios were calculated to evaluate the diagnostic significance of individual parameters. The analyses included those parameters which were found to be significantly different between PE+ and PE– groups. Variables were analyzed both as quantitative values and as qualitative parameters (above/below cutoff value, as determined by ROC analysis). Stepwise regression analysis was applied to identify a practical method for diagnosing PE in CHF patients. The analysis was restricted to those parameters which were found to be statistically significant in univariate analyses. The study protocol was approved by a local ethics committee (no. 637/08). Statistical analyses were conducted using Statistica 6 software.
Results
The studied population comprised 75 patients with CHF, admitted due to acute resting dyspnea, and fulfilling the inclusion criteria. A total of 382 consecutive patients with symptoms of CHF exacerbation were evaluated to obtain this sample size. Clinical and echocardiographic characteristics of the study population are presented in Table I.
Table I.
General characteristics of the study population
| Variable | All group (n = 75) | PE+ (n = 35) | PE– (n = 40) | Value of p |
|---|---|---|---|---|
| Male gender, n (%) | 36 (48) | 14 (40) | 22 (55) | 0.19 |
| Age [years] | 73.7 ±11.8 | 75.6 ±10.9 | 72.0 ±12.4 | 0.18 |
| Hypertension, n (%) | 45 (60) | 22 (62.8) | 23 (57.5) | 0.63 |
| Atrial fibrillation, n (%) | 34 (45.3) | 13 (37.1) | 21 (52.5) | 0.18 |
| Previous MI, n (%) | 21 (28) | 8 (22.8) | 13 (32.5) | 0.35 |
| LVEF < 40%, n (%) | 25 (33.3) | 9 (25.7) | 16 (40) | 0.19 |
| RVEDD [cm] | 3.3 (1.85–4.78) | 3.4 (2.4–4.7) | 3.3 (1.85–4.78) | 0.11 |
| LVEDD [cm] | 5.33 ±1.0 | 5.08 ±1.0 | 5.5 ±0.97 | 0.04 |
| LVEF (%) | 48 (20–80) | 55 (20–80) | 45 (20–79) | 0.08 |
| TRPG [mm Hg] | 40 (35–84) | 45 (35–84) | 36 (35–83) | 0.02 |
| VCI [cm] | 2.42 ±0.4 | 2.39 ±0.33 | 2.44 ±0.47 | 0.62 |
| TAPSE [cm] | 1.50 ±0.39 | 1.38 ±0.35 | 1.61 ±0.4 | 0.01 |
PE – pulmonary embolism, MI – myocardial infarction, RVEDD – right ventricular end-diastolic diameter, LVEDD – left ventricular end-diastolic diameter, LVEF – left ventricular ejection fraction, TRPG – tricuspid regurgitation pressure gradient, VCI – vena cava inferior, TAPSE – tricuspid annular peak systolic excursion
The etiology of CHF was similar in both groups with and without PE, and included ischemic heart disease, hypertension, and atrial fibrillation. In patients with a history of myocardial infarction (MI), echocardiography additionally included the assessment of the localization of necrotic myocardium, especially the involvement of the LV lateral wall. The presence of contractile abnormalities in this region could significantly confound measurement of mitral annular velocity by tissue Doppler. Lateral wall MI was found in 2 of 8 PE+ patients, and 1 of 13 PE– patients.
In the PE+ group, 9 patients (26%) were found to have CHF with significant LV systolic dysfunction (EF < 40%), while 26 patients (74%) had CHF with preserved LV systolic function (EF ≥ 40%). In the PE– group, 16 patients (40%) were found to have CHF with significant LV systolic dysfunction (EF < 40%), while 24 patients (60%) had CHF with preserved LV systolic function (EF ≥ 40%).
Standard transthoracic echocardiography results
PE+ patients, compared to PE– subjects, were found to have a smaller LVEDD (5.08 ±1.0 cm vs. 5.5 ±0.97 cm, p = 0.04, respectively), higher TRPG (45 mm Hg (35–84 mm Hg) vs. 36 mm Hg (35–83 mm Hg), p = 0.019, respectively), and lower TAPSE (1.38 ±0.35 cm vs. 1.61±0.4 cm, p = 0.01, respectively). Patients in both groups were similar with respect to RVEDD, LVEF and IVC.
Tissue Doppler echocardiography results
PE+ patients, compared to PE– subjects, were found to have higher systolic mitral valve lateral annular velocities (SmLV, 6.0 cm/s (2.0–13.8 cm/s) vs. 4.2 cm/s (1.3–9.1 cm/s), p = 0.003). Systolic tricuspid valve lateral annular velocities were not significantly different between groups (SmRV: 6.6 ±2.6 cm/s vs. 7.2 ±2.5 cm/s, p = 0.36). Peak tricuspid-to-mitral valve systolic lateral annular velocity ratios (SmRV/SmLV) were 1.05 (0.50–2.50) in PE+ subjects and 1.56 (0.62–4.30) in PE– subjects (p < 0.0001). The RV MPI and LV MPI were not significantly different between groups. However, the RV/LV MPI ratio was higher in the group with confirmed PE than in the group without PE (1.08 ±0.17 vs. 1.0 ±0.15, respectively, p = 0.03).
ROC analysis
The area under the ROC curve for SmLV in diagnosing PE was 0.700 with 95% CI 0.584–0.801. The corresponding cutoff value of SmLV = 5.5 cm/s demonstrated a sensitivity of 57.1%, specificity of 80.0%, positive predictive value (PPV) of 71.4%, and negative predictive value (NPV) of 68.1%.
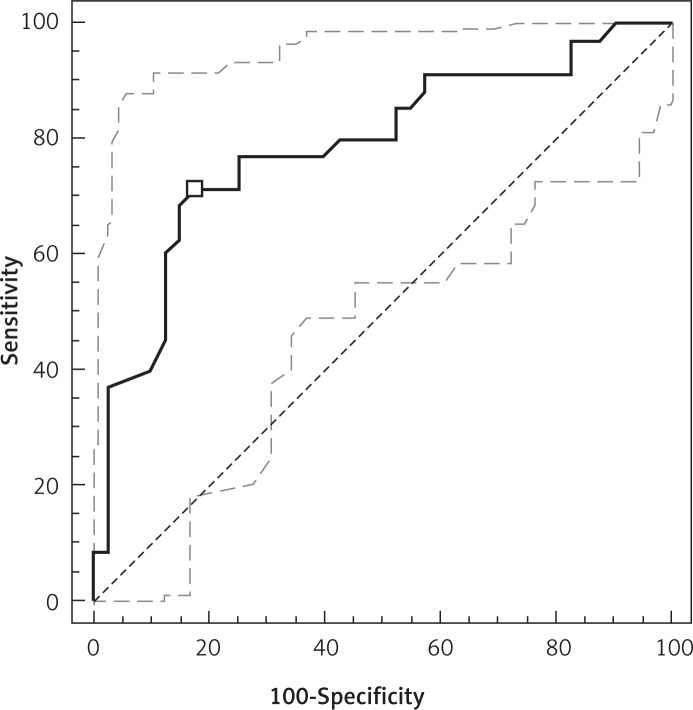
The area under the ROC curve for SmRV/SmLV was 0.789 with 95% CI 0.670–0.874. For the selected value of SmRV/SmLV = 1.23, sensitivity was found to be 71.4% and specificity equaled 82.5%; PPV and NPV were 78.1% and 76.7%, respectively (Figure 1).
Figure 1.
ROC analysis evaluating the usefulness of SmRV/SmLV in the diagnosis of PE
Due to a heterogeneous population of patients with respect to LV systolic function (LVSF), especially considering patients with a history of myocardial infarction and impaired LVEF, additionally systolic velocities of the examined annuli were measured in patients with LVEF < 40% and ≥ 40%.
The population of patients with LVEF < 40% comprised 9 subjects in the PE+ group and 16 subjects in the PE– group. Compared to patients without PE, subjects with confirmed PE were found to have a smaller SmRV/SmLV ratio (1.2 (0.5–2.5) vs. 1.6 (1.0–4.3), p = 0.018) and SmRV (4.3 ±1.0 cm/s vs. 5.8 ±2.0 cm/s, respectively, p = 0.05), whereas SmLV did not present a statistically significant difference (3.9 ±1.3 cm/s vs. 3.3 ±1.3 cm/s, respectively, p = 0.3). The area under the ROC curve for SmRV/SmLV was 0.788 with 95% CI 0.580–0.924, and the obtained value of SmRV/SmLV = 1.3 demonstrated a sensitivity of 88.9% and specificity of 75%; PPV and NPV were 66.7% and 92.3%, respectively.
The population of patients with LVEF ≥ 40% comprised 26 subjects in the PE+ group and 24 subjects in the PE- group. Compared to the PE– group, PE+ patients were also found to have a smaller SmRV/SmLV ratio (1.0 (0.6–2.2) vs. 1.55 (0.6–3.0) respectively, p = 0.0008) and higher SmLV (7.0 (3.8–13.8) vs. 4.8 (2.9–9.1) cm/s, respectively, p = 0.009), whereas SmRV was similar in both groups (7.4 ±2.5 cm/s vs. 8.1 ±2.3 cm/s, respectively, p = 0.36). The area under the ROC curve for SmLV was 0.716 with 95% CI 0.570–0.834, and the value of SmLV = 5.5 cm/s, selected on the basis of the analysis, demonstrated a sensitivity of 69.2% and specificity of 70.8%; PPV and NPV were 72% and 68%, respectively. The area under the ROC curve for SmRV/SmLV in the diagnosis of PE was 0.776 with 95% CI 0.636–0.881. For the selected SmRV/SmLV = 1.2, the sensitivity was found to be 73% and specificity 87.5% in the diagnosis of PE, whereas PPV and NPV were 86.4% and 75%, respectively.
Univariate logistic regression analysis
In univariate analyses, tissue Doppler echocardiographic parameters were found to have the highest odds ratios for diagnosis of PE: SmRV/SmLV ≤ 1.23 (OR: 11.78, 95% CI: 3.86–35.95, p < 0.0001), SmLV > 5.5 cm/s (OR: 5.33, 95% CI: 1.88–15.10, p = 0.0013). Results are presented in Table II.
Table II.
Univariate logistic regression analysis of echocardiographic variables
| Variables | Odds ratio | 95% CI | Value of p |
|---|---|---|---|
| LVEDD [cm] | 0.61 | (0.37–1.00) | 0.046 |
| TAPSE ≤ 1.4 [cm] | 3.11 | (1.19–8.15) | 0.018 |
| TRPG [mm Hg] | 1.03 | (0.99–1.07) | 0.053 |
| SmLV > 5.5 [cm/s] | 5.33 | (1.88–15.10) | 0.0013 |
| SmRV/SmLV ≤ 1.23 | 11.78 | (3.86–35.95) | < 0.0001 |
CI – confidence interval, LVEDD – left ventricular end-diastolic diameter, TAPSE – tricuspid annular peak systolic excursion, TRPG – tricuspid regurgitation pressure gradient, RV – right ventricular, LV – left ventricular, SmLV – peak systolic velocity of mitral annulus, SmRV/SmLV – ratio of peak systolic velocity of tricuspid annulus to peak systolic velocity of mitral annulus
Multivariate logistic regression analysis
In multivariate analyses, the only parameter which reached statistical significance was SmRV/SmLV ≤ 1.23, with an odds ratio for PE of 6.26 (95% CI: 1.53–25.59; p = 0.009). Results are presented in Table III.
Table III.
Multivariate logistic regression analysis of echocardiographic variables
| Variables | Odds ratio | 95% CI | Value of p |
|---|---|---|---|
| LVEDD [cm] | 0.68 | (0.35–1.34) | 0.26 |
| TAPSE ≤ 1.4 [cm] | 3.32 | (0.94–11.7) | 0.057 |
| SmLV > 5.5 [cm/s] | 1.71 | (0.36–8.0) | 0.48 |
| SmRV/SmLV ≤ 1.23 | 6.26 | (1.53–25.59) | 0.009 |
CI – confidence interval, LVEDD – left ventricular end-diastolic diameter, TAPSE – tricuspid annular peak systolic excursion, SmLV – peak systolic velocity of mitral annulus, SmRV/SmLV – ratio of peak systolic velocity of tricuspid annulus to peak systolic velocity of mitral annulus
Discussion
Chronic heart failure is an important risk factor for PE. Since the signs and symptoms of CHF can be similar to those of PE, differentiation between the two is particularly difficult [19]. Furthermore, PE is an independent risk factor for mortality or readmission in CHF patients [3]. According to the current guidelines of the European Society of Cardiology, echocardiographic examination is not a diagnostically significant study for patients with non-high risk, i.e. hemodynamically stable, PE [1].
Right ventricular overload occurs in most patients with CHF, usually secondary to LV failure or left-heart valvular defects. Tissue Doppler echocardiography allows for the analysis of regional wall function, based on the measurement of the velocity of specific areas of myocardium or the atrioventricular valve annuli. This method has been validated in several echocardiographic studies [20–26].
The current study focused on patients with CHF and PE; the control group comprised patients with CHF, in whom PE had been excluded, with an increased tricuspid regurgitation peak gradient (TRPG ≥ 35 mm Hg). In tissue Doppler imaging, peak systolic tricuspid valve lateral annular velocities did not differ between the groups. However, peak systolic mitral valve lateral annular velocities were found to be higher in patients with confirmed PE than in patients with excluded PE.
A few recently published studies have assessed the value of tissue Doppler echocardiography in the diagnosis of PE [27–30]. Kjaergaard et al. [27] measured systolic velocities of the basal and middle segments of the RV free wall in patients with confirmed PE and without PE. The results were not found to differ between groups, which supports our findings. Hsiao et al. [28] found that patients with PE had a lower early-diastolic mitral inflow velocity and higher RV myocardial performance index (MPI), compared to patients with pulmonary hypertension and healthy subjects. The investigators calculated the M index, defined as the ratio of early-diastolic mitral inflow velocity to RV MPI, and found that a cutoff of < 112 had a sensitivity and specificity of 92% each for the diagnosis of PE. No significant differences were noted between patients with PE and pulmonary hypertension with respect to systolic tricuspid valve lateral annular velocity, which again validates our results. However, systolic velocities of the basal segment of the interventricular septum (IVS) were found to be higher in patients with PE than in those with pulmonary hypertension. A similar increase in systolic velocity of the basal segment of the IVS was observed in another study by Hsiao et al. [29]. This could be explained by an additional effect of LV contraction on the IVS, which could in turn be due to an increase in peak systolic mitral valve lateral annular velocity, as observed in the setting of PE in the current study. This is reflected in the values of peak systolic mitral valve lateral annular velocity (SmLV) in patients with PE (6.0 cm/s (2.0–13.8 cm/s)) and without PE (4.2 cm/s (1.3–9.1 cm/s)); p = 0.003.
Therefore, it seems that the assessment of LV function may play a significant role in the diagnosis of PE. In the aforementioned study by Hsiao et al. [29], peak systolic mitral valve lateral annular velocity was found to be higher in patients with PE than those with pulmonary hypertension (9.3 ±2.6 cm/s and 8.8 ±3.0 cm/s, respectively), but this difference was not statistically significant. The decreased early-diastolic mitral inflow velocity, observed by Hsiao et al. [28] using spectral Doppler imaging, suggests that hyperkinetic LV function in PE is a result of decreased LV preload. It appears that LV contractile activity is increased in patients with PE, which would explain the higher systolic mitral valve lateral annular velocities (i.e. “hyperkinetic” contraction) observed in PE patients in our study, compared to patients with CHF and pulmonary hypertension.
This hypothesis is supported by the findings of Hsiao, Hsiao et al. [30], who assessed RV and LV MPI in the diagnosis of PE in patients with pulmonary hypertension. Patients with confirmed PE were found to have higher RV MPI, but lower LV MPI, than patients with pulmonary hypertension. The decreased LV MPI in subjects with PE would indicate an improvement in LV mechanical function, relative to patients with pulmonary hypertension. In order to compare RV and LV function, the authors calculated the V index, defined as the ratio of RV MPI to LV MPI. Values of > 1.2 identified patients with PE with a sensitivity of 82% and a specificity of 83%. In our material, the value of LV MPI was similar for both studied groups. The value of RV MPI in both groups of patients was at the limit of statistical significance (p = 0.08), with a trend towards higher values in patients with confirmed PE. However, the ratio of RV/LV MPI was greater in patients with confirmed PE compared to subjects without PE, and was 1.08 ±0.17 and 1.00 ±0.15, p = 0.03, respectively, whereas the RV/LV MPI ratio = 1.11 identified patients with PE with a sensitivity of 41.9% and specificity of 79.5%. In the current study, the ratio of systolic tricuspid valve lateral annular velocity to peak systolic mitral valve lateral annular velocity (SmRV/SmLV) was a relatively sensitive and specific indicator of PE in patients with CHF. The area under the ROC curve for SmRV/SmLV ≤ 1.23 was 0.789, sensitivity and specificity for diagnosis of PE were 71.4% and 82.5%, respectively, and PPV and NPV were 78.1% and 76.7%, respectively. In multivariate regression analysis, this was the only parameter that was found to be statistically significant, with an OR for diagnosis of PE of 6.25 (95% CI: 1.53–25.59). In both groups of patients – those with impaired LV systolic function (LVEF < 40%) and those with preserved LV systolic function (LVEF ≥ 40%) – the evaluated ratio of SmRV/SmLV was still a rather sensitive and specific indicator of PE. This was the case especially in patients with LVEF < 40%, i.e., in patients with a history of myocardial infarction. In this group of patients, we considered whether the infarct scar impacted on the evaluated systolic mitral valve lateral annular velocities. On the one hand, systolic mitral valve lateral annular velocity could be lowered due to the infarct scar in the lateral wall in 2 patients out of 8 subjects in the PE+ group and in 1 out of 13 patients in the PE– group. On the other hand, the systolic velocity of this annulus could be higher due to a non-lateral infarct scar in 6 out of 8 PE+ patients and in 12 out of 13 PE– patients. The higher systolic velocity of this annulus could be caused by the compensating reaction of the LV in order to retain an adequate ejection volume of the damaged LV. In our study, in patients with LVEF < 40%, despite the presence of infarct scars, the evaluated ratio of SmRV/LV still remained a rather sensitive prognostic factor for diagnosing PE. The area under the ROC curve for SmRV/SmLV = 1.3 was 0.788, whereas the sensitivity and specificity of this parameter for diagnosing PE were 88.9% and 75%, respectively, with PPV and NPV being 66.7% and 92.3%, respectively.
In tissue Doppler echocardiography systolic movement velocities were measured only within the lateral portion of the mitral annulus at the posterior leaflet of the mitral valve and within the lateral portion of the tricuspid annulus at the anterior leaflet of the tricuspid valve. The analysis of systolic movement velocities concerning the remaining portions of both annuli could improve the precision of this study. The hypothesis that APE leads to the so-called “hyperkinetic LV” by decreasing LV preload has not been confirmed by other noninvasive or invasive methods. Moreover, the diagnostic value of the SmRV/SmLV ratio in diagnosing PE was only compared to the V index (RV/LV MPI). Comparing it to other known echocardiographic parameters, e.g. 60/60 parameter, McConnell's sign, and the M index, would allow us to evaluate its diagnostic value much better. The most significant limitation of this study is that it is a single-center study, conducted on a relatively small group of subjects.
Potential clinical relevance: Measurement of mitral and systolic tricuspid valve lateral annular velocities by tissue Doppler echocardiography in the four-chamber view is simple, fast, and can be conducted at the bedside during a standard echocardiographic examination. This can potentially be applied to patients with CHF, with or without LV systolic dysfunction, with increased RV systolic pressure as assessed by tricuspid regurgitation peak gradient. In our opinion, measurement of SmLV and SmRV/SmLV can help identify patients with a high probability of PE in a diagnostically difficult group, thereby improving their prognosis.
In conclusion, tissue Doppler imaging of the lateral tricuspid and mitral annuli is a useful clinical tool that can help identify PE in CHF patients. Those patients who fulfill these criteria should be considered for further diagnostic studies in order to confirm PE.
Acknowledgments
The authors of this manuscript have certified that they have complied with the Principles of Ethical Publishing present in the Declaration of Helsinki and that the study protocol was approved by a local ethics committee. There are no financial or other relationship considerations that could lead to any conflict of interest.
References
- 1.Torbicki A, Perrier A, Konstantinides S, et al. Guidelines on the diagnosis and management of acute pulmonary embolism. Eur Heart J. 2008;29:2276–315. doi: 10.1093/eurheartj/ehn310. [DOI] [PubMed] [Google Scholar]
- 2.Monreal M, Munoz-Torrero JF, Naraine VS, et al. Pulmonary embolism in patients with chronic obstructive pulmonary disease or congestive heart failure. Am J Med. 2006;119:851–8. doi: 10.1016/j.amjmed.2005.11.035. [DOI] [PubMed] [Google Scholar]
- 3.Darze ES, Latado AL, Guimaraes AG, Guedes RA, Santos AB, Passos ML. Acute pulmonary embolism is an independent predictor of adverse events in severe decompensated heart failure patients. Chest. 2007;131:1838–43. doi: 10.1378/chest.06-2077. [DOI] [PubMed] [Google Scholar]
- 4.Gromadzinski L, Ciurzynski M, Januszko-Giergielewicz B, Targonski R, Cygański P, Pruszczyk P. Diagnostic value of mitral and tricuspid annular excursion in the diagnostics of acute pulmonary embolism patients with chronic heart failure. Int J Cardiol. 2011;149:118–9. doi: 10.1016/j.ijcard.2011.01.070. [DOI] [PubMed] [Google Scholar]
- 5.Sutherland GR, Steward MJ, Groundstroem WE, et al. Color Doppler myocardial imaging: a new technique for the assessment of myocardial function. J Am Soc Echocardiography. 1994;7:441–58. doi: 10.1016/s0894-7317(14)80001-1. [DOI] [PubMed] [Google Scholar]
- 6.Witte KK, Nikitin NP, de Silva R, Cleland JG, Clark AL. Exercise capacity and cardiac function assessed by tissue Doppler imaging in chronic heart failure. Heart. 2004;90:1144–50. doi: 10.1136/hrt.2003.025684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ozdemir K, Altunkeser BB, Icli A, Ozdil H, Gok H. New parameters in identification of right ventricular myocardial infarction and proximal right coronary artery lesion. Chest. 2003;124:219–26. doi: 10.1378/chest.124.1.219. [DOI] [PubMed] [Google Scholar]
- 8.Tuller D, Steiner M, Wahl A, Kabok M, Seiler C. Systolic right ventricular function assessment by pulsed wave tissue Doppler imaging of tricuspid annulus. Swiss Med Wkly. 2005;135:461–8. doi: 10.4414/smw.2005.11043. [DOI] [PubMed] [Google Scholar]
- 9.Kaul S, Tei CH, Hopkins JM, Shah PM. Assessment of right ventricular function using two-dimensional echocardiography. Am Heart J. 1984;107:526–31. doi: 10.1016/0002-8703(84)90095-4. [DOI] [PubMed] [Google Scholar]
- 10.Meluzin J, Spinarova J, Bakala J, et al. Pulsed Doppler tissue imaging of the velocity of tricuspid annular systolic motion. Eur Heart J. 2001;22:340–8. doi: 10.1053/euhj.2000.2296. [DOI] [PubMed] [Google Scholar]
- 11.Saxena N, Rajagopalan N, Edelman K, Lopez-Candales A. Tricuspid annular systolic velocity: a useful measurement in determining right ventricular systolic function regardless of pulmonary artery pressures. Echocardiography. 2006;23:750–4. doi: 10.1111/j.1540-8175.2006.00305.x. [DOI] [PubMed] [Google Scholar]
- 12.Gurudevan SV, Malouf PJ, Kahn AM, et al. Noninvasive assessment of pulmonary vascular resistance using Doppler tissue imaging of the tricuspid annulus. J Am Soc Echocardiography. 2007;20:1167–71. doi: 10.1016/j.echo.2007.02.004. [DOI] [PubMed] [Google Scholar]
- 13.Gromadziński L, Targoński R. The role of tissue colour Doppler imaging in diagnosis of segmental pulmonary embolism in congestive heart failure patients. Kardiol Pol. 2007;65:1433–9. [PubMed] [Google Scholar]
- 14.Wells PS, Anderson DR, Rodger M, et al. Derivation of a simple clinical model categorize patients probability of pulmonary embolism: increasing the models utility with the SimpliRed D-dimer. Thromb Haemost. 2000;83:416–20. [PubMed] [Google Scholar]
- 15.Kearon C, Ginsberg JS, Hirsh J. The role of ultrasonography in the diagnosis of suspected deep venous thrombolism and pulmonary embolism. Ann Intern Med. 1998;129:1044–9. doi: 10.7326/0003-4819-129-12-199812150-00009. [DOI] [PubMed] [Google Scholar]
- 16.Lang RM, Bierig M, Devereux RB, et al. Recommendations of chamber quantification: a report from the American Society of Echocardiography's Guidelines and Standards Committee and the Chamber Quantification Writing Group, developed in conjunction with the European Association of Echocardiography, a branch of the European Society of cardiology. J Am Soc Echocardiogr. 2005;18:1440–63. doi: 10.1016/j.echo.2005.10.005. [DOI] [PubMed] [Google Scholar]
- 17.Isaaz K, Thompson A, Ethevenot G, Cloez JL, Brembilla B, Pernot C. Doppler echocardiographic measurement of low velocity motion of the left ventricular posterior wall. Am J Cardiol. 1989;64:66–75. doi: 10.1016/0002-9149(89)90655-3. [DOI] [PubMed] [Google Scholar]
- 18.Miyatake K, Yamagishi M, Tanaka N, et al. New method for evaluating left ventricular wall motion by color-coded tissue Doppler imaging: in vitro and in vivo studies. J Am Coll Cardiol. 1995;25:717–24. doi: 10.1016/0735-1097(94)00421-L. [DOI] [PubMed] [Google Scholar]
- 19.Godycki-Ćwirko M, Bratkowska A. An 89-year-old patient with acquired murmur associated with pulmonary embolism. Arch Med Sci. 2011;7:902–4. doi: 10.5114/aoms.2011.25569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Derumeaux G, Ovize M, Loufoua J, et al. Doppler tissue imaging quantitates regional wall motion during myocardial ischemia and reperfusion. Circulation. 1998;97:1970–7. doi: 10.1161/01.cir.97.19.1970. [DOI] [PubMed] [Google Scholar]
- 21.Garcia-Fernandez MA, Azevedo J, Morena M, et al. Regional diastolic function in ischaemic heart disease using pulsed wave Doppler tissue imaging. Eur Heart J. 1999;20:496–505. doi: 10.1053/euhj.1998.1278. [DOI] [PubMed] [Google Scholar]
- 22.Meluzin J, Spinarova L, Dusek L, Toman J, Hude P, Krejci J. Prognostic importance of the right ventricular function assessed by Doppler tissue imaging. Eur J Echocardiography. 2003;4:262–71. doi: 10.1016/s1525-2167(02)00171-3. [DOI] [PubMed] [Google Scholar]
- 23.Mishiro Y, Oki T, Yamada H, Wakatsuki T, Ito S. Evaluation of left ventricular contraction abnormalities in patients with dilated cardiomyopathy with the use of pulsed tissue Doppler imaging. J Am Soc Echocardiography. 1999;12:913–20. doi: 10.1016/s0894-7317(99)70143-4. [DOI] [PubMed] [Google Scholar]
- 24.Oki T, Tabata T, Yamada H, et al. Clinical application of pulsed Doppler tissue imaging for assessing abnormal left ventricular relaxation. Am J Cardiol. 1997;79:921–8. doi: 10.1016/s0002-9149(97)00015-5. [DOI] [PubMed] [Google Scholar]
- 25.Severino S, Caso P, Galderisi W, et al. Use of pulsed Doppler tissue imaging to assess regional left ventricular diastolic dysfunction in hypertrophic cardiomyopathy. Am J Cardiol. 1998;82:1394–8. doi: 10.1016/s0002-9149(98)00648-1. [DOI] [PubMed] [Google Scholar]
- 26.Sohn DW, Song JM, Zo JH, et al. Mitral annulus velocity in the evaluation of left ventricalar diastolic function in atrial fibrillation. J Am Soc Echocardiography. 1999;12:927–31. doi: 10.1016/s0894-7317(99)70145-8. [DOI] [PubMed] [Google Scholar]
- 27.Kjaergaard J, Schaadt BK, Lund JO, Hassager C. Quantitative measures of right ventricular dysfunction by echocardiography in the diagnosis of acute nonmassive pulmonary embolism. J Am Soc Echocardiography. 2006;19:1264–71. doi: 10.1016/j.echo.2006.04.037. [DOI] [PubMed] [Google Scholar]
- 28.Hsiao SH, Chang SM, Lee CY, Yang SH, Lin SK, Chiou KR. Usefulness of tissue Doppler parameters for identifying pulmonary embolism in patients with sings of pulmonary hypertension. Am J Cardiol. 2006;98:685–90. doi: 10.1016/j.amjcard.2006.03.053. [DOI] [PubMed] [Google Scholar]
- 29.Hsiao SH, Lee CY, Chang SM, Yank SH, Lin SK, Huang WC. Pulmonary embolism and right heart function: insights from myocardial Doppler tissue imaging. J Am Soc Echocardiography. 2006;19:822–8. doi: 10.1016/j.echo.2006.01.011. [DOI] [PubMed] [Google Scholar]
- 30.Hsiao SH, Yang SH, Wang WC, Lee CY, Lin SK, Liu CP. Usefulness of regional myocardial performence index to diagnose pulmonary embolism in patients with echocardiographic signs of pulmonary hypertension. Am J Cardiol. 2006;98:1652–5. doi: 10.1016/j.amjcard.2006.07.051. [DOI] [PubMed] [Google Scholar]