Abstract
OBJECTIVE
Serum cystatin C is an alternative to serum creatinine for estimating glomerular filtration rate (GFR), since cystatin C is less influenced by age and muscle mass. Among persons with diabetes, we compared the performance of GFR estimated using cystatin C (eGFRcys) with that using creatinine (eGFRcr) for the identification of reduced kidney function and its association with diabetes complications.
RESEARCH DESIGN AND METHODS
We analyzed data from adult participants from the 1999–2002 National Health and Nutrition Examination Survey with available cystatin C (N = 4,457). Kidney function was dichotomized as preserved (eGFR ≥60 mL/min/1.73 m2) or reduced (eGFR <60 mL/min/1.73 m2) using the 2012 Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) cystatin C and the 2009 CKD-EPI creatinine equations.
RESULTS
Among 778 persons with diabetes, the prevalence of reduced kidney function was 16.5% using eGFRcr and 22.0% using eGFRcys. More persons with diabetes were reclassified from preserved kidney function by eGFRcr to reduced kidney function by eGFRcys than persons without diabetes (odds ratio 3.1 [95% CI 1.9–4.9], P < 0.001). The associations between lower eGFR and higher prevalence of albuminuria, retinopathy, peripheral arterial disease, and coronary artery disease were robust regardless of filtration marker. Similarly, the risk of all-cause mortality increased with lower eGFRcr and eGFRcys. Only lower eGFRcys was significantly associated with cardiovascular mortality.
CONCLUSIONS
More persons with diabetes had reduced kidney function by eGFRcys than by eGFRcr, and lower eGFRcys was strongly associated with diabetes complications. Whether eGFRcys is superior to eGFRcr in approximating true kidney function in a diabetic population requires additional study.
Introduction
Diabetes is the leading cause of chronic kidney disease (CKD) in developed countries, including the U.S. (1,2). Diabetic kidney disease accounts for 40% of prevalent CKD and 50% of incident end-stage renal disease, and it has increased in direct proportion to the increasing prevalence of diabetes (2–4). People with diabetes often suffer from microvascular and macrovascular complications, including retinopathy, nephropathy, coronary artery disease, peripheral arterial disease, and stroke, as well as early mortality (5,6). Compared with persons with diabetes and preserved kidney function, those with diabetes and CKD face even higher risks of morbidity and mortality. Indeed, both reduced kidney function and albuminuria are independent predictors for cardiovascular disease as well as all-cause mortality (7,8). Accurate estimation of glomerular filtration rate (GFR) and identification of CKD are important.
In clinical practice, kidney function is estimated rather than measured. Glomerular filtration rate estimated using serum creatinine (eGFRcr) is the most common approach; however, creatinine is influenced by age, muscle mass, sex, and race (9). Given these limitations, serum cystatin C has been proposed as an alternative filtration marker (10). Cystatin C, an endogenous protein believed to be produced by all nucleated cells, is less affected by age, race, and muscle mass and, in the general population, associates more strongly with all-cause and cardiovascular mortality than does serum creatinine (11,12). However, BMI, diabetes, and inflammation may affect cystatin C levels independent of kidney function (13). Given the high prevalence of obesity in the population with diabetes, as well as the suggestion that cystatin may perform differently in patients with diabetes, there is controversy as to whether cystatin C–based or creatinine-based estimated GFR (eGFR) equations should be used to estimate kidney function in this population (14,15). Furthermore, it is unknown whether the associations of diabetes complications with kidney function estimated using cystatin C (eGFRcys) are similar to those observed using eGFRcr.
Using nationally representative data from the 1999–2002 National Health and Nutrition Examination Survey (NHANES), we estimated the prevalence of reduced kidney function (eGFR <60 mL/min/1.73 m2) among persons with diabetes using the 2012 CKD-EPI cystatin C (16) and 2009 CKD-EPI creatinine (17) equations and investigated the discordance in CKD classification by the two filtration markers. We also compared the associations of eGFRcr and eGFRcys with prevalent complications of diabetes, including albuminuria, peripheral arterial disease, retinopathy, and coronary artery disease, as well as incident all-cause and cardiovascular mortality.
Research Design and Methods
Study Population
NHANES is an ongoing cross-sectional, multistage, stratified, clustered probability sample of the U.S. civilian noninstitutionalized population conducted by the National Center for Health Statistics. Cystatin C concentrations were measured in a subsample of the NHANES 1999–2002 participants aged 12 years and older who were not missing serum creatinine (18). For the current study, we included all participants in the cystatin C subsample aged 20 years or older (N = 4,457; 778 of whom had diabetes). The ankle-brachial index (ABI), used to define peripheral arterial disease, was measured only in persons aged 40 years or older (N = 556 of the 778 participants with diabetes, 10 of whom with ABI >1.5 were excluded owing to concern for calcified atherosclerosis) (19,20).
Assessment of Diabetes and Kidney Function
We classified persons as having diabetes if they reported a physician diagnosis of diabetes, took antidiabetes pills or insulin injections, or had a glycated hemoglobin (HbA1c) value of ≥6.5%. No distinction was made with regard to type of diabetes. Preserved/reduced kidney function was defined as eGFR ≥60 mL/min/1.73 m2 / eGFR <60 mL/min/1.73 m2, estimated using standardized creatinine and cystatin C values and the CKD-EPI 2009 and 2012 equations, respectively (16,17). The term “advanced CKD” was used to indicate CKD stage 4 or 5 (eGFR <30 mL/min/1.73 m2). Creatinine values from NHANES 1999–2000 were standardized [standard creatinine (mg/dL) = 0.147 + 1.013 × (NHANES 1999–2000 uncalibrated serum creatinine [mg/dL]), whereas no correction to the creatinine values in the 2001–2002 survey was needed (21,22). Cystatin C values were recalibrated and standardized to the International Federation of Clinical Chemistry and Laboratory Medicine (IFCC) standard: IFCC standard cystatin C (mg/L) = 1.12 × (cystatin C [mg/L] − 0.12) (22).
Other Variables of Interest
Hypertension was defined as mean systolic pressure ≥140 mmHg, mean diastolic pressure ≥90 mmHg, self-reported hypertension, or the use of an antihypertensive medication. Hyperlipidemia was defined as total cholesterol concentration ≥200 mg/dL. Low HDL was defined as serum HDL <40 mg/dL. Information on age, sex, race, and smoking was self-reported. Smoking status was determined using answers to the questions, “Have you smoked at least 100 cigarettes in your life?” and “Do you now smoke cigarettes?”
Prevalent Micro- and Macrovascular Outcomes
Coronary artery disease was defined on the basis of a self-reported history of coronary heart disease, angina, or previous heart attack. Albuminuria was defined as a urinary albumin-to-creatinine ratio (ACR) ≥30 mg/g. Peripheral arterial disease was defined by an ABI <0.90 in either leg (23). Diabetic retinopathy was self-reported (“Has a doctor ever told you that diabetes has affected your eyes or that you had retinopathy?”).
Mortality Follow-up
Information on all-cause and cardiovascular mortality was obtained using the linkage of NHANES data to death certificate data from the National Death Index (24). The underlying cause of death was coded according to the ICD-10. Outcomes of interest included all-cause and cardiovascular (ICD-10 code I00–I78) mortality. Length of follow-up for each participant was calculated as the date of the NHANES examination to date of death or 31 December 2006 (whichever occurred first).
Statistical Analyses
All statistical analyses incorporated modified sampling weights, primary sampling units, and strata specific to the sample with available cystatin C in order to generate nationally representative estimates of the U.S. population (18). SEs were estimated using the Taylor series (linearization) method. Kidney function was analyzed both as a continuous measure using restricted cubic splines and as a categorical measure according to K/DOQI classification (eGFR <15, 15–29, 30–59, 60–89, and 90–200 mL/min/1.73 m2) for both eGFRcr and eGFRcys. Individuals with eGFR values >200 mL/min/1.73 m2 were reassigned a value of 200 mL/min/1.73 m2 (two persons with eGFRcr >200 mL/min/1.73 m2).
Modified Poisson regression models were used to examine the relationship of eGFR with the prevalence of coronary artery disease, peripheral arterial disease, albuminuria, and retinopathy (25). Associations of eGFR and mortality were examined using multivariable Cox proportional hazards models. The proportional hazards assumption was tested using log-log plots by category of eGFR. All multivariable models were adjusted for age (years), female sex (yes, no), and race (non-Hispanic black, non-Hispanic white, Hispanic, other) as well as current smoking status (current, former, never), BMI (measured as weight in kilograms divided by the square of height in meters), hypercholesterolemia (yes, no), low HDL (yes, no), hypertension (yes, no), coronary artery disease (yes, no), and albuminuria (yes, no; only in analyses where albuminuria was not the outcome). Given the possible relationship between cystatin C and obesity, interactions between BMI, eGFRcys, and adverse outcomes were tested in multivariable models. All analyses were conducted using Stata, version 12 (Stata, College Station, TX).
Results
Study Population
The prevalence of diabetes among U.S. adults was 8.2% (95% CI 7.4–9.0). Persons with diabetes were older, more likely to be black, and more likely to be male than those without diabetes (Table 1). Persons with diabetes also had poorer lipid profiles. Two-thirds of persons with diabetes had hypertension, which was nearly twice as prevalent compared with those without diabetes (63.8% vs. 33.4%). There was also a substantial difference in the distribution of BMI: 50% of the U.S. population with diabetes had a BMI >30 kg/m2 compared with 28% without diabetes, and 28% of persons with diabetes had a BMI ≥35 kg/m2 compared with 12% of those without diabetes. The prevalence of reduced kidney function was almost three times higher in persons with diabetes compared with those without diabetes (eGFRcr 16.5% vs. 5.8%, eGFRcys 22.0% vs. 7.9%).
Table 1.
Characteristics of U.S. adults aged ≥20 years by diabetes status: NHANES 1999–2002 cystatin C subsample (N = 4,457)
Prevalence of Reduced Kidney Function in Persons With Diabetes by Filtration Marker
The trend of higher prevalence of reduced kidney function by eGFRcys persisted across subgroups of sex, race, age, and BMI (Supplementary Fig. 1). On the absolute scale, the discrepancy in eGFRcys versus eGFRcr was largest in older individuals. The absolute difference in reduced kidney function prevalence estimated by cystatin C versus creatinine was 6.9% in those aged 60–80 years and 10.3% in those aged 80 years and older. Similarly, the absolute difference was larger among persons with BMI >30 kg/m2 (reduced kidney function by eGFRcys vs. eGFRcr: 20.0% vs. 13.1%) than among those with BMI <30 kg/m2 (reduced kidney function by eGFRcys vs. eGFRcr: 21.3% vs. 16.9%).
Reclassification of Reduced Kidney Function by Filtration Marker
Discordance between eGFRcr and eGFRcys in the classification of reduced kidney function was 11.8% in persons with diabetes and 4.7% in persons without diabetes. In the population with diabetes, 10.4% of those classified as having preserved kidney function by eGFRcr (8.7% of the 83.6% with eGFRcr ≥60 mL/min/1.73 m2) were reclassified as having reduced kidney function by eGFRcys (Supplementary Table 1). In the population without diabetes, 3.6% of those classified as having preserved kidney function by eGFRcr (3.4% of the 94.2% with eGFRcr ≥60 mL/min/1.73 m2) were reclassified as having reduced kidney function by eGFRcys (Supplementary Table 1). Within the overall population, diabetes status was significantly associated with reclassification from preserved kidney function by eGFRcr to reduced kidney function by eGFRcys (odds ratio [OR] 3.1 [95% CI 1.9–4.9], P < 0.001) (Supplementary Table 2). This association was attenuated but still significant after sequential adjustment for eGFRcr and BMI but not after further adjustment for age. In contrast, eGFRcr, BMI, age, and albuminuria were all significantly associated with reclassification by eGFRcys in multivariable regression (eGFRcr, OR 0.9 per 1 mL/min/1.73 m2 increase, P < 0.001; BMI, OR 1.5 per 5 kg/m2 increase, P < 0.001; age, OR 1.8 per decade increase, P < 0.001; ACR >30 mg/g, OR 2.2, P = 0.01).
Association of Kidney Function With Micro- and Macrovascular Complications by Filtration Marker
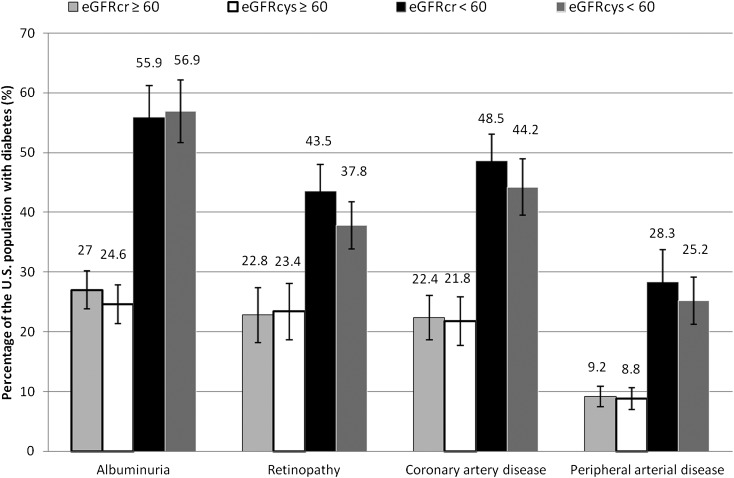
In the population with diabetes, the prevalence of albuminuria, retinopathy, coronary artery disease, and peripheral arterial disease was high, even among those with preserved kidney function (Fig. 1). The most common of these complications was albuminuria, present in 27.0% of those with eGFRcr ≥60 mL/min/1.73 m2 and 55.9% of those with eGFRcr <60 mL/min/1.73 m2. The prevalence of coronary artery disease, retinopathy, and peripheral artery disease was also two to three times higher among those with reduced kidney function than those with preserved kidney function. In general, the probability of microvascular and macrovascular complications increased with lower eGFR; above eGFR 90 mL/min/1.73 m2, relationships between eGFR and vascular complications were more variable. The unadjusted relationships between eGFR, coronary artery disease, peripheral arterial disease, albuminuria, and retinopathy were similar using eGFRcr and eGFRcys, although there was suggestion of a “U-shape” (with higher risk at both higher and lower levels of GFR) in the association between albuminuria, retinopathy, and eGFRcr but not eGFRcys (Supplementary Fig. 2A–D).
Figure 1.
Prevalence of microvascular and macrovascular conditions among U.S. adults with diabetes according to the presence or absence of reduced kidney function (estimated using creatinine and cystatin C).
After adjustment for demographic and traditional cardiovascular risk factors, the prevalence ratios for vascular complications (coronary artery disease, peripheral arterial disease, albuminuria, retinopathy) by eGFR category were similar using creatinine or cystatin C (Table 2). Compared with a reference group of eGFR 60–90 mL/min/1.73 m2, persons with advanced CKD (eGFR 15–30 mL/min/1.73 m2) had a higher prevalence of coronary artery disease, albuminuria, and retinopathy, regardless of filtration marker used. The adjusted relationships between eGFR category and peripheral arterial disease were similar in magnitude but not statistically significant. There were no significant interactions between BMI, eGFRcys, and vascular complications.
Table 2.
Adjusted prevalence ratios (95% CI) for categories of kidney function with complications of diabetes, by filtration marker, among U.S. adults with diabetes (N = 778)*
All-Cause and Cardiovascular Mortality in Those With and Without Reduced Kidney Function
There were 153 deaths (63 from a cardiovascular cause) during a median follow-up of 5.3 years among the 778 participants with diabetes. The risk of both all-cause and cardiovascular mortality increased with lower eGFR, regardless of filtration marker used (Table 3). Compared with the reference group of eGFR 60–90 mL/min/1.73 m2, persons with eGFR 15–30 mL/min/1.73 m2 had a significantly higher risk of all-cause mortality (eGFRcys, hazard ratio [HR] 3.8, P = 0.007; eGFRcr, HR 2.6, P = 0.04). By contrast, eGFR 15–30 mL/min/1.73 m2 was significantly associated with increased cardiovascular mortality when estimated by cystatin C (HR 5.3, P = 0.007) but not by creatinine (HR 2.0, P = 0.3). There were no significant interactions between BMI, eGFRcys, and either all-cause or cardiovascular mortality.
Table 3.
Adjusted hazard ratios (95% CI) for categories of kidney function with all-cause and cardiovascular mortality, by filtration marker, among U.S. adults with diabetes (N = 778)*
Conclusions
This nationally representative study of persons with diabetes suggests that the use of cystatin C to estimate kidney function would result in a higher prevalence of reduced kidney function than would estimates using serum creatinine. Reclassification from preserved kidney function using creatinine to reduced kidney function using cystatin C occurred more commonly among persons with diabetes than those without, but this observation was explained by differences in the distributions of eGFR, BMI, age, and albuminuria between the two populations: reclassification was significantly associated with lower eGFRcr, higher BMI, older age, and ACR >30 mg/g. Lower eGFR as determined by either creatinine or cystatin was associated with higher odds of prevalent vascular complications; however, the shape of the relationship with albuminuria and retinopathy at higher levels of eGFR differed slightly by filtration marker in unadjusted analysis. Similarly, while low eGFR was robustly associated with all-cause mortality, only eGFRcys showed significant association with cardiovascular mortality.
Differences in eGFRcr and eGFRcys have been noted previously in the general population (26,27). In the U.S. noninstitutionalized civilian population, kidney function estimated using cystatin C resulted in a reduced kidney function prevalence of 8.7% compared with an estimated 6.5% using creatinine (26). In a cross-sectional study of 1,360 inhabitants of the Alpine region in Europe, Pattaro et al. (28) noted that the Lin concordance correlation coefficient of eGFRcr and eGFRcys was 0.56, with significant differences by age (0.57 in those ≥65 years old vs. 0.38 in those <65 years old) but not by diabetes status. Our results differ somewhat from those of this prior study; the presence of diabetes differentially affected kidney disease classification by eGFRcr and eGFRcys, at least in univariable analysis—an observation that may be attributable to our larger sample size and distinct American population.
Neither creatinine nor cystatin C is a perfect marker of glomerular filtration; each has non-GFR determinants. Some have argued that neither marker can adequately estimate true GFR in persons with diabetes; however, this concern is primarily relevant for those with high GFR—not reduced GFR as in the current study (29,30). The strong relationship between reclassification to reduced kidney function by eGFRcys and age may be due to inherent properties of the filtration markers. In the case of creatinine, muscle mass and diet are significantly associated with creatinine levels (31). Older persons may be sicker than their younger peers; thus, kidney function estimated using serum creatinine may be confounded by cachexia and muscle wasting. A similar explanation could apply to the differences seen with albuminuria (i.e., those with albuminuria are sicker than their peers without albuminuria). To our knowledge, the observation that reclassification by cystatin C occurs more frequently among those with albuminuria is novel; however, it is fully consistent with a recent study demonstrating that the decrement in eGFRcys associated with 24-h albuminuria >30 mg was greater than that of eGFRcr or measured GFR (27).
In the population with diabetes, both serum creatinine and cystatin C may have drawbacks. Serum creatinine may poorly estimate kidney function given the tendency of persons with diabetes to have a lower than average muscle mass. Cystatin C may be directly affected by both BMI and diabetes (13,32,33). In obese individuals, cystatin C levels are higher, and eGFRcys significantly underestimates true kidney function (27,31). Indeed, our study suggests that much of the association between diabetes and cystatin C is driven by differences in the distribution of age and BMI. Additional work is needed to determine whether an approach using cystatin C or both filtration markers (the latter of which better approximates measured GFR in the overall population) would improve kidney function estimates in the population with diabetes (16).
Conventional wisdom is that, among persons with diabetes, kidney function decline and vascular complications go hand in hand. Certainly, our results support the association of prevalent complications with very low eGFR whether estimated by creatinine or cystatin C. Interestingly, in the upper ranges of preserved kidney function, the association between eGFRcr and retinopathy reversed, with higher levels of eGFRcr conferring increased odds of retinopathy, although this was not statistically significant in adjusted analysis. A similar pattern was seen in the univariate association of eGFRcr and albuminuria. These observations are consistent with previous studies demonstrating weaker-than-expected correlation between eGFRcr, albuminuria, and retinopathy (34). Because of the more monotonic relationship seen between eGFRcys, albuminuria, and retinopathy, it is possible that kidney function based on cystatin C may prove a better predictor of diabetes complications than that based on creatinine. However, this may be more useful in defining a low-risk group than a high-risk group, given the larger differences in the upper ranges of eGFR. We also observed that the relationship between eGFRcys and all-cause and cardiovascular mortality was stronger than the corresponding relationship with eGFRcr, similar to findings in the general population (11,12,35) and previous studies of persons with diabetes (36). Additional prospective studies are needed to determine whether eGFRcys provides better risk stratification for subsequent diabetes complications.
Our study has certain limitations. We relied on a single measurement of creatinine and cystatin; GFR is estimated from these filtration markers and not measured directly. As such, we cannot assess which filtration marker most closely approximates true kidney function. Additional research is needed to determine whether the confounding by age and BMI (or other unmeasured confounders such as thyroid disease) in diabetes favors eGFRcr, eGFRcys, or perhaps a combination of the two. Additionally, a single random sample of urine was used to quantify albuminuria. Next, despite being nationally representative, the subset of NHANES with diabetes was relatively small, with a short duration of follow-up for mortality outcomes. Some of the vascular complications were self-reported, and insofar as reporting may vary by level of eGFR, this may lead to bias. Finally, persons with more severe kidney disease are likely underrepresented in NHANES, thus limiting accuracy in the very low ranges of GFR.
In summary, this study demonstrates that using cystatin C to classify kidney function among persons with diabetes results in a higher prevalence of reduced kidney function yet the same or stronger associations with vascular complications and mortality. Future studies are needed to determine whether incorporating cystatin C measurement into clinical care and kidney function estimation would improve outcomes in persons with diabetes.
Supplementary Material
Article Information
Funding. This study was supported by the National Kidney Foundation (grant to E.S. at Johns Hopkins) and Merck Sharp and Dohme Corp., a subsidiary of Merck & Co, Inc., Whitehouse Station, NJ (professional service agreement to the National Kidney Foundation). M.E.G. receives support from the National Institute of Diabetes and Digestive and Kidney Diseases (K08-DK-092287).
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
Author Contributions. C.-W.T. and M.E.G. researched data and wrote the manuscript. L.A.I. and J.C. reviewed and edited the manuscript. E.S. contributed to the discussion and reviewed and edited the manuscript. M.E.G. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
This article contains Supplementary Data online at http://care.diabetesjournals.org/lookup/suppl/doi:10.2337/dc13-1910/-/DC1.
References
- 1.National Kidney Foundation KDOQI clinical practice guideline for diabetes and CKD: 2012 update. Am J Kidney Dis 2012;60:850–886 [DOI] [PubMed] [Google Scholar]
- 2.U.S. Renal Data System. USRDS 2012 Annual Data Report: Atlas of End-Stage Renal Disease in the United States Bethesda, MD, National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, 2012 [Google Scholar]
- 3.de Boer IH, Rue TC, Hall YN, Heagerty PJ, Weiss NS, Himmelfarb J. Temporal trends in the prevalence of diabetic kidney disease in the United States. JAMA 2011;305:2532–2539 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cowie CC, Rust KF, Byrd-Holt DD, et al. Prevalence of diabetes and high risk for diabetes using A1C criteria in the U.S. population in 1988-2006. Diabetes Care 2010;33:562–568 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Goff DC, Jr, Gerstein HC, Ginsberg HN, et al. ACCORD Study Group Prevention of cardiovascular disease in persons with type 2 diabetes mellitus: current knowledge and rationale for the Action to Control Cardiovascular Risk in Diabetes (ACCORD) trial. Am J Cardiol 2007;99:4i–20i [DOI] [PubMed] [Google Scholar]
- 6.Tandon N, Ali MK, Narayan KM. Pharmacologic prevention of microvascular and macrovascular complications in diabetes mellitus: implications of the results of recent clinical trials in type 2 diabetes. Am J Cardiovasc Drugs 2012;12:7–22 [DOI] [PubMed] [Google Scholar]
- 7.Matsushita K, van der Velde M, Astor BC, et al. Chronic Kidney Disease Prognosis Consortium Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: a collaborative meta-analysis. Lancet 2010;375:2073–2081 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Fox CS, Matsushita K, Woodward M, et al. Chronic Kidney Disease Prognosis Consortium Associations of kidney disease measures with mortality and end-stage renal disease in individuals with and without diabetes: a meta-analysis. Lancet 2012;380:1662–1673 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ferguson MA, Waikar SS. Established and emerging markers of kidney function. Clin Chem 2012;58:680–689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Stevens LA, Coresh J, Schmid CH, et al. Estimating GFR using serum cystatin C alone and in combination with serum creatinine: a pooled analysis of 3,418 individuals with CKD. Am J Kidney Dis 2008;51:395–406 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Astor BC, Levey AS, Stevens LA, Van Lente F, Selvin E, Coresh J. Method of glomerular filtration rate estimation affects prediction of mortality risk. J Am Soc Nephrol 2009;20:2214–2222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shlipak MG, Wassel Fyr CL, Chertow GM, et al. Cystatin C and mortality risk in the elderly: the health, aging, and body composition study. J Am Soc Nephrol 2006;17:254–261 [DOI] [PubMed] [Google Scholar]
- 13.Stevens LA, Schmid CH, Greene T, et al. Factors other than glomerular filtration rate affect serum cystatin C levels. Kidney Int 2009;75:652–660 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Iliadis F, Didangelos T, Ntemka A, et al. Glomerular filtration rate estimation in patients with type 2 diabetes: creatinine- or cystatin C-based equations? Diabetologia 2011;54:2987–2994 [DOI] [PubMed] [Google Scholar]
- 15.Silveiro SP, Araújo GN, Ferreira MN, Souza FD, Yamaguchi HM, Camargo EG. Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation pronouncedly underestimates glomerular filtration rate in type 2 diabetes. Diabetes Care 2011;34:2353–2355 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Inker LA, Schmid CH, Tighiouart H, et al. CKD-EPI Investigators Estimating glomerular filtration rate from serum creatinine and cystatin C. N Engl J Med 2012;367:20–29 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Levey AS, Stevens LA, Schmid CH, et al. CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration) A new equation to estimate glomerular filtration rate. Ann Intern Med 2009;150:604–612 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Köttgen A, Selvin E, Stevens LA, Levey AS, Van Lente F, Coresh J. Serum cystatin C in the united states: The third national health and nutrition examination survey (NHANES III). Am J Kidney Dis 2008;51:385–394 [DOI] [PubMed] [Google Scholar]
- 19.Centers for Disease Control and Prevention (CDC). Survey operations manuals, brochures, and consent documents: 1999-2000 NHANES. Lower extremity disease procedures manual [Internet], 2001. Available from http://www.cdc.gov/nchs/data/nhanes/le.pdf Accessed 28 April 2013
- 20.Feringa HH, Bax JJ, van Waning VH, et al. The long-term prognostic value of the resting and postexercise ankle-brachial index. Arch Intern Med 2006;166:529–535 [DOI] [PubMed] [Google Scholar]
- 21.Selvin E, Manzi J, Stevens LA, et al. Calibration of serum creatinine in the national health and nutrition examination surveys (NHANES) 1988-1994, 1999-2004. Am J Kidney Dis 2007;50:918–926 [DOI] [PubMed] [Google Scholar]
- 22.Selvin E, Juraschek SP, Eckfeldt J, Levey AS, Inker LA, Coresh J. Calibration of cystatin C in the National Health and Nutrition Examination Surveys (NHANES). Am J Kidney Dis 2013;62:253–260 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Newman AB, Siscovick DS, Manolio TA, et al. Cardiovascular Heart Study (CHS) Collaborative Research Group Ankle-arm index as a marker of atherosclerosis in the Cardiovascular Health Study. Circulation 1993;88:837–845 [DOI] [PubMed] [Google Scholar]
- 24.NHANES (1999-2004) public use linked mortality files [article online]. Available from http://www.cdc.gov/nchs/data_access/data_linkage/mortality/nhanes_99_04_linkage.htm#description Accessed 31 July 2013
- 25.Zou G. A modified poisson regression approach to prospective studies with binary data. Am J Epidemiol 2004;159:702–706 [DOI] [PubMed] [Google Scholar]
- 26.Grams ME, Juraschek SP, Selvin E, et al. Trends in the prevalence of reduced GFR in the United States: a comparison of creatinine- and cystatin C-based estimates. Am J Kidney Dis 2013;62:253–260 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rule AD, Bailey KR, Lieske JC, Peyser PA, Turner ST. Estimating the glomerular filtration rate from serum creatinine is better than from cystatin C for evaluating risk factors associated with chronic kidney disease. Kidney Int 2013;83:1169–1176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pattaro C, Riegler P, Stifter G, Modenese M, Minelli C, Pramstaller PP. Estimating the glomerular filtration rate in the general population using different equations: effects on classification and association. Nephron Clin Pract 2013;123:102–111 [DOI] [PubMed] [Google Scholar]
- 29.Gaspari F, Ruggenenti P, Porrini E, et al. GFR Study Investigators The GFR and GFR decline cannot be accurately estimated in type 2 diabetics. Kidney Int 2013;84:164–173 [DOI] [PubMed] [Google Scholar]
- 30.Rossing P, Rossing K, Gaede P, Pedersen O, Parving HH. Monitoring kidney function in type 2 diabetic patients with incipient and overt diabetic nephropathy. Diabetes Care 2006;29:1024–1030 [DOI] [PubMed] [Google Scholar]
- 31.Chew-Harris JS, Florkowski CM, George PM, Elmslie JL, Endre ZH. The relative effects of fat versus muscle mass on cystatin C and estimates of renal function in healthy young men. Ann Clin Biochem 2013;50:39–46 [DOI] [PubMed] [Google Scholar]
- 32.Knight EL, Verhave JC, Spiegelman D, et al. Factors influencing serum cystatin C levels other than renal function and the impact on renal function measurement. Kidney Int 2004;65:1416–1421 [DOI] [PubMed] [Google Scholar]
- 33.Macdonald J, Marcora S, Jibani M, et al. GFR estimation using cystatin C is not independent of body composition. Am J Kidney Dis 2006;48:712–719 [DOI] [PubMed] [Google Scholar]
- 34.Wolf G, Müller N, Mandecka A, Müller UA. Association of diabetic retinopathy and renal function in patients with types 1 and 2 diabetes mellitus. Clin Nephrol 2007;68:81–86 [DOI] [PubMed] [Google Scholar]
- 35.Menon V, Shlipak MG, Wang X, et al. Cystatin C as a risk factor for outcomes in chronic kidney disease. Ann Intern Med 2007;147:19–27 [DOI] [PubMed] [Google Scholar]
- 36.de Boer IH, Katz R, Cao JJ, et al. Cystatin C, albuminuria, and mortality among older adults with diabetes. Diabetes Care 2009;32:1833–1838 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.