Summary
Two mechanisms safeguard the bipolar attachment of chromosomes in mitosis. A correction mechanism destabilizes erroneous attachments that do not generate tension across sister kinetochores [1]. In response to unattached kinetochores, the mitotic checkpoint delays anaphase onset by inhibiting the anaphase-promoting complex/cyclosome (APC/CCdc20) [2]. Upon satisfaction of both pathways, the APC/CCdc20 elicits the degradation of securin and cyclin B [3]. This liberates separase triggering sister chromatid disjunction and inactivates cyclin-dependent kinase 1 (Cdk1) causing mitotic exit. How eukaryotic cells avoid the engagement of attachment monitoring mechanisms when sister chromatids split and tension is lost at anaphase is poorly understood [4]. Here we show that Cdk1 inactivation disables mitotic checkpoint surveillance at anaphase onset in human cells. Preventing cyclin B1 proteolysis at the time of sister chromatid disjunction destabilizes kinetochore-microtubule attachments and triggers the engagement of the mitotic checkpoint. As a consequence, mitotic checkpoint proteins accumulate at anaphase kinetochores, the APC/CCdc20 is inhibited, and securin reaccumulates. Conversely, acute pharmacological inhibition of Cdk1 abrogates the engagement and maintenance of the mitotic checkpoint upon microtubule depolymerization. We propose that the simultaneous destruction of securin and cyclin B elicited by the APC/CCdc20 couples chromosome segregation to the dissolution of attachment monitoring mechanisms during mitotic exit.
Graphical Abstract
Highlights
-
•
Cyclin B degradation stabilizes kinetochore attachments in anaphase
-
•
Cdk1 inactivation prevents mitotic checkpoint engagement after anaphase onset
-
•
Chromatid disjunction by separase causes APC/C inhibition if Cdk1 remains active
-
•
APC/C links separase activation to dissolution of attachment monitoring mechanisms
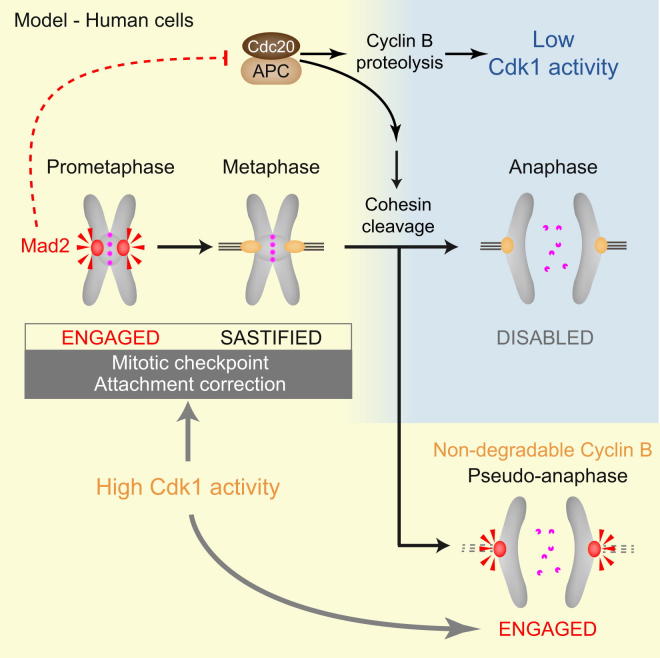
Vázquez-Novelle et al. propose that Cdk1-cyclin B activity sets the temporal window for microtubule-kinetochore attachment surveillance. Failure to degrade cyclin B at anaphase onset destabilizes attachments and engages the mitotic checkpoint. APC/C-induced proteolysis could couple sister chromatid disjunction to checkpoint dissolution when sister chromatids split.
Results and Discussion
Sister chromatids are held together until the metaphase-to-anaphase transition by a protein complex called cohesin [5]. After the destruction of securin, cleavage of cohesin by separase triggers the segregation of sister chromatids during anaphase. Cohesin cleavage also presumably abrogates the kinetochore tension that satisfies kinetochore attachment surveillance pathways. This raises the question of how eukaryotic cells avoid the potentially catastrophic action of these pathways during sister chromatid segregation at anaphase [4]. Recent work has shown that the artificial cleavage of cohesin in mitosis engages the mitotic checkpoint and destabilizes microtubule-kinetochore attachments [6, 7]. Eukaryotic cells may have solved this “anaphase problem” by entirely disabling these surveillance mechanisms at anaphase onset [8, 9]. The removal of Aurora B kinase, the tension sensor at kinetochores, from chromosomes at anaphase onset and the degradation of the checkpoint kinase Mps1 could contribute to this phenomenon [6, 8, 9]. Work in yeast cells, Drosophila embryos, and frog extracts has suggested that Cdk1-cyclin B, the enzyme that drives cells into mitosis, could act as an upstream regulator of the mitotic checkpoint [6, 7, 10–13]. The inactivation of Cdk1 through cyclin B proteolysis at anaphase onset could therefore disable attachment-monitoring pathways when sister chromatids split.
To investigate this hypothesis, we expressed a nondegradable (ND) version of cyclin B1 in HeLa Kyoto cells (ND cyclin B1-mCherry) (Figure 1). This mutant cyclin B1 protein (R42A and L45A) cannot be marked for proteolytic degradation by the anaphase-promoting complex/cyclosome (APC/CCdc20) ubiquitin ligase [3, 14]. Although Cdk1-cyclin B can inhibit separase [15], previous studies in Drosophila embryos and human cells have found that expression of a moderate level of ND cyclin B arrests cells in mitosis with separated sister chromatids [10, 16, 17]. Image quantification in our experiments revealed that both wild-type (WT) and ND cyclin B1 transgenes were expressed at levels close to the endogenous counterpart in the majority of cells (Figure S1A available online) (0.5-fold to 2-fold of endogenous cyclin B1).
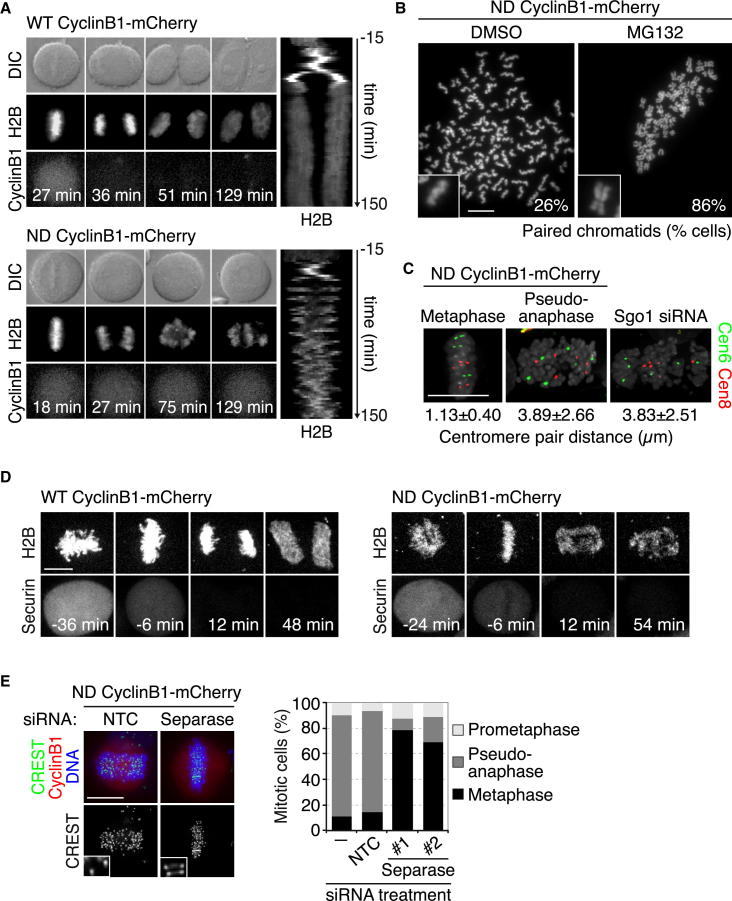
Figure 1.
Expression of Moderate Levels of Nondegradable Cyclin B1 Separates APC/CCdc20 Activation and Sister Chromatid Disjunction from Cdk1 Inactivation
(A) Time-lapse series and kymograph of H2B-EGFP HeLa cells expressing wild-type (WT) or nondegradable (ND) cyclin B1-mCherry. While 93% of cells expressing ND cyclin B1 arrested in mitosis, all cells expressing WT cyclin B1 exited mitosis (n > 40 cells from three independent experiments). The first time frame after nuclear envelope breakdown (NEBD) corresponds to t = 0 min. See also Figure S1.
(B) Chromosome spreads of cells expressing ND cyclin B1 that were synchronously released into mitosis and treated with DMSO (control) or the proteasome inhibitor MG132 (10 μM) for 4 hr (n > 299 spreads from 3 independent experiments). See also Figures S1E and S1F.
(C) Fluorescence in situ hybridization (FISH) analysis of the centromeres of trisomic chromosomes 6 and 8. Distance between the closest centromeres was measured on 3D pictures (n > 29 cells). Depletion of Sgo1 was used as a control for loss of sister chromatid cohesion.
(D) Analysis of securin degradation. Confocal live-cell imaging of H2B-iRFP HeLa cells expressing securin-EGFP and WT or ND cyclin B1-mCherry is shown. Time = 0 min was set to the first frame after anaphase onset. See also Figure 3A.
(E) Immunofluorescence (IF) analysis and quantification of mitotic cells expressing ND cyclin B1 and transfected with either nontargeting control (NTC) small interfering RNA (siRNA) or siRNA duplexes targeting separase (n > 152 cells from three independent experiments). See also Figure S1G.
Scale bars represent 10 μm. See also Figure S1.
WT cyclin B1 was degraded at the metaphase-to-anaphase transition, and cells underwent successful cell division before exiting mitosis (Figure 1A). In contrast, after alignment of chromosomes at the metaphase plate with normal kinetics, cells expressing ND cyclin B1 failed to degrade the transgenic protein. Cells with moderate levels of ND cyclin B1 subsequently displayed erratic chromosome movements and remained permanently arrested in mitosis (Figure 1A) (38/41 cells). The maintenance of this state, which we refer to as pseudoanaphase, was dependent on continuous Cdk1 activity (Figure S1B). The onset of pseudoanaphase in cells expressing ND cyclin B1 occurred with a timing that was comparable to anaphase onset in control cells (Figure S1C). This is in stark contrast to chromosome scattering caused by cohesion fatigue [18, 19], which takes several hours to occur in metaphase-arrested cells (Figure S1D).
Chromosome spread analysis and fluorescence in situ hybridization experiments revealed that sister chromatids were split in pseudoanaphase cells (Figures 1B and 1C). The loss of sister chromatid cohesion in pseudoanaphase cells, but not in cells lacking the cohesin protector protein Sgo1 [5], could be suppressed by addition of the proteasome inhibitor MG132 (Figures 1B, S1E, and S1F). Furthermore, chromosome scattering occurred concomitantly with the degradation of securin and within a short time after removal of MG132 (Figures 1D, 3A, and S1D). Importantly, chromatid scattering in cells expressing ND cyclin B1 was abolished by depletion of separase (Figures 1E and S1G). Our observations suggest that the transition from metaphase to pseudoanaphase in cells expressing ND cyclin B1 involves APC/C-dependent proteolysis and separase-dependent disjunction of sister chromatids, a situation closely resembling normal anaphase onset. Thus, expression of moderate levels of ND cyclin B1 separates APC/CCdc20 action and anaphase onset from the inactivation of Cdk1. In line with this interpretation, the tension sensor at kinetochores, Aurora B kinase [1], whose removal from chromosomes at the metaphase-to-anaphase transition depends on Cdk1 inactivation [20], remained associated with centromeres in pseudoanaphase cells (Figure S2A). Cells expressing ND cyclin B1 therefore provided a suitable model to investigate the consequences of separase-induced sister chromatid disjunction in the absence of Cdk1 inactivation.
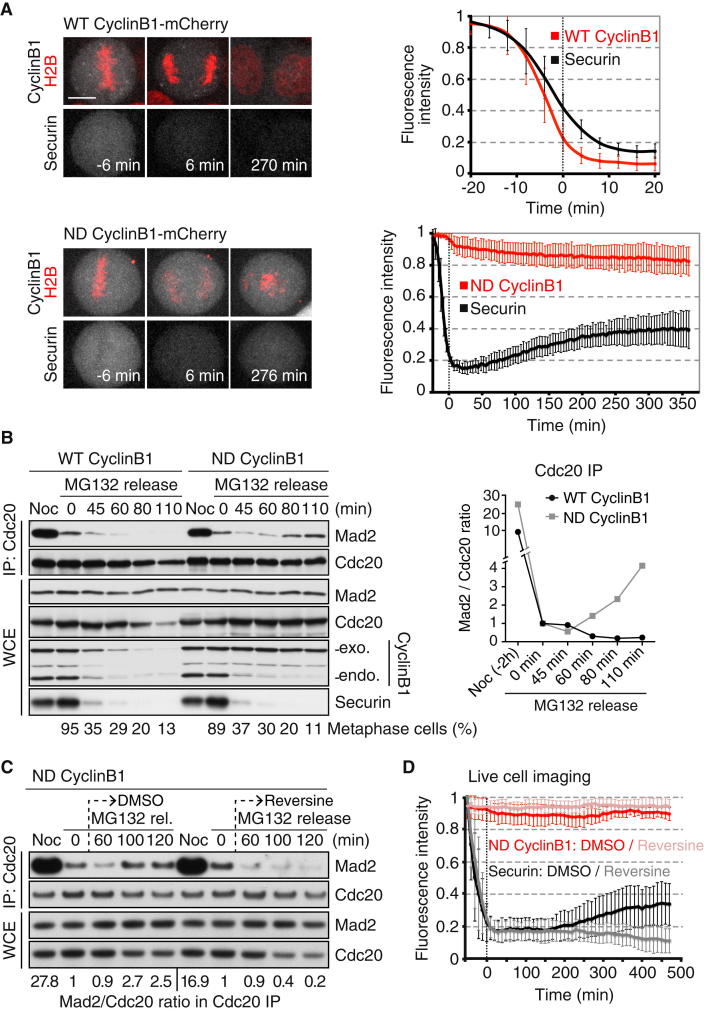
Figure 3.
Reaccumulation of Securin and MCC after Sister Chromatid Disjunction in the Absence of Cyclin B1 Proteolysis
(A) Frames of confocal live-cell imaging of H2B-iRFP HeLa cells expressing securin-EGFP and WT or ND cyclin B1-mCherry are shown (left). The integrated intensities of securin-EGFP and cyclin B1-mCherry were measured, background corrected, and normalized to the maximum-intensity value obtained per cell. Measurements in different cells were aligned to t = 0 min as the first frame after anaphase onset. The graph displays the mean intensity of securin and cyclin B1 (right). Error bars represent the SD of the analysis of nine cells per condition (from three experiments). The scale bar represents 10μm.
(B) Coimmunoprecipitation analysis of mitotic checkpoint complex (MCC) formation in cells expressing WT or ND cyclin B1. Cdc20 was immunoprecipitated from extracts prepared from nocodazole-arrested (Noc) and MG132-arrested cells at t = 0 min as well as from cells released from metaphase for the indicated times. Mad2 and Cdc20 intensities were analyzed in whole-cell extracts (WCE) and the precipitated fraction (IP) by fluorescent immunoblotting. The Mad2/Cdc20 ratio in the IP fraction was determined, normalized to the ratio at 0 min, and plotted (right). Due to the degradation of Cdc20 in WT cyclin B1-expressing cells, the amount of IP fraction loaded was adjusted for comparable amounts of Cdc20. The percentage of metaphase cells was determined using time-lapse analysis (n > 102 cells).
(C) Analysis of MCC formation as in (B). Cells expressing ND cyclin B1 were treated with DMSO or 0.5 μM reversine 40 min after MG132 release.
(D) Securin-EGFP accumulation in pseudoanaphase cells depends on Mps1 kinase. One hour after release from nocodazole (30 ng/ml), cells expressing ND cyclin B1 were treated with DMSO or reversine (0.5 μM). The graph is as in (A) (n = 9 cells per condition from three independent experiments).
See also Figure S3.
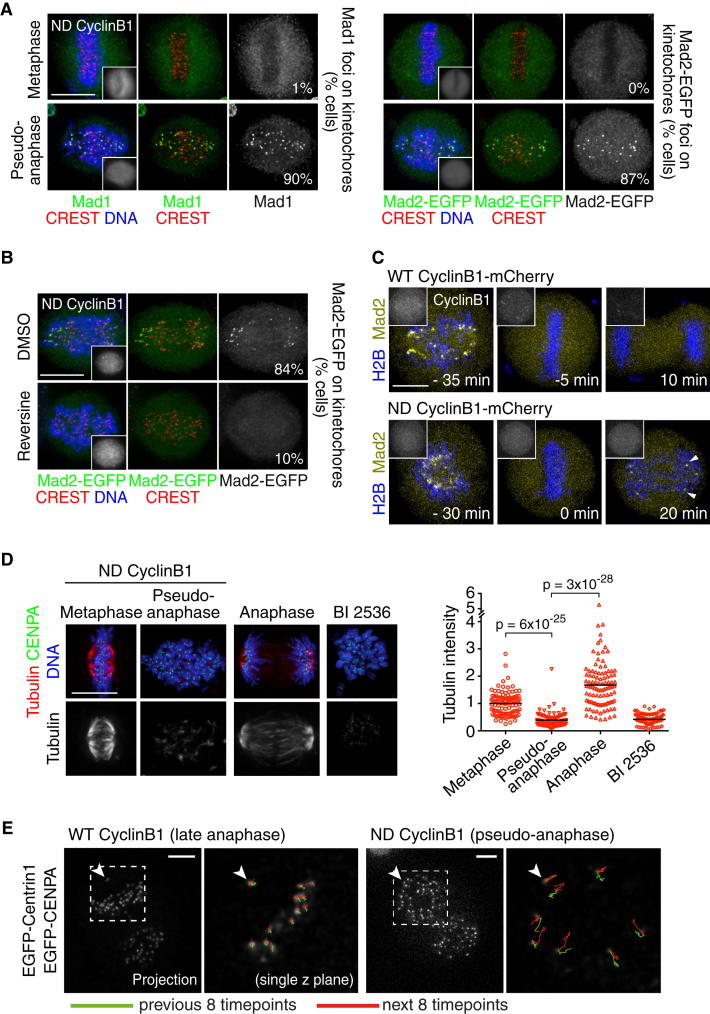
First, we scrutinized the localization of mitotic checkpoint proteins, which in early mitosis mark unattached kinetochores or kinetochores that lack tension [1, 2]. When stable bipolar attachments are formed at metaphase, mitotic checkpoint proteins dissociate from kinetochores and subsequently become undetectable at this location in anaphase [8]. Accordingly, kinetochore localization of mitotic checkpoint proteins was undetectable (Mad1, Mad2, and Mps1) or low (Bub1 and BubR1) in ND cyclin B1-positive metaphase cells (Figures 2A and S2B–S2D). In contrast, we found that Mad1, Mad2, Bub1, BubR1, and Mps1 were all recruited to kinetochores in pseudoanaphase cells that express ND cyclin B1 (Figures 2A and S2B–S2D). Kinetochore localization of Mad2 in pseudoanaphase cells was abrogated upon treatment of cells with reversine, an inhibitor of the mitotic checkpoint kinase Mps1 [21] (Figure 2B), and upon inhibition of Aurora B kinase (Figure S2E). Imaging of cells expressing ND cyclin B1 revealed the formation of Mad2-EGFP foci on chromosomes within 10 min (12/16 cells) or 15 min (4/16 cells) of pseudoanaphase onset (Figure 2C). Mad2-EGFP remained undetectable on chromosomes in cells that underwent anaphase while degrading WT cyclin B1, despite marking unaligned chromosomes in early mitosis (Figure 2C). These results suggest that separase-dependent cohesin cleavage and sister chromatid separation cause the re-engagement of the mitotic checkpoint pathway if cyclin B1 proteolysis is prevented and Cdk1 remains active at anaphase onset.
Figure 2.
Preventing Cyclin B1 Proteolysis at the Time of Sister Chromatid Separation Causes Recruitment of Mitotic Checkpoint Proteins to Kinetochores and the Destabilization of Microtubule-Kinetochore Attachments
(A) Mad1 and Mad2 localization was analyzed by IF in cells expressing ND cyclin B1-mCherry (n > 149 cells from three independent experiments). See also Figures S2A–S2D for Aurora B, Bub1, BubR1, and Mps1 localization.
(B) Mad2 localization was analyzed by IF 2 hr after MG132 release in cells expressing ND cyclin B1-mCherry. DMSO or reversine (0.5 μM) was added 40 min after release from MG132 arrest (n > 149 cells from three independent experiments). See also Figure S2E for effect of Aurora B inhibition.
(C) Confocal live-cell imaging of Mad2-EGFP H2B-iRFP HeLa cells expressing WT or ND cyclin B1-mCherry. Mad2 foci on chromosomes, indicated by arrowheads, are detected 4.7 ± 3.8 min after anaphase onset (t = 0 min) (n = 16 cells from more than three independent experiments).
(D) Microtubule stability was analyzed by IF in cells using cold treatment. Integrated intensities of cold-resistant α-tubulin were measured and background corrected in EGFP-CENPA HeLa cells that were either transfected with ND cyclin B1-mCherry or treated for 2 hr with 100 nM BI 2536 (n > 101 cells from three independent experiments). Tubulin intensities per cell are normalized to the average tubulin intensity in metaphase cells in each experiment and displayed in a scatter dot plot (right). Horizontal black lines represent the mean. p values were calculated using a two-tailed t test. Scale bars represent 10 μm.
(E) Analysis of kinetochore and spindle pole (indicated by arrowheads) trajectories in late anaphase cells (WT cyclin B1) and in pseudoanaphase cells (ND cyclin B1). Images are representative of six late-anaphase and nine pseudoanaphase cells analyzed. Scale bars represent 5 μm. See also Figure S2F.
See also Figure S2.
Mad1 and Mad2 kinetochore recruitment is thought to reflect partial or complete lack of microtubule attachment [1, 2]. The chaotic chromatid movements and the localization of Mad1 and Mad2 to multiple kinetochores that is observed upon sister splitting in cells expressing ND cyclin B1 raise the possibility that kinetochore-microtubule attachments are unstable in pseudoanaphase. To test this, we analyzed the stability of kinetochore fibers using cold treatment. Exposure to low temperature allows the preservation of stable kinetochore fibers in metaphase and anaphase cells but eliminates compromised attachments, such as those in cells treated with the Polo-like kinase 1 inhibitor BI 2536 (Figure 2D). Although kinetochore fibers in cells expressing ND cyclin B1 were cold resistant at metaphase, microtubule stability was dramatically reduced in the pseudoanaphase state (Figure 2D). Furthermore, tracking experiments revealed that kinetochores in pseudoanaphase cells failed to show persistent directional movement toward the closest spindle pole and underwent directional switches (Figures 2E and S2F). In vivo analysis of kinetochore architecture in yeast found that anaphase kinetochores adopted a relaxed configuration, possibly reflecting a loss of tension [22]. Our results suggest that, if cyclin B is not degraded, sister chromatid separation at anaphase onset could cause the erroneous destabilization of microtubule attachments at tensionless kinetochores and, subsequently, the untimely recruitment of mitotic checkpoint proteins.
Next, we tested whether these responses to sister chromatid separation in pseudoanaphase cells can lead to the inhibition of the APC/CCdc20. To address this question, we analyzed the level of the APC/CCdc20 substrate securin and the assembly of the mitotic checkpoint complex (MCC) over time in cells expressing either WT or ND cyclin B1 (Figure 3). The MCC is a key biochemical effector of the mitotic checkpoint and is composed of BubR1, Mad2, Bub3, and the APC/C cofactor Cdc20 [2]. In cells expressing WT cyclin B1, both securin and cyclin B1 were degraded at the metaphase-to-anaphase transition (Figure 3A). In cells expressing ND cyclin B1, securin was degraded at the time of chromosome scattering, but subsequently its protein levels rose again, albeit slowly and not reaching mitotic peak levels (Figure 3A). Coimmunoprecipitation of Cdc20 and Mad2 suggested that cells expressing ND cyclin B1 reassemble the APC/CCdc20 inhibitor MCC after undergoing sister chromatid separation (Figure 3B). Importantly, inhibition of Mps1 kinase at the time of pseudoanaphase onset prevented subsequent MCC formation and securin accumulation (Figures 3C, 3D, and S3). These data suggest that although both WT and ND cyclin B1-expressing cells activate the APC/CCdc20 and degrade APC/C substrates at the time of sister disjunction, maintenance of Cdk1 activity in ND cyclin B1-positive cells past the metaphase-to-anaphase transition elicits inhibition of the APC/CCdc20. Prevention of cyclin B1 proteolysis at anaphase onset appears to be sufficient to cause mitotic checkpoint engagement and APC/CCdc20 inhibition in response to cohesin cleavage. The corollary of this conclusion is that the inactivation of Cdk1 just prior to anaphase onset in normal cells could be responsible for preventing untimely checkpoint engagement caused by sister chromatids splitting.
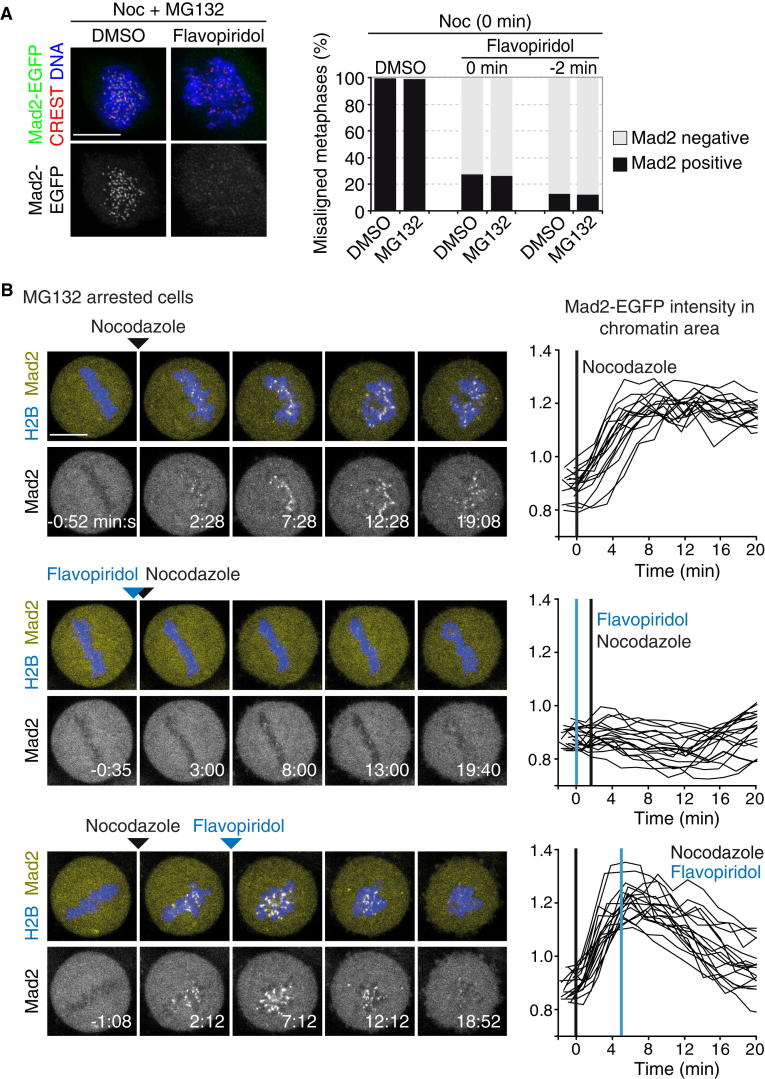
To test this hypothesis and corroborate the link between Cdk1 activity and mitotic checkpoint surveillance, we conducted acute pharmacological perturbation experiments in mitotic cells. As a proxy for mitotic checkpoint engagement, we used the rapid recruitment of Mad2-EGFP to kinetochores upon microtubule depolymerization by nocodazole (Figures 4A, 4B [upper panel], and S4A). Addition of the Cdk inhibitor flavopiridol about 2 min prior to or at the time of nocodazole application prevented the localization of Mad2-EGFP to kinetochores (Figures 4A and 4B [middle panel]). We noticed that flavopiridol treatment also slowed down the depolymerization of microtubules and dissolution of the metaphase plate after nocodazole treatment (Figure S4B). This effect, however, is unlikely to account for the absence of Mad2 recruitment, because the protein also failed to accumulate at kinetochores at time points when the spindle was largely disassembled (Figures 4B [middle panel] and S4B) and in cells with a disorganized metaphase plate (Figure 4A). Furthermore, we found that chemical inhibition of Cdk1 after microtubule depolymerization resulted in the loss of Mad2-EGFP from chromosomes within 15 min (Figure 4B, lower panel). These results suggest that Cdk1 activity is required for both mounting and maintaining a mitotic checkpoint response in human cells. The effects of chemical Cdk1 inhibition did not depend on proteolysis of other factors because addition of flavopiridol prevented or abolished the localization of Mad2-EGFP to kinetochores in the presence of MG132 (Figure 4). This indicates that the dephosphorylation of Cdk1 substrates causes the direct or indirect dissolution of mitotic checkpoint surveillance and highlights cyclin B as a key APC/CCdc20 substrate for this regulation at the metaphase-to-anaphase transition.
Figure 4.
Cdk1 Activity Is Required for Mad2 Recruitment and Retention at Kinetochores upon Microtubule Depolymerization
(A) IF analysis of Mad2-EGFP kinetochore localization. HeLa cells were arrested in metaphase by addition of MG132 for 2 hr and subsequently released (DMSO) or kept in presence of MG132 during the experiment. DMSO or flavopiridol (15 μM) was added 2 min prior to addition of nocodazole (1 μg/ml) or at the time of nocodazole addition. Kinetochore localization of Mad2-EGFP was analyzed in cells with disrupted chromosome alignment 10 min after treatment with nocodazole (n > 300 cells from three independent experiments).
(B) Mad2-EGFP kinetochore localization in live cells. HeLa cells expressing H2B-mCherry and Mad2-EGFP were arrested in metaphase with MG132 (30–90 min) and imaged by three-dimensional confocal live-cell microscopy. Nocodazole (1 μg/ml) and flavopiridol (20 μM) were acutely added during imaging as indicated by the arrowheads and vertical lines in black and blue, respectively. Images show single confocal z sections of representative cells. Graphs show quantified kinetics of Mad2 intensity on chromatin regions over cytoplasm in individual cells (n > 15 cells from three independent experiments). Time = 0:00 min:s at the first drug addition.
Scale bars represent 10 μm. See also Figure S4 for microtubule depolymerization kinetics.
We found that the central event of anaphase, the separase-dependent disjunction of sister chromatids, can lead to the destabilization of kinetochore attachments and the engagement of a mitotic checkpoint response with potentially deleterious consequences. Our experiments suggest that eukaryotic cells avoid this by coordinating separase activation with the termination of attachment surveillance mechanisms. This is achieved through the action of the APC/CCdc20 that elicits the simultaneous destruction of securin and cyclin B. This coupling could render late metaphase cells refractory to the loss of tension caused by cohesin cleavage upon anaphase initiation and contribute to the irreversibility of mitotic exit [23]. Experiments in mouse zygotes have led to similar conclusions, suggesting conservation of this mechanism [24]. While cell-cycle surveillance mechanisms are generally considered to exist in two states, engaged or satisfied, our work highlights a third state in which a mechanism has been disabled and is no longer monitoring cellular events. The transition from the satisfied to the disabled state of the mitotic checkpoint most likely occurs very briefly before anaphase entry, as acutely detached chromosomes remain proficient in recruiting Mad2-EGFP and inhibiting APC/CCdc20 throughout most of metaphase [25, 26].
Studies in Drosophila and yeast cells have shown that reversal of Cdk1 activity prevents engagement of the mitotic checkpoint in response to artificial sister chromatid disjunction [6, 7]. Our work suggests that cyclin B1’s destruction stabilizes kinetochore attachments and terminates mitotic checkpoint surveillance after anaphase onset. Separase-induced sister chromatid separation can elicit the inhibition of APC/CCdc20 if Cdk1 is not inactivated concomitantly. This raises the possibility that if the mitotic checkpoint is not disabled in anaphase, the reaccumulation of securin and cyclin B could interfere with chromatid separation and mitotic exit. However, after sister chromatid splitting in ND cyclin B1-expressing cells, securin-EGFP, as well as the endogenous APC/CCdc20 substrates cyclin B1 and securin, did not reaccumulate significantly within a short timeframe (Figures 3A and 3B). The slow reaccumulation could be linked to the requirement for resynthesis and to only partial and slow inhibition of APC/CCdc20 [26, 27], which may help protect mitotic exit from the adverse effects of untimely mitotic checkpoint engagement in anaphase. Nevertheless, failure to degrade cyclin B at anaphase onset in mouse zygotes causes mitotic checkpoint-dependent inhibition of APC/CCdc20 substrate degradation [24], suggesting physiological importance of mitotic checkpoint dissolution by Cdk1 inactivation.
The loss of stable kinetochore attachments that we observed upon chromatid separation in the presence of Cdk1 activity in human cells could result in chromosome nondisjunction events and lead to aneuploidy. Recent work showed that the degradation of cyclin A increases the stability of kinetochore-microtubule attachments between prometaphase and metaphase [28]. Kinetochore fiber stability further increases in anaphase cells [29]. We detected destabilization of kinetochore-microtubule attachments, as judged by Mad2 recruitment to unattached kinetochores, within 10 min of sister chromatid splitting in the presence of ND cyclin B1. Thus, cyclin B proteolysis and the reversal of Cdk1-dependent phosphorylation are likely to be crucial for the generation of stable kinetochore-microtubule attachments that underlie the correct partitioning of sister chromatids in anaphase [30].
In combination with previous studies [6–8, 10–12], this work establishes Cdk1-cyclin B as a switch that sets up mitotic checkpoint surveillance as cells enter mitosis and disables surveillance during anaphase. Cdk1-dependent localization of Aurora B to centromeres [8], together with additional Cdk1-controlled effects at kinetochores, is likely to sustain the mitotic checkpoint. In the future, it will be important to identify these additional substrates whose phosphoregulation confers direct or indirect Cdk1 control over mitotic checkpoint surveillance and the stability of kinetochore-microtubule attachments.
Acknowledgments
We would like to thank Julian Gannon, Silke Hauf, Tony Hyman, Peter Lenart, Andrea Musacchio, Jan-Michael Peters, Jonathon Pines, Kuan-Chung Su, and Stephen Taylor for reagents and advice. We are grateful to Peter Jordan and Daniel Zicha for help with light microscopy, Richard Mitter for bioinformatic support, and Sriramkumar Sundaramoorthy for comments on the manuscript. Research in the Gerlich laboratory has received funding from the European Community’s Seventh Framework Programme FP7/2007-2013 under grant agreement numbers 241548 (MitoSys) and 258068 (Systems Microscopy), an ERC Starting Grant (agreement number 281198), the EMBO Young Investigator Programme, the Swiss National Science Foundation, the Austrian Science Fund (FWF)-funded project “SFB Chromosome Dynamics,” and a Summer Research Award of the Marine Biology Laboratory Woods Hole (Laura and Arthur Colwin Endowed Summer Research Fellowship Fund). A.E.D. is a fellow of the Zurich Ph.D. Program in Molecular Life Sciences and has received funding from a Ph.D. fellowship by the Boehringer Ingelheim Fonds and from a Peter Müller fellowship. A.D.M. was supported by a Biotechnology and Biological Sciences Research Council (BBSRC) project grant (grant number BB/I021353/1). C.A.S. was supported by the Engineering and Physical Sciences Research Council (EPSRC) (grant number EP/F500378/1) via the MOAC doctoral training center. M.D.V.-N. acknowledges support by a Ramón Areces Foundation Fellowship. L.S. was supported by fellowships from the European Molecular Biology Organization (EMBO) and the Canadian Institutes of Health Research (CIHR). Work in the lab of M.P. is supported by Cancer Research UK and the EMBO Young Investigator Programme.
Footnotes
This is an open-access article distributed under the terms of the Creative Commons Attribution-NonCommercial-No Derivative Works License, which permits non-commercial use, distribution, and reproduction in any medium, provided the original author and source are credited.
Contributor Information
María Dolores Vázquez-Novelle, Email: lolavazquez.novelle@cancer.org.uk.
Mark Petronczki, Email: mark.petronczki@cancer.org.uk.
Supplemental Information
References
- 1.Nezi L., Musacchio A. Sister chromatid tension and the spindle assembly checkpoint. Curr. Opin. Cell Biol. 2009;21:785–795. doi: 10.1016/j.ceb.2009.09.007. [DOI] [PubMed] [Google Scholar]
- 2.Lara-Gonzalez P., Westhorpe F.G., Taylor S.S. The spindle assembly checkpoint. Curr. Biol. 2012;22:R966–R980. doi: 10.1016/j.cub.2012.10.006. [DOI] [PubMed] [Google Scholar]
- 3.Peters J.M. The anaphase promoting complex/cyclosome: a machine designed to destroy. Nat. Rev. Mol. Cell Biol. 2006;7:644–656. doi: 10.1038/nrm1988. [DOI] [PubMed] [Google Scholar]
- 4.Vázquez-Novelle M.D., Mirchenko L., Uhlmann F., Petronczki M. The ‘anaphase problem’: how to disable the mitotic checkpoint when sisters split. Biochem. Soc. Trans. 2010;38:1660–1666. doi: 10.1042/BST0381660. [DOI] [PubMed] [Google Scholar]
- 5.Nasmyth K., Haering C.H. Cohesin: its roles and mechanisms. Annu. Rev. Genet. 2009;43:525–558. doi: 10.1146/annurev-genet-102108-134233. [DOI] [PubMed] [Google Scholar]
- 6.Mirchenko L., Uhlmann F. Sli15(INCENP) dephosphorylation prevents mitotic checkpoint reengagement due to loss of tension at anaphase onset. Curr. Biol. 2010;20:1396–1401. doi: 10.1016/j.cub.2010.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Oliveira R.A., Hamilton R.S., Pauli A., Davis I., Nasmyth K. Cohesin cleavage and Cdk inhibition trigger formation of daughter nuclei. Nat. Cell Biol. 2010;12:185–192. doi: 10.1038/ncb2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Vázquez-Novelle M.D., Petronczki M. Relocation of the chromosomal passenger complex prevents mitotic checkpoint engagement at anaphase. Curr. Biol. 2010;20:1402–1407. doi: 10.1016/j.cub.2010.06.036. [DOI] [PubMed] [Google Scholar]
- 9.Palframan W.J., Meehl J.B., Jaspersen S.L., Winey M., Murray A.W. Anaphase inactivation of the spindle checkpoint. Science. 2006;313:680–684. doi: 10.1126/science.1127205. [DOI] [PubMed] [Google Scholar]
- 10.Parry D.H., Hickson G.R., O’Farrell P.H. Cyclin B destruction triggers changes in kinetochore behavior essential for successful anaphase. Curr. Biol. 2003;13:647–653. doi: 10.1016/s0960-9822(03)00242-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.D’Angiolella V., Mari C., Nocera D., Rametti L., Grieco D. The spindle checkpoint requires cyclin-dependent kinase activity. Genes Dev. 2003;17:2520–2525. doi: 10.1101/gad.267603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yamaguchi S., Decottignies A., Nurse P. Function of Cdc2p-dependent Bub1p phosphorylation and Bub1p kinase activity in the mitotic and meiotic spindle checkpoint. EMBO J. 2003;22:1075–1087. doi: 10.1093/emboj/cdg100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Morin V., Prieto S., Melines S., Hem S., Rossignol M., Lorca T., Espeut J., Morin N., Abrieu A. CDK-dependent potentiation of MPS1 kinase activity is essential to the mitotic checkpoint. Curr. Biol. 2012;22:289–295. doi: 10.1016/j.cub.2011.12.048. [DOI] [PubMed] [Google Scholar]
- 14.Gavet O., Pines J. Progressive activation of CyclinB1-Cdk1 coordinates entry to mitosis. Dev. Cell. 2010;18:533–543. doi: 10.1016/j.devcel.2010.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Stemmann O., Zou H., Gerber S.A., Gygi S.P., Kirschner M.W. Dual inhibition of sister chromatid separation at metaphase. Cell. 2001;107:715–726. doi: 10.1016/s0092-8674(01)00603-1. [DOI] [PubMed] [Google Scholar]
- 16.Hagting A., Den Elzen N., Vodermaier H.C., Waizenegger I.C., Peters J.M., Pines J. Human securin proteolysis is controlled by the spindle checkpoint and reveals when the APC/C switches from activation by Cdc20 to Cdh1. J. Cell Biol. 2002;157:1125–1137. doi: 10.1083/jcb.200111001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wolf F., Wandke C., Isenberg N., Geley S. Dose-dependent effects of stable cyclin B1 on progression through mitosis in human cells. EMBO J. 2006;25:2802–2813. doi: 10.1038/sj.emboj.7601163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Daum J.R., Potapova T.A., Sivakumar S., Daniel J.J., Flynn J.N., Rankin S., Gorbsky G.J. Cohesion fatigue induces chromatid separation in cells delayed at metaphase. Curr. Biol. 2011;21:1018–1024. doi: 10.1016/j.cub.2011.05.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Stevens D., Gassmann R., Oegema K., Desai A. Uncoordinated loss of chromatid cohesion is a common outcome of extended metaphase arrest. PLoS ONE. 2011;6:e22969. doi: 10.1371/journal.pone.0022969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hümmer S., Mayer T.U. Cdk1 negatively regulates midzone localization of the mitotic kinesin Mklp2 and the chromosomal passenger complex. Curr. Biol. 2009;19:607–612. doi: 10.1016/j.cub.2009.02.046. [DOI] [PubMed] [Google Scholar]
- 21.Santaguida S., Tighe A., D’Alise A.M., Taylor S.S., Musacchio A. Dissecting the role of MPS1 in chromosome biorientation and the spindle checkpoint through the small molecule inhibitor reversine. J. Cell Biol. 2010;190:73–87. doi: 10.1083/jcb.201001036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Joglekar A.P., Bloom K., Salmon E.D. In vivo protein architecture of the eukaryotic kinetochore with nanometer scale accuracy. Curr. Biol. 2009;19:694–699. doi: 10.1016/j.cub.2009.02.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.He E., Kapuy O., Oliveira R.A., Uhlmann F., Tyson J.J., Novák B. System-level feedbacks make the anaphase switch irreversible. Proc. Natl. Acad. Sci. USA. 2011;108:10016–10021. doi: 10.1073/pnas.1102106108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rattani A., Vinod P.K., Godwin J., Tachibana-Konwalski K., Wolna M., Malumbres M., Novák B., Nasmyth K. Dependency of the spindle assembly checkpoint on Cdk1 renders the anaphase transition irreversible. Curr. Biol. 2014 doi: 10.1016/j.cub.2014.01.033. Published online February 27, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Clute P., Pines J. Temporal and spatial control of cyclin B1 destruction in metaphase. Nat. Cell Biol. 1999;1:82–87. doi: 10.1038/10049. [DOI] [PubMed] [Google Scholar]
- 26.Dick A.E., Gerlich D.W. Kinetic framework of spindle assembly checkpoint signalling. Nat. Cell Biol. 2013;15:1370–1377. doi: 10.1038/ncb2842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Collin P., Nashchekina O., Walker R., Pines J. The spindle assembly checkpoint works like a rheostat rather than a toggle switch. Nat. Cell Biol. 2013;15:1378–1385. doi: 10.1038/ncb2855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kabeche L., Compton D.A. Cyclin A regulates kinetochore microtubules to promote faithful chromosome segregation. Nature. 2013;502:110–113. doi: 10.1038/nature12507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhai Y., Kronebusch P.J., Borisy G.G. Kinetochore microtubule dynamics and the metaphase-anaphase transition. J. Cell Biol. 1995;131:721–734. doi: 10.1083/jcb.131.3.721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Higuchi T., Uhlmann F. Stabilization of microtubule dynamics at anaphase onset promotes chromosome segregation. Nature. 2005;433:171–176. doi: 10.1038/nature03240. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.