Abstract
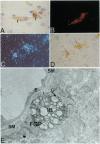
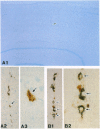
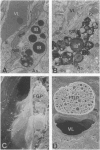
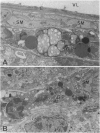
The transport of solutes between blood and brain is regulated by a specific barrier. Capillary endothelial cells of brain are known to mediate barrier function and facilitate transport. Here we report that specific cells surrounding arterioles, known as Mato's fluorescent granular perithelial (FGP) cells or perivascular microglial cells, contribute to the barrier function. Immunohistochemical and in situ hybridization studies indicate that, in normal brain cortex, type I and type II macrophage scavenger receptors are expressed only in FGP/perivascular microglial cells, and surface markers of macrophage lineage are also detected on them. These cells mediate the uptake of macromolecules, including modified low density lipoprotein, horseradish peroxidase, and ferritin injected either into the blood or into the cerebral ventricles. Accumulation of scavenged materials with aging or after the administration of a high-fat diet results in the formation of honeycomb-like foam cells and the narrowing of the lumen of arterioles in the brain cortex. These results indicate involvement of FGP/perivascular microglial cells in the barrier and scavenger functions in the central nervous system.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bell M. D., Lopez-Gonzalez R., Lawson L., Hughes D., Fraser I., Gordon S., Perry V. H. Upregulation of the macrophage scavenger receptor in response to different forms of injury in the CNS. J Neurocytol. 1994 Oct;23(10):605–613. doi: 10.1007/BF01191555. [DOI] [PubMed] [Google Scholar]
- Betz A. L., Goldstein G. W. Specialized properties and solute transport in brain capillaries. Annu Rev Physiol. 1986;48:241–250. doi: 10.1146/annurev.ph.48.030186.001325. [DOI] [PubMed] [Google Scholar]
- Dimitriadou V., Lambracht-Hall M., Reichler J., Theoharides T. C. Histochemical and ultrastructural characteristics of rat brain perivascular mast cells stimulated with compound 48/80 and carbachol. Neuroscience. 1990;39(1):209–224. doi: 10.1016/0306-4522(90)90234-u. [DOI] [PubMed] [Google Scholar]
- Farkas-Bargeton E., Arsenio-Nunes M. L. Maturation de l'équipement enzymatique des parois vasculaires du système nerveux. Etude histochimique. Acta Neuropathol. 1970;15(3):251–271. doi: 10.1007/BF00686771. [DOI] [PubMed] [Google Scholar]
- Fraser I., Hughes D., Gordon S. Divalent cation-independent macrophage adhesion inhibited by monoclonal antibody to murine scavenger receptor. Nature. 1993 Jul 22;364(6435):343–346. doi: 10.1038/364343a0. [DOI] [PubMed] [Google Scholar]
- Fruebis J., Parthasarathy S., Steinberg D. Evidence for a concerted reaction between lipid hydroperoxides and polypeptides. Proc Natl Acad Sci U S A. 1992 Nov 15;89(22):10588–10592. doi: 10.1073/pnas.89.22.10588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graeber M. B., Streit W. J., Büringer D., Sparks D. L., Kreutzberg G. W. Ultrastructural location of major histocompatibility complex (MHC) class II positive perivascular cells in histologically normal human brain. J Neuropathol Exp Neurol. 1992 May;51(3):303–311. doi: 10.1097/00005072-199205000-00009. [DOI] [PubMed] [Google Scholar]
- Graeber M. B., Streit W. J., Kreutzberg G. W. Identity of ED2-positive perivascular cells in rat brain. J Neurosci Res. 1989 Jan;22(1):103–106. doi: 10.1002/jnr.490220114. [DOI] [PubMed] [Google Scholar]
- Hickey W. F., Kimura H. Perivascular microglial cells of the CNS are bone marrow-derived and present antigen in vivo. Science. 1988 Jan 15;239(4837):290–292. doi: 10.1126/science.3276004. [DOI] [PubMed] [Google Scholar]
- Ibrahim M. Z. The mast cells of the mammalian central nervous system. 2. The effect of proton irradiation in the monkey. J Neurol Sci. 1974 Apr;21(4):479–499. doi: 10.1016/0022-510x(74)90044-6. [DOI] [PubMed] [Google Scholar]
- Kida S., Steart P. V., Zhang E. T., Weller R. O. Perivascular cells act as scavengers in the cerebral perivascular spaces and remain distinct from pericytes, microglia and macrophages. Acta Neuropathol. 1993;85(6):646–652. doi: 10.1007/BF00334675. [DOI] [PubMed] [Google Scholar]
- Kodama T., Freeman M., Rohrer L., Zabrecky J., Matsudaira P., Krieger M. Type I macrophage scavenger receptor contains alpha-helical and collagen-like coiled coils. Nature. 1990 Feb 8;343(6258):531–535. doi: 10.1038/343531a0. [DOI] [PubMed] [Google Scholar]
- Kodama T., Reddy P., Kishimoto C., Krieger M. Purification and characterization of a bovine acetyl low density lipoprotein receptor. Proc Natl Acad Sci U S A. 1988 Dec;85(23):9238–9242. doi: 10.1073/pnas.85.23.9238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawson L. J., Perry V. H., Dri P., Gordon S. Heterogeneity in the distribution and morphology of microglia in the normal adult mouse brain. Neuroscience. 1990;39(1):151–170. doi: 10.1016/0306-4522(90)90229-w. [DOI] [PubMed] [Google Scholar]
- Luoma J., Hiltunen T., Särkioja T., Moestrup S. K., Gliemann J., Kodama T., Nikkari T., Ylä-Herttuala S. Expression of alpha 2-macroglobulin receptor/low density lipoprotein receptor-related protein and scavenger receptor in human atherosclerotic lesions. J Clin Invest. 1994 May;93(5):2014–2021. doi: 10.1172/JCI117195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mato M., Aikawa E., Mato T. K., Kurihara K. Tridimensional observation of fluorescent granular perithelial (FGP) cells in rat cerebral blood vessels. Anat Rec. 1986 Aug;215(4):413–419. doi: 10.1002/ar.1092150413. [DOI] [PubMed] [Google Scholar]
- Mato M., Ookawara S. A simple method for observation of capillary nets in rat brain cortex. Experientia. 1979 Apr 15;35(4):501–503. doi: 10.1007/BF01922731. [DOI] [PubMed] [Google Scholar]
- Mato M., Ookawara S., Aikawa E., Kawasaki K. Studies on fluorescent granular perithelium (F.G.P.) of rat cerebral cortex - especially referring to morphological changes in aging. Anat Anz. 1981;149(5):486–501. [PubMed] [Google Scholar]
- Mato M., Ookawara S., Kurihara K. Uptake of exogenous substances and marked infoldings of the fluorescent granular pericyte in cerebral fine vessels. Am J Anat. 1980 Mar;157(3):329–332. doi: 10.1002/aja.1001570308. [DOI] [PubMed] [Google Scholar]
- Mato M., Ookawara S., Mato T. K., Namiki T. An attempt to differentiate further between microglia and fluorescent granular perithelial (FGP) cells by their capacity to incorporate exogenous protein. Am J Anat. 1985 Feb;172(2):125–140. doi: 10.1002/aja.1001720203. [DOI] [PubMed] [Google Scholar]
- Mato M., Ookawara S., Saito-Taki T. Serological determinants of fluorescent granular perithelial cells along small cerebral blood vessels in rodent. Acta Neuropathol. 1986;72(2):117–123. doi: 10.1007/BF00685972. [DOI] [PubMed] [Google Scholar]
- Mato M., Ookawara S., Sano M., Fukuda S., Sokabe H. Behavior of fluorescent granular perithelium (FGP) in cerebral cortex of SHR-SP rats under some conditions. Exp Mol Pathol. 1983 Aug;39(1):100–109. doi: 10.1016/0014-4800(83)90044-8. [DOI] [PubMed] [Google Scholar]
- Mato M., Ookawara S., Sano M., Fukuda S. Uptake of fat by fluorescent granular perithelial cells in cerebral cortex after administration of fat rich chow. Experientia. 1982 Dec 15;38(12):1496–1498. doi: 10.1007/BF01955791. [DOI] [PubMed] [Google Scholar]
- Mato M., Ookawara S., Sugamata M., Aikawa E. Evidence for the possible function of the fluorescent granular perithelial cells in brain as scavengers of high-molecular-weight waste products. Experientia. 1984 Apr 15;40(4):399–402. doi: 10.1007/BF01952574. [DOI] [PubMed] [Google Scholar]
- Mato M., Ookawara S., Tooyama K., Ishizaki T. Chronobiological studies on the blood-brain barrier. Experientia. 1981;37(9):1013–1015. doi: 10.1007/BF01971811. [DOI] [PubMed] [Google Scholar]
- Matsumoto A., Naito M., Itakura H., Ikemoto S., Asaoka H., Hayakawa I., Kanamori H., Aburatani H., Takaku F., Suzuki H. Human macrophage scavenger receptors: primary structure, expression, and localization in atherosclerotic lesions. Proc Natl Acad Sci U S A. 1990 Dec;87(23):9133–9137. doi: 10.1073/pnas.87.23.9133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naito M., Kodama T., Matsumoto A., Doi T., Takahashi K. Tissue distribution, intracellular localization, and in vitro expression of bovine macrophage scavenger receptors. Am J Pathol. 1991 Dec;139(6):1411–1423. [PMC free article] [PubMed] [Google Scholar]
- Naito M., Suzuki H., Mori T., Matsumoto A., Kodama T., Takahashi K. Coexpression of type I and type II human macrophage scavenger receptors in macrophages of various organs and foam cells in atherosclerotic lesions. Am J Pathol. 1992 Sep;141(3):591–599. [PMC free article] [PubMed] [Google Scholar]
- Ookawara S., Mitsuhashi U., Suminaga Y., Mato M. Study on distribution of pericyte and fluorescent granular perithelial (FGP) cell in the transitional region between arteriole and capillary in rat cerebral cortex. Anat Rec. 1996 Feb;244(2):257–264. doi: 10.1002/(SICI)1097-0185(199602)244:2<257::AID-AR13>3.0.CO;2-O. [DOI] [PubMed] [Google Scholar]
- Perry V. H., Andersson P. B., Gordon S. Macrophages and inflammation in the central nervous system. Trends Neurosci. 1993 Jul;16(7):268–273. doi: 10.1016/0166-2236(93)90180-t. [DOI] [PubMed] [Google Scholar]
- Perry V. H., Gordon S. Macrophages and the nervous system. Int Rev Cytol. 1991;125:203–244. doi: 10.1016/s0074-7696(08)61220-6. [DOI] [PubMed] [Google Scholar]
- Reese T. S., Karnovsky M. J. Fine structural localization of a blood-brain barrier to exogenous peroxidase. J Cell Biol. 1967 Jul;34(1):207–217. doi: 10.1083/jcb.34.1.207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steinberg D., Parthasarathy S., Carew T. E., Khoo J. C., Witztum J. L. Beyond cholesterol. Modifications of low-density lipoprotein that increase its atherogenicity. N Engl J Med. 1989 Apr 6;320(14):915–924. doi: 10.1056/NEJM198904063201407. [DOI] [PubMed] [Google Scholar]
- Yamashita A., Hattori Y., Kotani M., Miyasaka M., Fukumoto T. A novel monoclonal antibody, Mar 1, directed specifically against mononuclear phagocyte system cells in rats. Immunology. 1990 Jun;70(2):262–271. [PMC free article] [PubMed] [Google Scholar]