Abstract
Purpose
To investigate the adverse effect of intravitreal injection of normal saline (NS) and phosphate buffered saline (PBS) in mouse eyes.
Methods
NS or PBS was injected intravitreally into C57BL/6J mouse eyes. Retinal lesions were monitored by fundus imaging, spectral-domain optical coherence tomography (SD-OCT), and histological investigations. Retinal immune gene expression was determined by real-time polymerase chain reaction (PCR). The toxic effect of NS and PBS or retinal protein from NS- or PBS-injected eyes on retinal pigment epithelium (RPE) was tested in B6-RPE-07 mouse RPE cell cultures.
Results
Intravitreal injection of NS dose-dependently induced localized retinal lesion in mice. Histological investigations revealed multiple vacuoles in photoreceptor outer segments and RPE cells. The lesions recovered over time and by 3 weeks post injection the majority of lesions vanished in eyes receiving 1 μl NS. Inflammatory genes, including TNF-α, IL-1β, IL-6, iNOS, and VEGF were upregulated in NS injected eyes. Intravitreal injection of PBS did not cause any pathology. The treatment of B6-RPE07 cells with 30% PBS or 30% NS did not affect RPE viability. However, incubation of 1-μg/ml retinal protein from NS-injected eyes, but not PBS-injected eyes induced RPE cell death.
Conclusion
NS is toxic to the C57BL/6J mouse retina and should not be used as a vehicle for intraocular injection. PBS is not toxic to the retina and is a preferred vehicle.
Translational Relevance
NS is not a physiological solution for intraocular injection in the C57BL/6J mice and questions its suitability for intraocular injection in other species, including human.
Keywords: intraocular injection, retinal pigment epithelium, toxicity, photoreceptors, saline
Introduction
Intravitreal injection is an accepted route of drug delivery for the management of retinal and choroidal diseases. Therefore, injection of the drug or drug-containing devices directly into the vitreous cavity allows high levels of drug to be achieved in the posterior segment of the eye (retina and choroid), improving the efficacy of therapy for vitreoretinal diseases. Currently, the most common intravitreal injection in the clinic is the delivery of anti–vascular endothelial growth factor (VEGF) medications (bevacizumab [Genentech, San Francisco, CA] or ranibizumab [Novartis, Basel, Switzerland]), for the management of various retinal vascular diseases, including neovascular age-related macular degeneration (AMD),1,2 diabetic macular edema (DME),3–5 retinal vein occlusion,6,7 and neovascular glaucoma.8 In addition, intravitreal injection of steroids or steroid implants is widely used to treat various types of chronic intraocular inflammation,9–11 and the injection is considered relatively safe with only a very low risk of ocular damage and intraocular inflammation.12,13 In fact, the intravitreal drug delivery has revolutionized the management of the major sight-threatening conditions including AMD and DME. Hence, there is an insurgence in the use of intravitreal injection in preclinical studies, including intravitreal injection of different types of therapeutic compounds, molecular probes, and various gene transfection reagents.
In the clinic, different drugs use different vehicles (i.e., triamcinolone is in benzyl alcohol; Ranibizumab is in a solution containing histidine HCl, α, α-trehalose dehydrate, and polysorbate) and the drug companies normally test the toxicity of the vehicles before clinical applications. If further dilution is needed, normal saline (NS, 0.9% NaCl) or Hank's balanced salt solution (HBSS) would normally be used. NS and phosphate buffered saline (PBS) are the two most commonly used vehicles for intravitreal injection in preclinical studies. Surprisingly, we found that intravitreal injection of NS resulted in acute, localized retinal and retinal pigment epithelial (RPE) damage in the C57BL/6J mouse. On the other hand, intravitreal injection of PBS did not cause any detachable retinal damage.
Methods
Animal Handling
Adult (3-month old) C57BL/6J (Harlan, Indianapolis, IN) mice were used in this study. All animals were housed and bred in a normal experimental room and exposed to a 12 hour light/dark cycle with free access to food and water. All procedures were conducted under the regulation of the United Kingdom (UK) Home Office Animals (Scientific Research) Act 1986 and were in compliance with the ARVO Statement for the Use of Animals in Ophthalmology and Vision Research.
Reagents Used for Intravitreal Injection
Normal saline (NS): normal saline containing 0.9% NaCl was purchased from Fannin Healthcare Co. (Belfast, Northern Ireland). This commercial NS is a medical product and routinely used in the hospital for venous infusion. Phosphate Buffered Saline (PBS): 0.01M PBS was prepared freshly (0.8% NaCl, 0.02% KCl, 0.178% Na2HPO4.2H2O KH2PO4, pH 7.4). In addition, home-made 0.9% NaCl (0.9 g NaCl dissolved in 100 mL distilled water), and PBS adjusted to pH 6.0 were also used in the study. Home-made saline and PBS were sterilized before use.
Intravitreal Injection
Intravitreal injections were performed under a surgical microscope. Animals were anaesthetized with the gas anesthesia isofluorane (Merial; Animal Health Ltd., Essex, UK). Pupils were dilated using 1% tropicamide and 2.5% phenylephrine (Chauvin, Essex, UK). Viscotears Liquid Gel (Novartis Pharmaceuticals UK Ltd., Surrey, UK) and a microscope coverslip were used to improve the visibility of the fundus. A 33-gauge needle (Hamilton Bonaduz AG, Bonaduz, Switzerland) was inserted from the limbus with a 45° injection angle into the vitreous. The direction and location of the needle was monitored through the microscope. NS or PBS (1 or 2 μL) was injected using a repeating dispenser (PB-600-1; Hamilton Bonaduz). In the “needle punch group”, animals underwent the sample procedure without injecting any solution to the vitreous cavity. The control animals did not undergo any of the above procedures. All procedures were conducted by the same surgeon.
Clinical Investigations
Animals were anaesthetized and pupils dilated as described above. A topic endoscopic fundus imaging (TEFI) system, described previously,14,15 was used to obtain fundus images at baseline level (day 0), 1 day, and 1, 2, and 3 weeks after intravitreal injection. Images were captured using the Nikon D90 camera (Nikon UK Ltd., Surrey, UK) and saved in TIFF format. The brightness and contrast of the images were adjusted using Adobe Photoshop CS4 (Adobe Systems, Mountain View, CA).
Spectral-domain optical coherence tomography (SD-OCT) was conducted at 1 day and 3 weeks after intravitreal injection using the Spectralis Heidelberg OCT system (Heidelberg Engineering, Heidelberg, Germany) set to 30° field of view.
Retinal Morphometry
Retinal thickness was measured from the SD-OCT images using the Spectralis Heidelberg Eye Explorer software (Heidelberg Engineering). We made three types of measurements: (1) the total retinal thickness, from ganglion cell layer (GCL) to the outer limiting membrane (OLM), (2) inner retinal thickness, from GCL to the margin between the inner nuclear layer (INL) and the outer plexiform layer (OPL), and (3) outer retinal thickness, from OPL to OLM. Lesion and nonlesion regions from NS-injected eyes were measured separately and compared with other experimental groups at equivalent retinal eccentricities.
Histology
Mouse eyes were collected at 1 day and 3 weeks post eye procedures and fixed in 4% formalin for at least 24 hours. Samples were embedded in paraffin, sectioned at a thickness of 5 μm and processed for standard hematoxylin and eosin (H&E) staining. Sections were then examined for signs of pathology using a light microscope (Nikon Eclipse E400 light microscope; Nikon UK Ltd.).
Measure of Vitreous Humor pH
Animals were anaesthetized with an intraperitoneal injection of ketamine hydrochloride (60 mg/kg; Fort George Animal Centre, Southampton, UK) and xylazine (5mg/kg; Pharmacia & Veterinary Products, Kiel, Germany) and pupils dilated as described above. The vitreous humor was extracted from mouse eyes 1 hour after intravitreal injection (1 or 2 μL) of PBS or NS. A 33-gauge needle was inserted from the limbus with a 45° injection angle into the vitreous and cannulated with a 5-μm diameter glass capillary. Three- to 4-μL vitreous/eye was extracted by using a 1-mL insulin syringe coupled to the glass capillary. The vitreous pH was measured using pH test strips (6.0–7.7 pH, sensitivity 0.3 pH unit; Sigma, Cambridge, UK).
Real-Time Reverse Transcription Polymerase Chain Reaction (RT-PCR)
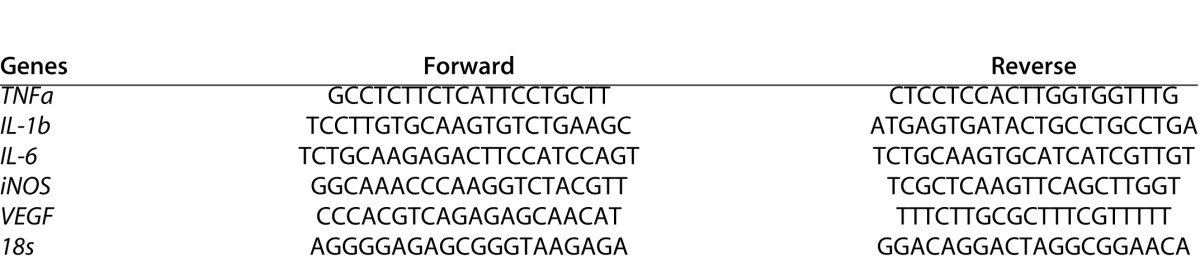
Total RNA was isolated from mouse retina and RPE/choroidal tissue using the RNeasy Mini Kit (Qiagen, West Sussex, UK) at 1 day after intraocular injection (n = 6 eyes for each group) and then reverse transcribed into complementary DNA (cDNA; SuperScript II reverse transcriptase; Invitrogen, Paisley, UK). All procedures were conducted following manufacturer's instructions. The quantity and quality of RNA were determined using a NanoDrop ND-1000 spectrophotometer (NanoDrop Technologies, Wilmington, DE). The same amount of total RNA was used for reverse transcription using Super-ScripTM II Reverse Transcriptase kit and random primers (Invitrogen). Real-time RT-PCR was performed using SYBR Green Master (Roche Diagnostics GmbH, Mannheim, Germany) in Light-CyclerH 480 system (Roche Diagnostics GmbH). 18s was used as a housekeeping gene to normalize the relative expression levels of target gene mRNA. The primer sequences used in this study are listed in Table 1.
Table 1. .
Primers Used for Real Time RT-PCR
Retinal Pigment Epithelial Cell Culture
The mouse RPE cell line B6-RPE07 were cultured in complete Dulbecco's modified Eagle medium (DMEM; PAA Laboratories Ltd., Yeovil, UK), supplemented with 10% fetal calf serum (FCS) (Sigma) and 0.2% primocin (InvivoGen, Tolouse, France) as detailed in our previous study.16 The cells were subcultured at the ratio 1:3 every 3 days.
Extraction of Retinal Protein
Retinal proteins were extracted from mouse eyes 24 hours after intravitreal injection (1 μL) of PBS or NS. For this purpose, retinas were dissected under a dissecting microscope within 2 to 3 minutes after eyes were collected. Retinal tissues were homogenized in distilled water (100 μL water/retina) and lysates centrifuged at 15,000g for 15 minutes. Only the soluble protein fraction was collected and the total protein concentration determined by the BCA Assay (Thermo Fisher Scientific, Waltham, MA).
Cytokine Measurement
Cytokines, including tumor necrosis factor (TNF)-α, interleukin (IL)-1β, and IL-6 were measured in retinal extracts of noninjected controls, PBS-injected and NS-injected mice (n ≥ 4) using the Cytometric Bead Array (CBA; BD Biosciences, Oxford, UK) kit according to manufacturer's instructions. Cytokine levels were normalized by total protein levels of the samples and expressed as nanogram cytokine per gram of total protein (ng/g).
RPE Viability Assay
The effect of PBS, NS, and retinal protein extracts on RPE cell viability was tested in B6-RPE07 cells. RPE cells were seeded into 24-well plates and incubated with 30% PBS or 30% NS in DMEM supplemented with 10% FCS for 1 or 24 hours. The concentration of PBS and NS used in the in vitro study was determined from the estimated in vivo concentration after intravitreal injection of 2 μL PBS or NS (mouse vitreous humor ∼5–6 μL/eye). In another group, RPE cells were treated with 1 μg/mL retinal extracts in 10% FCS-DMEM for 24 hours. Cell viability was determined in each group immediately after treatment, by incubating cells with 100 nM 4′,6-diamidino-2-phenyilindole (DAPI; Sigma-Aldrich) and 2 μg/mL Propidium Iodide (PI; Invitrogen) for 15 minutes. Cells were sampled and imaged by confocal microscopy (C1 Nikon Confocal Microscope, Eclipse TE200-U; Nikon UK Ltd.). For each well, five images from the left, right, up, down, and central areas were taken and then quantified by using the ImageJ software (National Institutes of Health, Bethesda, MD). The average of dead and live cell from five images was used as the number of the dead or live cell of the well. The percentage of dead cells (PI staining) was calculated by dividing the number of PI/DAPI dual-positive cells with the total number of nuclei in the field of view (DAPI staining).
Data Analysis
Gene fold changes, cytokine levels, retinal thickness, and RPE cell viability of different experimental groups were compared with nontreated controls using unpaired Student's t-test. Data were expressed as mean ± SE (mRNA expression, RPE cell viability) or ± SD (retinal thickness). A difference between the mean of separate experimental conditions was considered significant at P less than 0.05.
Results
Fundus Abnormalities in C57BL/6J Mouse Eyes Following Normal Saline Injection
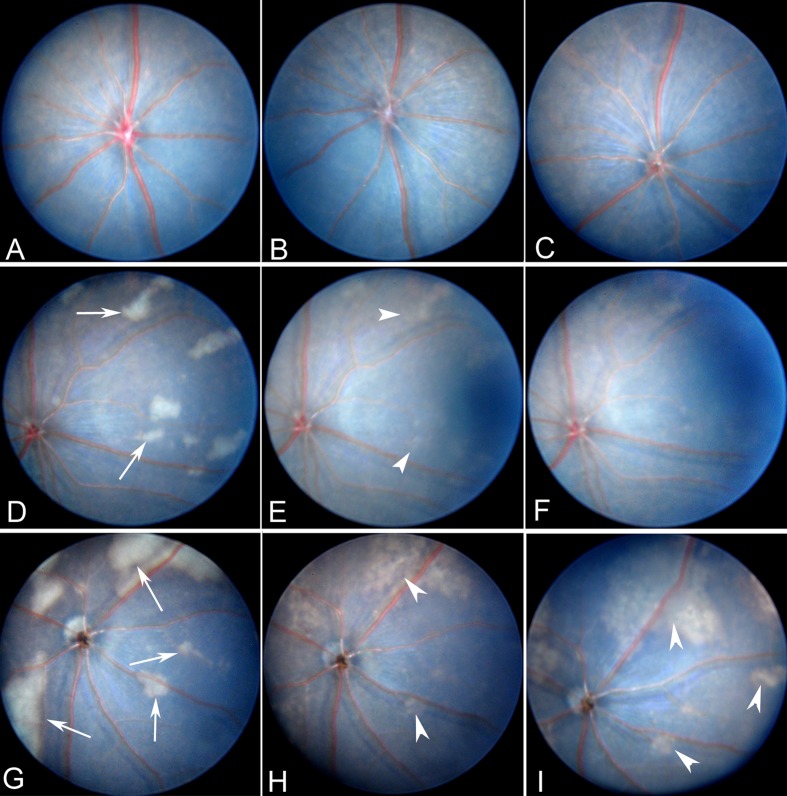
Fundus examinations at 24 hours, 1 and 3 weeks post intravitreal injection revealed no abnormalities in eyes from control (Fig. 1A), needle punch (Fig. 1C), and PBS-injected (1 μL/eye, Fig. 1B; 2 μL/eye, not shown) groups. Interestingly, 24 hours following intravitreal injection of NS (Fannin Healthcare Co.), patches of whitish lesions were observed in 40% and 58.33% of eyes receiving 1 μL (n = 20, Fig. 1D) and 2 μL (n = 12, Fig. 1G), respectively. Lesions were randomly spread throughout all retinal sectors, varying in size from 0.7 to 10 optic disc diameter. In general, lesion size was larger in eyes receiving 2 μL NS compared with eyes receiving 1 μL NS. By 1 week post injection (p.i.), whitish lesions partially disappeared and fundus began to recover its normal appearance in eyes receiving 1 μL NS injection (Fig. 1E). The majority of lesions vanished by 3 weeks p.i., and only a few lesions remained visible (Fig. 1F). However, in eyes receiving 2 μL NS injection, many lesions remained visible at 1 week (Fig. 1H) and 3 weeks (Fig. 1I) p.i. A repeated experiment using home-made NS (n = 14, 2 μL/eye) revealed similar results (10/14 eyes developed lesions).
Figure 1. .
Fundus images of C57BL/6J mouse eyes following intravitreal injection. (A) Fundus image from a normal C57BL/6J mouse. (B) Fundus image from PBS-injected mouse eyes at 24 hours p.i.. (C) Fundus image from needle-punched eye. (D, E) Fundus images from mice receiving 1 μL NS intravitreal injection at 24 hours (D), 1 (E) and 3 weeks (F) p.i. (G–I) Fundus images from mice receiving 2 μL NS intravitreal injection at 24 hours (G), 1 week (H), and 3 weeks (I) p.i. Arrows indicate whitish lesions. Arrowheads show the same lesions over time.
Acidic load can cause photoreceptor degeneration.17 The pH in NS used in this study was 6.0, whereas the pH in PBS is 7.4. To understand whether NS-induced fundus lesions were promoted by the acidic load, 1 (n = 4) and 2 μL (n = 4) of PBS with pH 6.0 was injected intravitreally. Interestingly, no fundus lesions were observed (data not shown). In addition, 1 hour after intravitreal injection of NS (1 and 2 μL) the vitreous humor had physiological pH values (pH ∼7.4, data not shown). These results suggest that the lower level of pH in NS is unlikely responsible for the fundus lesions.
Normal Saline-Induced Retinal Lesion in SD-OCT Investigation
To further understand the nature of fundus whitish lesions, we carried out SD-OCT examinations at 24 hours and 3 weeks p.i. in eyes receiving 1 μL of NS. All layers of the retina are clearly visible and no abnormalities were detected in nontreatment control (Fig. 2A), intravitreal PBS injection (Fig. 2B), and needle punch mice (Fig. 2C). In NS-injected mice, areas of hyperreflection (correlate to whitish lesions in fundus images [Fig. 2E; TEFI]) were detected at 24 hours p.i. (inserted image, Fig. 2D). The lesions were located in the outer retina, between photoreceptor IS/OS and RPE layers (Fig. 2D). The lesion size varied from 150 to approximately 500 μm in width and 20 to approximately 50 μm in depth. By 3 weeks p.i., the retinal structure appeared normal in those regions affected by lesions (Fig. 2F). Moreover, the retina presented no structural anomalies in regions unaffected by lesions, in neither 24 hours nor 3 weeks p.i. (data not shown).
Figure 2. .
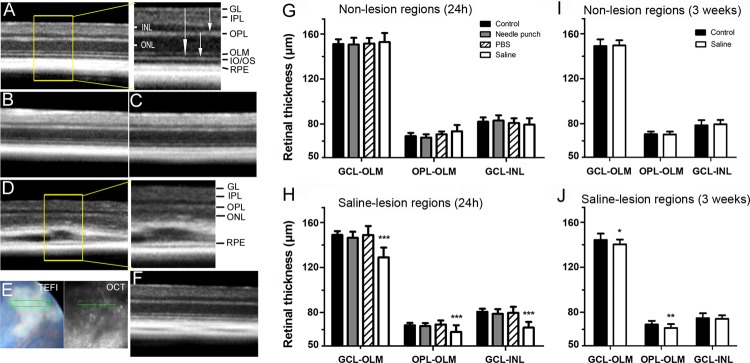
Evaluation of fundus lesions by SD-OCT. (A–C) SD-OCT sections showing normal morphology of retina and RPE in control nontreated mice (A) and 24 hours following intravitreal PBS-injection (B) or needle eye punch (C). The enlarged image in (A) shows a detailed view of retinal layers. (D, F) SD-OCT sections from intravitreal NS-injected (1 μL) eyes at 24 hours (D) and 3 weeks (F) p.i. (E) Fundus TEFI images were used as reference images to identify lesion areas during OCT scanning. Enlarged image in (D) shows lesions in outer retinal layers. (G–J) The bar diagram shows retinal thickness of nonlesion (G, I) and lesion (H, J) areas at 24 hours (G, H) and 3 weeks (I, J) after NS injection. The thicknesses between OPL-OLM, GCL-INL, or GCL-OLM were acquired from 6 to 10 eyes. * P < 0.05, ** P < 0.01, and *** P < 0.001 compared with nontreated control group, unpaired Student t-test's. Data are expressed as mean ± SD.
We carried out a morphometric analysis to investigate whether intravitreal injection of NS-induced retinal degeneration. PBS injection and needle punch did not affect retinal thickness in OCT measurement (Figs. 2G–J). In NS-injected mice, retinal thickness remained unchanged in nonlesion areas at 24 hours (Fig. 2G) and 3 weeks (Fig. 2I) p.i. However, a significant reduction of retinal thickness was detected at the lesion site at 24 hours p.i. (15% GCL-OLM, 17% GCL-INL, and 10% OPL-OLM; all values P < 0.001; Fig. 2H). By 3 weeks p.i., although retinal structure had recovered in the majority of lesion areas, a slight but statistically significant reduction of thickness was observed in the outer retina (P < 0.01, Fig. 2J). These data suggest that intravitreal injection of NS induces localized outer retinal degeneration.
Normal Saline Injection Mediated Retinal Damage in Histological Investigation
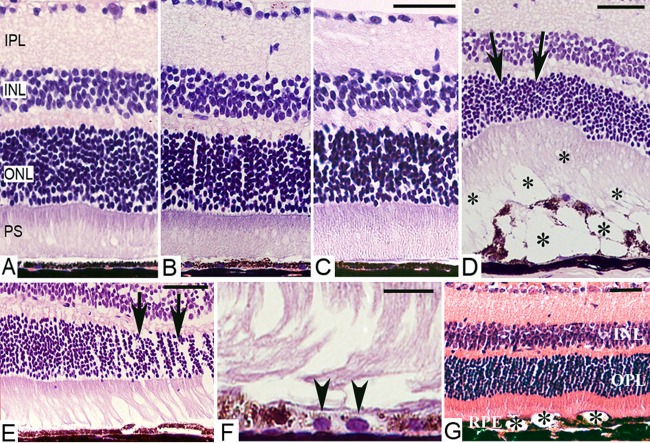
H&E staining of mouse eyes at 24 hours showed no abnormalities in nontreatment control (Fig. 3A), intravitreal PBS injection (Fig. 3B), and needle punch (Fig. 3C) groups. In NS-injected eyes where clinical investigations showed fundus abnormalities, patches of lesions were detected 24 hours p.i., characterized by multiple vacuolization in photoreceptor outer segments (OS) and RPE layers (asterisks, Fig. 3D). Despite the thickness of corresponding ONL was reduced (arrows, Fig. 3D), inner retinal layers appeared normal. At 3 weeks p.i., the majority of vacuoles disappeared in eyes receiving 1 μL NS injection (Fig. 3E), but RPE depigmentation was evident in some sectors (Fig. 3F). However, in eyes receiving 2 μL of NS, vacuolization of photoreceptor OS/RPE was still evident (Fig. 3G). In NS-injected eyes that had a normal fundus appearance in clinical investigations, H&E staining did not show any structural damage (data not shown).
Figure 3. .
Histology of C57BL/6J mouse eyes following intravitreal injection. (A–D) H&E sections of retinas from normal nontreated control mouse (A) or 24 hours after needle eye punch (B), PBS intravitreal injection (1 μL, [C]) or NS intravitreal injection (1 μL, [D]). (D) Vacuolization of photoreceptor OS and RPE layers (asterisks) 24 hours after NS injection. Note the thinning of the ONL (arrows) in areas corresponding to RPE damage. (E–G) Retinal sections 3 weeks after NS intravitreal injection (1 μL [E, F]; 2 μL [G]). Localized ONL thickness reduction (arrows, [E]) and RPE cell depigmentation (arrow heads, [G]) in recovered-lesion areas. (F) Vacuolization of the RPE layer is still evident in 2 μL NS injected eyes (asterisks). Scale bars, 50 μm (A–F); 20 μm (G). PS, photoreceptor segments.
Immune Gene and Cytokine Expression Following Normal Saline Injection
To understand the underlying mechanism related to NS intravitreal injection–mediated retinal damage, we evaluated immune gene expression in the retina and RPE/choroid by RT-PCR (Fig. 4). PBS injection or needle punch groups did not affect the expression of the immune related genes tested (TNF-α, IL-1β, IL-6, iNOS, VEGF), in both the retina (Fig. 4A) and RPE/choroid (Fig. 4B). However, 24 hours after NS injection, the expression of inflammatory genes TNF-α, IL-1β, IL-6, iNOS, and VEGF was significantly increased in the retina (Fig. 4A). In RPE/choroid, the expression of IL-1β and IL-6, but not of TNF-α, iNOS, and VEGF was upregulated (Fig. 4B). These results suggest that NS intravitreal injection induced inflammatory gene expression more in the retina than in the RPE/choroid.
Figure 4. .
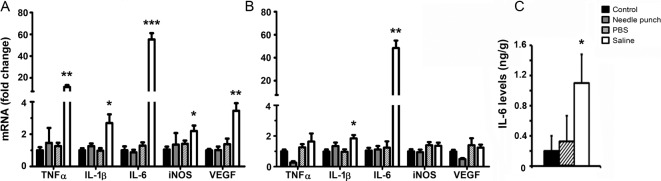
Immune gene expression in retina and RPE/choroid of C57BL/6J mice. Twenty-four hours following intravitreal injection of PBS or NS or needle eye punch, retinas, and RPE/choroidal tissues were collected and processed for RT-PCR analysis (A, B) and IL-6 cytokine levels (C). Gene fold changes and IL-6 levels are compared with untreated age-matched controls using unpaired Student t-test. * P < 0.05, ** P < 0.01, *** P < 0.001. Data are expressed as mean ± SE. (A, B) n = 6, (C) n ≥ 4.
Further analysis of cytokine levels using CBA revealed a significant increment of IL-6 in the NS injected retinas (1.1 ng/g, P < 0.05) compared with that in PBS-injected and control retinas (0.32 ng/g and 0.2 ng/g, respectively; Fig. 4C). TNF-α and IL-1β were undetectable in all samples (data not shown).
The Effect of Normal Saline in RPE Cell Cultures
RPE damage constitutes a major part of intravitreal NS injection–induced ocular pathology. NS is known to have direct effects on immune cells18,19 and peritoneal mesothelial cells.20 To further understand whether RPE damage is due to the direct effect of NS or secondary to retina-released cytotoxic factors, we carried out an in vitro RPE viability assay, using NS or retinal protein extracts from eyes injected with 1 μL of NS or PBS. In vitro incubation of RPE cells with 30% PBS or 30% NS for 1 or 24 hours did not affect RPE cell viability (data not shown), suggesting that both PBS and NS are not toxic to RPE cells. When RPE cells were incubated with 1 μg/mL retinal protein extracts from eyes receiving 1 μL NS injection, an increment in the number of dead cells (identified as PI+ DAPI+ cells, Fig. 5) were observed. Statistical analysis revealed a 15% reduction of cell viability in these RPE cultures, compared with those treated with protein extracts from PBS injected eyes or nontreatment controls (P < 0.05, Fig. 5). These results suggest that intravitreal injection of NS-mediated RPE cell death may be indirectly caused by cytotoxic mediators released from damaged retinal cells.
Figure 5. .
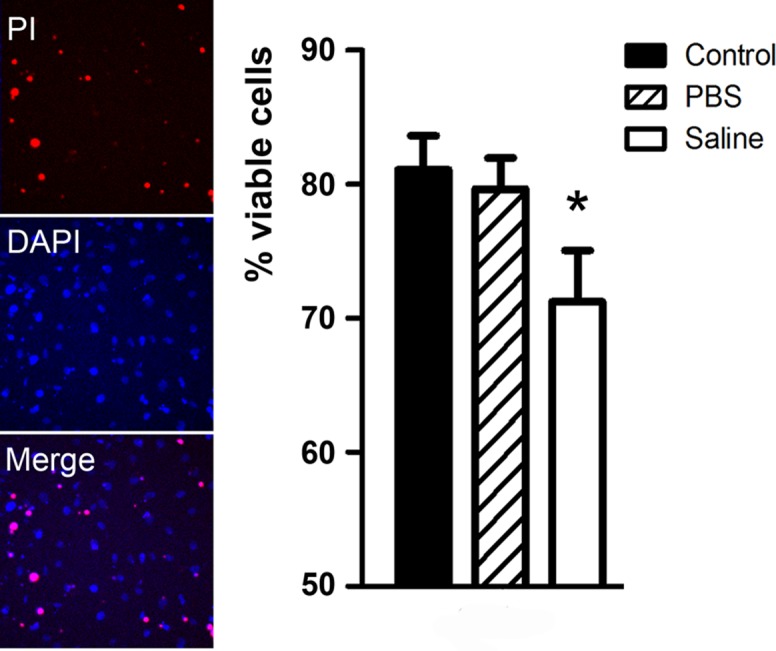
In vitro RPE viability. B6-RPE07 cells were treated with 1 μg/mL of retinal protein extracts, from eyes receiving 1 μL PBS or 1 μL NS intravitreal injection for 24 hours (see Methods). RPE cell cultures were incubated with DAPI and PI for 15 minutes and nonviable cells identified as PI+DAPI+ by confocal microscopy. A significant 15% reduction in cell viability was detected in RPE cultures incubated with retinal protein extracts from eyes receiving NS injection. * P < 0.05 compared with nontreatment control group, unpaired Student t-test. n = 3. Data are expressed as mean ± SE.
Discussion
In this study, we show that intravitreal injection of 0.9% normal saline dose-dependently induced retinal degeneration in C57BL/6J mouse eyes and that intravitreal injection of PBS had no significant adverse effect. NS injection appears to damage predominately outer retinal layers, including photoreceptors and RPE cells and the damage is accompanied by increased inflammatory cytokine expressions.
Both PBS and NS are isotonic solutions, and are widely used in various in vivo and in vitro studies. Why NS but not PBS is toxic to the retina is unknown. This may be related to different salt compositions in the two solutions. NS only contains 154 mM Na+ and 154 mM Cl−, whereas PBS contains 157 mM Na+, 139.7 mM Cl−, 4.7 mM K+, and 12 mM P−. Although the 0.9% NaCl is considered as “physiological” or “isotonic” saline, the solution is, however, neither “normal” nor “physiological” when comparing with extracellular fluid.21 Although infusing an appropriate volume of NS into the blood stream is safe, excessive infusion is harmful even in healthy volunteers.22,23 The adverse effect is believed to be related to the lower [Na+]:[Cl−] ratio in NS (1:1) than in human extracellular fluid or plasma (1.38:1)24 and the development of hyperchloraemia.25 The [Na+]:[Cl−] ratio in PBS is 1.13:1, slightly closer to that found in physiological conditions. At the cellular level, there is increasing evidence that NS may have adverse effects on immune cells. For example, 0.9% saline can induce neutrophil activation.18,19 Furthermore, NS is known to have toxic effects to the peritoneal cavity.25 Normal saline is commonly used in clinical practice to wash the peritoneal cavity during abdominal surgery or after chronic peritoneal dialysis.26 Accumulating evidence suggests that the use of NS in those conditions is more likely to produce adhesion than in no irrigation, and chronic exposure of the peritoneum to NS causes overgrowth of the connective tissue and formation of new blood vessels with that tissue.26 Further mechanistic studies suggest that NS damages peritoneal mesothelial cells20 through inducing oxidative stress.27
The adverse effects of NS in the retina have been observed in a number of in vivo studies, although the authors only used the data as vehicle controls and did not pay attention to the saline-mediated side effects. In a mouse model of endophthalmitis, intravitreal injection of NS as a vehicle control promoted a significant reduction in the electroretinogram (ERG) responses compared with contralateral noninjected eyes at 24 hours p.i.28,29 Although ERG was not tested in our NS-injected mice, the profound damage in photoreceptor and RPE cells (Figs. 1, 3) is likely to affect the ERG response, which would support those previous observations.28,29
Our investigations in OCT and histology show that saline-mediated photoreceptor damage is restricted to photoreceptor OS/RPE layers (Figs. 2, 3). Although the precise mechanism underlying saline-mediated OS damage remains elusive, it is possible that ion channels, in particular chloride channels may be involved. The OS is specialized for phototransduction, and is packed with a number of ion channels that are important for the transduction of light-induced responses, including the voltage-gated sodium channels, calcium channels, potassium channels,30–32 and chloride channels.33 The high ratio of chloride over sodium (compared with extracellular fluids) in the NS may affect ion homeostasis resulting in Cl− channel disruption. The Cl− channel is critically involved in many physiological processes (e.g., transepithelial H2O transport, retinal adhesion, or regulation of RPE volume34). It is well established that delicate electrolyte balance (mainly Cl−, Na+, and K+) is essential to maintain proper function of chloride channels.34 In this sense, the better [Na+]:[Cl−] ratio (1.13:1) and the presence of K+ in PBS would not disrupt the electrolyte composition of the vitreous and retina as the NS would.
The lower level of pH in NS (6.0) is unlikely to promote retinal damage, since the injection of PBS with pH 6.0 did not show any toxicity to the retina. Furthermore, vitreous pH was rapidly normalized after injection with 1 or 2 μL of NS (15% and 30% of total mouse vitreous volume, respectively). These data highlights the rapid pH buffering capacity of the mouse vitreous humor in response to a relatively large volume of acid load. A similar response may also exist in the human vitreous humor, as different ophthalmic drugs formulated in acidic buffers are well tolerated (e.g., Lucentis [Novartis] is at pH 5.5, Avastin [Genentech] at pH 6.2), although the injected volume of drugs in patients (1% of the total human vitreous volume) is significantly lower than that in mice, 15%–30%).
RPE damage in saline-injected eyes may be related to the release of cytotoxic mediators by damaged photoreceptors. Our histological investigation showed that RPE vacuolization is always associated with photoreceptor damage. Furthermore, NS did not affect RPE viability in vitro, whereas retinal extracts from NS-injected but not PBS-injected eyes induced RPE cell death. This is further supported by a more pronounced saline-induced inflammatory gene expression in retinal tissues compared with RPE/choroidal tissues.
In summary, we showed in this study that intravitreal injection of NS is toxic to the C57BL/6J mouse retina. Although the underlying mechanism remains elusive, the adverse effect appears to be related to photoreceptor and RPE damage. Despite that further investigations are needed to examine the adverse effect of NS to the retina of other species, including humans, we would urge not to use NS as a vehicle for intraocular injection in C57BL/6J mice, and should be used with caution in other species including humans.
Acknowledgments
The authors thank Judith Lechner for helping with the Cytometric Bead Array assay and Tom Friedel and Rosana Penalva for giving support in measuring mouse vitreous pH.
Supported by grants from Diabetes UK (11/0004230) and Fight for Sight (1362).
Disclosure: J.R. Hombrebueno, None; C. Luo, None; L. Guo, None; M. Chen, None; H. Xu, Diabetes UK (F), Fight for Sight (F)
Footnotes
heping.xu@qub.ac.uk
References
- 1.Avery RL, Pearlman J, Pieramici DJ, et al. Intravitreal bevacizumab (avastin) in the treatment of proliferative diabetic retinopathy. Ophthalmology. 2006;113:1695, e1–1695.15. doi: 10.1016/j.ophtha.2006.05.064. [DOI] [PubMed] [Google Scholar]
- 2.Brown DM, Kaiser PK, Michels M, et al. Ranibizumab versus verteporfin for neovascular agerelated macular degeneration. N Engl J Med. 2006;355:1432–1444. doi: 10.1056/NEJMoa062655. [DOI] [PubMed] [Google Scholar]
- 3.Haritoglou C, Kook D, Neubauer A, et al. Intravitreal bevacizumab (avastin) therapy for persistent diffuse diabetic macular edema. Retina. 2006;26:999–1005. doi: 10.1097/01.iae.0000247165.38655.bf. [DOI] [PubMed] [Google Scholar]
- 4.Rodriguez-Fontal M, Alfaro V, Kerrison JB, Jablon EP. Ranibizumab for diabetic retinopathy. Curr Diabetes Rev. 2009;5:47–51. doi: 10.2174/157339909787314239. [DOI] [PubMed] [Google Scholar]
- 5.Wang H, Sun X, Liu K, Xu X. Intravitreal ranibizumab (lucentis) for the treatment of diabetic macular edema: a systematic review and meta-analysis of randomized clinical control trials. Curr Eye Res. 2012;37:661–670. doi: 10.3109/02713683.2012.675616. [DOI] [PubMed] [Google Scholar]
- 6.Iturralde D, Spaide RF, Meyerle CB, et al. Intravitreal bevacizumab (avastin) treatment of macular edema in central retinal vein occlusion: a short-term study. Retina. 2006;26:279–284. doi: 10.1097/00006982-200603000-00005. [DOI] [PubMed] [Google Scholar]
- 7.Spaide RF, Chang LK, Klancnik JM, et al. Prospective study of intravitreal ranibizumab as a treatment for decreased visual acuity secondary to central retinal vein occlusion. Am J Ophthalmol. 2009;147:298–306. doi: 10.1016/j.ajo.2008.08.016. [DOI] [PubMed] [Google Scholar]
- 8.Iliev ME, Domig D, Wolf-Schnurrbursch U, Wolf S, Sarra GM. Intravitreal bevacizumab (avastin) in the treatment of neovascular glaucoma. Am J Ophthalmol. 2006;142:1054–1056. doi: 10.1016/j.ajo.2006.06.066. [DOI] [PubMed] [Google Scholar]
- 9.Hazirolan D, Pleyer U. Think global–act local: intravitreal drug delivery systems in chronic noninfectious uveitis. Ophthalmic Res. 2013;49:59–65. doi: 10.1159/000345477. [DOI] [PubMed] [Google Scholar]
- 10.van Kooij B, Rothova A, de Vries P. The pros and cons of intravitreal triamcinolone injections for uveitis and inflammatory cystoid macular edema. Ocul Immunol Inflamm. 2006;14:73–85. doi: 10.1080/09273940500545684. [DOI] [PubMed] [Google Scholar]
- 11.Yeh S, Albini TA, Moshfeghi AA, Nussenblatt RB. Uveitis, the comparison of age-related macular degeneration treatments trials (CATT), and intravitreal biologics for ocular inflammation. Am J Ophthalmol. 2012;154:429–435. doi: 10.1016/j.ajo.2012.05.011. e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group. Martin DF, Maguire MG, et al. Ranibizumab and bevacizumab for treatment of neovascular agerelated macular degeneration: two-year results. Ophthalmology. 2012;119:1388–1398. doi: 10.1016/j.ophtha.2012.03.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.IVAN Study Investigators. Chakravarthy U, Harding SP, et al. Ranibizumab versus bevacizumab to treat neovascular age-related macular degeneration: one-year findings from the IVAN randomized trial. Ophthalmology. 2012;119:1399–1411. doi: 10.1016/j.ophtha.2012.04.015. [DOI] [PubMed] [Google Scholar]
- 14.Paques M, Guyomard JL, Simonutti M, et al. Panretinal, high-resolution color photography of the mouse fundus. Invest Ophthalmol Vis Sci. 2007;48:2769–2774. doi: 10.1167/iovs.06-1099. [DOI] [PubMed] [Google Scholar]
- 15.Xu H, Koch P, Chen M, Lau A, Reid DM, Forrester JV. A clinical grading system for retinal inflammation in the chronic model of experimental autoimmune uveoretinitis using digital fundus images. Exp Eye Res. 2008;87:319–326. doi: 10.1016/j.exer.2008.06.012. [DOI] [PubMed] [Google Scholar]
- 16.Chen M, Muckersie E, Robertson M, Fraczek M, Forrester JV, Xu H. Characterization of a spontaneous mouse retinal pigment epithelial cell line B6-RPE07. Invest Ophthalmol Vis Sci. 2008;49:3699–3706. doi: 10.1167/iovs.07-1522. [DOI] [PubMed] [Google Scholar]
- 17.Yang Z, Alvarez BV, Chakarova C, et al. Mutant carbonic anhydrase 4 impairs pH regulation and causes retinal photoreceptor degeneration. Hum Mol Genet. 2005;14:255–265. doi: 10.1093/hmg/ddi023. [DOI] [PubMed] [Google Scholar]
- 18.Horton JW, Maass DL, White DJ. Hypertonic saline dextran after burn injury decreases inflammatory cytokine responses to subsequent pneumonia-related sepsis. Am J Physiol Heart Circ Physiol. 2006;290:H1642–H1650. doi: 10.1152/ajpheart.00586.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rhee P, Wang D, Ruff P, et al. Human neutrophil activation and increased adhesion by various resuscitation fluids. Crit Care Med. 2000;28:74–78. doi: 10.1097/00003246-200001000-00012. [DOI] [PubMed] [Google Scholar]
- 20.Polubinska A, Winckiewicz M, Staniszewski R, Breborowicz A, Oreopoulos DG. Time to reconsider saline as the ideal rinsing solution during abdominal surgery. Am J Surg. 2006;192:281–285. doi: 10.1016/j.amjsurg.2005.05.047. [DOI] [PubMed] [Google Scholar]
- 21.Wakim KG. “Normal” 0.9 per cent salt solution is neither “normal” nor physiological. JAMA. 1970;214:1710. doi: 10.1001/jama.214.9.1710b. [DOI] [PubMed] [Google Scholar]
- 22.Reid F, Lobo DN, Williams RN, Rowlands BJ, Allison SP. (Ab)normal saline and physiological hartmann's solution: a randomized double-blind crossover study. Clin Sci (Lond) 2003;104:17–24. doi: 10.1042/. [DOI] [PubMed] [Google Scholar]
- 23.Williams EL, Hildebrand KL, McCormick SA, Bedel MJ. The effect of intravenous lactated ringer's solution versus 0.9% sodium chloride solution on serum osmolality in human volunteers. Anesth Analg. 1999;88:999–1003. doi: 10.1097/00000539-199905000-00006. [DOI] [PubMed] [Google Scholar]
- 24.Veech RL. The toxic impact of parenteral solutions on the metabolism of cells: a hypothesis for physiological parenteral therapy. Am J Clin Nutr. 1986;44:519–551. doi: 10.1093/ajcn/44.4.519. [DOI] [PubMed] [Google Scholar]
- 25.Scheingraber S, Rehm M, Sehmisch C, Finsterer U. Rapid saline infusion produces hyperchloremic acidosis in patients undergoing gynecologic surgery. Anesthesiology. 1999;90:1265–1270. doi: 10.1097/00000542-199905000-00007. [DOI] [PubMed] [Google Scholar]
- 26.Breborowicz A, Oreopoulos DG. Is normal saline harmful to the peritoneum? Perit Dial Int. 2005;25((suppl 4)):S67–S70. [PubMed] [Google Scholar]
- 27.Polubinska A, Breborowicz A, Staniszewski R, Oreopoulos DG. Normal saline induces oxidative stress in peritoneal mesothelial cells. J Pediatr Surg. 2008;43:1821–1826. doi: 10.1016/j.jpedsurg.2008.02.010. [DOI] [PubMed] [Google Scholar]
- 28.Sugi N, Whiston EA, Ksander BR, Gregory MS. Increased resistance to staphylococcus aureus endophthalmitis in BALB/c mice: Fas ligand is required for resolution of inflammation but not for bacterial clearance. Infect Immun. 2013;81:2217–2225. doi: 10.1128/IAI.00405-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Whiston EA, Sugi N, Kamradt MC, et al. alphaB-crystallin protects retinal tissue during staphylococcus aureus-induced endophthalmitis. Infect Immun. 2008;76:1781–1790. doi: 10.1128/IAI.01285-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.MacLeish PR, Nurse CA. Ion channel compartments in photoreceptors: evidence from salamander rods with intact and ablated terminals. J Neurophysiol. 2007;98:86–95. doi: 10.1152/jn.00775.2006. [DOI] [PubMed] [Google Scholar]
- 31.Minke B, Selinger Z. The roles of trp and calcium in regulating photoreceptor function in drosophila. Curr Opin Neurobiol. 1996;6:459–466. doi: 10.1016/s0959-4388(96)80050-x. [DOI] [PubMed] [Google Scholar]
- 32.Wensel TG. Signal transducing membrane complexes of photoreceptor outer segments. Vision Res. 2008;48:2052–2061. doi: 10.1016/j.visres.2008.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pal R, Sekharan S, Batista VS. Spectral tuning in halorhodopsin: the chloride pump photoreceptor. J Am Chem Soc. 2013;135:9624–9627. doi: 10.1021/ja404600z. [DOI] [PubMed] [Google Scholar]
- 34.Wimmers S, Karl MO, Strauss O. Ion channels in the RPE. Prog Retin Eye Res. 2007;26:263–301. doi: 10.1016/j.preteyeres.2006.12.002. [DOI] [PubMed] [Google Scholar]