Summary
The prevalence of hearing loss after damage to the mammalian cochlea has been thought to be due to a lack of spontaneous regeneration of hair cells, the primary receptor cells for sound. Here, we show that supporting cells, which surround hair cells in the normal cochlear epithelium, differentiate into new hair cells in the neonatal mouse following ototoxic damage. Using lineage tracing, we show that new hair cells, predominantly outer hair cells, arise from Lgr5-expressing inner pillar and third Deiters cells and that new hair cell generation is increased by pharmacological inhibition of Notch. These data suggest that the neonatal mammalian cochlea has some capacity for hair cell regeneration following damage alone and that Lgr5-positive cells act as hair cell progenitors in the cochlea.
Graphical Abstract
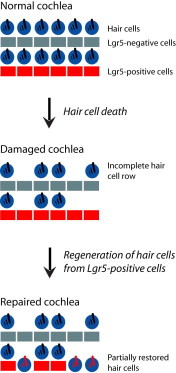
Highlights
-
•
Hair cells regenerate after damage to the neonatal mouse cochlea
-
•
Generation of hair cells is increased by inhibition of Notch
-
•
Hair cells regenerate by transdifferentiation with minimal proliferation
-
•
Regenerated hair cells arise from Lgr5-positive supporting cells
Although hair cells, the primary receptor cells for sound, had been thought to be incapable of regeneration, Edge and colleagues find that supporting cells, which surround hair cells in the normal cochlear epithelium, include a subset of Lgr5-expressing cells that act as progenitor cells, differentiating into hair cells in the neonatal mouse after damage.
Introduction
Permanent damage to the hair cells of the inner ear results in sensorineural hearing loss, leading to communication difficulties in a large percentage of the population. Hair cells are the receptor cells that transduce the acoustic stimulus. Regeneration of damaged hair cells could potentially yield a cure for a condition that currently has no therapies other than prosthetic devices. Although hair cells do not regenerate in the mammalian cochlea, new hair cells in lower vertebrates are generated from epithelial cells, called supporting cells, that surround hair cells (Balak et al., 1990; Raphael, 1992; Stone and Cotanche, 1994; Stone and Rubel, 2000; Warchol and Corwin, 1996).
In this study, we found newly formed hair cells after loss of the original hair cells in the newborn mammalian cochlea. The differentiation of new hair cells in response to damage occurred spontaneously and without significant proliferation of supporting cells. Previous work had shown generation of supernumerary hair cells in embryonic and neonatal mammals (Doetzlhofer et al., 2009; Hayashi et al., 2008; Kiernan et al., 2005; Takebayashi et al., 2007; Yamamoto et al., 2006) after inhibition of Notch signaling, which blocks differentiation of hair cells in the embryo (Lanford et al., 1999; Yamamoto et al., 2006; Zine et al., 2000).
The production of new hair cells from supporting cells could be increased by inhibition of Notch signaling in the damaged cochlea. We found, by the use of lineage tracing and confocal microscopy in the newborn cochlea after damage and Notch inhibition, that the capacity for supporting cell transdifferentiation to hair cells was not equally shared but rather occurred preferentially in a subset of these cells. In previous work, we had shown that supporting cells expressing Lgr5, a known marker of intestinal stem cells and a target of the Wnt pathway (Barker et al., 2007), had the capacity to differentiate into hair cells (Shi et al., 2012). In that study, we were not able to show that the cells identified retrospectively as progenitor cells after sorting had the capacity to regenerate hair cells in a damaged organ of Corti. Here, we demonstrate regenerative potential in Lgr5-positive supporting cells, using Sox2 and Lgr5 lineage tracing, in a damage model in the newborn cochlea. These results confirm that an Lgr5-expressing subset of supporting cells in the cochlea act as progenitors for hair cells after damage.
Results
Supporting Cells Transdifferentiated into Hair Cells at Low Levels following Damage
Because hair cell number can be increased by inhibition of Notch in the neonatal organ of Corti, we decided to employ lineage tracing using Lgr5 and Sox2-Cre expressing cells to identify cell populations within the mammalian organ of Corti that could generate these new hair cells. We tested whether the two Cre lines accurately reflected Lgr5 and Sox2 expression after crossing to reporters (Figure S1 and Table S1 available online). We chose to use newborn tissue with drug-induced hair cell damage as a model for hair cell regeneration that could be combined with lineage tracing. Organ of Corti explant cultures treated with 50 μM gentamicin overnight and examined 72 hr later showed significant outer hair cell (OHC) damage in the middle and basal regions, limited damage in the apex, and limited inner hair cell (IHC) loss (Figure S2).
We first tested whether the model we had selected for lineage tracing was viable by assessing the fate of the lineage-tagged cells in organs of Corti treated with tamoxifen at postnatal day 1 (P1) and exposed to gentamicin at P2 in the absence of Notch inhibition. Unexpectedly, we observed MYO7A-expressing cells in the damaged organ of Corti that were positive for Sox2 and Lgr5 lineage tags. The number of hair cells that expressed the lineage tag was small, and the presence of the reporter and unusual location in the pillar cell region suggested that some of the MYO7A-expressing cells were not simply surviving hair cells but had differentiated from supporting cells (Figures 1A and 1B). In addition, unlike native hair cells, these cells exhibited antibody staining for SOX2 in their nuclei (Figures 1C and 1E), consistent with immature hair cells (J.S. Kempfle et al., 2012, Molecular Biology of Hearing and Deafness, conference). Many of the new hair cells in the pillar region stained for PRESTIN (Zheng et al., 2000), a motor protein expressed only in OHCs (Figures 1D and 1F). The new hair cells were found in the apex and middle turns of the cochlea, but not in the base (Figure 1H), and the number of new hair cells was significantly increased relative to the undamaged control. The expression pattern of Lgr5 (inner pillar cells, third Deiters cells, inner border cells) and location of the new hair cells indicated that they were derived from inner pillar cells.
Figure 1.
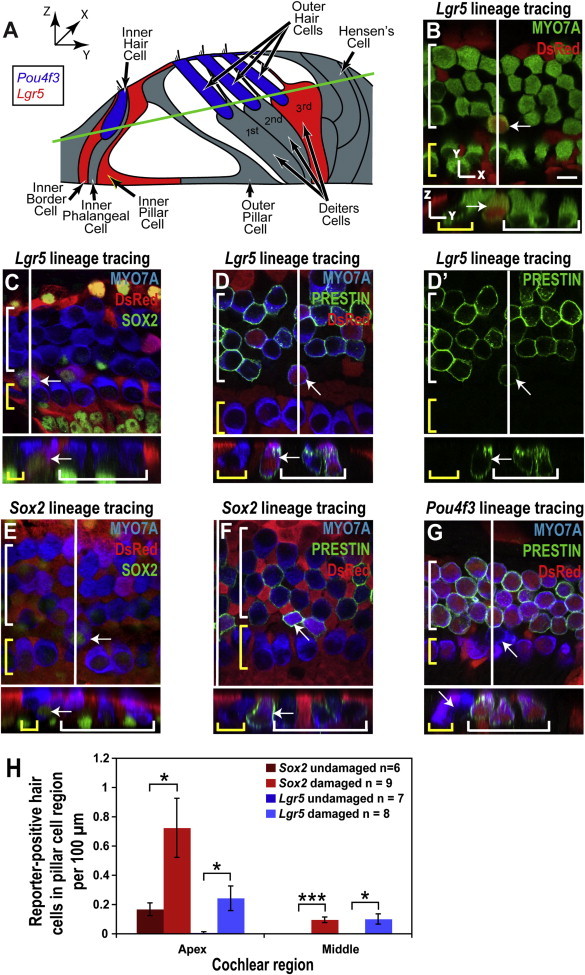
New Hair Cells in the Pillar Cell Region after Gentamicin Damage
(A) Illustration of organ of Corti structure showing the Pou4f3-positive hair cells (blue), the Lgr5-positive supporting cells (red), and the remaining supporting cells in gray. Both the red and gray supporting cells are Sox2 positive. The green line indicates the xy plane from which the confocal slices in (B)–(G) are taken.
(B–G) Confocal slices and cross sections from the midapex of neonatal organ of Corti explant cultures, treated with gentamicin and lineage-traced using the CAG-tdTomato reporter, were stained for DsRed (red). A white line on the whole-mount image shows the location of the cross section, and yellow and white brackets indicate IHCs and OHCs, respectively. Arrows point to new reporter-positive (or reporter-negative for Pou4f3) hair cells in the pillar cell region. Scale bar, 10 μm.
(B) A reporter-positive hair cell from the Lgr5 lineage (such as those counted in H) was visible in the pillar cell region.
(C and D) Reporter staining identified the hair cells marked by the white arrows as derived from Lgr5-positive cells; costaining for SOX2 (C) and location in the pillar cell region indicated that they were newly differentiated, and an OHC phenotype was suggested by the expression of PRESTIN (D). (D′) PRESTIN channel from (D) shows staining in the membrane and cuticular plate of the new hair cell.
(E and F) Staining for the Sox2 lineage reporter identified the hair cells marked by the white arrows as derived from supporting cells; their location (pillar cell region) and costaining for SOX2 (E) identified them as newly differentiated cells, and costaining for PRESTIN (F) indicated an OHC identity.
(G) The lack of Pou4f3 lineage reporter staining and the location in the pillar region identified the hair cell marked by the white arrow as a new hair cell, and costaining for PRESTIN indicated an OHC identity.
(H) Increased numbers of Lgr5 (blue bars) and Sox2 (red bars) reporter-positive hair cells were observed in the pillar cell region of the organ of Corti after gentamicin treatment (mean ± SEM per 100 μm; ∗p < 0.05, ∗∗∗p < 0.001).
To confirm the presence of new hair cells in the pillar cell region, Pou4f3 was employed for hair cell lineage tracing (Figure S1). Following gentamicin damage, reporter-negative hair cells were observed in the pillar cell region of Pou4f3 lineage-tagged cultures (Figure 1G). The absence of reporter expression in these hair cells, in contrast to native hair cells, again indicated that they had differentiated to hair cells from reporter-negative cells.
The appearance of new MYO7A-expressing cells in the newborn organ of Corti was surprising considering the previously observed resistance of the postnatal sensory epithelium to hair cell regeneration. Previous studies had showed no regeneration and in fact no supporting cell mitosis after birth.
In Vitro Damage followed by Notch Inhibition Resulted in Transdifferentiation of Supporting Cells into Hair Cells
The results showing newly generated MYO7A-positive cells in the damaged organ of Corti suggested that the postnatal organ of Corti had some regenerative capacity. However, because the number of new MYO7A-positive cells was small, we proceeded with Notch inhibition to study the lineage tracing of transdifferentiated cells.
Treatment of gentamicin-damaged cochlea with LY411575, a γ-secretase inhibitor, increased hair cell numbers (Figure 2A). The restoration of hair cells was not complete (80.5% and 36.3% of the normal hair cell numbers in the middle and base, respectively), but the increase was significant (28.3% and 154.7% in the middle and base, respectively; Figure 2B).
Figure 2.
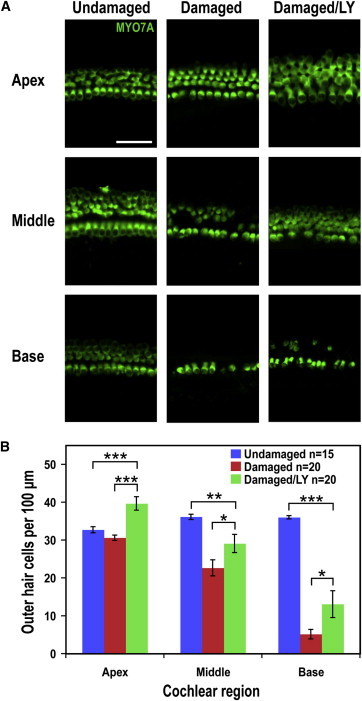
Damage followed by Notch Inhibition Leads to an Increase in Hair Cell Numbers
(A) Images of hair cells in the apex, middle, and base of an undamaged control, a damaged control, and a damaged/LY411575-treated explant culture show hair cell loss following damage and increased hair cell numbers with LY411575 treatment. Scale bar, 50 μm.
(B) Hair cell counts in the apex, middle, and base indicated significantly more OHCs in the damaged/LY411575-treated cultures than in the damaged controls (mean ± SEM per 100 μm; ∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001).
Lineage tracing (exposure to tamoxifen at P0 and P1) showed an increase in reporter-labeled hair cells in gentamicin-damaged (at P2), γ-secretase inhibitor-treated (at P3) cultures (Figure 3A; culture is shown at P6). The treatment resulted in significantly more Sox2 lineage-tagged hair cells in all cochlear regions and Lgr5 lineage-tagged hair cells in the apical and middle regions (Figure 3B). Based on the location of the reporter-positive hair cells and the expression pattern of Lgr5, the new hair cells were likely descended from Lgr5-positive inner pillar and third Deiters cells. Quantification of the percentage of total red OHCs located in the pillar region (corrected for red OHCs in the undamaged controls) indicated that reporter-positive OHCs occurred most frequently in the pillar cell region (62.4% ± 15.6% of the total in the apex, 88.1% ± 11.6% in the middle, and 40% ± 24.5% in the base). These data confirmed that the additional hair cells were newly differentiated and suggested that inner pillar cells were the source of the new hair cells.
Figure 3.
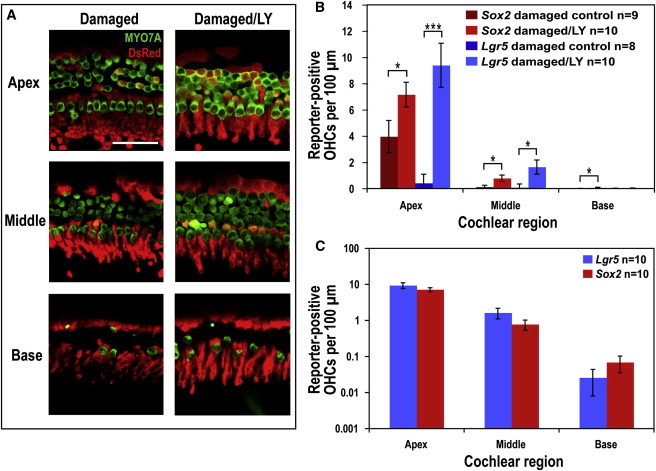
LY411575 Treatment after Damage Results in Transdifferentiation of Lgr5- and Sox2-Positive Supporting Cells into Hair Cells
(A) Images of the three regions of the organ of Corti from a damaged control and a damaged/LY411575-treated explant culture that were Lgr5 lineage traced using the CAG-tdTomato reporter and stained for DsRed (red). Hair cells were stained for MYO7A (green). Many more reporter-positive hair cells (costained for DsRed and MYO7A) were visible in the apical and middle regions of the culture treated with LY411575 than the damaged control. Lgr5 lineage tracing indicated that these reporter-labeled hair cells were derived from Lgr5-positive supporting cells. Scale bar, 50 μm.
(B) Reporter-positive OHC counts for Sox2 and Lgr5 lineage tracing of gentamicin/LY411575-treated and control explants showed significantly more reporter-labeled OHCs following LY411575 treatment (mean ± SEM per 100 μm; ∗p < 0.05, ∗∗∗p < 0.001).
(C) Reporter-positive OHC counts following gentamicin/LY411575 treatment revealed no significant difference between Sox2 and Lgr5 lineage tracing in any of the cochlear regions, suggesting that lineage tracing with both markers labeled the same population of cells (mean ± SEM per 100 μm, plotted on a logarithmic scale).
After correction for the number of recombined OHCs in undamaged controls, a comparison of the total Lgr5 and Sox2 lineage-tagged OHCs following gentamicin/LY411575 treatment indicated no significant difference between the number of OHCs from supporting cells labeled by the two markers in any cochlear region (Figure 3C). If a supporting cell type other than inner pillar and third Deiters cells had contributed to the production of new hair cells, we would have expected more Sox2 than Lgr5 lineage-tagged OHCs due to the relatively restricted expression of Lgr5. The similarity in the number of Lgr5 and Sox2 lineage-tagged OHCs suggested that Lgr5-positive supporting cells were the source of all new OHCs.
Many new hair cells in both the pillar cell and OHC regions stained for PRESTIN, suggesting that they were OHCs (white arrows, Figures 4A, 4C, 4E, and 4F). Other new hair cells did not appear to be PRESTIN-positive (yellow arrows, Figure 4C). Unlike preexisting hair cells, many hair cells with reporter expression were positive for SOX2, confirming that they were new (Figures 4B and 4D). Some new hair cells were observed in areas of extensive damage (Figures 4E and 4F). Immature stereocilia bundles were noted on many new hair cells in the pillar cell region, further indicating they were newly generated (Figures 4G–4G″).
Figure 4.
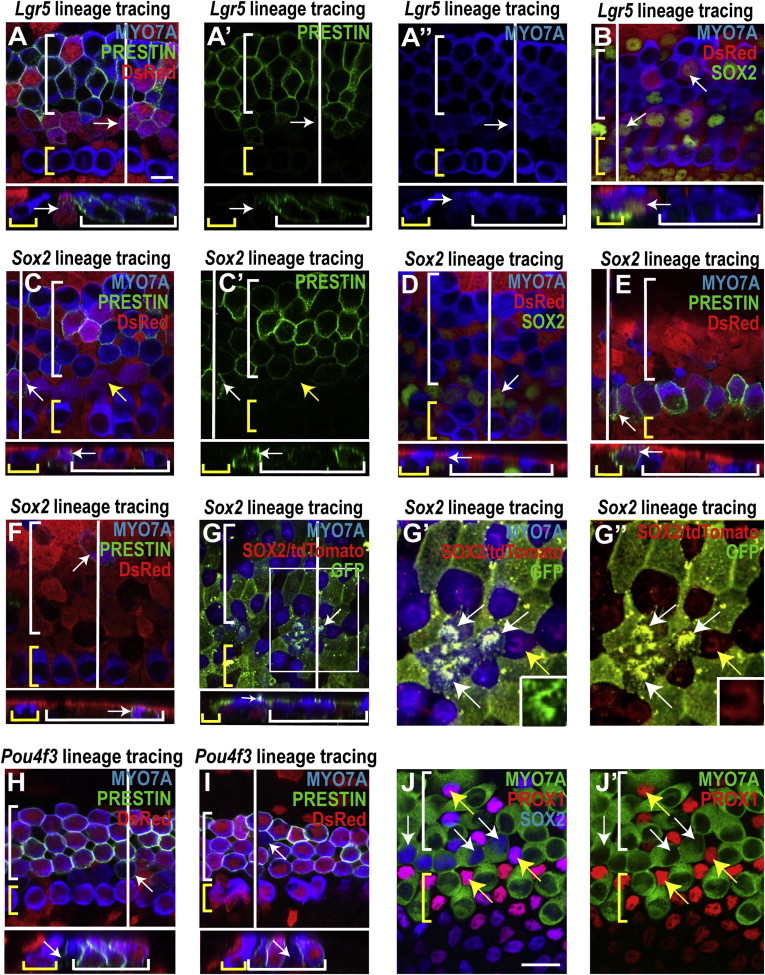
New Hair Cells Have Characteristics of Both Supporting Cells and Immature Hair Cells
(A and B) In the midapex from lineage-traced organ of Corti explant cultures treated with gentamicin/LY411575, reporter expression (DsRed staining) identified the hair cells indicated by white arrows as derived from Lgr5-positive cells. A white line shows the location of the cross section and yellow and white brackets indicate IHCs and OHCs, respectively. Many of the lineage-tagged hair cells costained for PRESTIN (A), indicating an OHC identity (PRESTIN and MYO7A channels shown in A′ and A″, respectively) and SOX2 (B), confirming that the lineage-tagged hair cells in the pillar and OHC regions were newly differentiated. Scale bar, 10 μm.
(C–G) Sox2 lineage tracing using either the CAG-tdTomato reporter stained for DsRed (C–F) or the CAG-tdTomato-EGFP reporter stained for GFP (G). Reporter-positive hair cells marked with white arrows are descended from Sox2-positive cells.
(C and D) Lineage-tagged hair cells visible in the pillar and OHC regions of the midapex (white arrows) costained for PRESTIN (C; PRESTIN channel shown in C′), suggesting an OHC phenotype. Other reporter-expressing hair cells were negative for PRESTIN (C, yellow arrows). Lineage tagged hair cells were also costained for SOX2 (D), indicating that they were newly differentiated.
(E and F) In the middle region where hair cell damage was extensive, reporter-positive hair cells were observed in what appeared to be the pillar cell region (E) and the OHC area (F).
(G) Immature stereocilia bundles were observed on some reporter-positive hair cells in the pillar cell region (white arrow, midapex), further suggesting they were newly differentiated. Note that because the CAG-tdTomato-EGFP reporter has a membrane localization sequence, the GFP signal is highly visible in the membrane-rich stereocilia bundles located atop hair cells that were derived from SOX2-positive supporting cells. SOX2 staining in the nucleus of the cell indicated by the white arrow is consistent with a new hair cell.
(G′ and G″) Higher magnification of the region outlined with a white box in (G) shows several GFP-positive stereocilia bundles (white arrows), indicating reporter recombination, whereas the bundles of the original hair cells remain positive for tdTomato (yellow arrow). The insets show stereociliary bundles of individual hair cells, either new, and therefore recombined (G′, GFP), or original (G″, tdTomato).
(H and I) The absence of reporter expression in the hair cells marked with white arrows from Pou4f3 lineage-traced cultures using the CAG-tdTomato reporter (red) indicated that they were new hair cells located in the pillar (H) and OHC (I) regions. Costaining for PRESTIN suggested an OHC identity in some of these hair cells (H).
(J and J′) SOX2/MYO7A double-positive hair cells in the pillar cell region are PROX1 negative (white arrows), whereas the surrounding supporting cells are SOX2/PROX1 positive (yellow arrows). Scale bar, 20 μm.
Hair cells without the lineage tag were also observed in the pillar and OHC regions of damaged organ of Corti cultures treated with LY411575 after Pou4f3 lineage tracing (Figures 4H and 4I), again indicating that new hair cells had been generated. SOX2-positive hair cells in the pillar cell region were not PROX1 positive (Figure 4J), unlike surrounding supporting cells that were both SOX2 and PROX1 positive. This suggests that the SOX2/MYO7A-positive cells in the pillar cell region had begun to lose some supporting cell markers during their differentiation to hair cells.
Generation of New Hair Cells after γ-Secretase Inhibition was Dependent on Wnt Signaling
To ask whether transdifferentiation of the Lgr5-positive supporting cells into hair cells in the cochlea could be Wnt dependent, we inactivated β-catenin, the intracellular mediator of canonical Wnt signaling. Organ of Corti cultures from β-cateninflox(exon2–6);Sox2-Cre-ER pups were treated with tamoxifen at P0 to delete β-catenin in supporting cells. The organ of Corti was treated in vitro with gentamicin as in the damage model described above and then with LY411575, and treated mutant tissues were compared to their β-cateninflox(exon2–6)-negative littermates. β-cateninflox(exon2–6);Sox2-Cre-ER cultures showed significantly fewer MYO7A/SOX2 double-positive cells in the apical region than control cultures (Figure 5). This indicates that β-catenin and Wnt signaling are necessary for transdifferentiation of supporting cells into hair cells following damage and Notch inhibition. Thus, the effects of Notch inhibition are β-catenin dependent and the response of Lgr5-positive cells to damage is attributable to Wnt signaling.
Figure 5.
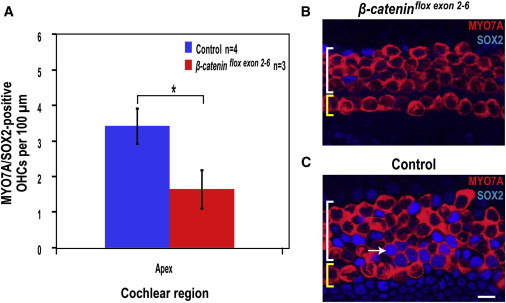
Wnt Signaling Is Necessary for Supporting Cell Transdifferentiation
(A) Double-positive MYO7A/SOX2 OHC counts for organ of Corti cultures from β-cateninflox(exon2–6);Sox2-Cre-ER pups and their β-cateninflox(exon2–6)-negative littermates (control) after gentamicin and LY411575 treatment show significantly less double-positive OHCs in the apex (mean ± SEM per 100 μm; ∗p < 0.05).
(B and C) Images from the apex of the organ of Corti after gentamicin and LY411575 treatment of β-cateninflox(exon2–6);Sox2-Cre-ER cultures (B) and β-cateninflox(exon2–6)-negative controls (C). Yellow and white brackets indicate IHCs and OHCs, respectively. Scale bar, 10 μm. MYO7A/SOX2-positive cells are absent in (B), whereas the white arrow indicates one of several double-positive cells in (C).
A Small Number of New Hair Cells Were Generated through Proliferation after Damage
Organ of Corti cultures treated with gentamicin followed by bromodeoxyuridine (BrdU) exhibited a small number of MYO7A-positive cells in the pillar cell region that were also labeled for SOX2 and BrdU (Figure 6A). These cells were seen only in the apical end of the cochlea and often occurred in pairs. In 16 organ of Corti cultures, MYO7A/SOX2/BrdU-positive cells were observed with a mean frequency of 0.62 ± 0.22 per organ of Corti, which is significantly more (p < 0.01) than in undamaged controls, which had no BrdU-positive cells. Cultures damaged with gentamicin and then treated with LY411575 also displayed occasional MYO7A/SOX2/BrdU/-positive cells in the pillar cell region (Figure 6B). Among seven explant cultures, an average of 15.57 ± 12.66 triple-positive cells were observed per organ of Corti. The variability was high, with some cultures lacking any BrdU-positive hair cells. These results suggest that cell division can precede transdifferentiation in inner pillar cells in response to damage.
Figure 6.
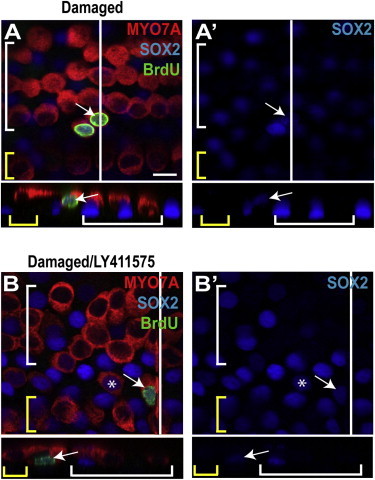
Supporting Cells Proliferate to Generate New Hair Cells
(A) A gentamicin- and BrdU-treated organ of Corti explant from the midapex showed a pair of triple-labeled hair cells in the pillar cell region. A white line shows the location of the cross section, and yellow and white brackets indicate IHCs and OHCs, respectively. The arrow points to a triple-labeled (BrdU/SOX2/MYO7A) cell in the pillar cell region. Its unusual location and costaining indicated a newly differentiated hair cell generated through proliferation.
(A′) The SOX2 channel from (A) is shown.
(B) The arrow points to a single triple-labeled cell in the pillar region of a gentamicin/LY411575-treated explant culture, indicating a newly differentiated hair cell generated through proliferation, and the asterisk indicates a BrdU-negative, SOX2-positive hair cell, suggesting generation through transdifferentiation rather than proliferation.
(B′) The SOX2 channel from (B) is shown. Scale bar, 10 μm.
Discussion
We have demonstrated generation of new hair cells in the neonatal mouse cochlea following gentamicin damage. The new hair cells were derived from supporting cells by transdifferentiation and limited proliferation. Appearance of new hair cells has not been observed in studies that have assessed regeneration in the mammalian cochlea after damage. In contrast, spontaneous transdifferentiation of supporting cells in response to aminoglycoside damage resulted in vestibular hair cell replacement, and hair cell number could be increased by Notch inhibition (Lin et al., 2011), consistent with previous studies showing some capacity for regeneration in the vestibular, as opposed to the hearing, organs of the mammalian inner ear (Forge et al., 1993; Rubel et al., 1995; Warchol et al., 1993).
The sensitive lineage tracing tools used in this study allowed for the unexpected discovery that hair cells regenerated spontaneously in the postnatal mammalian cochlea. New hair cells were detected by the presence of both the reporter from the tagged supporting cell and hair cell markers in the same cell. Supporting cell transdifferentiation was thus shown to be the mechanism for the generation of hair cells, whether after damage alone or after damage and blocking Notch signaling by treatment with a γ-secretase inhibitor. The organ of Corti’s regenerative response, whether occurring spontaneously or after pharmacological inhibition of Notch, appeared to be restricted to early postnatal times and gave rise to OHCs, even when the new hair cells were derived from cells closer to IHCs. Surprisingly, we saw less generation of new hair cells in areas with the most damage. The damage effect was thus not confined to the areas of cell loss but extended to undamaged regions. Apical areas may maintain their capacity for transdifferentiation of supporting cells longer because of their later development, as compared to the base and middle regions.
In addition to demonstrating spontaneous supporting cell transdifferentiation to hair cells after damage, we showed that the capacity to generate hair cells was limited to a subset of supporting cells: inner pillar cells and third-row Deiters cells (Figure 7). This was a striking set of supporting cells, because the same cells were identified as Lgr5-expressing progenitor cells by their capacity for self-renewal and generation of hair cells in vitro compared to a general supporting cell population (Shi et al., 2012). Inner pillar cells were the source of most of the hair cells after damage in the absence of Notch inhibition. Inner pillar cells and third-row Deiters cells were the major contributors to new hair cells after treatment of the damaged cochlea with a γ-secretase inhibitor. These cell types showed the highest expression of Lgr5 in neonatal animals in the previous study (Shi et al., 2012). They also maintained expression into adulthood.
Figure 7.
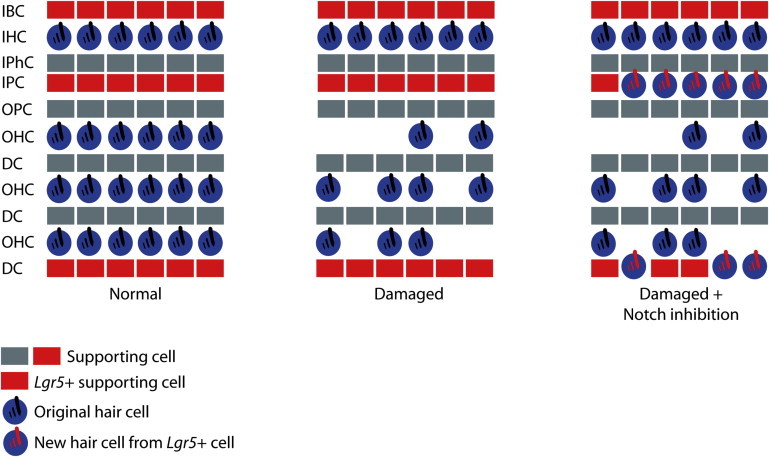
Schematic Showing the Normal Organ of Corti, the Organ of Corti following Gentamicin Damage, and the Organ of Corti following Gentamicin Damage and Notch Inhibition
Cell types are labeled as follows: inner border cell (IBC), inner hair cell (IHC), inner phalangeal cell (IPhC), inner pillar cell (IPC), outer pillar cell (OPC), outer hair cell (OHC), and Deiters cell (DC). Supporting cells are indicated in gray or red. Red supporting cells are Lgr5 positive. Original hair cells are indicated as blue cells with black stereocilia bundles. Following damage, outer hair cells are missing and are replaced spontaneously or after Notch inhibition by new hair cells derived from Lgr5-positive inner pillar and third Deiters cells (blue cells with red stereocilia bundles).
A recent study on hair cell differentiation in adult animals with hair cell loss after Notch inhibition did not identify the supporting cells that responded (Mizutari et al., 2013). The results we describe here are surprising, because we did not expect that the transdifferentiation of supporting cells to hair cells after Notch inhibition would be related to Lgr5 expression. The current work, however, implies that hair cell differentiation after Notch inhibition was not only due to overcoming the inhibition imposed by Notch on supporting cell conversion to hair cells but also was dependent on Wnt. Both cell division and differentiation resulted from forced expression of β-catenin in Lgr5-positive cells in the newborn cochlea in vivo (Shi et al., 2013), consistent with the known relationship between LGR5 expression and Wnt signaling (de Lau et al., 2011). The lack of a proliferative response despite the dependence on Wnt signaling seen here suggested a low level of signaling, because proliferation was predominant only at high levels of Wnt expression (Shi et al., 2013).
The capacity of inner pillar cells to transdifferentiate into hair cells appears to contradict findings that pillar cells, as compared to other supporting cells, resisted transdifferentiation in response to Notch inhibition (Doetzlhofer et al., 2009). Cells that remained after treatment with a γ-secretase inhibitor in that paper were identified as pillar cells (Figure 2A; yellow bracket in Doetzlhofer et al., 2009); however, the markers used to identify pillar cells (PROX1, p75ntr and HEY2) did not distinguish between inner and outer pillar cells. In addition, PROX1 is expressed in both pillar and Deiters cells (Bermingham-McDonogh et al., 2006). Our results would be consistent if the two rows of resistant cells were outer pillar and first-row Deiters cells rather than two rows of pillar cells. Due to their apparent disappearance following Notch inhibition, supporting cells have been assumed to be the source of these new hair cells (Doetzlhofer et al., 2009; Kiernan et al., 2005; Takebayashi et al., 2007), and identification of inner pillar cells as the source of most new hair cells is in agreement with previous data showing increased hair cell numbers and evidence of pillar cell division but no concomitant increase in pillar cell numbers in Notch mutants, suggesting possible transdifferentiation of pillar cells into new hair cells (Kiernan et al., 2005). Similarly, a population of sensory epithelial cells enriched for pillar and Hensen cells gave rise to significantly more hair cells than an unfractionated population of sensory epithelial cells (White et al., 2006), even without Notch inhibition.
Hair cells express Notch ligands, interacting with Notch receptors on neighboring cells, thus activating Hes family genes and blocking supporting cell conversion to a hair cell fate. Previous studies on the generation of new cochlear hair cells in embryonic and neonatal mammals have relied on deletion or inhibition of Notch (Doetzlhofer et al., 2009; Hayashi et al., 2008; Kiernan et al., 2005; Takebayashi et al., 2007; Yamamoto et al., 2006). Extra hair cells come from a combination of transdifferentiation and proliferation (Kiernan et al., 2005; Takebayashi et al., 2007), although proliferation was not observed in neonatal mice (Doetzlhofer et al., 2009; Kiernan et al., 2005; Takebayashi et al., 2007). The limited proliferation that we saw in neonatal explant cultures may have been facilitated by damage, which was not used in the studies on Notch inhibition. Although loss of Notch ligands due to damage or loss of hair cells could theoretically induce hair cells from neighboring cells, this has not appeared to be the case in the postnatal mammalian cochlea.
Notch and Wnt signaling are important for self-renewal and differentiation of adult stem cells in a variety of tissues. Notch signaling is required for the generation and maintenance of neural stem cells and proper control of neurogenesis and preserves neural stem cell characteristics in the adult brain (Piccin et al., 2013; Tanigaki et al., 2001). Notch and Wnt signaling regulate proliferation as well as acquisition of a mature phenotype by muscle, intestinal, and cardiac stem cells (Hirata et al., 2013; Klaus et al., 2012; Noah and Shroyer, 2013). The accurate expansion of Lgr5-expressing intestinal stem cells, with the correct timing of the switch to differentiation of goblet cells, in addition to being regulated by β-catenin (Barker et al., 2007; van Es et al., 2005a), is under Notch control (Stamataki et al., 2011; van Es et al., 2005b). The Lgr5-expressing progenitor cells identified in the cochlea are activated by Wnt signaling (Shi et al., 2012). Indeed, Wnt signaling has been shown to increase expression of a basic helix-loop-helix transcription factor, Atoh1, which is required for hair cell differentiation (Shi et al., 2010), and Wnt is necessary for upregulation of Atoh1 in response to Notch inhibition.
Inner pillar cells express Fgfr3, which may contribute to their capacity for regeneration (Jacques et al., 2007). Supporting cells in the chicken basilar papilla express Fgfr3, which is transiently downregulated during hair cell regeneration (Bermingham-McDonogh et al., 2001). Fgfr3−/− mice lack inner pillar cells in the apical and middle regions of the cochlea but exhibit an extra row of OHCs in these areas (Hayashi et al., 2007; Puligilla et al., 2007). Expression of both Lgr5 and Fgfr3 may be important for the transdifferentiation of cochlear cells after damage. We did not find any evidence of transdifferentiation in supporting cells that did not express Lgr5, suggesting that signaling through Lgr5 or Fgfr3 may be necessary to generate new hair cells with Notch inhibition. These studies therefore confirm the identification of Lgr5-expressing supporting cells as hair cell progenitors and show that damage to the newborn cochlea results in hair cell regeneration initiated by these Lgr5-positive cells.
Experimental Procedures
Animals
The Lgr5-EGFP-IRES-Cre- ER (Barker et al., 2007), β-cateninflox(exon2–6) (Brault et al., 2001), CAG-tdTomato (Madisen et al., 2010), and CAG-tdTomato-EGFP (Muzumdar et al., 2007) mice were obtained from The Jackson Laboratory. The Sox2-Cre-ER mouse was described previously (Arnold et al., 2011). The Pou4f3-Cre mouse (Sage et al., 2006) was a gift from Dr. Douglas Vetter (Tufts University, Boston, MA). The Sox2-Cre-ER and Pou4f3-Cre mice were genotyped with PCR using primers to amplify the Cre gene (forward: 5′-TGGGCGGCATGGTGCAAGTT-3′; reverse: 5′-CGGTGCTAACCAGCGTTTTC-3′). The Lgr5-EGFP-IRES-Cre-ER, CAG-tdTomato and CAG-tdTomato-EGFP mice were genotyped with PCR according to Jackson Laboratory recommendations. The β-cateninflox(exon2–6) mice were genotyped as described previously (Brault et al., 2001). All animal use was approved by the Massachusetts Eye and Ear Infirmary Animal Care Committee.
In Vitro Lineage Tracing
Sox2-Cre-ER mice were crossed with CAG-tdTomato-EGFP or CAG-tdTomato reporter mice. The CAG-tdTomato-EGFP reporter construct contains two loxP sites flanking both a STOP sequence and the red fluorescence gene tdTomato. Administration of tamoxifen induced Cre excision of the floxed region, allowing for permanent expression of EGFP. Lgr5-EGFP-IRES-Cre-ER and Pou4f3-Cre mice were crossed with CAG-tdTomato reporter mice. At P1 (or both P0 and P1 for Lgr5 lineage tracing), an intraperitoneal injection of 100 μl tamoxifen dissolved in corn oil (50 mg/ml) was administered to mothers of double-transgenic litters and transferred to the pups via the milk.
Organ of Corti Dissection
Mice positive for both Cre and the reporter construct were dissected at P2 and the cochlea was removed from the temporal bone. Cochleae for organ of Corti culture were transferred to Hanks’ balanced salt solution (Invitrogen) and the organ of Corti was isolated. The stria vascularis and basal hook region were removed and the organ of Corti was plated and cultured as described below. Cochleae not meant for culture were immediately fixed for 10 min in 4% paraformaldehyde (PFA; Electron Microscopy Sciences). The organ of Corti was then isolated and the stria vascularis and basal hook region removed. This was followed by additional fixation in 4% PFA for 30 min and then storage in 1× phosphate buffer solution at 4°C until staining.
Organ of Corti Culture
The organ of Corti was plated on a glass coverslip coated in poly-L-ornithine (Sigma) and laminin (BD Biosciences), given 100 μl Dulbecco’s modified Eagle’s medium (DMEM; Invitrogen) with 10% fetal bovine serum (FBS; Invitrogen) and 25 μg/ml ampicillin (Sigma), and cultured at 37°C with 5% CO2. One hour after dissection, cultures were treated with 50 μM gentamicin (Sigma) in DMEM with 10% FBS and ampicillin. The organ of Corti was cultured overnight (16 hr) and then placed in serum-free DMEM:F12 (Invitrogen) with 1% B27 supplement (Invitrogen), 25 μg/ml ampicillin, and either 5 μM dibenzazepine (deshydroxy LY411575; Santa Cruz) or 0.1% DMSO (Sigma) as a control. Undamaged controls were cultured under the same conditions without gentamicin or LY411575. LY411575 treatment continued for 72 hr. After 4 days in vitro, the cultures were fixed with 4% PFA (Electron Microscopy Sciences) and stained for MYO7A to identify hair cells and either DsRed or GFP to enhance the endogenous signal from the reporter.
To measure proliferation, cultures were treated with gentamicin as detailed above followed by 10 μM BrdU along with either LY411575 or 0.1% DMSO for 72 hr and then fixed, treated with 2 N HCl for 30 min at 37°C, and stained for BrdU.
Antibodies
The primary antibodies used in this study are as follows: anti-MYO7A (mouse, Developmental Studies Hybridoma Bank, 1:500; rabbit, Proteus Biosciences, 1:500), anti-BrdU (rat, ABD Serotec, 1:100), anti-GFP (rabbit, Invitrogen, 1:500), anti-SOX2 (goat, Santa Cruz Biotechnology, 1:500), anti-DsRed (rabbit, Clontech, 1:500), anti-PROX1 (mouse, Chemicon, 1:200), and anti-PRESTIN (goat, Santa Cruz Biotechnology, 1:400). Immunoreactivity was visualized using Alexa Fluor-conjugated secondary antibodies (Molecular Probes, 1:500).
Cell Counting
The organ of Corti was analyzed using a Leica TCS SP5 confocal microscope. Organ of Corti cross sections were obtained from confocal z stacks using Amira imaging software (Visage Imaging). High-magnification fluorescent images of the organs of Corti were merged in Photoshop and counted manually with ImageJ software (National Institutes of Health). Each organ of Corti was split into three regions for counting (apex, middle, and base), and hair cell counts were obtained per 100 μm. Hair cells in the pillar cell region were counted as OHCs. Each counted region was 1,900 ± 100 μm. Counts of reporter-positive hair cells for damaged and damaged/Notch-inhibited organs of Corti were corrected for residual recombination of native hair cells by subtracting the mean number of hair cells expressing the reporter in undamaged controls for each region (apex, middle, and base). Hair cell counts for the control and treated groups were compared using the Student’s t test.
Sox2 lineage tracing recombination rates were estimated by counting the percentage of inner pillar cells displaying reporter expression in each region. Lgr5 lineage tracing recombination rates were similarly calculated for inner pillar and third Deiters cells in each region.
Acknowledgments
We thank Drs. M. Charles Liberman, Cynthia Morton, and Matthew Kelley for helpful comments. This work was supported by grants RO1 DC007174, R21 DC010440, P30 DC05209, and R01 DK96034; an NIH Ruth L. Kirschstein National Research Service Award; and the Tillotson Corporation, the Shulsky Foundation, and Robert Boucai.
Footnotes
This is an open-access article distributed under the terms of the Creative Commons Attribution-NonCommercial-No Derivative Works License, which permits non-commercial use, distribution, and reproduction in any medium, provided the original author and source are credited.
Supplemental Information
References
- Arnold K., Sarkar A., Yram M.A., Polo J.M., Bronson R., Sengupta S., Seandel M., Geijsen N., Hochedlinger K. Sox2(+) adult stem and progenitor cells are important for tissue regeneration and survival of mice. Cell Stem Cell. 2011;9:317–329. doi: 10.1016/j.stem.2011.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balak K.J., Corwin J.T., Jones J.E. Regenerated hair cells can originate from supporting cell progeny: evidence from phototoxicity and laser ablation experiments in the lateral line system. J. Neurosci. 1990;10:2502–2512. doi: 10.1523/JNEUROSCI.10-08-02502.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barker N., van Es J.H., Kuipers J., Kujala P., van den Born M., Cozijnsen M., Haegebarth A., Korving J., Begthel H., Peters P.J., Clevers H. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature. 2007;449:1003–1007. doi: 10.1038/nature06196. [DOI] [PubMed] [Google Scholar]
- Bermingham-McDonogh O., Stone J.S., Reh T.A., Rubel E.W. FGFR3 expression during development and regeneration of the chick inner ear sensory epithelia. Dev. Biol. 2001;238:247–259. doi: 10.1006/dbio.2001.0412. [DOI] [PubMed] [Google Scholar]
- Bermingham-McDonogh O., Oesterle E.C., Stone J.S., Hume C.R., Huynh H.M., Hayashi T. Expression of Prox1 during mouse cochlear development. J. Comp. Neurol. 2006;496:172–186. doi: 10.1002/cne.20944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brault V., Moore R., Kutsch S., Ishibashi M., Rowitch D.H., McMahon A.P., Sommer L., Boussadia O., Kemler R. Inactivation of the beta-catenin gene by Wnt1-Cre-mediated deletion results in dramatic brain malformation and failure of craniofacial development. Development. 2001;128:1253–1264. doi: 10.1242/dev.128.8.1253. [DOI] [PubMed] [Google Scholar]
- de Lau W., Barker N., Low T.Y., Koo B.K., Li V.S., Teunissen H., Kujala P., Haegebarth A., Peters P.J., van de Wetering M. Lgr5 homologues associate with Wnt receptors and mediate R-spondin signalling. Nature. 2011;476:293–297. doi: 10.1038/nature10337. [DOI] [PubMed] [Google Scholar]
- Doetzlhofer A., Basch M.L., Ohyama T., Gessler M., Groves A.K., Segil N. Hey2 regulation by FGF provides a Notch-independent mechanism for maintaining pillar cell fate in the organ of Corti. Dev. Cell. 2009;16:58–69. doi: 10.1016/j.devcel.2008.11.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forge A., Li L., Corwin J.T., Nevill G. Ultrastructural evidence for hair cell regeneration in the mammalian inner ear. Science. 1993;259:1616–1619. doi: 10.1126/science.8456284. [DOI] [PubMed] [Google Scholar]
- Hayashi T., Cunningham D., Bermingham-McDonogh O. Loss of Fgfr3 leads to excess hair cell development in the mouse organ of Corti. Dev. Dyn. 2007;236:525–533. doi: 10.1002/dvdy.21026. [DOI] [PubMed] [Google Scholar]
- Hayashi T., Kokubo H., Hartman B.H., Ray C.A., Reh T.A., Bermingham-McDonogh O. Hesr1 and Hesr2 may act as early effectors of Notch signaling in the developing cochlea. Dev. Biol. 2008;316:87–99. doi: 10.1016/j.ydbio.2008.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirata A., Utikal J., Yamashita S., Aoki H., Watanabe A., Yamamoto T., Okano H., Bardeesy N., Kunisada T., Ushijima T. Dose-dependent roles for canonical Wnt signalling in de novo crypt formation and cell cycle properties of the colonic epithelium. Development. 2013;140:66–75. doi: 10.1242/dev.084103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacques B.E., Montcouquiol M.E., Layman E.M., Lewandoski M., Kelley M.W. Fgf8 induces pillar cell fate and regulates cellular patterning in the mammalian cochlea. Development. 2007;134:3021–3029. doi: 10.1242/dev.02874. [DOI] [PubMed] [Google Scholar]
- Kiernan A.E., Cordes R., Kopan R., Gossler A., Gridley T. The Notch ligands DLL1 and JAG2 act synergistically to regulate hair cell development in the mammalian inner ear. Development. 2005;132:4353–4362. doi: 10.1242/dev.02002. [DOI] [PubMed] [Google Scholar]
- Klaus A., Müller M., Schulz H., Saga Y., Martin J.F., Birchmeier W. Wnt/β-catenin and Bmp signals control distinct sets of transcription factors in cardiac progenitor cells. Proc. Natl. Acad. Sci. USA. 2012;109:10921–10926. doi: 10.1073/pnas.1121236109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lanford P.J., Lan Y., Jiang R., Lindsell C., Weinmaster G., Gridley T., Kelley M.W. Notch signalling pathway mediates hair cell development in mammalian cochlea. Nat. Genet. 1999;21:289–292. doi: 10.1038/6804. [DOI] [PubMed] [Google Scholar]
- Lin V., Golub J.S., Nguyen T.B., Hume C.R., Oesterle E.C., Stone J.S. Inhibition of Notch activity promotes nonmitotic regeneration of hair cells in the adult mouse utricles. J. Neurosci. 2011;31:15329–15339. doi: 10.1523/JNEUROSCI.2057-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madisen L., Zwingman T.A., Sunkin S.M., Oh S.W., Zariwala H.A., Gu H., Ng L.L., Palmiter R.D., Hawrylycz M.J., Jones A.R. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat. Neurosci. 2010;13:133–140. doi: 10.1038/nn.2467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mizutari K., Fujioka M., Hosoya M., Bramhall N., Okano H.J., Okano H., Edge A.S.B. Notch inhibition induces cochlear hair cell regeneration and recovery of hearing after acoustic trauma. Neuron. 2013;77:58–69. doi: 10.1016/j.neuron.2012.10.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muzumdar M.D., Tasic B., Miyamichi K., Li L., Luo L. A global double-fluorescent Cre reporter mouse. Genesis. 2007;45:593–605. doi: 10.1002/dvg.20335. [DOI] [PubMed] [Google Scholar]
- Noah T.K., Shroyer N.F. Notch in the intestine: regulation of homeostasis and pathogenesis. Annu. Rev. Physiol. 2013;75:263–288. doi: 10.1146/annurev-physiol-030212-183741. [DOI] [PubMed] [Google Scholar]
- Piccin D., Yu F., Morshead C. Notch signaling imparts and preserves neural stem characteristics in the adult brain. Stem Cells Dev. 2013;22:1541–1550. doi: 10.1089/scd.2012.0390. [DOI] [PubMed] [Google Scholar]
- Puligilla C., Feng F., Ishikawa K., Bertuzzi S., Dabdoub A., Griffith A.J., Fritzsch B., Kelley M.W. Disruption of fibroblast growth factor receptor 3 signaling results in defects in cellular differentiation, neuronal patterning, and hearing impairment. Dev. Dyn. 2007;236:1905–1917. doi: 10.1002/dvdy.21192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raphael Y. Evidence for supporting cell mitosis in response to acoustic trauma in the avian inner ear. J. Neurocytol. 1992;21:663–671. doi: 10.1007/BF01191727. [DOI] [PubMed] [Google Scholar]
- Rubel E.W., Dew L.A., Roberson D.W. Mammalian vestibular hair cell regeneration. Science. 1995;267:701–707. doi: 10.1126/science.7839150. [DOI] [PubMed] [Google Scholar]
- Sage C., Huang M., Vollrath M.A., Brown M.C., Hinds P.W., Corey D.P., Vetter D.E., Chen Z.Y. Essential role of retinoblastoma protein in mammalian hair cell development and hearing. Proc. Natl. Acad. Sci. USA. 2006;103:7345–7350. doi: 10.1073/pnas.0510631103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi F., Cheng Y.F., Wang X.L., Edge A.S. Beta-catenin up-regulates Atoh1 expression in neural progenitor cells by interaction with an Atoh1 3′ enhancer. J. Biol. Chem. 2010;285:392–400. doi: 10.1074/jbc.M109.059055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi F., Kempfle J.S., Edge A.S. Wnt-responsive Lgr5-expressing stem cells are hair cell progenitors in the cochlea. J. Neurosci. 2012;32:9639–9648. doi: 10.1523/JNEUROSCI.1064-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi F., Hu L., Edge A.S. Generation of hair cells in neonatal mice by β-catenin overexpression in Lgr5-positive cochlear progenitors. Proc. Natl. Acad. Sci. USA. 2013;110:13851–13856. doi: 10.1073/pnas.1219952110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stamataki D., Holder M., Hodgetts C., Jeffery R., Nye E., Spencer-Dene B., Winton D.J., Lewis J. Delta1 expression, cell cycle exit, and commitment to a specific secretory fate coincide within a few hours in the mouse intestinal stem cell system. PLoS ONE. 2011;6:e24484. doi: 10.1371/journal.pone.0024484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stone J.S., Cotanche D.A. Identification of the timing of S phase and the patterns of cell proliferation during hair cell regeneration in the chick cochlea. J. Comp. Neurol. 1994;341:50–67. doi: 10.1002/cne.903410106. [DOI] [PubMed] [Google Scholar]
- Stone J.S., Rubel E.W. Temporal, spatial, and morphologic features of hair cell regeneration in the avian basilar papilla. J. Comp. Neurol. 2000;417:1–16. doi: 10.1002/(sici)1096-9861(20000131)417:1<1::aid-cne1>3.0.co;2-e. [DOI] [PubMed] [Google Scholar]
- Takebayashi S., Yamamoto N., Yabe D., Fukuda H., Kojima K., Ito J., Honjo T. Multiple roles of Notch signaling in cochlear development. Dev. Biol. 2007;307:165–178. doi: 10.1016/j.ydbio.2007.04.035. [DOI] [PubMed] [Google Scholar]
- Tanigaki K., Nogaki F., Takahashi J., Tashiro K., Kurooka H., Honjo T. Notch1 and Notch3 instructively restrict bFGF-responsive multipotent neural progenitor cells to an astroglial fate. Neuron. 2001;29:45–55. doi: 10.1016/s0896-6273(01)00179-9. [DOI] [PubMed] [Google Scholar]
- van Es J.H., Jay P., Gregorieff A., van Gijn M.E., Jonkheer S., Hatzis P., Thiele A., van den Born M., Begthel H., Brabletz T. Wnt signalling induces maturation of Paneth cells in intestinal crypts. Nat. Cell Biol. 2005;7:381–386. doi: 10.1038/ncb1240. [DOI] [PubMed] [Google Scholar]
- van Es J.H., van Gijn M.E., Riccio O., van den Born M., Vooijs M., Begthel H., Cozijnsen M., Robine S., Winton D.J., Radtke F., Clevers H. Notch/gamma-secretase inhibition turns proliferative cells in intestinal crypts and adenomas into goblet cells. Nature. 2005;435:959–963. doi: 10.1038/nature03659. [DOI] [PubMed] [Google Scholar]
- Warchol M.E., Corwin J.T. Regenerative proliferation in organ cultures of the avian cochlea: identification of the initial progenitors and determination of the latency of the proliferative response. J. Neurosci. 1996;16:5466–5477. doi: 10.1523/JNEUROSCI.16-17-05466.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warchol M.E., Lambert P.R., Goldstein B.J., Forge A., Corwin J.T. Regenerative proliferation in inner ear sensory epithelia from adult guinea pigs and humans. Science. 1993;259:1619–1622. doi: 10.1126/science.8456285. [DOI] [PubMed] [Google Scholar]
- White P.M., Doetzlhofer A., Lee Y.S., Groves A.K., Segil N. Mammalian cochlear supporting cells can divide and trans-differentiate into hair cells. Nature. 2006;441:984–987. doi: 10.1038/nature04849. [DOI] [PubMed] [Google Scholar]
- Yamamoto N., Tanigaki K., Tsuji M., Yabe D., Ito J., Honjo T. Inhibition of Notch/RBP-J signaling induces hair cell formation in neonate mouse cochleas. J. Mol. Med. 2006;84:37–45. doi: 10.1007/s00109-005-0706-9. [DOI] [PubMed] [Google Scholar]
- Zheng J., Shen W., He D.Z., Long K.B., Madison L.D., Dallos P. Prestin is the motor protein of cochlear outer hair cells. Nature. 2000;405:149–155. doi: 10.1038/35012009. [DOI] [PubMed] [Google Scholar]
- Zine A., Van De Water T.R., de Ribaupierre F. Notch signaling regulates the pattern of auditory hair cell differentiation in mammals. Development. 2000;127:3373–3383. doi: 10.1242/dev.127.15.3373. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


