Abstract
Rationale
Following cardiac injury, cardiac progenitor cells are acutely reduced, and are replenished in part by regulated self-renewal and proliferation, which occurs through symmetric and asymmetric cellular division. Understanding the molecular cues controlling progenitor cell self-renewal and lineage commitment is critical towards harnessing these cells for therapeutic regeneration. We have previously found that the cell surface ATP binding cassette (ABC)-transporter, Abcg2, influences the proliferation of cardiac side population (CSP) progenitor cells, though through unclear mechanisms.
Objective
To determine the role of Abcg2 on cell cycle progression and mode of division in mouse CSP cells.
Methods and Results
Herein, using CSP cells isolated from wild-type and Abcg2-knockout mice, we find that Abcg2 regulates G1-S cell cycle transition by FUCCI cell cycle indicators, cell cycle-focused gene expression arrays and confocal live cell fluorescent microscopy. Moreover, we find that modulation of cell cycle results in transition from symmetric to asymmetric cellular division in CSP cells lacking Abcg2.
Conclusions
Abcg2 modulates CSP cell cycle progression and asymmetric cell division, establishing a mechanistic link between this surface transporter and cardiac progenitor cell function. Greater understanding of progenitor cell biology, and in particular the regulation of resident progenitor cell homeostasis, is vital for guiding the future development of cell-based therapies for cardiac regeneration.
Keywords: ABC transporter, cardiac side population cells, asymmetric division, adult stem cell, cell cycle
INTRODUCTION
Adult cardiac stem cells have recently been introduced in the treatment of cardiovascular diseases with encouraging results.1 During the course of tissue repair, stem/progenitor cells self-renew to expand their pool, and differentiate to create a specialized progeny. Stem/progenitor cells modulate their cell fate decision through their modality of replication, since they can divide symmetrically and asymmetrically. With symmetric stem cell division, two identical daughter cells are formed that retain both stem cell properties or become both early committed cells. With asymmetric stem cell division, two daughter cells with divergent fate are generated, one capable of self-renewal and the other committed to differentiation.2 While during physiological tissue homeostasis progenitor cells divide asymmetrically, these progenitor cells revert to symmetric division and rapid proliferation following tissue injury, a pattern of cell growth postulated for the damaged human heart and in neuronal progenitor cells.3-5 Similarly, following cardiac injury, we have previously found that endogenous cardiac progenitor cell populations are acutely reduced, and are replenished within days by self-renewal and proliferation.6 The molecular cues that dictate the mechanisms of stem cell division, however, remain unclear.
The cell surface ABC-transporter, Abcg2, is highly expressed in several reported stem/progenitor cell populations and is responsible for the DNA-binding dye extrusion that marks the population phenotype of cardiac side population (CSP) progenitor cells.7, 8 Moreover, we have found that expression of Abcg2 may influence CSP proliferation.9 Herein, we provide evidence demonstrating that Abcg2 directly regulates cell cycle progression in CSP cells with loss of Abcg2 resulting in delayed G1-S transition. Additionally, Abcg2 regulates in CSP cells the switch between symmetric and asymmetric cell division, determining progenitor cell fate decisions. These findings establish that the Abcg2 transporter is a critical determinant of cardiac progenitor cell function and may be essential for cardiac regeneration.
METHODS
Cell cycle analysis using the Fluorescence Ubiquitination Cell Cycle Indicator (FUCCI) lentiviral system
FUCCI-expressing CSP cells were synchronized in G1 and analyzed by flow cytometry or confocal live-cell imaging for up to 40hrs following synchronization for the expression of Cdt1 and geminin.
An expanded Methods section describing all procedures and protocols is available in the Online Data Supplement at http://circres.ahajournals.org.
RESULTS
Abcg2 controls cell cycle progression of CSP cells
To dissect the role that Abcg2 has in the growth of CSP cells, the progression of the cell cycle was measured in CSP cells isolated from transgenic mice lacking Abcg2 (Abcg2-KO) and wild-type mice (WT). Deletion of Abcg2 was associated with a reduction in the number of CSP cells in S and G2/M phases of the cell cycle, while the fraction of cells in G0/G1 increased (Figure 1A-C). To characterize the length of the cell cycle in WT CSP cells and Abcg2-KO CSP cells, a lentivirus-based fluorescent ubiquitination-dependent cell cycle indicator (FUCCI) was utilized. Flow cytometric analysis of pre-sorted FUCCI-expressing WT and Abcg2-KO CSP cells 24 hours post-synchronization revealed striking differences in the distribution of cells within the various phases of the cell cycle. WT CSP cells were actively cycling with the vast majority of cells residing within the G1-S transition. In contrast, most Abcg2-KO CSP cells were in G1 and only a small fraction was in G1-S or S-G2-M phases (Figure 2A and B). Greater than 50% of Abcg2-KO CSP cells were in early G1.
Figure 1. Abcg2-KO CSP cells exhibit altered cell cycle profile.
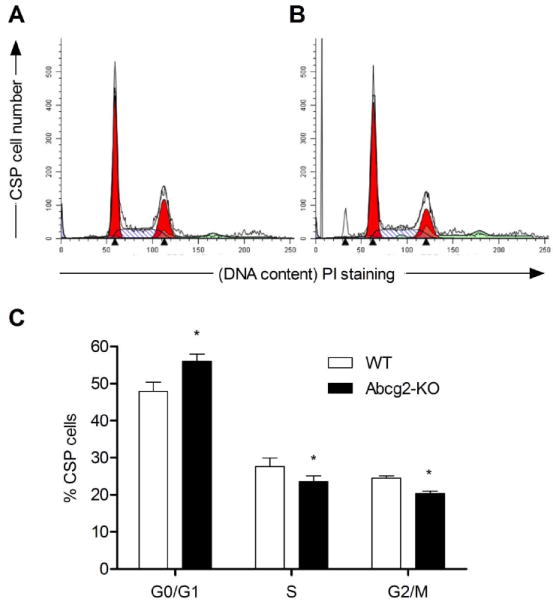
Representative flow cytometric analysis of (A) WT and (B) Abcg2-KO CSP cells stained with PI. (C) Quantification of propidium iodide (PI) staining.
Figure 2. Lack of Abcg2 prolongs cell cycle duration in CSP cells.
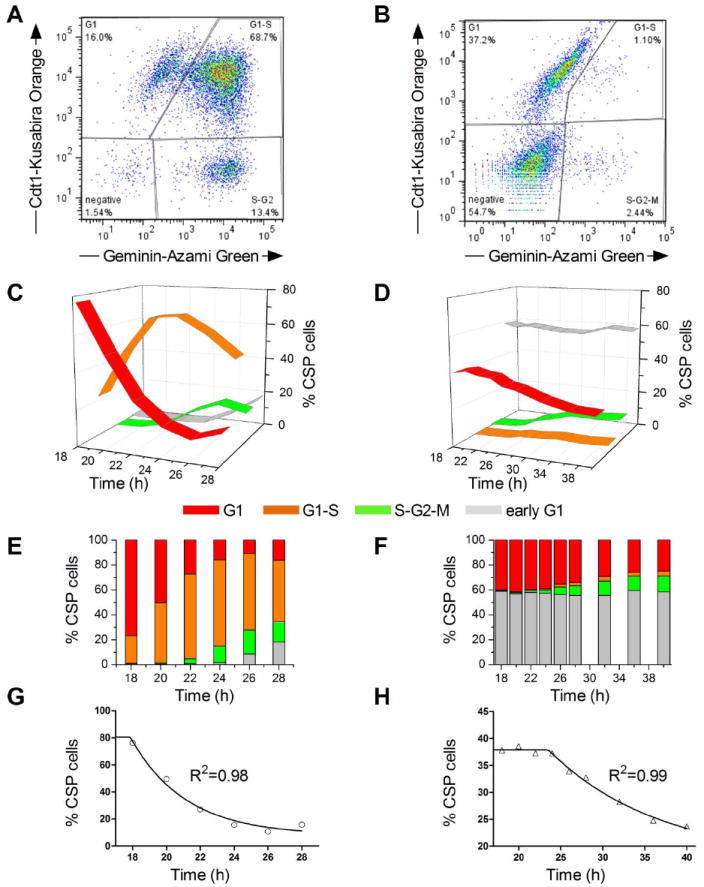

Representative flow cytometric analysis of Fucci+ (A) WT and (B) Abcg2-KO CSP cells 24 hours post-synchronization. Time course analysis of Fucci+ (C) WT and (D) Abcg2-KO CSP cells over a period of 10 and 22 hours respectively, starting at 18 hours post-synchronization. Bar graphs indicate the percentage of (E) WT and (F) Abcg2-KO CSP cells residing in each cell cycle phase during the specified time course. (G-H) Mathematical calculation of the G1-phase T50 value following a “plateau followed by exponential decay” model. The T50 for (G) WT and (H) Abcg2-KO CSP cells is 20.2hr and 32.6hr respectively. (I-J) Sequential (2 hours interval) DIC and fluorescent confocal microscopy images of Fucci+ (I) WT and (J) Abcg2-KO CSP cells 18-40 hours following synchronization. Cells were infected with lentiviruses expressing both Cdt1-Kusabira Orange probe (red) and Geminin-Azami Green (green) present in the G1 and the S-G2-M phases respectively. Cells residing in the G1-S transition phase appear as orange. Cells immediately after cytokinesis or in early G1 phase lose the fluorescence. Scale bars 20μm. Yellow box indicates the position of representative (I) WT and (J) Abcg2-KO CSP cells.
Abcg2 regulates G1-S transition in CSP cells
To elucidate the effects of Abcg2 on cell cycle progression, we monitored cell cycle kinetics in WT and Abcg2-KO CSP cells. WT CSP cells entered G1-S transition as early as 14 hours post synchronization (data not shown). Within 24 hours, WT cells in G1 reached a peak followed by a progressive decrease and concomitant entry into S-G2-M. Entry in G2-M was rapidly followed by evidence for cell division (Figures 2C and E). In contrast, Abcg2-KO CSP cells were unable to progress through the cell cycle and remained largely in G1 40 hours post-synchronization (Figures 2D and F). Moreover, only 4% of Abcg2-KO CSP cells were in G1-S transition and 12% in S/G2/M at 40 hours; these values were negligible at 18 hours (Figures 2D and F). Modeling of cell exit from the G1 phase using a “plateau followed by exponential decay” model10 revealed that the T50 for WT CSP cells was 20.2 hours while Abcg2-KO CSP cells had a T50 of 32.6 hours (Figure 2G and H). Confocal time-lapse, live-cell imaging documented that WT CSP cells cycled normally, while Abcg2-KO CSP cells remained predominantly confined to G1 (Figures 2I, J and Online Videos I, II). Collectively, these data strongly suggest that Abcg2 regulates the cell cycle in CSP cells.
Abcg2 and cell cycle gene expression profile of CSP cells
Based on these observations, we next determined the expression of cell cycle regulatory genes in WT and Abcg2-KO CSP cells. Following shRNA-lentivirus mediated silencing of Abcg2 (sh-Abcg2), the expression of cyclin C, cyclin D1 and cyclin E was downregulated (Figure 3A and Online Table I). Moreover, negative regulators of the cell cycle, including p27, were up-regulated (Figure 3A and Online Table I). Strikingly, the absence of Abcg2 increased significantly the expression of DNA-damage related genes such as Atm, Msh2, p53, Smc1a, Rad51, Brca1 and Brca2 (Figure 3B and Online Table I).
Figure 3. Abcg2 deficiency alters the gene expression profile of CSP cells.

Gene expression analysis of (A) cell cycle regulators and (B) DNA-damage responsive genes in sh-Abcg2 CSP cells compared to Scramble cells (n=3). Data are mean ± s.e.m. *p<0.05.
Abcg2 and asymmetric stem cell division
The dual fundamental property of stem cells is to self-renew and differentiate.2 Immunolabeling for the cell fate determinant α-adaptin demonstrated that sh-Abcg2 CSP cells preferentially divided asymmetrically (65.3±5.4% vs. scramble 27.9±6.6%) (Figure 4A-D). Moreover, in comparison to sh-Abcg2 CSP cells, a higher number of scramble CSP cells expressed the cell-fate determinant, numb (Figure 4E-G), supporting the concept that during division in Abcg2-deficient cells, the fate determinants segregate asymmetrically to one daughter cell.
Figure 4. Lack of Abcg2 favors asymmetric cell division in CSP cells.

Representative immuno-fluorescent images of (A) WT and (B) Abcg2-KO CSP cells stained for phospho-Histone-H3 (pH3, green), α-adaptin (red) and DAPI (blue) (Scale bars 10μm). Quantification of symmetric and asymmetric cell division mode of pH3+ (C) scramble and (D) sh-Abcg2 CSP cells, based on the distribution of α-adaptin. Representative flow cytometric analysis of (E) scramble and (F) sh-Abcg2 CSP stained with numb. (G) Quantification of numb-expressing scramble and sh-Abcg2 CSP cells. Data are mean ± s.e.m. *p<0.05.
DISCUSSION
Despite accumulating evidence as to the therapeutic benefits of stem cell therapy, little is known regarding the molecular mechanisms that dictate progenitor cell homeostasis. The identification of novel regulators of cardiac stem/progenitor cell self-renewal is of paramount importance for the development of new strategies for treatment of chronic heart failure. Such regulators may allow for manipulation of endogenous progenitors enhancing tissue regeneration following injury. The expansion of stem/progenitor cells through symmetric divisions, followed by a switch towards a differentiated progeny via asymmetric division would greatly enhance myocardial repair. In this context, the identification of the molecular variables that regulate the balance between symmetric and asymmetric stem cell division are of great relevance for the expansion and lineage specification of resident stem cells. In this report, we have identified that the cell surface transporter, Abcg2, is a critical determinant of cell cycle progression and transition between symmetric and asymmetric stem cell division in cardiac progenitor cells. Regulation of cardiac progenitor cells either by direct modulation of Abcg2 expression or indirectly through manipulation of Abcg2 substrates may represent a promising approach for cell-based therapy. Abcg2 activation following cardiac injury could allow the restoration and increase of progenitor cell numbers while inhibition of the transporter and activation of asymmetric cell division of cardiac progenitors would promote tissue regeneration.
Stem cell fate decisions such as proliferation, quiescence and differentiation are influenced by the cell cycle.11 Interestingly, an association may be present between the length of G1 and asymmetric cell division, further highlighting the significance of the effects of Abcg2 in CSP cell growth and commitment.11-14 Cells exhibiting high self-renewal ability, such as embryonic stem cells, have a short G1 phase, which is significantly prolonged with differentiation.11 Similarly, inhibition of G1 in adult neural stem cells results in neurogenesis, while shortening of G1 promotes their expansion through symmetric divisions.12, 15 By a number of techniques and approaches, our data show that the majority of Abcg2-KO CSP cells reside in the G1 phase of the cell cycle. In addition to the prolonged G1 phase and delayed entry in S phase, crucial cell fate determinants such as α-adaptin and numb were asymmetrically segregated in Abcg2-deficient CSP cells. These results strengthen the notion that the length of the cell cycle is linked to the mode of cell division in cardiac progenitor cells. We cannot exclude the possibility that a fraction of CSP cells negative for both probes could exit the cell cycle and reside in G0. These cells would correspond to daughter cells acquiring a committed fate or cells undergoing apoptosis. In fact, lack of Abcg2 increases apoptotic and necrotic cell death in basal conditions.9 Evidence of apoptotic cells can also be seen in the live cell imaging data. Interestingly, the role of Abcg2 in the regulation of the mode of division may extend beyond progenitor cells to transformed cancer cells, in which high expression of Abcg2 and unchecked symmetric division have been described.16-19
Importantly, it remains to be determined whether loss of Abcg2 influences the replication of CSP cells and the replenishment of endogenous progenitor cell niches following cardiac injury. Abcg2 is a membrane bound transporter that is primarily responsible for the cytoplasmic clearance of a number of substrates.8 We have previously shown that Abcg2 expression is regulated in a developmental manner in CSP cells.9 Abcg2 is highly expressed in neonatal CSP cells and its expression decreases with age. Moreover, during hypoxia, Abcg2 has been shown to be regulated by HIF-1α signaling,20 as well as upregulated by EGF 21, 22 and EGFR23 in transformed cells. The regulation of Abcg2 expression may, in part, be modulated by use of alternative 5’UTR leader exons.24 Abcg2 downstream signaling, however, remains completely undefined and the exact mechanisms by which Abcg2 regulates cell cycle and mode of division are unclear at the present time. It is likely that levels of certain molecules are altered in cells lacking Abcg2, and such yet to be defined molecules may subsequently reprogram the cell cycle mode of CSP cells directly or indirectly toward asymmetric division. For instance, Abcg2 has been shown to regulate embryonic stem cell function through maintenance of porphyrin homeostasis.25 A number of reports suggest that p53 inhibits proliferation while promoting asymmetric cell divisions.26, 27 Notably, lack of Abcg2 in CSP cells increases the expression of several cell cycle inhibitors such as p27 as well as p53, a well-known stress sensor. At the present time, we cannot exclude the possibility that Abcg2 may directly interact with other signaling cascades to influence cell division. Further investigation is required to address the exact role of Abcg2 in the regulation of progenitor cell mode of division. Stem/progenitor cell function, including both proliferation and mode of division, is highly sensitive to external signals from the stem cell microenvironment, or stem cell niche,28 including those effector pathways originating from neighboring progenitors, tissue stromal and parenchymal cells, and potentially extra-tissue infiltrating cells. Given that Abcg2 is a cell surface transporter, it may function to link internal and external signals and thereby regulate the function of progenitor cell populations via the microenvironment.
Supplementary Material
Novelty and Significance.
What Is Known?
Stem/progenitor cells modulate their cell fate decision in part through a balance between symmetric stem cell division, in which two identical daughter cells are formed, versus asymmetric stem cell division, in which two daughter cells with divergent fates are generated, one capable of self-renewal and one committed to differentiation.
Abcg2 regulates cardiac side population (CSP) progenitor cell homeostasis by promoting proliferation and survival while inhibiting differentiation, but mechanisms underlying this relationship remain unknown.
What New Information Does This Article Contribute?
Abcg2 regulates cell cycle duration and CSP proliferation through G1-S transition.
Abcg2 deficiency promotes asymmetric division in CSP cells.
Adult cardiac stem/progenitor cells represent a promising therapeutic option for the treatment of heart failure. Understanding the molecular mechanisms regulating adult progenitor cell self-renewal and lineage commitment is key towards their therapeutic utilization. The maintenance of progenitor cell pools and the production of differentiated progeny are ensured by a balance between symmetric and asymmetric division. Progenitor cell homeostasis is regulated by expression of Abcg2 cell surface transporter through unresolved mechanisms. This study finds that Abcg2 controls cell cycle progression and CSP cell proliferation as well as the balance between symmetric and asymmetric division. Manipulation of Abcg2 could represent a novel therapeutic strategy for enhancing efficacy of cell-based therapy for cardiovascular diseases.
Acknowledgments
We thank G. Losyev at the Cardiovascular FACS Core at Brigham and Women’s Hospital and Harvard Medical School for assistance with CSP cells sorting.
SOURCES OF FUNDING
This study was supported in part by NIH grants (HL086967 and HL093148) to R.L. K.-I.S. was supported by the Manasaki fellowship from the University of Crete-Greece.
Non-standard Abbreviations
- CSP
Cardiac Side Population
- Abcg2
ATP-Binding Cassette G-subfamily transporter 2
- FUCCI
Fluorescence Ubiquitination Cell Cycle Indicator
- PI
Propidium Iodide
Footnotes
DISCLOSURES
No disclosures.
References
- 1.Bolli R, Chugh AR, D’Amario D, Loughran JH, Stoddard MF, Ikram S, Beache GM, Wagner SG, Leri A, Hosoda T, Sanada F, Elmore JB, Goichberg P, Cappetta D, Solankhi NK, Fahsah I, Rokosh DG, Slaughter MS, Kajstura J, Anversa P. Cardiac stem cells in patients with ischaemic cardiomyopathy (scipio): Initial results of a randomised phase 1 trial. Lancet. 2011;378:1847–1857. doi: 10.1016/S0140-6736(11)61590-0. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 2.Knoblich J. Mechanisms of asymmetric stem cell division. Cell. 2008;132:583–680. doi: 10.1016/j.cell.2008.02.007. [DOI] [PubMed] [Google Scholar]
- 3.Zhang R, Zhang Z, Zhang C, Zhang L, Robin A, Wang Y, Lu M, Chopp M. Stroke transiently increases subventricular zone cell division from asymmetric to symmetric and increases neuronal differentiation in the adult rat. J Neurosci. 2004;24:5810–5815. doi: 10.1523/JNEUROSCI.1109-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Morrison S, Kimble J. Asymmetric and symmetric stem-cell divisions in development and cancer. Nature. 2006;441:1068–1142. doi: 10.1038/nature04956. [DOI] [PubMed] [Google Scholar]
- 5.Leri A, Kajstura J, Anversa P. Role of cardiac stem cells in cardiac pathophysiology: A paradigm shift in human myocardial biology. Circ Res. 2011;109:941–961. doi: 10.1161/CIRCRESAHA.111.243154. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 6.Mouquet Fdr, Pfister O, Jain M, Oikonomopoulos A, Ngoy S, Summer R, Fine A, Liao R. Restoration of cardiac progenitor cells after myocardial infarction by self-proliferation and selective homing of bone marrow-derived stem cells. Circulation research. 2005;97:1090–1092. doi: 10.1161/01.RES.0000194330.66545.f5. [DOI] [PubMed] [Google Scholar]
- 7.Bunting K. Abc transporters as phenotypic markers and functional regulators of stem cells. Stem cells (Dayton, Ohio) 2002;20:11–31. doi: 10.1002/stem.200011. [DOI] [PubMed] [Google Scholar]
- 8.Huls M, Russel F, Masereeuw R. The role of atp binding cassette transporters in tissue defense and organ regeneration. The Journal of pharmacology and experimental therapeutics. 2009;328:3–12. doi: 10.1124/jpet.107.132225. [DOI] [PubMed] [Google Scholar]
- 9.Pfister O, Oikonomopoulos A, Sereti KI, Sohn RL, Cullen D, Fine GC, Mouquet F, Westerman K, Liao R. Role of the atp-binding cassette transporter abcg2 in the phenotype and function of cardiac side population cells. Circ Res. 2008;103:825–835. doi: 10.1161/CIRCRESAHA.108.174615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Schauble S, Klement K, Marthandan S, Munch S, Heiland I, Schuster S, Hemmerich P, Diekmann S. Quantitative model of cell cycle arrest and cellular senescence in primary human fibroblasts. PLoS One. 2012;7:e42150. doi: 10.1371/journal.pone.0042150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Malumbres M. Physiological relevance of cell cycle kinases. Physiol Rev. 2011;91:973–1007. doi: 10.1152/physrev.00025.2010. [DOI] [PubMed] [Google Scholar]
- 12.Calegari F, Huttner WB. An inhibition of cyclin-dependent kinases that lengthens, but does not arrest, neuroepithelial cell cycle induces premature neurogenesis. Journal of cell science. 2003;116:4947–4955. doi: 10.1242/jcs.00825. [DOI] [PubMed] [Google Scholar]
- 13.Orihara-Ono M, Toriya M, Nakao K, Okano H. Downregulation of notch mediates the seamless transition of individual drosophila neuroepithelial progenitors into optic medullar neuroblasts during prolonged g1. Developmental biology. 2011;351:163–175. doi: 10.1016/j.ydbio.2010.12.044. [DOI] [PubMed] [Google Scholar]
- 14.Tsunekawa Y, Britto JM, Takahashi M, Polleux F, Tan SS, Osumi N. Cyclin d2 in the basal process of neural progenitors is linked to non-equivalent cell fates. Embo J. 2012;31:1879–1892. doi: 10.1038/emboj.2012.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lange C, Huttner W, Calegari F. Cdk4/cyclind1 overexpression in neural stem cells shortens g1, delays neurogenesis, and promotes the generation and expansion of basal progenitors. Cell stem cell. 2009;5:320–351. doi: 10.1016/j.stem.2009.05.026. [DOI] [PubMed] [Google Scholar]
- 16.Tsunoda S, Okumura T, Ito T, Kondo K, Ortiz C, Tanaka E, Watanabe G, Itami A, Sakai Y, Shimada Y. Abcg2 expression is an independent unfavorable prognostic factor in esophageal squamous cell carcinoma. Oncology. 2006;71:251–258. doi: 10.1159/000106787. [DOI] [PubMed] [Google Scholar]
- 17.Yoh K, Ishii G, Yokose T, Minegishi Y, Tsuta K, Goto K, Nishiwaki Y, Kodama T, Suga M, Ochiai A. Breast cancer resistance protein impacts clinical outcome in platinum-based chemotherapy for advanced non-small cell lung cancer. Clin Cancer Res. 2004;10:1691–1697. doi: 10.1158/1078-0432.ccr-0937-3. [DOI] [PubMed] [Google Scholar]
- 18.Kim JE, Singh RR, Cho-Vega JH, Drakos E, Davuluri Y, Khokhar FA, Fayad L, Medeiros LJ, Vega F. Sonic hedgehog signaling proteins and atp-binding cassette g2 are aberrantly expressed in diffuse large b-cell lymphoma. Mod Pathol. 2009;22:1312–1320. doi: 10.1038/modpathol.2009.98. [DOI] [PubMed] [Google Scholar]
- 19.Knoblich JA. Asymmetric cell division: Recent developments and their implications for tumour biology. Nat Rev Mol Cell Biol. 2010;11:849–860. doi: 10.1038/nrm3010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Krishnamurthy P, Ross D, Nakanishi T, Bailey-Dell K, Zhou S, Mercer K, Sarkadi B, Sorrentino B, Schuetz J. The stem cell marker bcrp/abcg2 enhances hypoxic cell survival through interactions with heme. The Journal of biological chemistry. 2004;279:24218–24243. doi: 10.1074/jbc.M313599200. [DOI] [PubMed] [Google Scholar]
- 21.Meyer zu Schwabedissen HE, Grube M, Dreisbach A, Jedlitschky G, Meissner K, Linnemann K, Fusch C, Ritter CA, Volker U, Kroemer HK. Epidermal growth factor-mediated activation of the map kinase cascade results in altered expression and function of abcg2 (bcrp) Drug Metab Dispos. 2006;34:524–533. doi: 10.1124/dmd.105.007591. [DOI] [PubMed] [Google Scholar]
- 22.Chen JS, Pardo FS, Wang-Rodriguez J, Chu TS, Lopez JP, Aguilera J, Altuna X, Weisman RA, Ongkeko WM. Egfr regulates the side population in head and neck squamous cell carcinoma. Laryngoscope. 2006;116:401–406. doi: 10.1097/01.mlg.0000195075.14093.fb. [DOI] [PubMed] [Google Scholar]
- 23.Huang WC, Chen YJ, Li LY, Wei YL, Hsu SC, Tsai SL, Chiu PC, Huang WP, Wang YN, Chen CH, Chang WC, Chen AJ, Tsai CH, Hung MC. Nuclear translocation of epidermal growth factor receptor by akt-dependent phosphorylation enhances breast cancer-resistant protein expression in gefitinib-resistant cells. J Biol Chem. 2011;286:20558–20568. doi: 10.1074/jbc.M111.240796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zong Y, Zhou S, Fatima S, Sorrentino BP. Expression of mouse abcg2 mrna during hematopoiesis is regulated by alternative use of multiple leader exons and promoters. J Biol Chem. 2006;281:29625–29632. doi: 10.1074/jbc.M606314200. [DOI] [PubMed] [Google Scholar]
- 25.Susanto J, Lin YH, Chen YN, Shen CR, Yan YT, Tsai ST, Chen CH, Shen CN. Porphyrin homeostasis maintained by abcg2 regulates self-renewal of embryonic stem cells. PLoS One. 2008;3:e4023. doi: 10.1371/journal.pone.0004023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rambhatla L, Ram-Mohan S, Cheng J, Sherley J. Immortal DNA strand cosegregation requires p53/impdh-dependent asymmetric self-renewal associated with adult stem cells. Cancer research. 2005;65:3155–3216. doi: 10.1158/0008-5472.CAN-04-3161. [DOI] [PubMed] [Google Scholar]
- 27.Sherley J. Guanine nucleotide biosynthesis is regulated by the cellular p53 concentration. The Journal of biological chemistry. 1991;266:24815–24843. [PubMed] [Google Scholar]
- 28.Hsu YC, Fuchs E. A family business: Stem cell progeny join the niche to regulate homeostasis. Nat Rev Mol Cell Biol. 2012;13:103–114. doi: 10.1038/nrm3272. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


