Abstract
Circulating cells, proteins, microparticles, and DNA in cerebrospinal fluid (CSF) are excellent biomarkers of many diseases, including cancer and infections. However, the sensitivity of existing methods is limited in their ability to detect rare CSF biomarkers at the treatable, early-stage of diseases. Here, we introduce novel CSF tests based on in vivo multicolor photoacoustic flow cytometry (PAFC) and ex vivo photothermal scanning cytometry. In the CSF of tumor-bearing mice, we molecularly detected in vivo circulating tumor cells (CTCs) before the development of breast cancer brain metastasis with 20-times higher sensitivity than with current assays. For the first time, we demonstrated assessing three pathways (i.e., blood, lymphatic, and CSF) of CTC dissemination, tracking nanoparticles in CSF in vivo and their imaging ex vivo. In label-free CSF samples, we counted leukocytes, erythrocytes, melanoma cells, and bacteria and imaged intracellular cytochromes, hemoglobin, melanin, and carotenoids, respectively. Taking into account the safety of PAFC, its translation for use in humans is expected to improve disease diagnosis beyond conventional detection limits.
Keywords: cerebrospinal fluid, ultrasensitive detection in vivo, photoacoustic and photothermal methods, circulating tumor cells, nanoparticles, leukocytes, erythrocytes, bacteria
1. Introduction
Cerebrospinal fluid (CSF) of the central nervous system disseminates numerous cells, proteins, microparticles and DNA as potential biomarkers of many diseases and therapy efficacy, such as metastatic cancer (biomarker: circulating tumor cells [CTCs]), infectious meningitis and encephalitis (biomarkers: bacteria, viruses, fungus), trauma (erythrocytes) and Alzheimer’s disease (biomarker: tau-protein) [1–11]. CSF may also contain various drugs and nanoparticles as drug carriers [12–14]. Despite obvious diagnostic importance of CSF testing, currently available assays for detecting biomarkers in CSF are far from ideal. Instantly, autopsy data demonstrated that metastases of the central nervous system contribute to death in ~75% of cancer patients, but they are clinically diagnosed in only ~37% of patients [15–16]. A main drawback of existing tests is that they are performed in vitro, and their sensitivity is substantially limited by the volume of the sample [16]. Typically, 10 mL of CSF is used for sampling at the total volume of CSF of 135–150 mL in human [11, 16–17]. It means that current methods can assess only ~6–7% of CSF’s cells, and the earliest, rare circulating biomarkers (e.g., CTCs at the stage of latent metastasis) remain undetectable. Furthermore, cytology as a gold standard of CSF examination in clinics, is a relatively subjective method because its results depend on the ability of a laboratory technician to correctly identify morphological types of cells [10–11]. This may lead to diagnostic mistakes and delays in therapeutic interventions until other diagnostic criteria are determined and/or strong clinical symptoms present. More objective examination of CSF samples is achieved by flow cytometry, polymerase chain reaction (PCR) analysis and CellSearch assay (Veridex) [3, 16, 18]. However, most of these methods have higher false-negative results than cytology. For example, two of four specimens that yielded negative PCR results were determined as positive using a conventional cytology [16]. The problem can be solved by examination of a larger volume of CSF, potentially a whole CSF volume, in vivo. Despite significant progress in diagnostic techniques, no clinically relevant method has been developed for monitoring of cells (e.g., CTCs) in CSF in vivo. For example, computer tomography cannot operate at single cell level due to low spatial and temporal resolution [16, 19]. The intravital fluorescent microscopy has been applied for imaging fluid itself (so-called, cisternography), but not single biomarkers in CSF [20]. Photoacoustic flow cytometry (PAFC) based on detection of laser-induced acoustic waves, which is not sensitive to light scattering or autofluorescence, offers advanced alternatives for diagnosis diseases at single cell level in vivo including detection of CTCs and leukocytes in blood and lymph flows as well as bacteria in circulating blood [21–22]. Furthermore, this method provides higher sensitivity and resolution in deeper tissues than other optical modalities [21–23]. The diagnostic advantages of PAFC also include (1) label-free spectral and molecular identification of fast-moving cells; (2) multicolor detection; (3) use of functionalized nanoparticles as near-infrared-absorbing, low-toxicity, super-contrast photoacoustic and photothermal molecular agents; and (6) noninvasiveness for normal tissues due to the use of laser energy at levels that are safe for humans [21–24].
The goal of this study is to develop an in vivo ultrasensitive detection of cells and nanoparticles in CSF based on technical platform of PAFC. To extend diagnostic significance, PAFC was integrated with photothermal scanning cytometery/microscopy ex vivo using label-free mode or/and molecular targeting with low-toxicity bioconjugated nanoparticles.
2. Materials and methods
2.1 Multifunctional integrated technical platform
The integrated setup incorporated photoacoustic, photothermal, fluorescent and transmission microscopic and spectroscopic modules as previously described [23–26]. For analysis of photoacoustic, photothermal, fluorescent phenomena, this setup was equipped with a high-speed (200 MHz) analog-to-digital converter board (National Instruments Corp., PCI-5152, 12-bit card, 128 MB of memory), specialized software (LabVIEW; National Instruments), and a Dell Precision 690 workstation with a quadcore processor, 4 GB of RAM, and Windows Vista 64-bit operating system.
2.1.1 Multicolor PAFC
The physical mechanism of PAFC is associated with non-radiative relaxation of absorbed laser energy into heat and subsequent thermoelastic generation of sound. When laser irradiates CSF through skin, the laser-induced photoacoustic waves from individual cells can be detected with an ultrasound transducer attached to the tissue (e.g., skin or scull) over ventricles or spinal cord (Fig.1). To provide multicolor real-time detection of PA signals from different photoacoustic contrast agents (e.g., nanoparticles or CTCs targeted by functionalized nanoparticles), the cell of interest (e.g., CTCs) were irradiated by nanosecond (8–25 ns) high pulse rate (10 kHz – 100 kHz) laser pulses at different wavelengths (i.e., colors). A time delay between laser pulses (e.g., 25 µs) provided time-resolved detection of signals. In particular, to detect gold nanorods (GNRs) with absorption maximum at 670 nm and 820 nm, PAFC was equipped with lasers with the following parameters: (1) wavelength, 671 nm; pulse energy, 35 µJ; pulse width, 25 ns, and pulse rate, 10–30 kHz (QL671–500, CrystaLaser, USA); and (2) 820 nm, 75 µJ, 8 ns, and 30 kHz (LUCE 820, Bright Solutions).
Figure 1.

Integrated technical platform for testing CSF in vivo and ex vivo. (a) Schematic of multicolor PAFC with the example of real-time two-color PAFC tracings from CSF. (b) The heating stereotaxic table for in vivo PAFC and CSF sampling.
Delivery of laser radiation to the CSF was performed with a 300 µm diameter fiber with focusing optical tip. PA signals were detected with two ultrasound transducers: 1) unfocused, model 6528101, 3.5 MHz, 5.5 mm in diameter (Imasonic, Inc.); and 2) focused, model V316-SM, 20 MHz, focal length, 12.5 mm (Panametrics - NDT). Specifically, the unfocused transducer was used to assess cells in superficially located fluids (e.g., assessment of CSF after removing skin, muscles and scull) with a focused laser beam, while the focused transducer was applied to detect cells in deeply located fluids (e.g., assessment of CSF through muscles and scull) with acoustic resolution.
The warm water (or gel) was applied for the acoustic coupling between transducer and tissues. The signals after amplifier (model 5662: bandwidth, 5 MHz; gain, 54 dB; and model 5678: 40 MHz, 60 dB; both from Panametrics- NDT) were recorded with a PC and analyzed. The analysis included the identification of photoacoustic peaks with the amplitude exceeding the selected threshold, counting these peaks and calculating amplitude and width for each peak.
2.1.2 Integrated in vivo PAFC- fluorescence flow cytometry
As described previously [26], PAFC was integrated with fluorescence flow cytometer module for enumeration of blood CTCs expressing green fluorescent protein (GFP) with excitation at 488 nm and emission at 509 nm. The setup was built on the platform of Nikon Eclipse E400 microscope (Nikon Instruments Inc., USA) with a continuous wave (CW) diode laser operating at 488 nm, power in the sample ~ 1 mW (IQ1C45(488–60)G26, Power Tech., Alexander, AR). Laser beams for excitation of fluorescent signals were focused into the blood vessel by a 40× microobjective (NA 0.65; Olympus), which simultaneously collected fluorescence. Cylindrical lenses (f = 250 mm) provided the linear shape of the laser beams in the sample (10×80 µm). A dichroic mirror separated laser light and collected fluorescence detected by a photomultiplier tube (R928, Hamamatsu, Co., Bridgewater, NJ) after a bandpass filter having central wavelength of 520±15 nm (Semrock, Inc., Rochester, NY). Signals from the photomultiplier were continuously sampled at 4 MHz rate by a high-speed digitizer (PCI-5124, National Instruments, Austin, TX) and downsampled to 10 kHz rate with an average of 400 points.
2.1.3 Photothermal microscopy/cytometry ex vivo
The scanning photothermal microscope was build on the platform of an upright Olympus IX81 microscope (Olympus America, Inc., Central Valley, PA) as previously described [27]. To provide generation of photothermal phenomena, the setup was equipped with a tunable optical parametric oscillator (OPO; Opolette HR 355 LD, Opotek, Inc.) having a 5-ns wide laser pulse, a repetition rate up to 100 Hz, a wavelength range of 410–2,500 nm, and a fluence range of 1–104 mJ/cm2 on a sample surface.
In photothermal thermal-lens schematic, laser-induced temperature-dependent variation of the refractive index around absorbing zones was optically detected through defocusing of a second, probe beam. Both OPO (pump) and He-Ne (probe) collinear lasers (He-Ne: model 117A, Spectra-Physics, Inc.; wavelength, 633 nm; 1.4 mW) were focused into a sample by 100× oil-immersion microobjective (DPlan 100, NA 1.25, Olympus, Inc.) and collected after the sample by a 100× water-immersion microobjective (LUMPlanFl 100, NA 1.00, Olympus, Inc.). Probe beam after sample was focused by a 25-mm lens on a 50-µm pinhole confocal with the sample plane. Probe beam power was only 130 µW in the sample plane. Changes in probe beam intensity at the pinhole due to formation of a thermal-lens in the sample were detected by a photodetector (PDA10A, 150 MHz, Thorlabs Inc, Newton, NJ). In linear mode, photothermal signal showed a linear, positive, asymmetric component associated with fast heating (0.1–0.5 µs) and slower (0.5–1 µs) cooling effects. Images were constructed by acquiring photothermal signals from a sample as it undergoes X-Y stage scanning. The intensity of each pixel of the image represents the average of photothermal signals from several (2–4 averaging) OPO pulses. The lateral resolution of the photothermal imaging of 300 nm was determined by the focal spot size of the pump laser beam [27].
2.1.4 Optical imaging
High-resolution (~300 nm) transmission digital microscopy (TDM) module with a cooled, color CCD camera (DP72, Olympus) and high sensitive CCD (Cascade: 512; Photometrics, Roper Scientific, Inc.) camera were used for the navigation of laser beams and cell imaging.
2.1.5 Spectroscopy
The optical absorption spectra of CSF samples were acquired by Ultrospec 3300 PRO (Amersham Biosciences, Ltd., UK) spectrophotometer in a 1-cm cuvette, operating at the wavelength range of 200–1100 nm.
2.2 Nanoparticles
We explored two types of gold nanorods (GNRs, Nanopartz, Loveland, CO) with absorption maximum at 670 and 820 nm, respectively. These nanoparticles follow the main requirements (sizes less than 100 nm, PEG-coating, high absorption, low toxicity, and easy bioconjugation) for two color labeling of cells in CSF. The relatively narrow absorption spectra of GNRs (50–80 nm), compared to other nanoparticles (e.g., carbon nanotubes or magnetic nanoparticles), provided effective PA identification of two colors simultaneously. The GNRs were conjugated with folate (GNR670-Folate) and antibodies against EpCam (GNR670-EpCam) or CD45 (GNR820-CD45) receptors.
2.3 Cells
Human breast cancer cells (MDA-MB-231, American Type Culture Collection [ATCC] and MDA-MB-231-luc2-GFP Bioware® Ultra Green cell line, Caliper) and mouse melanoma cells (B16F10, American Type Culture Collection, Manassas, VA) were cultured according to the vendor’s specifications. Viable cells were resuspended in phosphate buffered saline (PBS) or in a sample of CSF.
Erythrocytes and leukocytes were prepared from the blood samples of donor mice using standard protocols. Packed cells were re-suspended in PBS or CSF.
Bacterial cells (Staphylococcus aureus [S. aureus]); strain designated as UAMS-1 and deposited with the ATCC and now available as strain ATCC 49230) were cultured according to a standardized procedure.
Each cell sample was either a subject of label-free detection (e.g., melanoma cells, WBCs, RBCs, St. aureus) or was labeled with bioconjugated GNRs (e.g., breast cancer cells and leukocytes) at different concentrations separately and in combination (30 min, 37°C, double washing). Specifically, the breast cancer cells were double labeled by cocktail of bioconjugated GNR670 (GNR670-Folate + GNR670-EpCam in equal proportions) while leukocytes were targeted by GNR820-CD45.
In ex vivo/in vitro studies, the equal aliquots (~8 µL) of the different cell samples were placed in the individual wells (S-24737, Molecular Probes) attached to the microscopic slide and were a subject of photothermal and optical microscopy.
2.4. Animal models
All animal work was done in accordance with protocols approved by the UAMS Institutional Animal Care and Use Committee. To test the PAFC of CSF in vivo, we used nude nu/nu mice weighing 20–25 g, purchased from Harlan Sprague-Dawley (Indianapolis, IN) (Fig. 1a). The animals were anesthetized by isoflurane and placed prone on a heated stereotaxic table (38°C). Using this instrument, the head was secured with the head adaptors so that the head formed approximately a 135° angle with the body. The PAFC of CSF with fiber–based delivery of laser energy was performed using two schematics: (1) through a skull and soft tissues when only small piece of skin was removed over the cisterna magna; and (2) directly through the transparent membrane which covered the cisterna magna (dura mater of the cisterna magna) after open-scull surgery. For the latter, a sagittal incision through the skin and muscles was made in the base of the skull. Pressure was applied with a gauze to control bleeding during all procedures. A pair of microretractors was used to hold the muscles apart. The dura mater of the cisterna magna appeared as a glistening and clear triangular space through which the CSF was visible to navigate the PAFC fiber.
The first set of experiments involved healthy mice after open-scull-surgery. The PAFC at 670 nm was applied to the CSF in the cisterna magna before and after injection of GNR670 (~109 nanoparticles in 2 µL of PBS per injection).
In the second set of experiments, we used the xenograft orthotopic mouse model of breast cancer. To establish primary tumors, 5×106 MDA-MB-231-luc2-GFP cells in 20 µL of PBS were inoculated into the mammary gland of each mouse. The mice were examined weekly for 10 weeks. The CTC rate in ear blood vessels with a diameter of 50–70 mm and blood velocities of 3–5 mm/s was measured noninvasively with fluorescent flow cytometry module. The growth of the primary tumor and metastases was imaged by an IVIS Spectrum imaging system (Caliper Life Sciences). At the end of week 10, the selected mice without detectable brain metastases were injected by cocktail of GNRs (GNR670-Folate + GNR670-EpCam + GNR820-CD45 in equal proportions; 5 µL per an injection) into primary tumor. Two days after injection CTCs were monitored in CSF of the cisterna magna with two-color (670 nm and 820 nm) PAFC.
Finally, the samples of blood and tissues (e.g., brain, liver, lymph nodes, and lungs) were extracted for ex vivo transmission and fluorescent (a 530/30 nm filter for GFP) optical imaging with different magnifications (objective lenses of ×20 and ×40). Multiple organs were also used for histological examination with hematoxylin and eosin (H&E) staining.
2.5 Sampling of CSF
Sprague–Dawley rats (Harlan Sprague-Dawley Indianapolis, IN) weighing 250–300 g were anesthetized by isoflurane and placed on a heated stereotaxic table (Fig. 1b). A 25-gauge needle connected through a plastic tube with a cuvette was inserted into the cisterna magna to collect ~100 µL of CSF.
2.6 Statistical analysis
Results are expressed as means plus/minus the standard error of at least three independent experiments (P <0.05). MATLAB 7.0.1 (MathWorks) were used for the statistical calculations.
3. Results
3.1 Spectroscopy of CSF
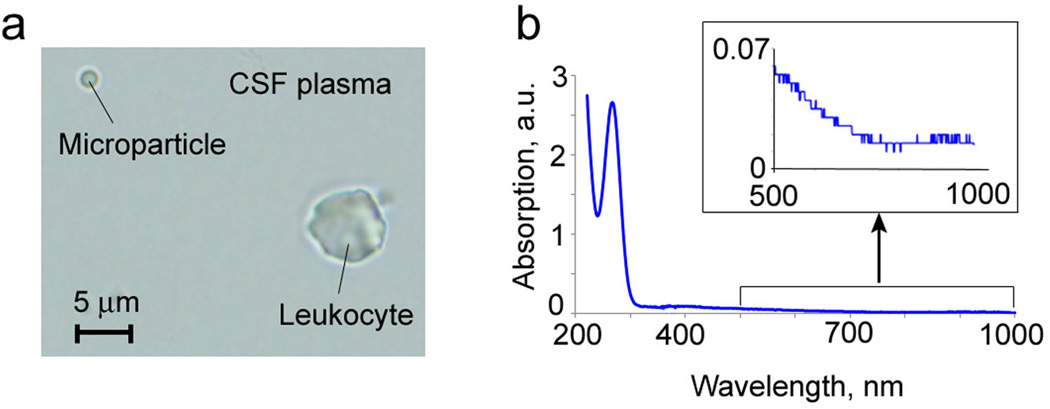
First, we tested the optical properties of CSF from healthy animals using conventional spectroscopy in vitro (Fig. 2). To obtain optical spectrum of CSF, measurements were conducted in increments of Δλ=1 nm. The absorption of CSF was normalized to the absorption of saline (PBS) solution. As expected, the optical properties of CSF were similar to the blood plasma and, mainly, provided by proteins diluted in CSF serum. Specifically, the absorption spectrum was relatively high in ultraviolet range (200–250 nm) and dramatically dropped (~15–20 times) to 300 nm. Then the absorption coefficient of CSF slowly and monotonically decreased in the wavelength’s range from 300–400 to 700 nm. Above 700 nm, the absorption coefficient was minimal and was not significantly changed. Thus, absorption of nanoparticles (e.g., GNRs) and cells with high-absorbing molecules (e.g., hemoglobin, melanin, caratenoid) [22, 24, 28] predominated over the absorption of CSF by a few orders of magnitude (e.g., three orders of magnitude for hemoglobin and one order of magnitude for caratenoids) especially in near-infrared range (NIR) of 700–1,000 nm.
Figure 2.
Ex vivo examination of normal CSF collected from healthy animals. (a) High resolution optical imaging; (b) optical spectroscopy.
The obtained low absorption background of CSF together with the well-known high absorption sensitivity of photoacoustic and photothermal methods encouraged us to explore various cells without any labeling, based on their natural absorption.
3.2 Ex vivo label-free photothermal imaging cells in CSF
Taking into account the absorption maximums of the hemoglobin in erythrocytes, cytochromes in leukocytes and staphyloxanthin (the type of carotenoids) in S. aureus [28] the photothermal imaging was performed at 530 nm, 550 nm and at 710 nm, respectively. In case of melanoma cells, the intrinsic melanin has a broad absorption spectrum, with slight decreases as the wavelength increases [24]. Therefore, any wavelength within the range of >400 nm was appropriate for photothermal imaging of melanoma cells.
First, we compared the high-resolution color-coded photothermal images of PBS, normal CSF, and CSF with S.aureus (imaging area of 15 µm × 15 µm, sample thickness of 120 µm). We used the laser wavelength of 710 nm corresponding to the maximum NIR absorption of staphyloxanthin in bacteria. The image of PBS showed almost no photothermal signals (the black square in Fig. 3a). The normal CSF without cells in the field of view represented blue spotty image associated with low-amplitude randomly distributed signals (Fig. 3b). In contrast, the presence of bacteria in CSF led to the appearance of the multicolor bright spots (Fig. 3c). The association of these spots with bacteria was verified by high-resolution optical imaging (Fig. 3c, insert). The single bacteria were distinguished from their clusters by spot’s size. The amount of staphyloxanthin in a single bacteria was estimated by the color (blue color is minimal, red color is maximum) and amplitude of the peak in 3D images (Fig. 3c).
Figure 3.

Detection and enumeration of S. aureus in CSF sample (15 µm × 15 µm × 120µm) using photothermal scanning cytometry. (a) PBS; (b) normal CSF; and (c) CSF with bacteria; high-resolution optical imaging in the insert (×100, oil immersion) confirmed the signals related to single bacteria. Laser wavelength and energy fluence are 710 nm and 100 mJ/cm2, respectively.
The same experimental design was used to detect and count leukocytes, erythrocytes and melanoma cells in CSF samples. Furthermore, the photothermal imaging revealed the distribution of the intracellular biomarkers (Fig. 4, right column) such as cytochrome (Fig 4a) and melanin (Fig 4c) clusters in the cytoplasm of leukocytes and melanoma cells, respectively. Melanin was detectable at a significantly lower laser energy than cytochromes, which can be used to distinguish melanin from cytochromes [22, 24]. In particular, the imaging of even low pigmented melanoma cells (so-called amelanotic and transparent in optical images) required four-times less laser energy in visible spectral range than it was used for imaging of cytochromes (Fig 4c). As expected, the images of high-absorbing erythrocytes’ hemoglobin which filled the entire cell volume were obtained at still lower laser energy and replicated the shape of the erythrocytes (e.g., biconcave in Fig. 4b). Notably, despite wide range of used laser energy, all types of detected cells did not demonstrate visible changes of their original shape and intracellular structure.
Figure 4.
Label-free optical and photothermal images of the leukocyte (a), erythrocyte (b) and melanoma cell (c). Laser wavelength is 530 nm; laser energy fluence is 400, 50, and 100 mJ/cm2, respectively.
3.4 Photothermal imaging nanoparticles in CSF
As a challenging test case with the nano-scale object of interest, we imaged free unbound GNR670 in live CSF specimen. Because these nanoparticles have absorption maximum at 670 nm and relatively high absorption in visible spectral range, photothermal microscopy of GNR670 can be performed at 670 nm or 520–550 nm. We obtained the images like “starred night sky” where the small blue spots (low-amplitude photothermal signals) were associated with single nanoparticles or their clusters randomly distributed in CSF’s plasma (Fig 5a, 530 nm).
Figure 5.

Detection of GNRs in CSF. (a) Photothermal images of GNRs color-coded by blue in CSF plasma (black). (b) Optical (left) and photothermal (right) images of the CSF sample with breast cancer cell labeled with GNRs. Laser wavelength and fluence are 530 nm and 50 mJ/cm2, respectively
3.5 Photothermal imaging and cytometry of breast cancer cells labeled by bioconjugated GNRs
Photothermal and photoacoustic methods are very similar in basic physical processes supplement each other in applications with a with a sensitivity advantage of photothermal techniques (up to 10–50-fold) in vitro at a sub-cellular level, while photoacoustic methods allow detection of individual cells in vivo in deep tissue. Therefore, we applied photothermal microscopy ex vivo to verify photoacoustic data of CTC diagnosis in CSF in vivo. For detection of labeled breast cancer cells we used laser energies that were several times below the detection thresholds for label-free breast cancer cells (control sample). Labeling specificity was provided through bioconjugation of GNR670 with molecular markers (e.g., antibodies, folate) against the surface receptors (e.g., EpCam and folic receptors) which are highly expressed in breast cancer cells, but almost absent in normal CSF cells (mainly, leukocytes). Labeling efficacy of breast cancer cells, which typically exhibit heterogeneous expression of receptors [29–31], was achieved using multiple-targeting strategy, as we demonstrated previously [23]. Specifically, the selection of two markers, Folate (with GNR670) and EpCam (GNR670) instead one, to label breast cancer cells allowed us to increase labeling efficacy to 99.7%.
We also explored the dependence of labeling efficiency from nanoparticle concentration. Breast cancer cells were labeled by GNR670-Folate at the concentrations ranging from 500 to 50,000 nanoparticles per a cell. Surprisingly, even 500 GNR670-Folate per a cell was enough for effective photothermal detection of 30–40 % cells. At the ~5,000 nanoparticles per a cell, the most cells (~92%) provided photothermal signals. The obtained results represented a good agreement with data from conventional flow cytometry (90–95% for folate). Increasing the number of GNRs to ~50,000 per a cell at even, decreasing laser energy led to a non-significant increased percentage of detected Folate+ cells to 97.5%. However, we observed simultaneously, false signals from the surrounding solution due to the likely presence of not perfectly washed NPs. Thus, the concentration of ~5,000 GNRs per cell was selected as the optimal for labeling breast cancer cells, and was used in experiments in vivo.
Finally, we obtained high-resolution photothermal images from labeled breast cancer cells spiked into the CSF. The images of GNRs molecularly attached to the cell membrane (Fig 5b) differed from the images of unbound GNRs (Fig 5a): the attached nanoparticles provided a 10–50-fold higher photothermal signal amplitude than the unbound ones. It can be explained by nanoparticle clustering around naturally densely packed cell markers leading to nonlinear signal amplification due to more effective laser-induced formation of nanobubble (as a signal amplifier) [32] around overheated nanoparticle clusters with enhanced local absorption. These phenomena may serve as the indicator of successful molecular targeting of cells.
3.6 Real-time photoacoustic monitoring of GNRs in circulating CSF in vivo
The PAFC was applied to monitor GNRs in CSF of cisterna magna that represents a small brain’s bath of 3–7 µL for mice (5 µL in average) with continuously renewed CSF. Typically, CSF produces at the rate of 0.33 µL/min for mice (800 µL/min for human) [11]. Based on these considerations, the concentration of injected NPs in cisterna magna suggested to be reduced in half during ~4.5–10 min and should be near zero ~9–20 min after.
To be absolutely sure of nanoparticle detection in CSF, we used the open-scull–surgery strategy when the fiber’s tip was directly attached to the membrane covered by the cisterna magna. Nevertheless, the PAFC could be still considered as was noninvasive tool for assessment of CSF. This allowed us to monitor natural kinetic of GNRs in circulating CSF. The CSF was monitored by PAFC at 670 nm before and after injection of GNR670 in the cisterna magna. We used laser pulse energy which provided a small background (signal-to-noise ratio of ~2–3) from the CSF before injection. The GNR670 injection enhanced photoacoustic signal amplitude in 20–30 times (pattern I in Fig. 6). Then the amplitude gradually decreased two times in one minute (pattern II in Fig. 6) and fifteen times in seven minutes (pattern III in Fig. 6). However, the photoacoustic signals were still readable above the CSF background. Thus, the experimentally obtained kinetics of GNRs was faster than theoretically estimated that requires further detailed studies to optimize nanoparticle delivery to the central nervous system.
Figure 6.
In vivo real-time tracing of CSF after injection of GNRs670 in cisterna magna. Patterns I, II and III are related to the different time points at the same position of laser beam over CSF. Laser wavelength is 670 nm; laser energy fluence is 30 mJ/cm2.
3.7 PAFC of CTCs in cerebrospinal fluid of breast-cancer-bearing mouse model
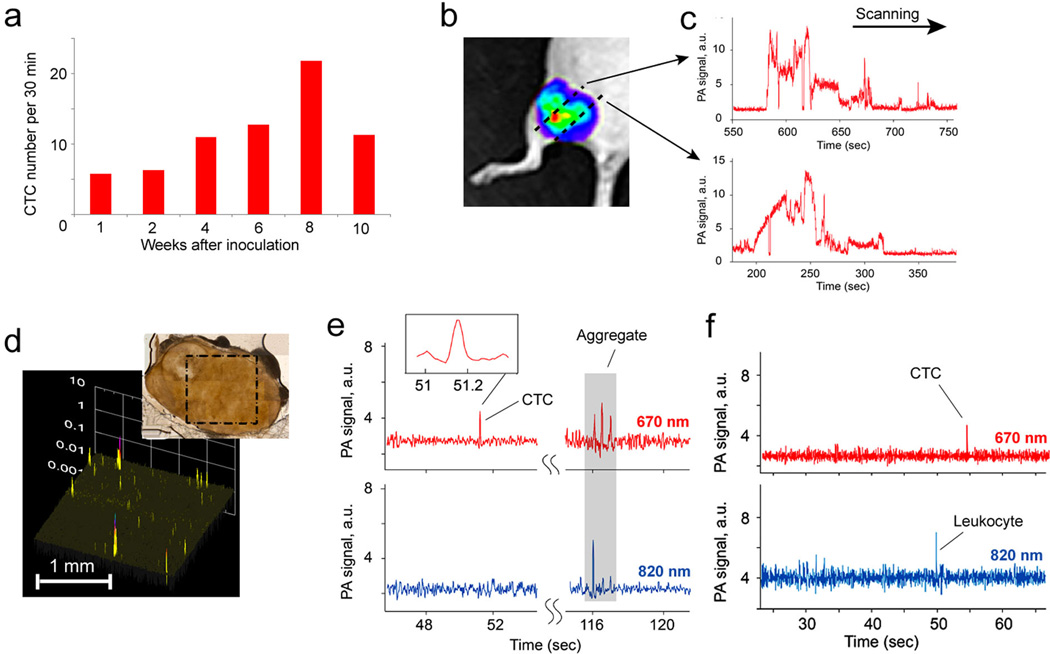
To test capability of PAFC to target real CTCs, we used orthotopic xenograft mouse model of human metastatic breast cancer. This model was characterized by the early production of blood CTCs (Fig 7a) and the development of multiple distant metastases over a period of 9–11 weeks. Because metastasis in the central nervous system is the late event in the breast cancer development [1–2], PAFC of CSF was performed at week 10 after tumor inoculation. To detect CTCs in CSF, we used the double-targeting strategy with GNR670-Folate and GNR670-EpCam which was optimized in vitro with photothermal microscopy (see above). To increase PAFC’s specificity and reduce the probability for false-positive signals, we added GNR820 bioconjugated with antibodies to CD45. This receptor was used as a common marker of leukocytes to distinguish CTCs (CD45−) from leukocytes (CD45+) [3, 30, 33] that may non-specifically uptake GNRs. Cells with profiles of Folate+/CD45−, EpCam+/CD45− and Folate+/EpCam+/CD45− were defined as CTCs.
Figure 7.
In vivo photoacoustic detection and counting of CTCs in CSF of tumor-bearing mice. (a) Dynamic of CTCs in blood circulation during tumor development measured by fluorescent flow cytometry. (b) Intravital whole-body imaging of primary tumor at week 10 after tumor inoculation. Black dashed lines indicate two photoacoustic scans (670 nm; 30 mJ/cm2) presented in (c). (d) Photoacoustic scanning of the sentinel lymph node; high-amplitude signals color-coded by yellow and red are associated with objects ≤15 µm. (e) two-color (670 nm and 820 nm) PAFC of CSF in vivo through scull. (f) In vivo two-color PAFC of CSF in cisterna magna after scull removing.
The CTCs in CSF were detected by two-color PAFC at 670 nm and 820 nm two days after injection of GNR cocktail into a primary tumor. The effective labeling was confirmed by photoacoustic scanning of the primary tumor (Fig 7c), which was identified by the bright spot in the fluorescent image (Fig 7b). Specifically, the tumor with GNR670 produced 5–10 times higher signal amplitudes at 670 nm than unlabeled tissues, even with well-vascularized skin.
The presence of labeled cells in the sentinel lymph nodes (Fig 7d) indicated, first, on the primary tumor ability to shed cells into circulation; and, second, on the participation of lymphatic system in dissemination of late breast CTCs. Further PAFC monitoring of the cisterna magna through the scull and muscles (just the skin was removed) revealed rare high-amplitude photoacoustic signals at 670 nm (Fig 7e) at the rate of ~1 signals every 20–30 min. The signal’s width [34] allowed us to associate these signals with the labeled CTCs.
Considering a cisterna magna volume turnover time of about 20 minutes (up to 7µL at the production rate of ~0.33 µL/min [11]), the production rate of CTCs in CSF was calculated as 0–1 CTCs/20 min. The total CTC amount was estimated at 4–6 CTCs in the entire CSF volume (~40 µL for a mouse [11]). The results were reproduced after removing the scull and the muscles over the detection area when the fiber was attached directly to the cisterna magna (Fig. 7f).
The examination of brain specimens ex vivo and in vitro by high-resolution optical microscopy and histology revealed that more than half mice had not brain’s micro- and macro-metastases as a potential source of CTCs while all mice had blood CTCs at the average rate of 8–12 CTCs per 20 min. Thus, pre-metastatic early CTCs in CSF could be originated from blood CTCs.
Finally, two findings were unexpected. First, PAFC detected rare, high amplitude signals at 820 nm (Fig. 7f) which were associated with leukocytes. Second, we monitored a few, high-amplitude signals together at 670 nm which sometimes coincided with high-amplitude signals at 820 nm (Fig. 7e, grey rectangular). Based on our previous experience [22], these combined signals were associated with circulating aggregates of CTCs and leukocytes.
4. Discussion
In this study we introduced in vivo molecular testing of CSF using multicolor PAFC. We demonstrated the capability of PAFC to significantly (at least 10–20-fold increase in sensitivity) overcome the detection limit of conventional CSF methods by monitoring nearly the entire CSF volume (vs. 6–7% in conventional tests). This is feasible for both, animals and humans. For example, PAFC of the entire CSF volume in a mouse can be performed in approximately two hours (a total volume of ~40 µL at the production rate of ~0.33 µL/min [11] has a total volume turnover time of about 2 hours). Beyond existing CSF tests, our approach can also estimate the real-time production of CTCs in CSF and can provide dynamic quantitative detection and tracking of CSF’s cells in the natural environment of the central nervous system.
Another advantage of this method includes a combination of high sensitivity with high specificity for the diagnosis of cells with specific molecular profiles (e.g., Folate+, EpCam+ and CD45− CTCs). We demonstrated that low absorbing cells of interest (e.g., breast cancer cells) molecularly labeled by strongly absorbing GNRs can be detected in CSF with PAFC at low (safe for humans [35]) laser energy. The narrow absorption spectra of GNRs make them a superior nanoprobe for a multicolor-multiple targeting strategy, with the possibility of using their extremely low concentration in vivo (in the picomolar, and potentially femtomolar, range; i.e., at least 5–7 orders of magnitude lower than the concentration of conventional agents) [22, 36], and ultrasharp nonlinear nanobubble-amplified phototothermal and photoacoustic plasmonic resonances to enhance spectral specificity [37].
In cancer research, overcoming technical limitations of CSF tests may open new avenues to expand the knowledge about the role of CTCs in the development of metastasis. Instantly, we have found that early rare CTCs enter CSF before developing of detectable brain metastasis. The integrative assessment of CTCs in CSF and blood allowed us to suggest that these early CTCs in the CSF are the result of their migration from blood circulation.
In addition, the detection of CTCs in blood, CSF and sentinel lymph nodes at the same time point of tumor development indicated that CTCs may disseminate by three body fluids – blood, lymph and CSF – simultaneously. These results are consistent with the well-known close interrelationships between these three systems in a whole organism. Notably, integration of blood, lymph, and CSF testing is unachievable with other existing methods.
Furthermore, finding aggregates of CTCs and leukocytes in the CSF suggested that CSF’s CTCs, similar to the CTCs in blood [38], may bring cells from the primary tumor stroma (e.g., tumor-associated macrophages and fibroblasts) and co-disseminate with them.
To extend diagnostic capability of our approach, we developed photothermal cytometry of CSF samples ex vivo, which has absorption sensitivity several orders of magnitude higher than conventional absorption/transmission spectroscopy. The capability of our platform to combine data on photothermal imaging of nanoparticles in SCF ex vivo with PAFC’s results about their kinetics in vivo holds promise to characterize biological processes related to nanoparticles in the central nervous system and has a great potential to improve drug delivery using advanced nanoparticles as drug carriers.
In label-free mode, photothermal scanning cytometry was capable to identify and count leukocytes, erythrocytes, bacteria (S. aureus) and melanoma cells. Moreover, imaging of intracellular cytochromes, hemoglobin, carotenoids and melanin, and potential to count their molecules (e.g., cytochrome at concentration up to 5 × 10−9 mol/L or 80 attomols in the signal-generation zone [39]) can improve disease’s diagnosis through advanced detection of a virulent factor (e.g., staphyloxanthin of S. aureus), a marker of apoptosis (e.g., cytochrome c in leukocytes) or metastatic activity (e.g., melanin in melanoma cells) [24, 40–42]. Furthermore, the targeted pigment neutralization (e.g., photothermal destroying of melanosomes) may represent a new advanced therapeutic concept.
Prospectively the integrated photoacoustic and photothermal platform can be easily developed for detection of any cells, microparticles, or biomolecules of interest in the CSF for which labeling agents or antibodies are available, as well as unlabeled cells that have intrinsic detectable markers (e.g., melanoma CTCs). Thus, the presented technical platform has a great potential to solve many clinical tasks in a wide spectrum of diseases. The future clinical applications include, but are not limited to, detection of viruses or fungus at early sign of infectious meningitis; tau protein, as a biomarker of Alzheimer’s disease; apoptotic cells of different types of inflammatory and non-inflammatory neurological diseases; normal and cancer stem cells.
The simplicity and clinical relevance of the PAFC give it the potential to be a novel, valuable diagnostic tool. We expect that our proposed novel diagnostic platform using safe laser parameters can be quickly translated to humans with the assessment of individual CTCs at least at 1 cm depth with potential for further depth improvement [24, 43–44]). The clinical prototype of PAFC could represent a portable, fiber-based optical device that overlies the spinal canal (e.g., typical depth of CSF in human spinal canal is ~1–3 cm in L4-5 or L3-4 interspaces), ventricular sites or arachnoid space [45]. We predict that the NIR range of wavelengths of 800–1,100 nm (e.g., 820 or 1064 nm) will be close to optimal because background from the skin is minimal and background from SCF plasma associated with water absorption is relatively low. In PAFC as a combination of laser and ultrasound methods, resolution in superficial spinal canal on small animal models (e.g., mouse), was determined by optical resolution (10 µm); in the deeper human SCF, it will be determined by ultrasound resolution (20–40 µm) with a focused cylindrical high frequency (20–70-MHz) ultrasound transducer. The cylindrical configuration of optical and ultrasound beams provides a minimal detection volume (due to high lateral resolution) with simultaneous assessment of an entire cross-section of the spinal canal (~15 mm for adults). From these calculations and from anatomo-physiological features of the CSF, the detected volume should contain ~1 rare abnormal cell. Thus, such approach may permit the detection and counting of rare single cells in human CSF.
Acknowledgement
This work was supported in part by the National Institute of Health grants R01 CA131164, R01 EB0005123, the Department of Defense grant W81XWH-11-1-0129 and the grant from the Arkansas Breast Cancer Research Program and the University of Arkansas for Medical Sciences Translational Research Institute (CTSA Grant Award # UL1TR000039). We thank L.J. Henning for histological examination, S. Foster and J. Ye for assistance with breast cancer cell culturing and imaging; and K. Beenken for culturing and preparation of bacteria.
REFERENCES
- 1.Gleissner B, Chamberlain MC. Neoplastic meningitis. Lancet Neurol. 2006;5:443–452. doi: 10.1016/S1474-4422(06)70443-4. [DOI] [PubMed] [Google Scholar]
- 2.Langley RR, Fidler IJ. Clin Chem. 2013;59:180–189. doi: 10.1373/clinchem.2012.193342. [DOI] [PubMed] [Google Scholar]
- 3.Patel AS, Allen JE, Dicker DT, Peters KL, Sheehan JM, Glantz MJ, El-Deiry WS. Oncotarget. 2011;2:752–760. doi: 10.18632/oncotarget.336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Burns TF, Wolff AC. Cell Cycle. 2012;11:203–204. doi: 10.4161/cc.11.2.18780. [DOI] [PubMed] [Google Scholar]
- 5.Chamberlain MC. Handb Clin Neurol. 2012;105:757–766. doi: 10.1016/B978-0-444-53502-3.00022-7. [DOI] [PubMed] [Google Scholar]
- 6.Ray P, Badarou-Acossi G, Viallon A, Boutoille D, Arthaud M, Trystram D, Riou B. Am J Emerg Med. 2007;25:179–84. doi: 10.1016/j.ajem.2006.07.012. [DOI] [PubMed] [Google Scholar]
- 7.Groves MD, Hess KR, Puduvalli VK, Colman H, Conrad CA, Gilbert MR, Weinberg J, Cristofanilli M, Yung WK, Liu TJ. J Neurooncol. 2009;94:229–234. doi: 10.1007/s11060-009-9819-2. [DOI] [PubMed] [Google Scholar]
- 8.Goertsches R, Knappe S, Mix E, Zettl UK. J Neuroimmunol. 2007;188:175–180. doi: 10.1016/j.jneuroim.2007.06.004. [DOI] [PubMed] [Google Scholar]
- 9.Formichi P, Battisti C, Radi E, Federico A. J Cell Physiol. 2006;208:39–46. doi: 10.1002/jcp.20602. [DOI] [PubMed] [Google Scholar]
- 10.Lee MC, Heaney LM, Jacobson RL, Klassen AC. Stroke. 1975;6:638–641. doi: 10.1161/01.str.6.6.638. [DOI] [PubMed] [Google Scholar]
- 11.Johanson CE, Duncan 3rd JA, Klinge PM, Brinker T, Stopa EG, Silverberg GD. Cerebrospinal Fluid Res. 2008;5:10. doi: 10.1186/1743-8454-5-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bhaskar S, Tian F, Stoeger T, Kreyling W, de la Fuente JM, Grazú V, Borm P, Estrada G, Ntziachristos V, Razansky D. Part Fibre Toxicol. 2010;7:3–25. doi: 10.1186/1743-8977-7-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Silva GA. BMC Neurosci. 2008;10(Suppl 3):S4. doi: 10.1186/1471-2202-9-S3-S4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wankhede M, Bouras A, Kaluzova M, Hadjipanayis CG. Expert Rev Clin Pharmacol. 2012;5:173–186. doi: 10.1586/ecp.12.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fidler IJ, Schackert G, Zhang RD, Radinsky R, Fujimaki T. Cancer Metastasis Rev. 1999;18:387–400. doi: 10.1023/a:1006329410433. [DOI] [PubMed] [Google Scholar]
- 16.Chamberlain MC, Glantz M, Groves MD, Wilson WH. Semin Oncol. 2009;36:S35–S45. doi: 10.1053/j.seminoncol.2009.05.005. [DOI] [PubMed] [Google Scholar]
- 17.Sweetman B, Linninger AA. Ann Biomed Eng. 2011;39:484–496. doi: 10.1007/s10439-010-0141-0. [DOI] [PubMed] [Google Scholar]
- 18.Hoon DS, Kuo CT, Wascher RA, Fournier P, Wang HJ, O'Day SJ. J Invest Dermatol. 2001;117:375–378. doi: 10.1046/j.0022-202x.2001.01417.x. [DOI] [PubMed] [Google Scholar]
- 19.Barajas R, Jr, Cha S. Prog Neurol Surg. 2012;25:55–73. doi: 10.1159/000331174. [DOI] [PubMed] [Google Scholar]
- 20.Shibata Y, Kruskal JB, Palmer MR. Neurol Sci. 2007;28:87–92. doi: 10.1007/s10072-007-0792-2. [DOI] [PubMed] [Google Scholar]
- 21.Zharov VP, Galanzha EI, Tuchin VV. J. Cell Biochem. 2006;97:916–932. doi: 10.1002/jcb.20766. [DOI] [PubMed] [Google Scholar]
- 22.Kim J-W, Galanzha EI, Zaharoff DA, Griffin RJ, Zharov VP. Mol Pharm. 2013;4:813–830. doi: 10.1021/mp300577s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Galanzha EI, Shashkov EV, Kelly T, Kim J-W, Yang L, Zharov VP. Nature of Nanotechnology. 2009;4:855–860. doi: 10.1038/nnano.2009.333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Galanzha EI, Shashkov EV, Spring P, Suen JY, Zharov VP. Cancer Res. 2009;69:7926–7934. doi: 10.1158/0008-5472.CAN-08-4900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nedosekin DA, Sarimollaoglu M, Shashkov EV, Galanzha EI, Zharov VP. Opt. Exp. 2010;18:8605–8620. doi: 10.1364/OE.18.008605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Nedosekin DA, Sarimollaoglu M, Galanzha EI, Sawant R, Torchilin VP, Verkhusha VV, Ma J, Frank MH, Biris AS, Zharov VP. J Biophotonics. 2012 Aug 20; doi: 10.1002/jbio.201200047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Nedosekin DA, Galanzha EI, Ayyadevara S, Shmookler Reis RJ, Zharov VP. Biophys J. 2012;102:672–681. doi: 10.1016/j.bpj.2011.12.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Galanzha EI, Shashkov E, Sarimollaoglu M, Beenken KE, Basnakian AG, Shirtliff ME, Kim JW, Smeltzer MS, Zharov VP. PLoS One. 2012;7:e45557. doi: 10.1371/journal.pone.0045557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Stingl J, Caldas C. Nat Rev Cancer. 2007;7:791–799. doi: 10.1038/nrc2212. [DOI] [PubMed] [Google Scholar]
- 30.Attard G, de Bono JS. Curr Opin Genet Dev. 2011;21:50–58. doi: 10.1016/j.gde.2010.10.010. [DOI] [PubMed] [Google Scholar]
- 31.Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature. 2012;490:61–70. doi: 10.1038/nature11412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kim J-W, Galanzha EI, Shashkov EV, Moon H-M, Zharov VP. Nature of Nanotechnology. 2009;4:688–694. doi: 10.1038/nnano.2009.231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hayes DF, Smerage JB. Prog Mol Biol Transl Sci. 2010;95:95–112. doi: 10.1016/B978-0-12-385071-3.00005-8. [DOI] [PubMed] [Google Scholar]
- 34.Sarimollaoglu M, Nedosekin DA, Simanovsky Y, Galanzha EI, Zharov VP. Opt Lett. 2011;36:4086–4088. doi: 10.1364/OL.36.004086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.American National Standard for Safe Use of Lasers. ANSI Z136.1. 2003 [Google Scholar]
- 36.He W, Wang H, Hartmann LC, Cheng JX, Low PS. Proc Natl Acad Sci U S A. 2007;104:11760–11765. doi: 10.1073/pnas.0703875104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zharov VP. Nat Photon. 2011;5:110–116. doi: 10.1038/nphoton.2010.280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Duda DG, Duyverman AM, Kohno M, Snuderl M, Steller EJ, Fukumura D, Jain RK. Proc Natl Acad Sci U S A. 2010;107:21677–21682. doi: 10.1073/pnas.1016234107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Brusnichkin AV, Nedosekin DA, Galanzha EI, Vladimirov YA, Shevtsova EF, Proskurnin MA, Zharov VP. J Biophotonics. 2010;3:791–806. doi: 10.1002/jbio.201000012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Tsukamoto K, Gersten DM, Law LW, Hearing VJ. Melanoma Res. 1991;1:223–230. [PubMed] [Google Scholar]
- 41.Caroppi P, Sinibaldi F, Fiorucci L, Santucci R. Curr Med Chem. 2009;16:4058–4065. doi: 10.2174/092986709789378206. [DOI] [PubMed] [Google Scholar]
- 42.Liu GY, Nizet V. Trends Microbiol. 2009;17:406–413. doi: 10.1016/j.tim.2009.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wang LV, Hu S. Science. 2012;335:1458–1462. doi: 10.1126/science.1216210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Razansky D, Distel M, Vinegoni C, Ma R, Perrimon N, Koster RW, Ntziachristos V. Nat Photon. 2009;3:412–417. [Google Scholar]
- 45.Boon JM, Abrahams PH, Meiring JH, Welch T. Clin Anat. 2004;17:544–553. doi: 10.1002/ca.10250. [DOI] [PubMed] [Google Scholar]