Abstract
Cells may lack eyes to see and ears to hear, but cells do seem to have a sense of ‘touch’ that allows them to feel their microenvironment. This is achieved in part through contractility-coupled adhesion to physically flexible ‘soft’ tissue. Here we summarize some of the known variations in elasticity of solid tissue and review some of the long-term effects of cells ‘feeling’ this elasticity, focusing on differentiation processes of both committed cell types and stem cells. We then highlight what is known of molecular remodeling in cells under stress on short time scales. Key roles for forces generated by ubiquitous and essential myosin-II motors in feedback remodeling are emphasized throughout.
Introduction
Solid tissues are soft, with the exception of bone, and they possess an elasticity E that not only varies between many different tissues (Fig. 1A) but also exerts a considerable influence on how resident tissue cells behave. Indeed, cells that are dissociated from a solid tissue are generally not viable in a fluid suspension – they must adhere to a “solid” which, by definition, recovers its shape after pushing and pulling, even at the scale of a cell. Solid tissues such as skin, muscle, and brain, are all relatively elastic, with the macroscale elasticity evident in their recovery of shape within seconds after mild poking and pinching or even after sustained compression, such as sitting. This is in contrast to fluid tissues such as blood and lymph which flow readily on a similar time scale and contain distinct cells, such as red and white blood cells, that are functional without sustained attachment. The impact of solid tissue elasticity on adherent cells is the focus here, with recent insights from stem cells and structural proteomics adding to past reviews (eg. [1]) of findings that indicate tissue elasticity E is felt by cells, affecting cell structure and function.
Figure 1.
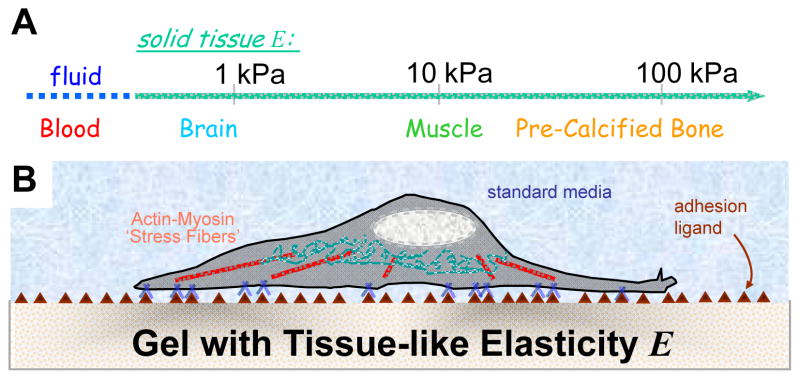
Tissue elasticity scale (A) and model elastic culture systems (B).
Matrix composition and cell activity both contribute to tissue elasticity or stiffness at a scale that cells can actively probe and sense. With collagen as an example: collagen type, amount, diameter of fibers, crosslinking (eg. cellular lysyl oxidase activity) plus non-covalent interactions with other matrix proteins will all contribute to the matrix elasticity. Recent measurements of the elasticity of zebrafish embryos [2] that were treated with the nonmuscle myosin-II (NMM II)-specific inhibitor blebbistatin also document a dramatic decrease in the effective elasticity, illustrating the contribution of myosin-derived tension (like tension in a guitar string) to the elasticity of the entire organism. While adherent tissue cells and extracellular matrix contribute to a characteristic if not strictly tissue-specific elastic micro-environment, cells generally anchor and pull on their surroundings through myosin-II based contractility and transcellular adhesions of integrins plus other adhesion molecules [3]. The resistance felt by a given cell derives from tissue matrix, an adjacent cell, or perhaps – in culture – a synthetic substrate intended to model soft tissue (Fig. 1B). Disease can bring significant changes in tissue elasticity: indeed, sclerosis – as in atherosclerosis, otosclerosis, scleroderma, and more – is greek for hardening of tissue.
Contractile forces generated by ubiquitous cross-bridging interactions of actin and myosin-II filaments in stress fibers are transmitted to the substrate as ‘traction’ forces that cause visible wrinkles in a thin film or lateral displacements of markers at the surface of a soft gel [4–7]. On gels with collagen-I covalently attached, epithelial cells and fibroblasts [8] were the first cells reported to detect and respond distinctly to soft versus stiff substrates; differences were suggested to depend on myosin-II as they were inhibited by BDM (2,3-butanedione monoxime) – although this drug is now known to have multiple effects beyond myosin inhibition. Since then, neurons [9,10], muscle cells of various types [11–13], mesenchymal stem cells [14], plus many other tissue cell types [15–18] have been shown to sense substrate stiffness, and at least some of the results have confirmed the importance of nonmuscle myosin-II through inhibition of elasticity-dependent behavior changes with blebbistatin. Most cell types are found to respond to the elasticity E of the substrate within hours by spreading and assembling both adhesions and cytoskeleton in proportion to E up to some saturating value beyond which changes in E exert no influence. Given that an isoform of myosin-II is also responsible for the work done by skeletal muscle, an analogy to lifting weights and exercise seems appropriate: to your bicep, a load of 1 kg undoubtedly feels very different from a load of 10 kg, whereas pushing or pulling on an immovable object like the handle of a locked door is a very distinct isometric exercise. Similar sensitivity to E seems to apply to most anchored cell types with similar implications for growth and remodeling within individual cells.
Soft tissue E Measurements and Model systems
The intrinsic resistance of a solid to a stress, regardless of topography and thickness (eg. basement membrane), is measured by the solid’s elastic modulus E, which is most simply obtained by applying a force – such as poking with an atomic force microscope (AFM) [19] – to a section of tissue or other substrate and then measuring the relative displacement. Tissues with small E show larger indentations or displacements under a given force.
E not only appears to adequately characterize many tissues at a macroscopic scale but also at the micro-scale of cell-exerted tractions – despite heterogeneity within tissue. Subtleties include matrix fiber effects and length scales of greatest relevance to cell sensing. Measurements issues include sample preparation; for example, E of whole brain in macroscopic measurements can vary by a factor of two or more depending on specifics of preparation, tissue perfusion, etc. [20]. In addition, many probing methods involve high frequency stressing [21] whereas relevant time scales for cell-exerted strains seem likely to range from seconds to hours with signal integration likely for longer timescales. Nonetheless, comparisons of three diverse tissues that contain a number of different and illustrative cell types show that brain tissue with E ~ 0.1-1 kPa [20] is softer than striated muscle with E ~ 10 kPa [12,22], which is softer than osteoid with E ~ 20–40 kPa [14]. Even though bone is about as rigid as glass and plastic, it begins as compliant, pre-calcified and heavily crosslinked collagenous ‘osteoid’ [14]. Fibrotic tissues and wounds have been measured to have E overlapping with this range [23,24]. Although mapping of soft tissue micro-elasticity at a resolution typical in histology seems important and likely to be revealing, the implication from current studies is that there are distinct elastic microenvironments for neurons in brain, for myotubes in fiber bundles, and for osteoblasts on osteoid.
Tactile sensing of tissue stiffness requires a matrix ligand such as collagen for attachment, but once the ligand density is no longer limiting on a substrate [11], this cell-driven sensing feeds back on adhesion and cytoskeleton as well as on net contractile forces. Inert polyacrylamide gels with covalently attached collagen-I [8] have emerged as the most common system for controlling E in cell biology. By controlling the extent of polymer crosslinking in the gels, E can be adjusted over several orders of magnitude from extremely soft to stiff. Whether the collagen is fibrillar or not in such systems does not appear to have been scrutinized, but the collagen film does not contribute significantly to micro-elasticity [11] based on AFM measurements that also show E does not change even after many days of exposure to serum proteins – contrary to recent speculations [25].
Focal adhesion proteins paxillin [11,14] and vinculin [8] are found in small, diffuse, and dynamic adhesion complexes in cells on soft, lightly crosslinked gels (E ~ 1 kPa). In contrast, stiff, highly crosslinked gels (E ≫ 20 kPa) show cells with stable focal adhesions, typical of those seen in cells attached to glass. Similarly, rigidification of cell-derived fibrillar matrices with glutaraldehyde shifts adhesions from fibrilllar to large, non-fibrillar focal adhesions similar to those found on fixed substrates of fibronectin [26]. Cytoskeletal assembly and cell tension [14] follow the same trends as adhesions. Thus the stiffer the matrix, the larger the adhesions and the stronger the cell pulls.
On a series of gel substrates, cell types that are committed to lineages such as neurons, muscle, and bone will modulate their phenotype in accord with tissue elasticity (Fig. 2A). Branching morphogenesis of neurons has been found to be maximal on soft matrices that mimic Ebrain and minimal on both stiffer gels and glass [9,10]. Likewise, striation of skeletal muscle myosin in muscle cells was maximal on stiffer matrices that mimic Emuscle, although myoblast fusion and expression levels of markers such as skeletal muscle myosin appeared otherwise unaffected [12]. Similar results were also obtained with multilayer cultures in which a first layer of cells grown on rigid glass showed no striation whereas an upper layer of cells that feel the soft bottom layer of cells showed strong striation. Lineage-specific secretions of osteoblasts [27] also showed a relative maximum near Eosteoid, suggesting that rigid bone is derived from remodeling of a suitably non-rigid matrix, although only three values of E were examined. The results of the osteoblast study were obtained with the non-collagenous ligand RGD attached to alginate gels rather than polyacrylamide. In addition, the elasticity dependence of the fibroblast-myofibroblast transition was demonstrated using a non-hydrous rubber rather than a hydrated gel like polyacrylamide [28]. These latter two studies reporting elasticity dependence on non-polyacrylamide substrates support the premise that the collective property E of a substrate rather than its detailed chemistry influences cell structure and function. It should be emphasized that the differentiating factor in all of these studies and those reviewed below was a property of the insoluble microenvironment rather than the soluble milieu.
Figure 2.
Matrix elasticity effects on differentiation of committed cells (A) and mesenchymal stem cells (B).
Stem cells are particularly E-Sensitive
Mesenchymal stem cells (MSCs) appear especially sensitive to tissue elasticity. These cells reside in the bone marrow and are believed to enter the circulation and contribute to tissue regeneration and repair after injury, such as a muscle tear. Bone marrow aspirates are either fluid, with an ‘intercellular substance’ measured decades ago to be about 100-fold more viscous than water [29], or else have a very small E [30], and the rare MSCs in marrow are generally separated from the many other marrow cell types by their differential adhesion to rigid plastic. Adhesion occurs within hours [31] and is likely mediated by serum matrix proteins such as fibronectin that commonly adsorb rapidly to plastic and are also prominent in bone marrow. When replated on collagen-I coated gels that mimic the soft tissue elasticities, a significant fraction of the slowly proliferating MSCs (in 10% serum) began within 4–24 hrs to take on cell shapes that resembled a branched neuronal lineage on Ebrain gels, a spindle-shaped myoblast on Emuscle gels, or a well-spread osteoblast on Eosteoid gels [14]. Collagen-I is not reported to be a matrix molecule within marrow, and while ligand might be necessary, collagen-I on glass was not sufficient to induce lineage-specific morphogenesis. Elasticity was clearly the differentiating factor, and the pluripotency of MSCs makes these cells especially sensitive to microenvironmental factors.
Expression of lineage-specific proteins such as MyoD occurred several days after plating, and a broader confirmation of lineage induction by 1 week was documented through both lineage-specific protein and transcript profiling (Fig. 2B). When calibrated against established differentiated cell lines, both protein and transcript levels for the three lineages proved consistent in showing that E-induction is about half that of the established cell lines. The MSCs would nonetheless commit to their E-induced lineages after several weeks in culture, based on the fact that induction media composed of potent synthetic glucocorticoids (eg. dexamethasone) could re-direct lineages at early but not late time points. Moreover, when myogenic or osteogenic induction media cocktails were added to the various cell on gel systems, the soluble and insoluble effects proved additive in inducing expression levels similar to committed cell lines.
One prediction from the cell biology studies of human-derived MSCs above follows from the fact that osteoid elasticity [14] possesses an elasticity E similar to that of a fibrotic infarct scar [23]. Osteogenesis is therefore predicted to occur if MSCs are injected into an infarct scar – this is exactly what was found in mouse studies in which MSCs and HSCs (hematopoietic stem cells) were injected into infarcts, with only the MSCs causing ossification [32]. The potential risks for human trials should not be neglected and neither should the need for basic studies of the mechanism of elasticity-induced signals in stem cell biology.
Matrix-coupled, Myosin-driven Remodeling
Molecular mechanisms of elasticity sensing by cells seem likely to be collective and dependent on many interacting components of the cyto-adhesion apparatus. Cell tension is expected to be important, and differentiation of MSCs was indeed blocked by myosin-II inhibition with blebbistatin [14]. In addition, lineage specification was associated with significant changes in the levels NMM IIB and C, with considerable down-regulation on soft matrices (0.1–1 kPa) and modest up-regulation on stiffer matrices (>10 kPa); NMM IIA showed the smallest variation with E among a dozen myosin motors studied, suggestive of a constitutive role in cell biological processes. The findings are consistent with the lack of differentiation in NMM IIA null mice, which are embryonic lethal at day ~6 with no signs of heart or vasculature development despite normal levels of expression of many key factors such the transcription factor GATA-4 [33]. Embryoid bodies of the NMM IIA null cells showed no proliferation defect but appeared flaccid rather than as the typical tensed spheroids.
In the earliest studies of stiffness sensing, tyrosine phosphorylation on multiple proteins (including paxillin) appeared broadly enhanced in cells on stiffer gel substrates [8]. Pharmacologically induced, non-specific hyper-phosphorylation could drive focal adhesion formation on soft materials, and key roles for membrane localized phosphatases have also emerged [34]. Likewise on soft substrates, overexpression of GFP-actin was found to over-ride the limited spreading, whereas overexpression of GFP-paxillin had no effect [11]. α5-integrin was reportedly downregulated on soft gels but overexpression had no effect on cell spreading [18]; in contrast, engineered clustering of integrins could strongly influence stiffness sensing [35]. Microtubules have been proposed to act as ‘struts’ in cells, but quantification of their contributions to cells on gels had shown that they provide only a minor fraction of the resistance (14%) to cell-generated contractile tension [36]; the majority of a cell’s tension or stress is thus resisted by the strain of the matrix and microenvironment.
The list of molecular players in E-sensing will undoubtedly grow as will a myriad of interacting binding partners, but understanding of the molecular dynamics of cell-driven matrix-coupled remodeling seems likely to depend as much on new approaches that can clarify how forces within cells remodel proteins and their assemblies through extension, conformation, and/or dissociation (Fig. 3). Extension of proteins should, under small forces, resemble stretching of a spring, but tension forces exerted on folded domains can in principle relieve the stress by unfolding one of many domains or else by dissociating one end of the protein from binding partners that anchor it down. Many ubiquitous cytoskeletal proteins including myosin’s rod domain [37], filamin [38], spectrin [39], and ankyrin [40] have been studied by AFM as purified single molecules and found to undergo forced unfolding; likewise, many adhesion proteins including VCAM [41] and integrins [42] unfold or dissociate from their ligands under force. Transitions clearly depend on the levels of force and the duration of stress, with typical transition timescales of <1 sec at forces of <100 pN that 1–20 myosin molecules can generate. Cyto-adhesion structures are therefore prone to force-f driven kinetics (i.e. rate ≈ rateo exp(f / fo) where fo is a molecule-specific constant), but identifying which proteins or complexes remodel or reorganize has of necessity motivated new methods.
Figure 3.
Schematic of how myosin forces strain the cell and also strain the matrix as resisted by matrix elasticity. The strain within the cell is manifested as protein extension, domain unfolding, and/or protein-protein dissociation. These processes might simply relieve stress in the cell, or they might create sites for new binding partners or new signals.
Cys Shotgun and other Methods begin clarifying Molecular Dynamics
Cysteine is a reactive but relatively hydrophobic amino acid that is often buried within tertiary or quaternary structures. Reactivity of cysteine’s thiol group had been exploited in solution to a limited extent to probe protein interactions [43] and folding [44], and in situ Cys labeling of membrane proteins such as GPCRs had yielded insights into accessibility and ligand-induced changes in individual proteins [45]. Proteomic-scale Cys Shotgun labeling of intact cells has now been shown to be feasible with Mass Spectrometry (MS) [46]. The basic premise is to label Cys with thiol-reactive, membrane-permeable fluorescent probes in both stressed and unstressed cells, and then search for differential labeling of proteins by multiple methods, including MS, to identify the proteins and the position of the cysteines that, under stress, have shifted from buried and inaccessible to exposed and accessible to a fluorescent reactant.
Cys Shotgun methods were first developed with the simplest possible mammalian cell, the red blood cell, which was sheared in a flow device while suspended with an encapsulated dye. The membrane-localized cytoskeleton of this cell is a simple but ubiquitous spectrin-actin network, and it showed shear-enhanced labeling within minutes as visualized by fluorescence microscopy. Both α and β spectrin showed shear-enhanced labeling in SDS-PAGE and MS analyses – although no differences were seen with actin, ankyrin, and other peripheral membrane proteins. Some of the Cys with increased labeling were clearly hidden in crystal structures or homology models of domains, and recombinant proteins studied with temperature-dependent labeling kinetics in native versus denaturing conditions proved consistent with domain unfolding as assessed by circular dichroism. Cys Shotgun methods were subsequently applied to MSCs on polyacrylamide gel substrates in tensed versus blebbistatin-relaxed states [46] and to cardiomyocytes exerting their rhythmic contractions on soft versus stiff substrates [47]. Among the most prominent differences induced by matrix elasticity were Cys sites that implicate differential unfolding in myosin-IIs, differential unfolding and dissociation of filamin, and shifts in the polymerization state of vimentin.
New binding partners and new phosphorylation-based signals (Fig. 3, right) might arise directly or indirectly from molecular extension, unfolding, or dissociation. Initial progress in finding such force-sensitive signaling proteins has identified p130-Cas. Within spread and fixed cells, binding of a conformation-sensitive antibody to p130Cas in regions of the cells expected to generate the highest traction forces has suggested force-induced extension of a central proline-rich region in p130-Cas making it available for phosphorylation by Src family kinases [48]. A role for p130-Cas in stiffness sensing by cells has yet to be demonstrated. Nonetheless, with the growing capabilities of Mass Spectrometry for detecting functional modifications that are either natural (phosphorylation) or synthetic (modified Cys), proteomic scale insights into the various pathways involved in how cells feel the elasticity of their microenvironment seem within reach.
Acknowledgments
We are grateful for grant support from the NSF, MDA, NIH (NHLBI, NIBIB, NIDDK) and NIH TG support (AZ).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
• of special interest
•• of outstanding interest
- 1.Discher DE, Janmey P, Wang YL. Tissue cells feel and respond to the stiffness of their substrate. Science. 2005;310:1139–1143. doi: 10.1126/science.1116995. [DOI] [PubMed] [Google Scholar]
- **2.Krieg M, Arboleda-Estudillo Y, Puech PH, Kafer J, Graner F, Muller DJ, Heisenberg CP. Tensile forces govern germ-layer organization in zebrafish. Nat Cell Biol. 2008;10:429–436. doi: 10.1038/ncb1705. The cellular tension as well as the adhesiveness of progenitor cells from the 3 germ-layers of zebrafish embryos were characterized by AFM. Insight into germ-layer sorting shows that cellular tension generated by acto-myosin contraction regulated by TGFβ-Nodal signaling is key in regulating cell-sorting during gastrulation of the embryo. [DOI] [PubMed] [Google Scholar]
- 3.Schwartz MA, Desimone DW. Cell adhesion receptors in mechanotransduction. Curr Opin Cell Biol. 2008 doi: 10.1016/j.ceb.2008.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Balaban NQ, Schwarz US, Riveline D, Goichberg P, Tzur G, Sabanay I, Mahalu D, Safran S, Bershadsky A, Addadi L, et al. Force and focal adhesion assembly: a close relationship studied using elastic micropatterned substrates. Nat Cell Biol. 2001;3:466–472. doi: 10.1038/35074532. [DOI] [PubMed] [Google Scholar]
- 5.Harris AK, Wild P, Stopak D. Silicone rubber substrata: a new wrinkle in the study of cell locomotion. Science. 1980;208:177–179. doi: 10.1126/science.6987736. [DOI] [PubMed] [Google Scholar]
- 6.Marganski WA, Dembo M, Wang YL. Measurements of cell-generated deformations on flexible substrata using correlation-based optical flow. Methods Enzymol. 2003;361:197–211. doi: 10.1016/s0076-6879(03)61012-8. [DOI] [PubMed] [Google Scholar]
- 7.Oliver T, Dembo M, Jacobson K. Separation of propulsive and adhesive traction stresses in locomoting keratocytes. J Cell Biol. 1999;145:589–604. doi: 10.1083/jcb.145.3.589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- **8.Pelham RJ, Jr, Wang Y. Cell locomotion and focal adhesions are regulated by substrate flexibility. Proc Natl Acad Sci U S A. 1997;94:13661–13665. doi: 10.1073/pnas.94.25.13661. First paper to demonstrate a role for substrate elasticity in the behavior of cells and to mimic it with polyacrylamide hydrogels,. Fibroblasts and epithelial cells grown on soft vs stiff gels have decreased area, small irregular focal adhesions, and increased migration rates. These substrate-induced changes were found to be regulated by phospho-tyrosine signaling and my. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Flanagan LA, Ju YE, Marg B, Osterfield M, Janmey PA. Neurite branching on deformable substrates. Neuroreport. 2002;13:2411–2415. doi: 10.1097/01.wnr.0000048003.96487.97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Georges PC, Miller WJ, Meaney DF, Sawyer ES, Janmey PA. Matrices with compliance comparable to that of brain tissue select neuronal over glial growth in mixed cortical cultures. Biophys J. 2006;90:3012–3018. doi: 10.1529/biophysj.105.073114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Engler A, Bacakova L, Newman C, Hategan A, Griffin M, Discher D. Substrate compliance versus ligand density in cell on gel responses. Biophys J. 2004;86:617–628. doi: 10.1016/S0006-3495(04)74140-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Engler AJ, Griffin MA, Sen S, Bonnemann CG, Sweeney HL, Discher DE. Myotubes differentiate optimally on substrates with tissue-like stiffness: pathological implications for soft or stiff microenvironments. J Cell Biol. 2004;166:877–887. doi: 10.1083/jcb.200405004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Peyton SR, Putnam AJ. Extracellular matrix rigidity governs smooth muscle cell motility in a biphasic fashion. J Cell Physiol. 2005;204:198–209. doi: 10.1002/jcp.20274. [DOI] [PubMed] [Google Scholar]
- **14.Engler AJ, Sen S, Sweeney HL, Discher DE. Matrix elasticity directs stem cell lineage specification. Cell. 2006;126:677–689. doi: 10.1016/j.cell.2006.06.044. Mesenchymal stem cells are shown to be extremely sensitive to the elasticty of their substrate. Collagen-I coated polyacrylamide hydrogels mimicing the elasticity of brain, muscle, or bone induce naive MSCs to differentiate to the respective lineage. The insoluble mechanical cue is also shown to be additive to chemical induction of differentiation. [DOI] [PubMed] [Google Scholar]
- 15.Beningo KA, Wang YL. Fc-receptor-mediated phagocytosis is regulated by mechanical properties of the target. J Cell Sci. 2002;115:849–856. doi: 10.1242/jcs.115.4.849. [DOI] [PubMed] [Google Scholar]
- 16.Deroanne CF, Lapiere CM, Nusgens BV. In vitro tubulogenesis of endothelial cells by relaxation of the coupling extracellular matrix-cytoskeleton. Cardiovasc Res. 2001;49:647–658. doi: 10.1016/s0008-6363(00)00233-9. [DOI] [PubMed] [Google Scholar]
- 17.Wang HB, Dembo M, Wang YL. Substrate flexibility regulates growth and apoptosis of normal but not transformed cells. Am J Physiol Cell Physiol. 2000;279:C1345–1350. doi: 10.1152/ajpcell.2000.279.5.C1345. [DOI] [PubMed] [Google Scholar]
- 18.Yeung T, Georges PC, Flanagan LA, Marg B, Ortiz M, Funaki M, Zahir N, Ming W, Weaver V, Janmey PA. Effects of substrate stiffness on cell morphology, cytoskeletal structure, and adhesion. Cell Motil Cytoskeleton. 2005;60:24–34. doi: 10.1002/cm.20041. [DOI] [PubMed] [Google Scholar]
- 19.Engler AJ, Rehfeldt F, Sen S, Discher DE. Microtissue elasticity: measurements by atomic force microscopy and its influence on cell differentiation. Methods Cell Biol. 2007;83:521–545. doi: 10.1016/S0091-679X(07)83022-6. [DOI] [PubMed] [Google Scholar]
- 20.Gefen A, Margulies SS. Are in vivo and in situ brain tissues mechanically similar? J Biomech. 2004;37:1339–1352. doi: 10.1016/j.jbiomech.2003.12.032. [DOI] [PubMed] [Google Scholar]
- 21.Hu S, Eberhard L, Chen J, Love JC, Butler JP, Fredberg JJ, Whitesides GM, Wang N. Mechanical anisotropy of adherent cells probed by a three-dimensional magnetic twisting device. Am J Physiol Cell Physiol. 2004;287:C1184–1191. doi: 10.1152/ajpcell.00224.2004. [DOI] [PubMed] [Google Scholar]
- 22.Collinsworth AM, Zhang S, Kraus WE, Truskey GA. Apparent elastic modulus and hysteresis of skeletal muscle cells throughout differentiation. Am J Physiol Cell Physiol. 2002;283:C1219–1227. doi: 10.1152/ajpcell.00502.2001. [DOI] [PubMed] [Google Scholar]
- 23.Berry MF, Engler AJ, Woo YJ, Pirolli TJ, Bish LT, Jayasankar V, Morine KJ, Gardner TJ, Discher DE, Sweeney HL. Mesenchymal stem cell injection after myocardial infarction improves myocardial compliance. Am J Physiol Heart Circ Physiol. 2006;290:H2196–2203. doi: 10.1152/ajpheart.01017.2005. [DOI] [PubMed] [Google Scholar]
- 24.Goffin JM, Pittet P, Csucs G, Lussi JW, Meister JJ, Hinz B. Focal adhesion size controls tension-dependent recruitment of alpha-smooth muscle actin to stress fibers. J Cell Biol. 2006;172:259–268. doi: 10.1083/jcb.200506179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Antia M, Baneyx G, Kubow KE, Vogel V. Fibronectin in aging extracellular matrix fibrils is progressively unfolded by cells and elicits an enhanced rigidity response. Faraday Discuss. 2008 doi: 10.1039/b718714a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- *26.Cukierman E, Pankov R, Stevens DR, Yamada KM. Taking cell-matrix adhesions to the third dimension. Science. 2001;294:1708–1712. doi: 10.1126/science.1064829. This study compares cell-matrix adhesions in traditional 2D culture to several more in vivo-like conditions using tissue-derived 3D matrices and 3D collagen gels. The characteristics of in vivo cell-matrix interaction were only found in vitro when the dimensionality, biological composition, and the pliability (although actual elasticity was not measured) were mimicked in culture. [DOI] [PubMed] [Google Scholar]
- *27.Kong HJ, Polte TR, Alsberg E, Mooney DJ. FRET measurements of cell-traction forces and nano-scale clustering of adhesion ligands varied by substrate stiffness. Proc Natl Acad Sci U S A. 2005;102:4300–4305. doi: 10.1073/pnas.0405873102. This study introduces a FRET-based method to visualize adhesion ligand clustering as well as traction forces generated by cells on hydrogels of tunable compliance. [DOI] [PMC free article] [PubMed] [Google Scholar]
- *28.Wipff PJ, Rifkin DB, Meister JJ, Hinz B. Myofibroblast contraction activates latent TGF-beta1 from the extracellular matrix. J Cell Biol. 2007;179:1311–1323. doi: 10.1083/jcb.200704042. This study examines the dual requirement of a fibrotic elasticity and TGFβ1 for induction of myofibroblast differentiation. TGFβ1 remains sequestered in the large latent complex in the matrix on soft substrates, but more rigid substrates similar to those of fibroblast-rich wounds induce increased cell forces on the matrix through integrins, allowing TGFβ1 to be pulled free. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Carter RE, Fleishacker B, Jackson WL. The Intercellular Substances of Bone Marrow. I. Preliminary Studies of the Physical and Chemical Character of the Intercellular Bone Marrow Substances in Normal Rabbits. Blood. 1961;17:650–656. [PubMed] [Google Scholar]
- 30.Winer JP, Janmey PA, McCormick ME, Funaki M. Bone Marrow-Derived Human Mesenchymal Stem Cells Become Quiescent on Soft Substrates but Remain Responsive to Chemical or Mechanical Stimuli. Tissue Eng Part A. 2008 doi: 10.1089/ten.tea.2007.0388. [DOI] [PubMed] [Google Scholar]
- 31.Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–147. doi: 10.1126/science.284.5411.143. [DOI] [PubMed] [Google Scholar]
- 32.Breitbach M, Bostani T, Roell W, Xia Y, Dewald O, Nygren JM, Fries JW, Tiemann K, Bohlen H, Hescheler J, et al. Potential risks of bone marrow cell transplantation into infarcted hearts. Blood. 2007;110:1362–1369. doi: 10.1182/blood-2006-12-063412. [DOI] [PubMed] [Google Scholar]
- 33.Conti MA, Even-Ram S, Liu C, Yamada KM, Adelstein RS. Defects in cell adhesion and the visceral endoderm following ablation of nonmuscle myosin heavy chain II-A in mice. J Biol Chem. 2004;279:41263–41266. doi: 10.1074/jbc.C400352200. [DOI] [PubMed] [Google Scholar]
- 34.Kostic A, Sap J, Sheetz MP. RPTPalpha is required for rigidity-dependent inhibition of extension and differentiation of hippocampal neurons. J Cell Sci. 2007;120:3895–3904. doi: 10.1242/jcs.009852. [DOI] [PubMed] [Google Scholar]
- *35.Paszek MJ, Zahir N, Johnson KR, Lakins JN, Rozenberg GI, Gefen A, Reinhart-King CA, Margulies SS, Dembo M, Boettiger D, et al. Tensional homeostasis and the malignant phenotype. Cancer Cell. 2005;8:241–254. doi: 10.1016/j.ccr.2005.08.010. This study investigates the relationship between rigidity of breast tumors and the malignancy of the cancer cells. The increased stiffness in tumors induces increased cytoskeletal tension in mammary epithelial cells, which disrupts their ability to form normal acini, leads them to be more proliferative, and more responsive to growth factors, implicating the elasticity of the tumor microenvironment in perpetuating the malignancy of cancer cells. [DOI] [PubMed] [Google Scholar]
- 36.Wang N, Naruse K, Stamenovic D, Fredberg JJ, Mijailovich SM, Tolic-Norrelykke IM, Polte T, Mannix R, Ingber DE. Mechanical behavior in living cells consistent with the tensegrity model. Proc Natl Acad Sci U S A. 2001;98:7765–7770. doi: 10.1073/pnas.141199598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Schwaiger I, Sattler C, Hostetter DR, Rief M. The myosin coiled-coil is a truly elastic protein structure. Nat Mater. 2002;1:232–235. doi: 10.1038/nmat776. [DOI] [PubMed] [Google Scholar]
- 38.Furuike S, Ito T, Yamazaki M. Mechanical unfolding of single filamin A (ABP-280) molecules detected by atomic force microscopy. FEBS Lett. 2001;498:72–75. doi: 10.1016/s0014-5793(01)02497-8. [DOI] [PubMed] [Google Scholar]
- 39.Law R, Harper S, Speicher DW, Discher DE. Influence of lateral association on forced unfolding of antiparallel spectrin heterodimers. J Biol Chem. 2004;279:16410–16416. doi: 10.1074/jbc.M313107200. [DOI] [PubMed] [Google Scholar]
- 40.Lee G, Abdi K, Jiang Y, Michaely P, Bennett V, Marszalek PE. Nanospring behaviour of ankyrin repeats. Nature. 2006;440:246–249. doi: 10.1038/nature04437. [DOI] [PubMed] [Google Scholar]
- 41.Bhasin N, Carl P, Harper S, Feng G, Lu H, Speicher DW, Discher DE. Chemistry on a single protein, vascular cell adhesion molecule-1, during forced unfolding. J Biol Chem. 2004;279:45865–45874. doi: 10.1074/jbc.M404103200. [DOI] [PubMed] [Google Scholar]
- 42.Puklin-Faucher E, Gao M, Schulten K, Vogel V. How the headpiece hinge angle is opened: New insights into the dynamics of integrin activation. J Cell Biol. 2006;175:349–360. doi: 10.1083/jcb.200602071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chiancone E, Currell DL, Vecchini P, Antonini E, Wyman J. Kinetics of the reaction of the “masked” and “free” sulfhydryl groups of human hemoglobin with p-mercuribenzoate. J Biol Chem. 1970;245:4105–4111. [PubMed] [Google Scholar]
- 44.Silverman JA, Harbury PB. Rapid mapping of protein structure, interactions, and ligand binding by misincorporation proton-alkyl exchange. J Biol Chem. 2002;277:30968–30975. doi: 10.1074/jbc.M203172200. [DOI] [PubMed] [Google Scholar]
- 45.Ward SD, Hamdan FF, Bloodworth LM, Siddiqui NA, Li JH, Wess J. Use of an in situ disulfide cross-linking strategy to study the dynamic properties of the cytoplasmic end of transmembrane domain VI of the M3 muscarinic acetylcholine receptor. Biochemistry. 2006;45:676–685. doi: 10.1021/bi051503q. [DOI] [PubMed] [Google Scholar]
- **46.Johnson CP, Tang HY, Carag C, Speicher DW, Discher DE. Forced unfolding of proteins within cells. Science. 2007;317:663–666. doi: 10.1126/science.1139857. This study introduces a method to detect force-induced protein conformational changes in the whole proteme of living cells and indentifies spectrin, myosin, filamin and vimentin as force sensitive proteins. Utilizing dyes reactive to the usually buried zzcysteine residues in proteins, increased labeling of proteins under force and the position of the differentially labeled cysteines are identified by mass spectrometry. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Engler A, Carag C, Johnson C, Speicher D, Sanger J, Sanger J, Discher DE. Cardiomyocyte beating is optimal on elastic matrices that mimic normal heart - Beating stops on mechanical mimics of fibrotic scars. Journal of Cell Science. to appear. [Google Scholar]
- **48.Sawada Y, Tamada M, Dubin-Thaler BJ, Cherniavskaya O, Sakai R, Tanaka S, Sheetz MP. Force sensing by mechanical extension of the Src family kinase substrate p130Cas. Cell. 2006;127:1015–1026. doi: 10.1016/j.cell.2006.09.044. This study implicates p130Cas as a link between mechanical stress and intracellular signaling. p130Cas is found to unfold under stress, exposing its substrate domain for phosphorylation by Src family kinases. [DOI] [PMC free article] [PubMed] [Google Scholar]




