Abstract
Purpose
Spontaneous superior ophthalmic vein thrombosis (SOVT) is a rare entity. We describe three patients with spontaneous ophthalmic vein thrombosis, each with various risk factors.
Patients and Methods
A retrospective review of three patients with a diagnosis of superior ophthalmic vein thrombosis. Clinical characteristics, radiographic features, management techniques and outcomes are described.
Results
All patients presented with unilateral painful proptosis. Two patients had intact light perception, whereas one patient presented with absent light perception. All patients had identifiable risk factors for thrombosis, which included sickle cell trait, hereditary hemorrhagic telangectasia and colon cancer with recurrent deep vein thrombosis. Anticoagulation was initiated in two patients. Resolution of proptosis was seen in all patients, with no recovery of vision in one patient.
Conclusions
Risk factors for spontaneous superior ophthalmic vein thrombosis are multifactorial. MRI and MRV confirm the diagnosis of SOVT. Despite urgent intervention devastating visual loss may occur.
Keywords: superior ophthalmic vein, thrombosis, orbital compartment syndrome, optic neuropathy, proptosis, coagulopathy
Introduction
Superior ophthalmic vein thrombosis (SOVT) is a rare entity predisposed by sino-orbital disease, vascular and coagulation anomalies.1, 2, 3 Signs of orbital congestion including proptosis, chemosis, and ophthalmoplegia are often present.
We present a series of spontaneous SOVT, highlighting potential life and vision threatening associations.
Materials and methods
Retrospective case series from the University of California, San Diego and University of Wisconsin-Madison. The medical records of three patients presenting with SOVT between August 2005–June 2010 were identified and reviewed.
Case reports
There were two males and one female. Mean age was 59.3 (range 42–77). No patient had previous trauma or eye surgery. Investigation for hypercoagulablity was performed on all patients, including anticardiolipin antibodies, protein-C, protein-S, antithrombin-III, homocysteine, antiphospholipid antibodies, and Factor-V-Leiden mutation. Autoimmune work-up was also performed including ANA, ds-DNA, ANCA, and ENA profile. All were negative.
Case 1
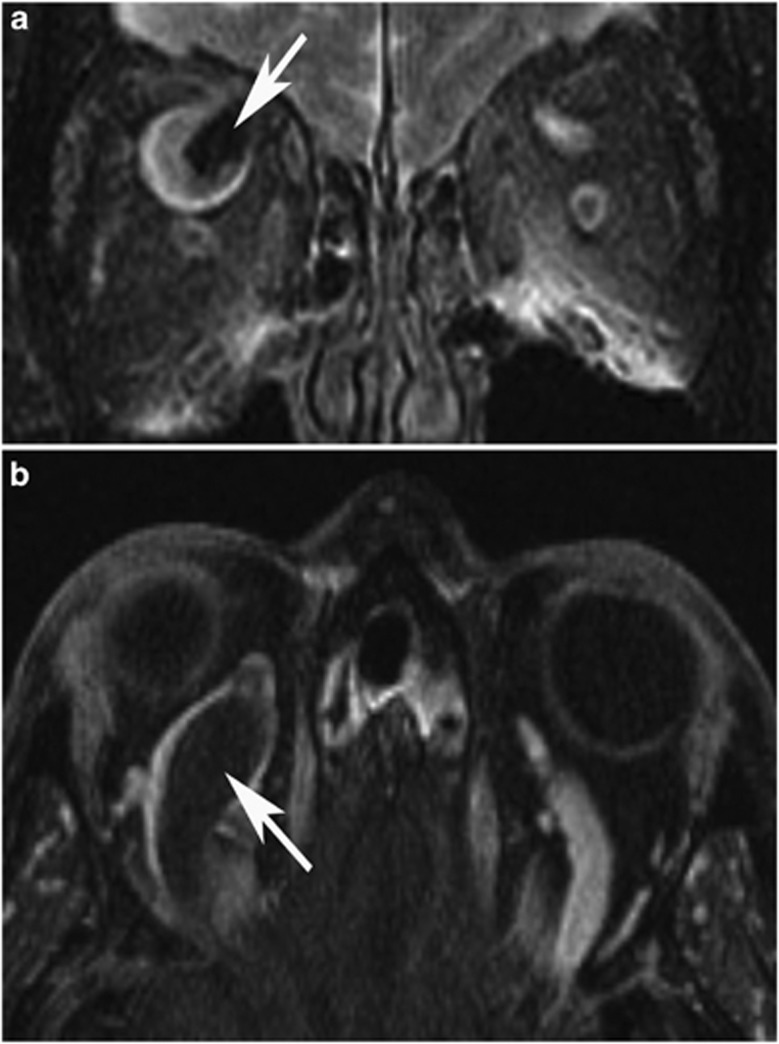
A 58-year-old African-American woman with sickle trait and hypertension awoke with right orbital pain and diplopia. Vision was OD 20/25 and OS 20/20. Pupils, color vision, and intraocular pressure (IOP) were normal. There was no conjunctival chemosis or vascular engorgement, but 4 mm proptosis with mild supraduction and adduction deficits were present. MRI imaging was performed (Figure 1a and b). Oral warfarin was instituted. Two weeks later, visual acuity was 20/20 OU, with full motility, proptosis resolution, and normal SOV flow (Figure 1c). The patient remained recurrence free at 6 years.
Figure 1.
(a and b) Case 1. Axial and coronal MRI and MRV showing intraluminal thrombosis and dilation of the right SOV (arrows). (c) Case 1. Axial CT angiogram showing resolution of SOV thrombosis 4 months later (arrow).
Case 2
A 77-year-old Caucasian male with a history of hereditary hemorrhagic telangiectasia (HHT), diabetes, hypertension, hypercholesterolemia and smoking awoke with right eye pain, blurred vision, and diplopia. Visual acuity was 20/25 OU, with normal color vision and pupillary function. Anterior segment examination showed episcleral vascular engorgement and tortuosity. IOP was normal but resistance to retropulsion was observed. Exophthalmometry revealed 3 mm proptosis. Motility was mildly limited in all positions. Oropharynx examination demonstrated multiple telangiectasias and mucosal vascular malformations, consistent with HHT.4 MRI demonstrated SOVT (Figure 2a and b).
Figure 2.
(a and b) Case 2. Axial and coronal MRI and MRV showing bilaterally enlarged SOV, more pronouced on the right with intraluminal thrombus (arrow).
Anticoagulation was not initiated due to the increased risk of systemic hemorrhage related to HHT. Proptosis and diplopia resolved spontaneously over 5 weeks.
Case 3
A 42-year-old Caucasian male awoke with pain and visual loss in the left eye 3 days after cessation of warfarin therapy. Anticoagulation had been initiated after two prior deep vein thromboses and hemicolectomy for colon carcinoma but was discontinued by his primary physician after 4 years of therapy.
On presentation, vision was no light perception (NLP) OS with a left afferent pupillary defect, diffuse hemorrhagic chemosis and an IOP of 55 mm Hg. The left orbit was tense to retropulsion with 3 mm proptosis and ophthalmoplegia. Urgent canthotomy with cantholysis was performed with IOP reduction to 25 mm Hg. International normalized ratio was 1.6 on admission and before warfarin cessation was 2.1. CT showed left SOVT (Figures 3a and b). Anticoagulation was reinstituted with intravenous heparin. Topical timolol 0.5%, latanaprost, dorzolamide, and oral acetazolamide were initiated alongside intravenous methylprednisolone 250 mg every 6 h. Despite proptosis and ophthalmoplegia resolution, vision remained NLP at 1 year with optic atrophy.
Figure 3.
(a and b) Case 3. Axial and coronal CT revealing enlarged and thrombosed left SOV (arrow) and diffusely enlarged extraocular muscles.
Discussion
The etiology of SOVT is multifactorial. Risk factors may be local or systemic, usually including at least one risk factor from Virchow's triad.5
Each case demonstrates differing predispositions. Sickle cell trait is a known procoagulant, although sequelae of clinical significance are unusual.6, 7 HHT (Osler-Weber-Rendu syndrome), an autosomal-dominant condition characterized by telangiectasia and arteriovenous malformations (AVMs) of the skin, mucosa, and viscera,4 is known to cause thrombosis but to our knowledge has never been associated with SOVT. Case 3 represents systemic malignancy associated hypercoagulability highlighting the risk of anticoagulation withdrawal in patients with recurrent thrombosis.
MRI and MRV confirm the diagnosis of SOVT8 and exclude carotid-cavernous sinus fistula, cavernous sinus thrombosis, and sino-orbital infection. Life threatening underlying systemic disease must also be excluded, and all patients received consultation with an internist. Patients with HHT require a multi-disciplinary screening for visceral AVMs particularly involving the pulmonary or central nervous system.
Although not noted in our series SOVT may occur in association with cavernous sinus thrombosis (CST), particularly in septic etiologies.9, 10 Potential consequences of CST include pituitary insufficiency, hemiparesis, and death.
Clinical presentation with proptosis, chemosis, conjunctival congestion, and ophthalmoplegia are common. In our series symptoms arose on awakening, evoking the possible influence of supine positioning contributing to venous stasis.
Management is guided by severity of findings and systemic considerations. Anticoagulation is considered after risk-benefit analysis. In our patient with HHT, spontaneous SOVT resolution occurred suggesting expectant management in patients without immediate vision compromise in whom anticoagulation therapy presents a higher risk. Despite visual loss being rare in SOVT literature1, 2, 3 this condition may not be benign, as illustrated by patient three who sustained profound vision loss from an orbital compartment syndrome despite immediate aggressive treatment.

Acknowledgments
This work was generously supported by Steve and Kathleen Flynn, the Bell Charitable Foundation, and an unrestricted grant from Research to Prevent Blindness (RPB), New York, NY, USA.
The authors declare no conflict of interest.
References
- Stiebel-Kalish H, Setton A, Nimii Y, Kalish Y, Hartman J, Huna Bar-On R, et al. Cavernous sinus dural arteriovenous malformations: patterns of venous drainage are related to clinical signs and symptoms. Ophthalmology. 2002;109:1685–1691. doi: 10.1016/s0161-6420(02)01166-1. [DOI] [PubMed] [Google Scholar]
- Schmitt NJ, Beatty RL, Kennerdell JS. Superior ophthalmic vein thrombosis in a patient with dacryocystitis-induced orbital cellulitis. Ophthal Plast Reconstr Surg. 2005;21:387–389. doi: 10.1097/01.iop.0000176269.84949.96. [DOI] [PubMed] [Google Scholar]
- Shinder R, Oellers P, Esmaeli B, Schiffman JS. Superior ophthalmic vein thrombosis in a patient with chronic myeloid leukemia receiving antifibrinolytic and thrombopoietin receptor agonist therapy. J Ocul Pharmacol Ther. 2010;26:293–296. doi: 10.1089/jop.2010.0008. [DOI] [PubMed] [Google Scholar]
- Shovlin CL, Guttmacher AE, Buscarini E, Faughnan ME, Hyland RH, Westermann CJ, et al. Diagnostic criteria for hereditary hemorrhagic telangiectasia (Rendu-Osler-Weber syndrome) Am J Med Genet. 2000;91:66–67. doi: 10.1002/(sici)1096-8628(20000306)91:1<66::aid-ajmg12>3.0.co;2-p. [DOI] [PubMed] [Google Scholar]
- Bagot CN, Arya R. Virchow and his triad: a question of attribution. Br J Haematol. 2008;143:180–190. doi: 10.1111/j.1365-2141.2008.07323.x. [DOI] [PubMed] [Google Scholar]
- Austin H, Key NS, Benson JM, Lally C, Downing NF, Whitsett C, et al. Sickle cell trait and the risk of venous thromboembolism among blacks. Blood. 2007;110:908–912. doi: 10.1182/blood-2006-11-057604. [DOI] [PubMed] [Google Scholar]
- Key NS, Derebail VK. Sickle-cell trait: novel clinical significance. Hematology Am Soc Hematol Educ Program. 2010;2010:418–422. doi: 10.1182/asheducation-2010.1.418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei R, Cai J, Ma X, et al. Imaging diagnosis of enlarged superior ophthalmic vein. Zhonghua Yan Ke Za Zhi. 2002;38:402–404. [PubMed] [Google Scholar]
- Lai PF, Cusimano MD. The spectrum of cavernous sinus and orbital venous thrombosis: a case and a review. Skull Base Surg. 1996;6 (1:53–59. doi: 10.1055/s-2008-1058913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desa V, Green R. Cavernous sinus thrombosis: current therapy. J Oral Maxillofac Surg. 2012;70 (9:2085–2091. doi: 10.1016/j.joms.2011.09.048. [DOI] [PubMed] [Google Scholar]